Abstract
Hypoglycemia can lead to seizures, unconsciousness, or death. Insulin pump treatment reduces the frequency of severe hypoglycemia compared with multiple daily injections treatment. The addition of a continuous glucose monitor, so-called sensor-augmented pump (SAP) treatment, has the potential to further limit the duration and severity of hypoglycemia as the system can detect and in some systems act on impending and prevailing low blood glucose levels. In this narrative review we summarize the available knowledge on SAPs with and without automated insulin suspension, in relation to hypoglycemia prevention. We present evidence from randomized trials, observational studies, and meta-analyses including nonpregnant individuals with type 1 diabetes mellitus. We also outline concerns regarding SAPs with and without automated insulin suspension. There is evidence that SAP treatment reduces episodes of moderate and severe hypoglycemia compared with multiple daily injections plus self-monitoring of blood glucose. There is some evidence that SAPs both with and without automated suspension reduces the frequency of severe hypoglycemic events compared with insulin pumps without continuous glucose monitoring.
Keywords
Almost 100 years after the discovery of insulin hypoglycemia is still a barrier for achieving optimal glycated hemoglobin for individuals with type 1 diabetes (T1D). If untreated, hypoglycemia can lead to seizures, unconsciousness or death. For many individuals with T1D fear of hypoglycemia is the main limiting factor in diabetes management, keeping them from adhering to an intensive insulin treatment regiment. 1 The physiological and psychological burdens of hypoglycemia are the basis for the continued development of strategies and tools to avoid it.
Insulin pump treatment reduces the frequency of severe hypoglycemia compared with multiple daily injections treatment.2-4 The addition of a continuous glucose monitor (CGM), so-called sensor-augmented pump (SAP) treatment, has the potential to further limit the duration and severity of hypoglycemia as the system can detect and in some systems act on impending and prevailing low blood glucose levels. In this review we define SAP as an insulin pump with a CGM sensor that transmits the glucose readings to the insulin pump. There are currently 2 generations of SAPs on the market: in the first, the insulin dosing software operates