Abstract
Background:
In type 1 diabetes (T1D), closed-loop systems provide excellent overnight fasting blood glucose control by adjusting the insulin infusion rate based on corresponding changes in sensor glucose levels. In patients on multiple daily insulin (MDI) injections, such control in overnight glucose levels has not been possible due to the inability to alter the absorption rate of long-acting insulin after injection. In this study, we tested the hypothesis that increases/decreases of fasting glucose levels could be achieved by cooling/warming the skin around the injection site, which would result in lower/higher Glargine absorption rates from its subcutaneous depot.
Methods:
Fourteen subjects with T1D (4 females; age 39.6 ± 16.7 years, HbA1c 7.8 ± 1.1%, BMI 25.4 ± 2.8 kg/m2) on MDI therapy underwent fasting pharmacokinetic and pharmacodynamic studies that started at ~8
Results:
Cooling the skin around the glargine injection site reduced insulin concentrations by >40% (P < .01 versus the warming study, P = .21 versus the control study), accompanied by a 55 mg/dL increase in serum glucose (P < .01 versus the control study). Conversely, skin warming prevented the fall in serum insulin (P = .2 versus the control study; P < .01 versus the cooling study), resulting in a 40 mg/dL reduction in serum glucose (P < .001 versus the cooling study, P = .11 versus the control study).
Conclusions:
This proof of concept study has shown that cooling and warming the skin around the injection site provides a means to decrease and increase the rate of absorption and action of insulin glargine from its subcutaneous depot.
Keywords
Nighttime is often the worst time for controlling glucose levels in patients with type 1 diabetes receiving either open-loop insulin pump or multiple daily insulin injection therapies. 1 With both treatments, most patients receive fixed overnight basal insulin doses; whereas, the amount of insulin required at any given time during the night varies widely from night to night. Patients are susceptible to hyperglycemia on some nights due to emotional stress, nighttime binging or delayed postprandial bolusing after dinner or bedtime snacks. 2 On other nights, patients are at greater risk for nocturnal hypoglycemia due to the delayed effects of antecedent exercise during the day 3 and because of impaired counterregulatory responses to falling glucose levels during sleep. 4 Consequently, patients with T1D on multiple injection regimens can take the same bedtime dose of a long-acting insulin analog over and over and get a different result almost every night.
The introduction of integrated insulin pump and continuous glucose monitoring systems that can automatically adjust the rate of infusion of rapid-acting insulin analogs to maintain sensor glucose levels within the target range have transformed the precision and safety of overnight glucose control in T1D. 5 In broad terms, these systems vary the rate of insulin delivery from night to night to achieve the same targeted level of glucose control every morning with little or no hypo- or hyperglycemia. However, in patients on multiple daily insulin injection regimens, such precision in regulating overnight glucose levels has not been possible due to the inability to alter the rate of absorption of long-acting insulin analogs following the bedtime subcutaneous injection. In this proof of concept study, we tested the hypothesis that cooling or warming the skin around the long-acting insulin injection site would provide a means to automatically decrease or increase the rate of systemic absorption of insulin glargine from its subcutaneous depot, which in turn would result in higher or lower blood glucose levels, respectively.
Research Design and Methods
Study Design
The study was an open-label, randomized, cross-over design in subjects with type 1 diabetes receiving multiple daily injection therapy. Each enrolled subject underwent fasting pharmacodynamic (PD) and pharmacokinetic (PK) studies on 3 separate days in random order: a control day without cooling or warming the injection site and two experimental days, one with injection site cooling and the other with injection site warming for 240 minutes.
The primary PD outcome of the study was the differences in the incremental changes in serum glucose levels from baseline to 240 minutes between the control versus the experimental studies, as well as between the cooling and warming experiments. We also measured the differences in the percentage change from baseline to 240 min in circulating serum insulin concentrations between the three studies. Evidence of any skin inflammation or irritation around the injection sites was also collected.
