Abstract
Background:
Continuous glucose monitoring (CGM)/flash glucose monitoring (FGM) use in diabetes management is increasing. Cutaneous complications associated with these devices were reported. We conducted a systematic review to provide an overview of cutaneous complications with CGM/FGM use.
Methods:
We identified observational studies and intervention trials that report on cutaneous complications with CGM/FGM use up to January 14, 2019. Studies were identified through Medline, Embase, and PubMed, or with hand searching of the previous publications. Screening was duplicated and data extracted to consider four main themes: incidence rate and severity, participant perspectives of cutaneous complications, potential solutions, and future directions in diabetic technology relevant to reducing cutaneous complications.
Results:
A total of 54 eligible studies were identified. The overall event rate of cutaneous complications reported from 19 trials was one event per eight weeks of sensor wear-time of which 1.5% were considered severe. The most common cutaneous complications were wear-related erythema, itching, and induration. Although skin irritations were the most common cause of CGM/FGM discontinuation, most users experienced less pain or discomfort with CGM/FGM than capillary blood glucose testing. Future technological advances may reduce, but not eliminate cutaneous complications.
Conclusion:
The incidence rate of reported cutaneous complications with CGM/FGM use from the available literature is low, with one event per eight weeks of sensor wear-time. Reported complication severity was also low, leading to low rates of CGM/FGM discontinuation. However, there appear to be discrepancies between reporting in trial and observational data. Greater constancy in reporting is necessary to understand the frequency of this issue.
Introduction
Achieving healthy glycemic control is a critical goal for those with diabetes given the increased risk of both micro- and macrovascular complications.1,2 Crucial to achieving this is frequent self-monitoring of glucose.3,4 Given the limitations of traditional capillary glucose monitoring, 5 interstitial glucose monitoring such as continuous glucose monitoring (CGM) and flash glucose monitoring (FGM) is growing in popularity. 6 These devices typically involve the insertion of a sensor under the skin that requires additional external securing with an adhesive cutaneous patch for periods usually of 6 to 14 days. Results from CGM/FGM trials indicate reductions in hypoglycemia and improvements in glycated hemoglobin (HbA1c)7-12 suggesting that their use in standard diabetes care is likely to increase, particularly as they are also a crucial component of automated insulin delivery technology. For the above reasons, newly published guidelines recommend CGM as standard care for children and adolescents with type 1 diabetes. 13
Cutaneous complications arising from insulin pump technology, which use similar adhesives, have been previously reported 14 and are now also emerging in the CGM/FGM literature.15-19 These often involve degrees of allergic contact dermatitis from CGM/FGM adhesives15-18 and pose a management challenge for both patients and healthcare professionals. 19 It is important to be able to counsel prospective users of CGM/FGM with accurate information on the potential of cutaneous complications, so that realistic expectations can be set prior to undertaking this mode of glucose monitoring. However, few resources exist to guide healthcare professionals on these issues. 20 We therefore conducted a systematic review to identify reported cutaneous complications related to CGM/FGM use, calculate a current incidence rate, describe apparent severity and implications, as well as document any additional themes that pertain to cutaneous complications with CGM/FGM use now, and in future glucose sensing technology.
Methods
We followed reporting standards for systematic reviews 21 (see Supplement 1 for PRISMA checklist). The protocol for this systematic review is registered (CRD42019121526).
Study Eligibility and Search Strategy
Peer-reviewed reports of clinical trials and observational studies of research or clinical practice with CGM/FGM that mention or consider cutaneous complications were eligible. Published opinions on solutions to cutaneous complications with CGM/FGM or future directions were also considered. Eligible reports were identified with an online search of Medline, Embase, and PubMed up to January 14, 2019. Search terms included (Diabetes OR T2D* OR T1D* OR GDM* OR impaired glucose OR blood glucose control OR normal glucose tolerance) AND (continuous glucose monitoring OR flash glucose monitoring OR CGM* OR FGM*) AND (skin OR cutaneous OR epidermis). No date or language limits were applied to our search. This online search was complemented by hand searching of the reference lists of relevant studies and systematic reviews. Two authors independently screened titles, abstracts, and full text articles to identify eligible studies.
Data Extraction
Data were extracted into a standardized spreadsheet used in the previous systematic reviews 22 and checked by a second reviewer. We extracted the following information for each article: geographical location, study design, interventions (if relevant), inclusion and exclusion criteria, mean age, type of diabetes, type of glucose monitor, duration of device wear, and details of any cutaneous complications. Corresponding authors of articles were contacted to request additional information.