independently of the CGM values so that the user has to make basal rate adjustments manually; in the second, the insulin dosing software and the CGM values are coupled which allows for automated suspension of basal insulin delivery in response to a predicted or detected low glucose level. For both SAP generations, however, meal and correction insulin delivery must be manually controlled, preferentially supported by the use of the insulin pump bolus calculator software.
In this narrative review we summarize the available knowledge on SAPs, with and without automated insulin suspension, in relation to hypoglycemia prevention. We present evidence from randomized trials (Table 1), observational studies and meta-analyses including nonpregnant individuals with T1D.
Studies Included in This Review.
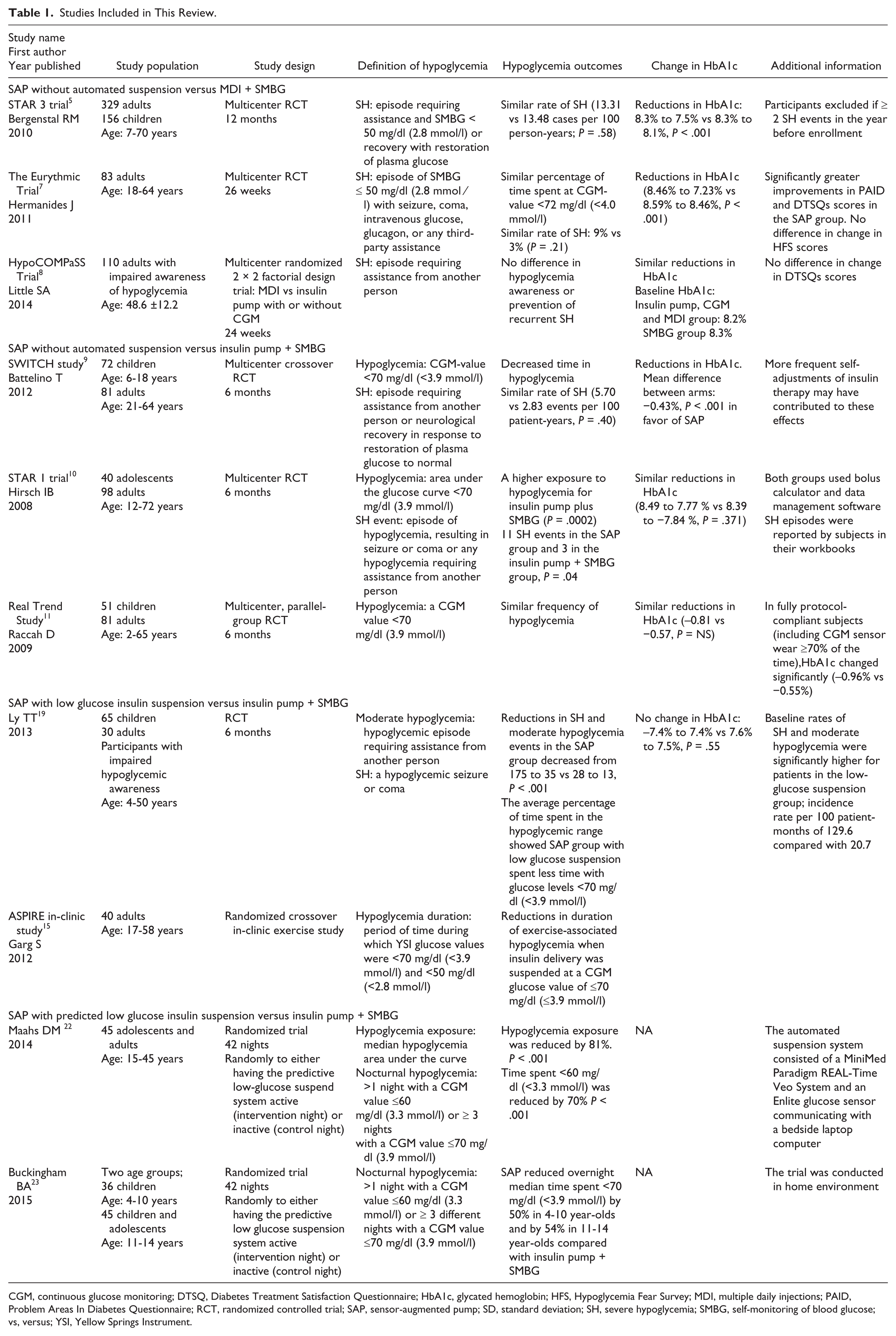
CGM, continuous glucose monitoring; DTSQ, Diabetes Treatment Satisfaction Questionnaire; HbA1c, glycated hemoglobin; HFS, Hypoglycemia Fear Survey; MDI, multiple daily injections; PAID, Problem Areas In Diabetes Questionnaire; RCT, randomized controlled trial; SAP, sensor-augmented pump; SD, standard deviation; SH, severe hypoglycemia; SMBG, self-monitoring of blood glucose; vs, versus; YSI, Yellow Springs Instrument.
Sensor-Augmented Pumps Without Automated Insulin Suspension
Obviously, the results of studies investigating the effectiveness of SAPs without automated insulin suspension depend on the reference treatment. We searched for studies comparing SAP treatment with multiple daily injections plus self-monitoring of blood glucose (SMBG), with insulin pump plus SMBG, and with multiple daily injections plus CGM.
We found 2 large multicenter randomized controlled trials comparing SAP treatment with multiple daily injection plus SMBG treatment. From the STAR 3 trial, we have solid evidence that SAP treatment reduces glycated hemoglobin without increasing the frequency of severe hypoglycemia. This trial included 329 adults and 156 children and showed greater reductions in glycated hemoglobin (8.3% to 7.5% vs 8.3% to 8.1%,
The findings of randomized trials comparing SAP treatment with treatment with insulin pump plus SMBG are more diverse.9-11 On one hand, the 6-month SWITCH study of 153 subjects aged 6-70 years concluded that SAP treatment was associated with decreased glycated hemoglobin and time spent with a sensor glucose level <70 mg/dl (<3.9 mmol/l).