Study Subjects
To be eligible for the study, the subjects had to be receiving insulin glargine (Lantus insulin, Sanofi-Aventis, France) for basal insulin replacement and have a clinical diagnosis of type 1 diabetes. They also had to be between 18 and <75 years of age and have HbA1c values <11% on enrollment. Exclusion criteria included pregnancy or breastfeeding, alcohol abuse or other serious medical or psychiatric problems. The IRB board of the Hadassah Medical Center in Jerusalem, Israel, approved the study and eligible subjects gave written informed consent for their participation in the study.
Study Procedures
On each study day, subjects reported to the clinical research center in the morning after an 8-10 hour overnight fast, without taking bedtime dose of insulin glargine on the previous night. An indwelling intravenous catheter was inserted and blood samples were obtained for measurement of serum glucose and insulin every 20 min throughout the study. The studies were performed in a sitting position in a temperature-controlled room (24 ± 3°C).
At the 8
During the 0 to 240 min cooling and warming experiments, the studies were prematurely terminated and exogenous glucose was administered intravenously if blood glucose meter measurements fell below 75 mg/dL. The studies were also prematurely terminated and intravenous insulin given if blood glucose meter measurements were greater than 250 mg/dL.
Cooling/Warming Device
A skin area of 3 × 2 cm over the injection site was warmed using the InsuPad device (6). During the warming period, the skin was warmed intermittently, warming to 40°C for 10 minutes followed by no warming for 10 minutes until the end of warming period. This warming profile enables establishment of increased local blood perfusion without increasing the temperature at the subcutaneous drug depot above 40°C. The skin area of 3 × 2 cm over the injection site was cooled using a modified InsuPad device attached to the skin by the InsuPad injection window. During the cooling period, skin was cooled to a temperature of 15°C constantly. Cooling was achieved by flowing cold water through the modified InsuPad device using Blanketrol III (CSZ, Cincinnati, OH, USA). The skin temperature during cooling or warming was monitored using a temperature sensor connected to a control circuit to ensure correct skin temperatures during warming/cooling periods.
Laboratory Methods
Blood samples for glucose and insulin measurements were collected in serum separating gel tubes and kept for 20 minutes on ice to allow for clotting and then were centrifuged and stored in -20 C° until measurement. Glucose levels were measured using hexokinase enzymatic method (Glucose HK gen.3 on Cobas 800 system, Roche Diagnostics, Manheim, Germany). Insulin assays were performed using an enzyme-linked immunosorbent assay test kit (Eliza assay kit from Crystal Chem, Elk Grove Village, IL, USA) which is sensitive to all insulin types. This broad sensitivity is not expected to add interferences to our basal insulin measurement as subjects received only iv insulin during the stabilization period beside the tested basal insulin. For safety reasons, blood glucose levels were also measured at the bedside in duplicate during the studies using the Accu-Chek© Glucometer (Roche Diagnostics, Manheim, Germany).
Data Analysis
Incremental increases or decreases in serum glucose levels over time in each study were calculated by subtracting the baseline levels at time 0 min from all subsequent measurements. Changes in serum insulin levels over time during the 3 studies were expressed as the percentage of baseline values. Namely, each baseline serum insulin level (Cins 0 min) was given a value of 100% and each subsequent serum insulin level (Cins n min) was expressed as the percentage of the baseline value by dividing it by the baseline insulin concentration multiplied by 100 (Cins n min/Cins 0 min × 100 = % of baseline levels). One-way analysis of variance (ANOVA) followed by Tukey’s B post hoc test were used for comparing the results obtained on all study days; namely cooling vs control, heating vs control and cooling vs heating. In studies that were prematurely terminated between 0 to 240 minutes due to hypo- and hyperglycemia, the last glucose measurement at the time of termination was carried forward to the end of the study. Data are given in the text as mean ± SD, and in figures bars as mean ± SEM, unless otherwise specified.
Results
Ten males and four females participated in the study: mean age was 40 ± 17 years, HbA1c: 7.8 ± 1.1%; BMI 25.4 ± 2.8 kg/m2 and diabetes duration: 20.1 ± 9.0 years; daily insulin glargine dose was 0.42 ± 0.14 IU/kg body weight. As shown in Table 1, baseline glucose and insulin levels were similar in all three studies (P = NS)
Baseline Serum Glucose and Insulin Levels, mean ± SD.