Data Analysis
We calculated an incidence rate (frequency of an event divided by total wear time) of CGM/FGM use from studies reporting subcutaneous complications. Severity was also analyzed. In addition, the data from studies identified by this systematic review are further described as they pertain to major themes: participant perspectives of these complications, potential solutions for users to mitigate or avoid cutaneous complications with CGM/FGM use, or cutaneous complications as a potential barrier to ongoing CGM/FGM use, and future developments in glucose sensing technology that may alter the future occurrence or experience of cutaneous complications.
Results
The process to identify eligible trials and studies is shown in Figure 1. Data from 46 observational studies and intervention trials of 5609 participants, and eight published opinions/reviews that discuss potential solutions and future directions were included. Identified studies were conducted in North America (50%), Europe (43%), and Asia or Australia (7%). A description of each study identified by this review is shown in Table 1. The majority (67%) of studies were conducted in the last decade.
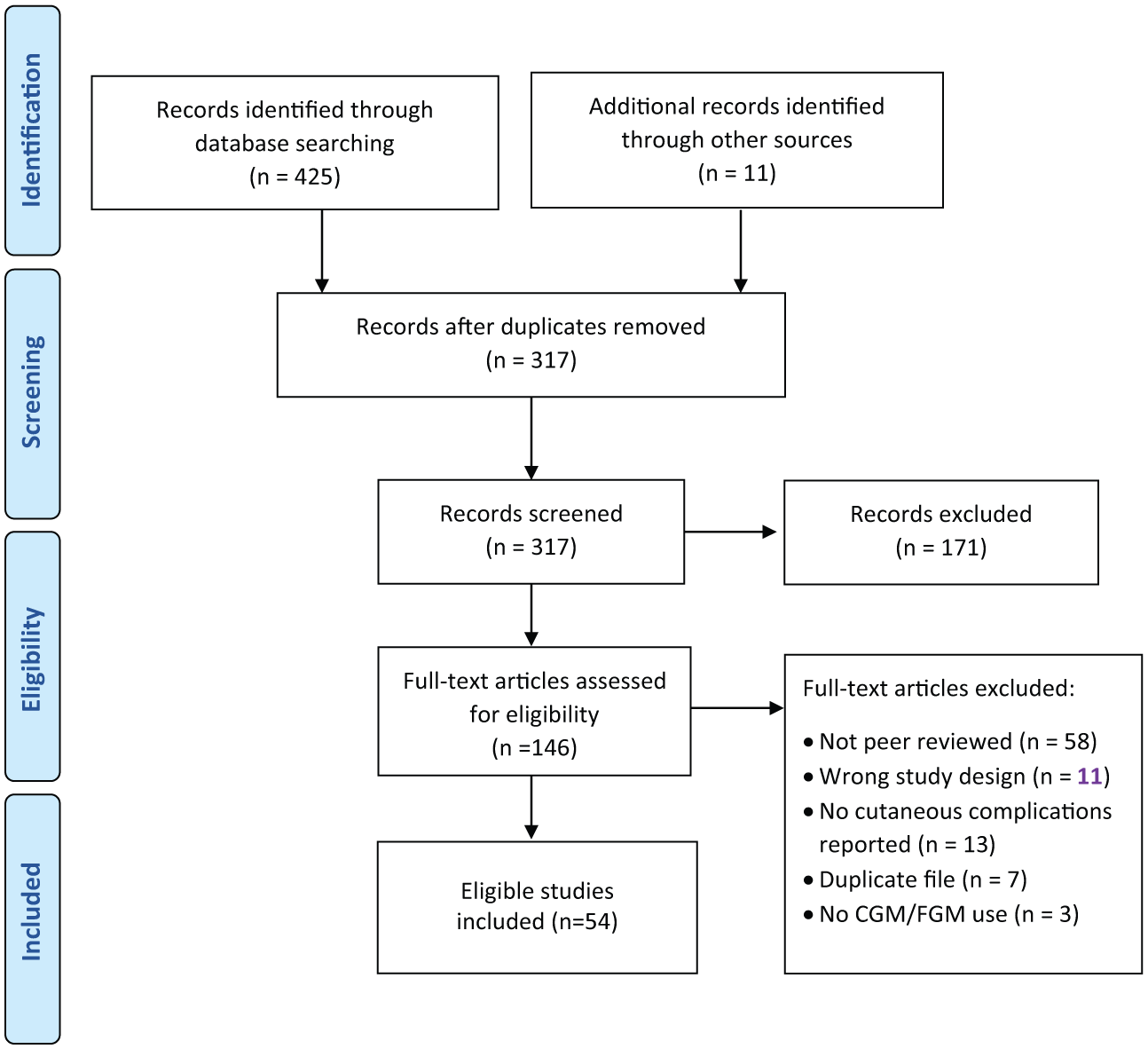
The study selection process to identify eligible trials and studies.
