Abstract

Continuous subcutaneous insulin infusion (CSII) and continuous glucose monitoring (CGM) can improve glucose control 1 with less hypoglycemic events, 2 decrease glycemic variability, and improve quality of life in type 1 diabetes (T1D) patients.
Health authorities in Qatar have made diabetes prevention and treatment a national priority. 3 CSII and CGM policy covers full reimbursement for nationals, reimbursement for insurance holders, and charity support. Sidra Medicine in Doha provides services for all T1D children.
The objective of our study is to describe CSII characteristics in T1D children and adolescent in routine clinical settings in Qatar.
The retrospective and cross-sectional study was conducted in 138 T1D patients (age 9.8 ± 3.4 years; M 62, F 76), who initiated CSII in 2016 and 2017. MiniMed 640G (Medtronic, Northridge, USA) was started in 89 patients, Medtronic Veo (Medtronic, Northridge, USA) with sensor in 11 patients, and Medtronic Veo (Medtronic, Northridge, USA) without sensor in 38 patients.
The data were collected through the electronic medical record (Cerner Millennium, North Kansas City, USA) and Carelink Therapy Management (Medtronic, Northridge, USA) on CSII initiation and one year later.
CSII education was performed during a 5-day group program (2-3 patients; 12 hours) in outpatient clinics. Standardized protocol for CSII initiation has been used for all patients: 0-20% reduction in total daily insulin (TDI), basal/bolus distribution 40/60%, five basal rates, insulin to carbohydrate ratio (ICHR) formula of 300-450/TDI, and insulin sensitivity factor (ISF) formula of 1800/TDI (mg/dl). The research plan has been approved by the local Ethics Committee.
We found average reduction of HbA1c levels by –1.6% after one year of CSII initiation with different reduction by CSII models: –1.9% (–17.5 mmol/mol) on MiniMed 640G; –1.7% (–18.6 mmol/mol) on MiniMed Veo with sensor, and –1.5% (–16.4 mmol/mol) on MiniMed Veo without sensor. Possible explanations of improved glucose control are structured approach in pre-CSII assessment, standardized CSII/CGM protocol, and frequent visits (6 visits per year). Our long-term plan is to achieve CSII utilization of 60% among T1D with HbA1c less than 7.5%.
CSII characteristics are shown in Table 1. We also found two ICHR (67%), two ISF (85%), active insulin time of 4 hours (86%), one target range, and five basal rates (92%). Low glucose suspend on Medtronic Veo was set at 78 ± 11 mg/dl with 0.3 ± 0.2 events per patient/day. Predictive low glucose suspend (PLGS) on MiniMed 640G was set at 63 ± 8 mg/dl and showed 2.1 ± 0.9 events with 94 ± 26 minute’s duration per patient/day. No significant difference was found in patient’s weight and total carbohydrate intake per day at the end of the study.
Follow up characteristics.
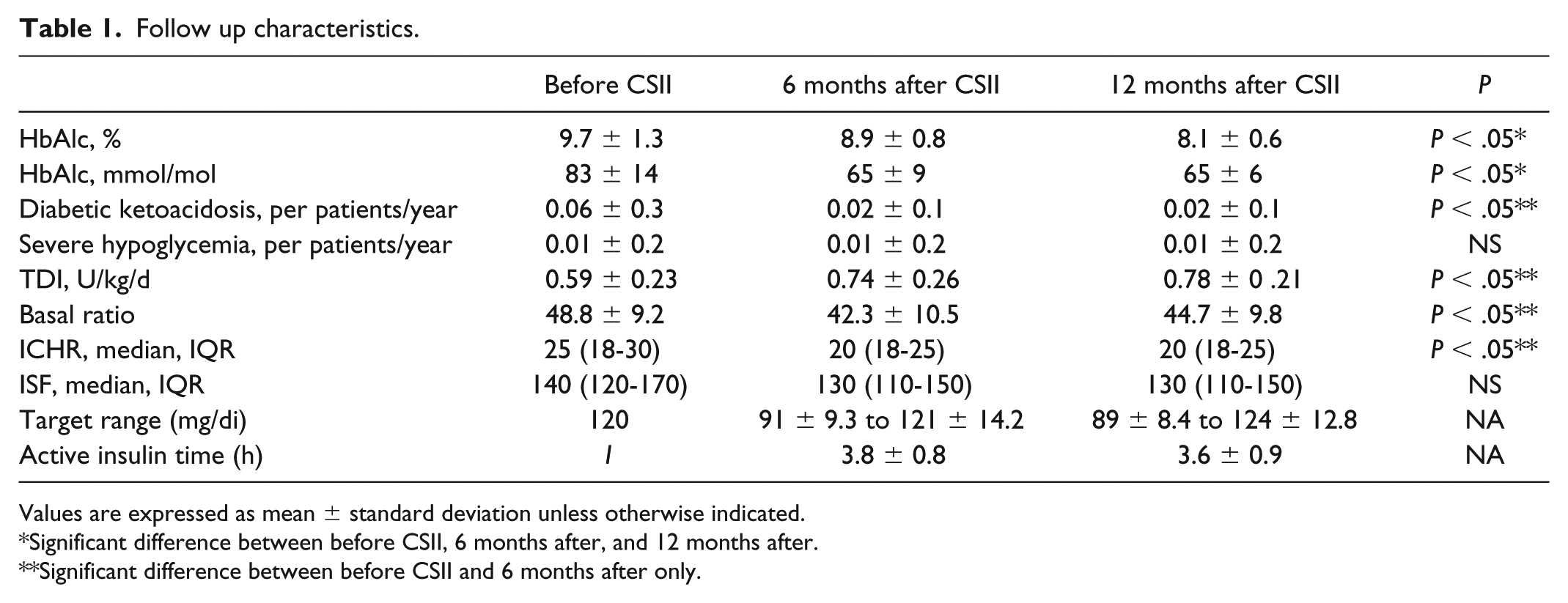
Values are expressed as mean ± standard deviation unless otherwise indicated.
Significant difference between before CSII, 6 months after, and 12 months after.
Significant difference between before CSII and 6 months after only.
The bolus wizard was used in 92% of patients, compared with 50-100% in other studies. 4
The low rates of severe hypoglycemic events and diabetic ketoacidosis were similar to other studies5,6 and can be explained by improved glucose control and patients’ motivation.
We showed improved glucose control in T1D children and adolescents initiated CSII with HbA1c level reduction by –1.6% (17.5 mmol/mol) in one year. HbA1c level of 7.7% (61 mmol/mol) was achieved in patients using sensor augmented pump with PLGS at the end of the study.
Footnotes
Abbreviations
CGM, continuous glucose monitoring; CSII, continuous subcutaneous insulin infusion; ICHR, insulin to carbohydrate ratio; ISF, insulin sensitivity factor; PLGS, predictive low glucose suspend; T1D, type 1 diabetes; TDI, total daily insulin.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