Pharmacodynamic Outcomes
Incremental changes in serum glucose on the three study days are shown in Figure 1. As shown in the figure, cooling the injection site for 240 min was accompanied by a sharp rise in serum glucose levels compared to the gradual decline in serum glucose during the control study. In contrast, the rate of fall in serum glucose was modestly accelerated by warming the injection site compared the control study after the first hour of skin warming.
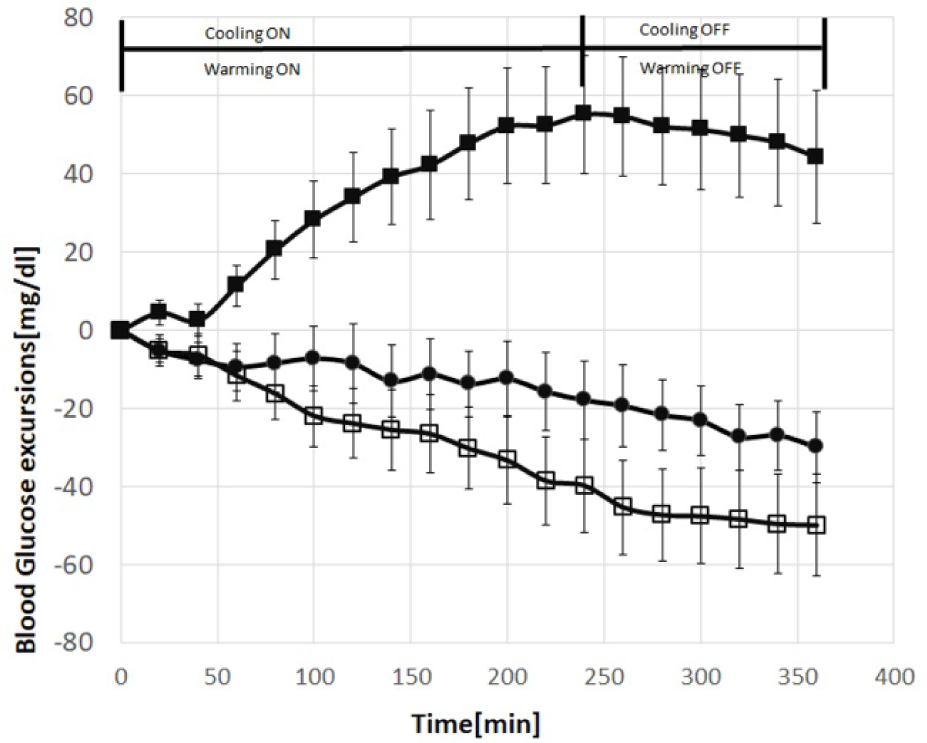
Incremental changes in serum glucose during the three studies (cooling in black squares, warming in open squares and control in black circles). Mean ± SEM.
The primary outcome of interest was differences in the changes in serum glucose levels from baseline to 240 min between the three studies. As shown in Figure 2, serum glucose levels increased by 55 ± 15 mg/dL during the cooling study, which differed significantly from the -18 ± 10) mg/dL fall in serum glucose in the control study (P < .01) and from the -40 ± 12 mg/dL fall in serum glucose during the warming study (P < .001); the fall in glucose during the warming study was not significantly different from the control study (P = .11). It is particularly noteworthy that serum glucose levels started to decline 20 minutes after the cooling element was shut off and that falling glucose levels stabilized 20 minutes after the warming element was shut off (Figure 1).