Basic Description of Studies and Trials Identified as Eligible by This Systematic Review.
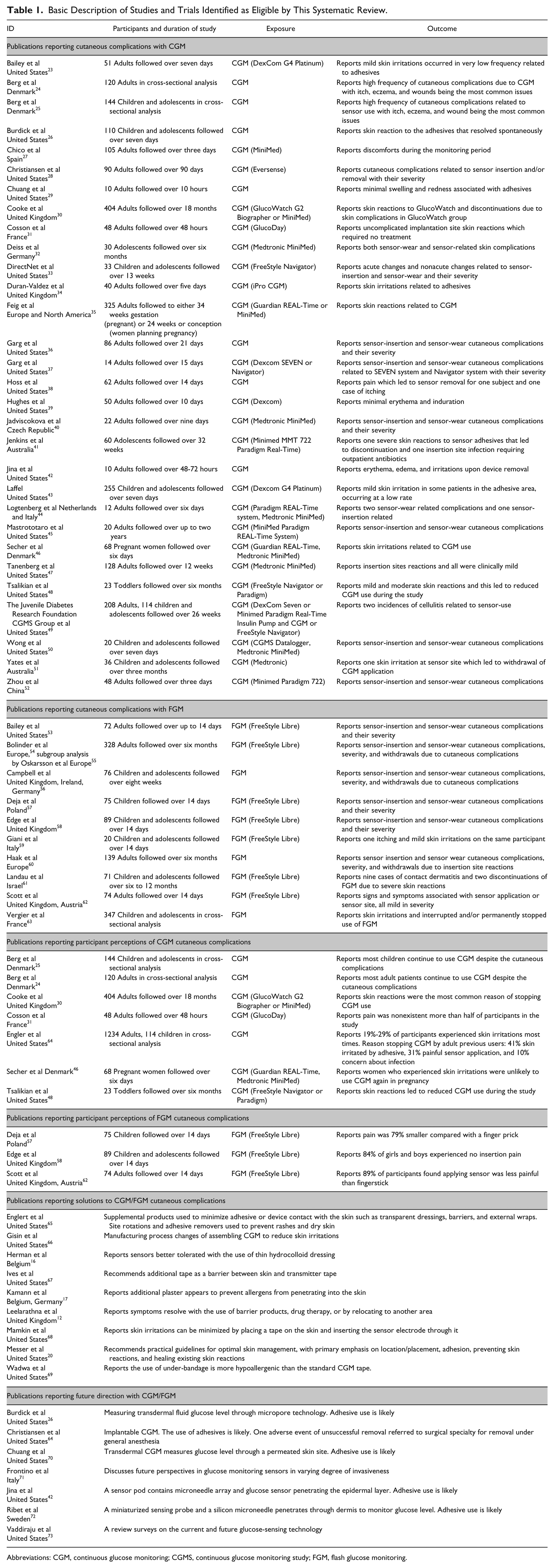
Abbreviations: CGM, continuous glucose monitoring; CGMS, continuous glucose monitoring study; FGM, flash glucose monitoring.
Cutaneous Complication Incidence
Six studies or trials with FGM53,54,56,59,60,62 and 13 studies or trials with CGM26,31,32,34,36-38,44,45,47-49,52 provided sufficient information to calculate an overall incidence rate. From 1158 participants of these trials, we identified 1090 events of cutaneous complication over 138 to 158 accumulated years of wear time. The cutaneous complication event rate was 0.13 to 0.15 for every week of wear-time, indicating one event every eight weeks. Reported occurrence varied considerably between trials with higher rates when researchers inspected the site (1.4 events per week of wear time) 53 than when patient reported adverse events (0.04 events per week of wear-time). 26 Many papers reported only moderate to severe events. The most common cutaneous reaction was erythema (55%), followed by itching/pruritus (11%) and induration (9%). A full summary of cutaneous complication frequency by type is shown in Table 2. Studies27,29,36-38,40,41,44,49,50,52,55,56,60,61 reported more adhesive or wear-associated cutaneous complications (80%) (eg, erythema and itch) than direct insertion-related complications (20%) (eg, bruising or bleeding).
