Abstract

Glucose meters remain an important means to measure glucose for people with diabetes. A recent study provided a thorough analysis of accuracy of four glucose meters in the hands of actual users and characterized the different errors made by users. 1 However, this study was typical in that the number of times that no glucose result was obtained was not reported.
The Food and Drug Administration (FDA) guidance for self-monitoring glucose meters 2 states that cases of “no result” should be included in the submission, but there is no requirement for including the frequency or percentage of no results.
An additional source of glucose meter performance data is the adverse event database from the FDA. I examined adverse events for glucose meters for the year 2021. Data were downloaded from the FDA website 3 into an SQL Server database and through queries—a table of 17 755 glucose meter adverse events was created with 9234 distinct events across all manufacturers. The extra records in the table (eg, for a total of 17 755) are responses by the manufacturer about each adverse event. The glucose meter events were classified by the manufacturer in the database as malfunction (8246), injury (984), and death (4). Further queries into the event text description suggested the problems that caused the events to be reported. Table 1 shows that a frequent reason for an adverse event was that no glucose result was obtained. (Another frequent complaint was an inaccurate result).
Causes for No Result by Event_Type.
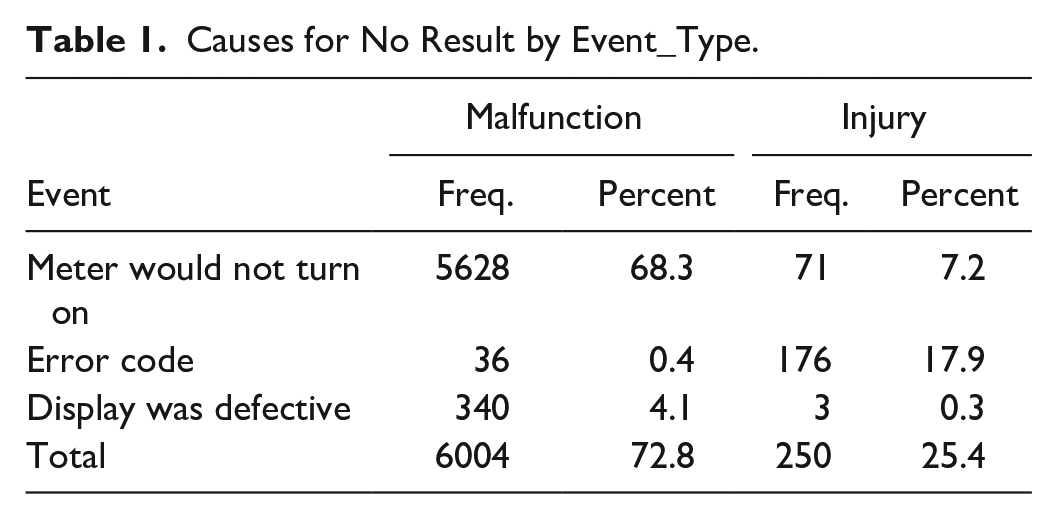
Glucose meters are a point of care (POC) device. These devices provide a result quickly and help people with diabetes to make treatment decisions. Failure to obtain a glucose result, especially during an emergent need is a serious problem. Here is an example from the 2021 adverse event database: A CALLER REPORTED ON BEHALF OF A CUSTOMER WHO RECEIVED AN UNSPECIFIED ERROR MESSAGE ON (B)(6) 2020 WHILE USING THE GLUCOSE METER AND THEREFORE WAS UNABLE TO MONITOR HIS GLUCOSE. THE CUSTOMER LOST CONSCIOUSNESS AND SUBSEQUENTLY A CONCUSSION. PARAMEDICS WERE CALLED AND THE CUSTOMER WAS TRANSPORTED TO THE HOSPITAL WHERE AN HCP READING OF “MORE THAN 900 MG/DL” WAS OBTAINED. THE CUSTOMER WAS DIAGNOSED WITH HYPERGLYCEMIA AND TREATED WITH HUMALOG AND LANTUS INSULIN. THE CUSTOMER WAS TREATED IN THE INTENSIVE CARE UNIT, DISCHARGED ON (B)(6) 2020, AND RECEIVED NEW PRESCRIPTIONS FOR HUMALOG AND LANTUS. THERE WAS NO REPORT OF DEATH OR PERMANENT INJURY ASSOCIATED WITH THIS EVENT.
This date of the report is April 1, 2021, and terms such as (B)(6) are redactions from the FDA. The rate of no glucose results should be a metric that is reported for glucose meter evaluations.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