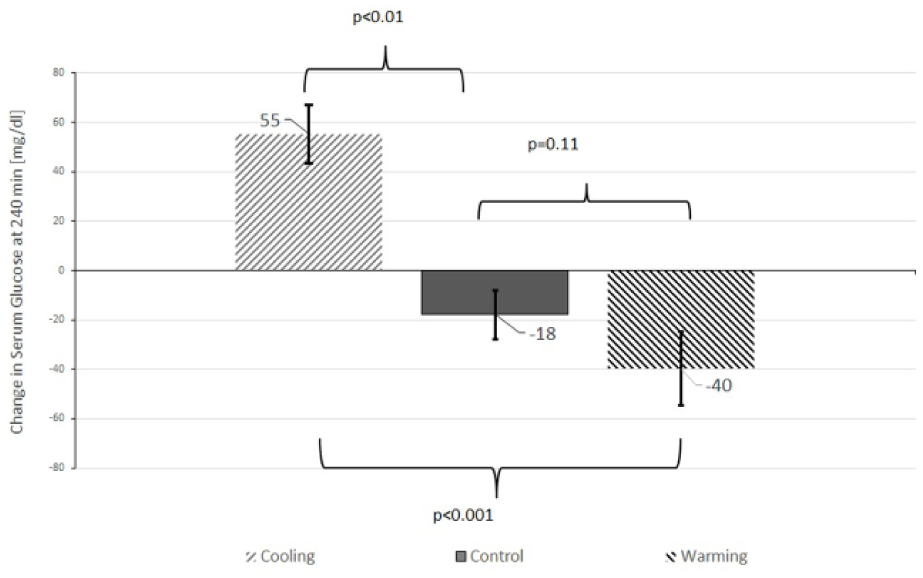
Incremental changes in serum glucose from 0 to 240 min in the three studies. Mean ± SEM.
Warming and cooling the injection site were found also to affect the area under the curve of blood glucose levels from 0-240 minutes. The results are shown in Table 2.
Area Under the Curve of Blood Glucose Levels Between 0-240 Minutes, Mean ± SD With and Without Baseline Subtraction, Mean ± SD.

Pharmacokinetic Outcomes
Serum insulin profiles, expressed as the % of baseline, in all 3 studies are shown in Figure 3. As shown in the figure, cooling the injection site caused a marked reduction in serum insulin levels compared to the more gradual reduction in serum insulin during the control study. In contrast, insulin levels remained at or slightly above baseline values during the warming study. As shown in Figure 4, there was a 41% fall from baseline in serum insulin levels at the end of the 240-min cooling study compared to a minimal increase in serum insulin during the warming study (P = .02). In addition, the percentage change from baseline in serum insulin levels was significantly greater in the cooling versus the control study between 100 and 200 minutes (P < .03). It is also noteworthy that insulin levels rebounded briskly to baseline during the 120 min after the cooling element was shut off (Figure 3).
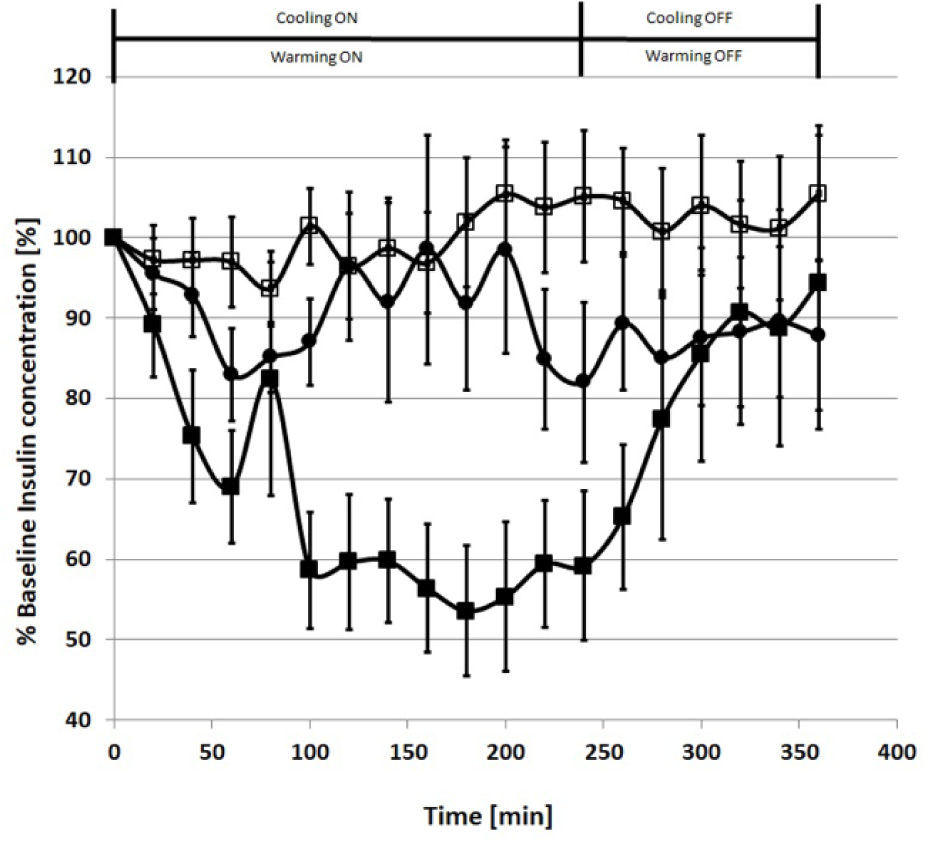
Serum insulin changes expressed as percentage of baseline levels during the cooling (black squares), warming (open squares), and control (black circles) studies. Mean ± SEM.