Reported Cutaneous Complications Related to Continuous Glucose Monitoring/Flash Glucose Monitoring Use.

Several studies reported severity of cutaneous complications.36,37,40,47,48,55,56,59,60,62 Overall, 78.6% of cutaneous complications were rated as mild, 19.8% were moderate, and only 1.5% were severe. Skin reactions or irritations were the most common reason for participants to withdraw from studies.30,41,51,54,60
Data from observational studies indicate that more than 70% of participants have experienced cutaneous complications related to CGM/FGM use at some point, with itch, eczema, and insertion wounds the most commonly reported complications.24,25,63
Participant Perspectives of Cutaneous Complications
Several studies assessed participant perspectives toward the use of CGM/FGM with questionnaires,24,25,30,31,46,48,53,57,58,62,65 primarily comparing CGM/FGM use with capillary blood glucose testing.31,46,48,53,57,58,62,64 Many studies indicated that both adults and children report less pain and bleeding associated with CGM/FGM use than capillary blood glucose testing. Sensor insertion pain or bleeding was not considered a deterrent of use for the majority of individuals.31,53
Cutaneous complications are often given as a reason for discontinuing CGM/FGM use. Skin irritation due to adhesives was given as the predominate reason for adults (42%) 64 and children (40%) 48 to stop CGM/FGM use. Two studies reported that many children and adult patients continue to use CGM despite experiencing cutaneous complications,24,25 but concern about infection accounted for 10% of the previous adult CGM/FGM users discontinuing CGM. 64
Potential Solutions
Data on this topic were largely from reviews or published opinions. The most recent of these provides a detailed discussion on the current solutions for preserving skin integrity for chronic device use for those wanting more detail. 74 In brief, this process should begin with setting appropriate expectations, and an assessment of current and past skin health. 74 Once that has been done, the current solutions for reducing cutaneous complications with CGM/FGM use relate to careful consideration for appropriate device placement, minimizing adhesive or sensor contact with the skin,65,67 or changing the adhesion process of CGM components. 66
In practical terms, options include adequate drying prior to insertion and the use of various supplemental products such as adhesive barriers, tackifiers (to improve adhesion), or possibly off-label steroid sprays prior to insertion for those with known prior reactions (eg, fluticasone).16,17,20,65,68,69 To minimize adhesive exposure, an external wrap (eg, cohesive bandage) to secure sensors without the use of additional adhesives can also be used. Dressings can also be trimmed to only cover sensor adhesive areas and not the transmitter, so as to prevent moisture buildup. Site rotations, adhesive remover, and the use of steroid cream and antihistamines have all been reported to prevent or manage rashes and dry skin. 74 Careful sensor removal so as to avoid further skin damage cannot be overemphasized; and if an injury is present, postremoval skin care for recovery may be necessary. 74 In addition, FGM/CGM suppliers have reportedly modified manufacturing to reduce skin reactions, however, such changes have not removed the need for an adhesive barrier between skin and device. 66
Future Technology to Mitigate Cutaneous Complications
Several publications have considered new technologies in glucose sensing that may modify the risk of cutaneous complications.71,73 These technologies can be considered by their degree of invasiveness.
Invasive
Implantable CGM (such as the recently approved Eversense device) sits under the skin surface and currently last for up to 90 days, 28 reducing the risk of cutaneous complications associated with changing sensors. An adhesive patch is then used to secure the transmitter over the sensor. Complications related to sensor insertion and removal (bruising, erythema, and pain or discomfort) have been reported. 28 In this pivotal trial for regulatory approval, one participant of 90 had to be referred to surgical specialty for removal under general anesthesia. No skin reactions were reported due to the adhesive patch.
Minimally invasive
Microneedle-based CGM involves two components: a miniaturized sensing probe and a silicon micro-needle 72 or an array of silicon microneedles 42 which penetrate the dermis 72 or epidermis 42 to monitor glucose level as compared with hypodermis, as in the current CGM/FGM devices. Adhesive is used to secure the device to the skin. Skin complications such as erythema and edema have been observed at the sensor site upon device removal. 42 This technology is currently not approved for general use and requires further trials and development to assess accuracy and safety.