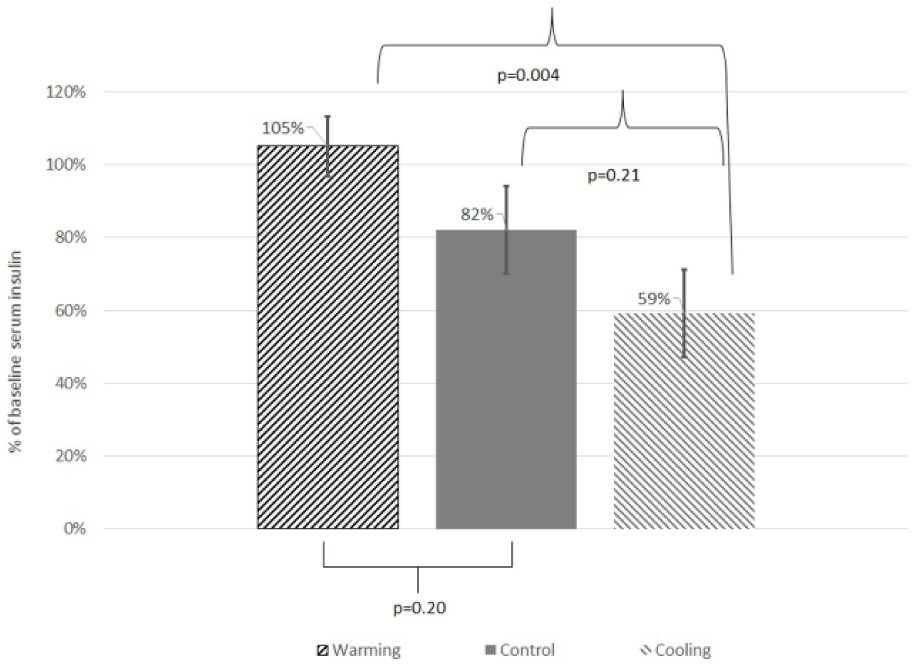
Percentage change in serum insulin from 0 to 240 min in the three studies. Mean ± SEM.
Warming and cooling the injection site were found also to affect the area under the curve of normalized serum insulin levels from 0-240 minutes. The results are shown in Table 3.
Area Under the Curve of Normalized Serum Insulin Levels Between 0-240 Minutes, Mean ± SD.

Tolerability
Local cooling and warming was well tolerated by all study participants. Comparison of the skin area at the injection site with or without use of the intervention device showed no occurrence of irritation or inflammation at the site. The cooling studies were prematurely terminated in 3 subjects due to hyperglycemia after 140, 120, and 180 min of cooling. During the warming studies, one subject was treated for hypoglycemia after 180 min of warming and one subject was treated for hyperglycemia after 145 min of warming.