Noninvasive
Transdermal CGM continuously measure glucose through ultrasonically permeated skin sites. 70 Alternatively, transdermal fluid glucose is measured by producing micropores in the skin and drawing transdermal fluid to the glucose sensor, 26 or with electrochemical sensing of the skin surface. 75 The use of adhesives for these options are likely, although not specifically mentioned in the current literature. Cutaneous complications have yet to be reported from the available studies on this emerging glucose-sensing technology.
Discussion
This systematic review and the number of studies available for inclusion highlight a significant interest in CGM/FGM technology for modern diabetes management. To our knowledge, this is the first systematic review to synthesize and focus on the occurrence of cutaneous complications in studies with CGM/FGM devices. As reported in the available trials, we determined an overall cutaneous complication incidence rate of one event/eight weeks wear time. Most complications were minor skin reactions/irritations related to wear time. Few participants in these trials ceased use due to these complications.
There was a great deal of variability in the incidence rate of cutaneous complications as reported in the individual trials and between trials where cutaneous adverse events were patient vs investigator reported. In addition, the primary objectives for these trials were not to examine cutaneous complication frequency, and most were of relatively short duration. All these factors suggest that not all minor or low severity complications have been reported as adverse events during the published trials, indicating that our trial-reported incidence rate is likely an underestimate of the true real-world incidence. Supporting this possibility, the data from observational studies indicate a likely high prevalence of experiencing cutaneous complications.24,25 To fully understand this important issue and allow developments for prevention and treatment, consistent reporting, including standardized definitions of severity, is required, and future reporting of trials should identify all occurrences of cutaneous complications by type and severity so as to better inform on this issue.
Crucial for perspective on this topic is the clearly increasing rates of CGM use worldwide6,13 and the overall high satisfaction seen with users of CGM/FGM in the available literature.76,77 These factors combined with our review findings would suggest that the perceived benefits to the user largely outweigh the downsides of cutaneous complications. Further solutions for prevention and management of cutaneous issues are required, 20 as the next generation of CGM still largely relies on both adhesive use and penetrating the skin barrier. Regarding the current adhesives, varying degrees of contact dermatitis can occur depending on the manufacturer, and reactions to isobornyl acrylate, colophonium, ethyl cyanoacrylate, and N,N-dimethylacrylamide have all been reported.78-80 There is also a recognized need for manufacturing changes to improve breathability and reduce trapped moisture that contribute to skin reactions with the current technologies. 81 Further research into hypoallergenic adhesives or the long-term possibility of glucose sensing without penetrating the skin barrier are necessary to reduce these barriers to CGM/FGM use. Integration of glucose sensing with insulin delivery may also decrease the risk of cutaneous complication by reducing the number of points of contact with adhesive.
Our review has several strengths. We have followed best practice guidelines for conducting and reporting of systematic reviews. Given the nature of our research question, we have presented results of our review under four themes, each necessary for the consideration of cutaneous complications with CGM/FGM use. Our search strategy also recognized many recent conference abstracts that have not yet resulted in a peer-reviewed publication, indicating the growing use of CGM/FGM, and therefore, future relevance of our review. Our review also has limitations. Not all trials utilizing CGM and FGM report adverse events, let alone cutaneous complications. Our incidence rate is based on trials that did report cutaneous complications and therefore may introduce bias into our estimates. Further, data on the specific type and severity of complications were not often available. We believe that this limitation can be minimized in further considerations of this topic should publications routinely report cutaneous complications with diabetic devices and emerging technologies.
Conclusion
This systematic review of the CGM/FGM literature focused on cutaneous complications highlights an overall trial-reported incidence rate of one cutaneous complication for every eight weeks of wear-time. These were typically mild in severity and resulted in minimal discontinuation. However, this rate is likely an underestimate, especially for mild events. There is a need for improved routine and consistent reporting of CGM/FGM related cutaneous complications in future CGM/FGM trials; as well for improved techniques and technology to improve the patient experience and reduce the future impact and occurrence of cutaneous complications.
Supplemental Material
MOHD_ASARANI_Supplement_1 – Supplemental material for Cutaneous Complications With Continuous or Flash Glucose Monitoring Use: Systematic Review of Trials and Observational Studies
Supplemental material, MOHD_ASARANI_Supplement_1 for Cutaneous Complications With Continuous or Flash Glucose Monitoring Use: Systematic Review of Trials and Observational Studies by Nurul A. Mohd Asarani, Andrew N. Reynolds, Sara E. Boucher, Martin de Bock and Benjamin J. Wheeler in Journal of Diabetes Science and Technology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Department of Women’s and Children’s Health and the Department of Medicine, University of Otago.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