Discussion and Conclusion
An important finding of this proof of concept study was the demonstration that cooling the skin around the glargine injection site provides a means to decrease the rate of absorption of this long-acting insulin analog from its subcutaneous depot. Even more important was the observation that the decrease in serum insulin concentrations due to injection site cooling was accompanied by a brisk rise in serum glucose levels during the 240-min cooling period. These alterations in serum glucose and insulin in response to turning on the cooling element are very similar to the time course of changes in serum insulin and glucose observed in sensor-augmented insulin pump systems that can suspend insulin delivery based on actual or predicted low sensor glucose levels. Thus, activation of an injection site cooling device in the face of falling sensor glucose concentrations could provide a means of decreasing the risk of hypoglycemia during the overnight period in patients receiving bedtime doses of glargine insulin. It is also noteworthy that serum insulin levels rebounded quickly to control values when the cooling devise was shut off, which prevented further increases in serum glucose levels.
We have previously shown that warming the skin around the injection site of rapid-acting analogs shortens the time and increases the magnitude of peak insulin concentrations in patients with type 2 diabetes receiving multiple daily injections 6 and in patients with type 1 diabetes on insulin pump therapy. 7 In this study, warming the skin around the injection site of insulin glargine appears to have been less effective in accelerating the absorption of glargine from its subcutaneous depot than in our prior experiments with rapid-acting insulin analogs. Nevertheless, injection site warming resulted in a fall in serum glucose that was significantly greater than that in the control experiments. This outcome was achieved by a gradual increase in serum insulin concentrations observed during the warming studies compared to the control studies as expressed in the average serum insulin levels in the different studies.
The prescribing information sheet for rapid-acting insulin analogs contain the warning that changes in skin temperature and local blood flow can alter the rate of absorption from subcutaneously depots. In this study, we demonstrated that cooling and heating the skin around the subcutaneous depot of glargine can also decrease or increase the rate of absorption of glargine presumably by decreasing or increasing local blood flow. In this study, the cooling element lowered skin temperature to 15°C or about 17°C below the normal skin temperature of ~32°C; whereas, the warming element increased skin temperature to 40°C, which is only 8°C above normal skin temperature. Hence the main effect on the absorption process appears to be the absolute temperature change that is applied to the skin. The rapid return of serum insulin to baseline concentrations once the cooling element was shut off was an unexpected benefit, since it served to limit further increases in serum glucose. We anticipate that the result seen in this study for insulin glargine can also be seen to some extent in other long acting insulins which form a subcutaneous drug depot during a significant part of the insulin life time.
A limitation of each of the studies were their relatively short, 8-hour duration. This made it impossible to determine the effects of 4 hours of cooling and warming the injection site on the half-life and total duration of action of glargine. We also had to terminate several studies prematurely due to hyperglycemia, which occurred primarily in the cooling studies, and hypoglycemia, which occurred primarily in the warming studies. Even though these premature terminations worked against our ability to show the full effects of cooling and warming the injection sites, the overall results remained striking. Another potential limitation we faced was the difficulties to achieve similar baseline glucose and insulin levels in type 1 diabetes patients on three different study days. While a few of the subjects required intravenous infusions of exogenous insulin or glucose, baseline glucose and insulin levels were similar on all three study days.
More research is needed to establish the clinical importance of the observed changes in serum glucose levels following these interventions in real-life settings. A first step might involve the development of an integrated CGM/skin cooling device that can prevent nocturnal hypoglycemia in type 1 diabetes patients receiving multiple injection regimens by reducing the rate of absorption of glargine automatically in the face of falling glucose levels. It would also be interesting to study the impact of controlling skin temperatures on reducing the PK variability of glargine. Clearly, much remains to be done to turn the results of this proof of concept into a practical device that can regulate the rate of glargine absorption in patients receiving multiple injection therapy.
Footnotes
Abbreviations
ADA, American Diabetes Association; BGM, blood glucose meter; DM, diabetes mellitus; FDA, Food and Drug Administration; HCP, health care professional; ISO, International Organization for Standardization; PD, pharmacodynamic; PK, pharmacokinetic; SMBG, self-monitoring of blood glucose.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: GB, VR, and UH are company employees. IR is a company consultant. SW and WVT serve on the Insuline-Medical scientific board.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Insuline-Medical Ltd.