9
In addition significant differences in the average daily time spent in euglycemia (70-180 mg/dl [3.9-10.0 mmol/l]) were observed in favor of the SAP group. A total of 4 severe hypoglycemia events (5.70 per 100 patient-years) occurred in the SAP group and 2 events (2.83 per 100 patient-years) occurred in the group with insulin pumps plus SMBG (
One may assume that decreased frequency of severe hypoglycemia leads to less fear of hypoglycemia. In the Interpret Study, fear of hypoglycemia was reduced at 12 months for subjects using SAP in real life settings as compared with baseline. In HypoCOMPaSS, frequencies of hypoglycemia varied between groups but worries about hypoglycemia were almost identical levels. More data are needed before we have a full understanding of the relation between occurrence of hypoglycemia and fear of hypoglycemia.
Sensor-Augmented Pumps With Automated Insulin Suspension
Two different automated insulin suspension features are currently commercially available. One feature suspends basal insulin delivery in response to a low CGM value (low glucose suspension [VEO in Europe]/threshold suspension [530G in USA]; Medtronic, Northridge, CA). Insulin delivery resumes automatically after 120 minutes irrespective of the glucose level but can be restarted beforehand by the user. The other feature can also suspend basal insulin delivery in response to a predicted low glucose value (predictive low glucose suspension/management; 640G/Not available in USA; Medtronic, Northridge, CA). As with the first feature, this system will also automatically resume basal insulin infusion after a maximum of 120 minutes in the absence of user intervention; however, if glucose values rise above a preset level, insulin can automatically start again already after 30 minutes. There are no valid clinical comparisons between these 2 features, which have only recently been introduced in the clinic. However, a simulation study has demonstrated that predicted low glucose suspension reduces hypoglycemia even more than low glucose suspension. 14
Several in-clinic and out-patient studies have shown that SAPs with low glucose suspension reduce the rate of severe hypoglycemia compared with insulin pump plus SMBG treatment.15-19 In addition, trial subjects reported alleviation to the psychological burden associated with the fear of nocturnal hypoglycemia when using the system. 20 A large, 6-month randomized trial in 95 individuals with T1D aged 4-50 years with impaired awareness of hypoglycemia concluded that treatment with SAPs with low glucose suspension reduced the combined rate of severe and moderate hypoglycemia. 19 The average percentage of time spent in the hypoglycemic range showed that the group using SAP with low glucose suspension spent less time with glucose levels <70 mg/dl (<3.9 mmol/l). During the trial, there were no episodes of ketoacidosis or hyperglycemia with ketosis and the subjects demonstrated overall improvement in hypoglycemia awareness scores. In the ASPIRE in-clinic study, 50 subjects with T1D aged 17-58 years, were studied while exercising on a stationary bicycle. The mean duration and severity of exercise-associated hypoglycemia were reduced when insulin delivery was suspended at a CGM value of ≤70 mg/dl (≤3.9 mmol/l). 15
In randomized controlled trials, home use of a predictive low-glucose suspension system substantially reduced the frequency and duration of nocturnal hypoglycemia compared with SAPs without automated insulin suspension function.21 -23 In a 42-night trial in 45 individuals with T1D aged 15-45 years, hypoglycemia exposure (median hypoglycemia area under the curve) was reduced by 81% and time spent <60 mg/dl (<3.3 mmol/l) was reduced by 70%. 22 The trial was conducted before the launch of a SAP with predictive low glucose suspension feature and the automated insulin suspension system consisted of a MiniMed Paradigm REAL-Time Veo System and an Enlite glucose sensor (Medtronic, Northridge, CA) communicating with a bedside laptop computer that contained the hypoglycemia prediction algorithm. Another 42-night study included 2 age groups; 36 children with 4-10 years of age and 45 children and adolescents with 11-14 years of age. This study found that the use of SAP with nocturnal predictive low-glucose suspend system reduced overnight median time spent <70 mg/dl (<3.9 mmol/l) by 50 % in 4-10 year-olds and by 54% in 11-14 year-olds compared with using SAP without the suspend mode. 23 In this trial the prediction algorithm was also running on a bedside laptop. A retrospective study of SAP with predictive low glucose suspension showed that adults and children could avoid mild, moderate and severe hypoglycemic events by using the automated suspension feature. 24
We found no studies comparing SAP with automated insulin suspension with multiple daily injections plus SMBG or CGM.
Concerns Regarding Sensor-Augmented Pumps With and Without Automated Insulin Suspension
As with all new technologies, the benefits of SAPs must be balanced against the concerns and potential disadvantages of the system (Table 2).
Concerns regarding sensor-augmented pumps.
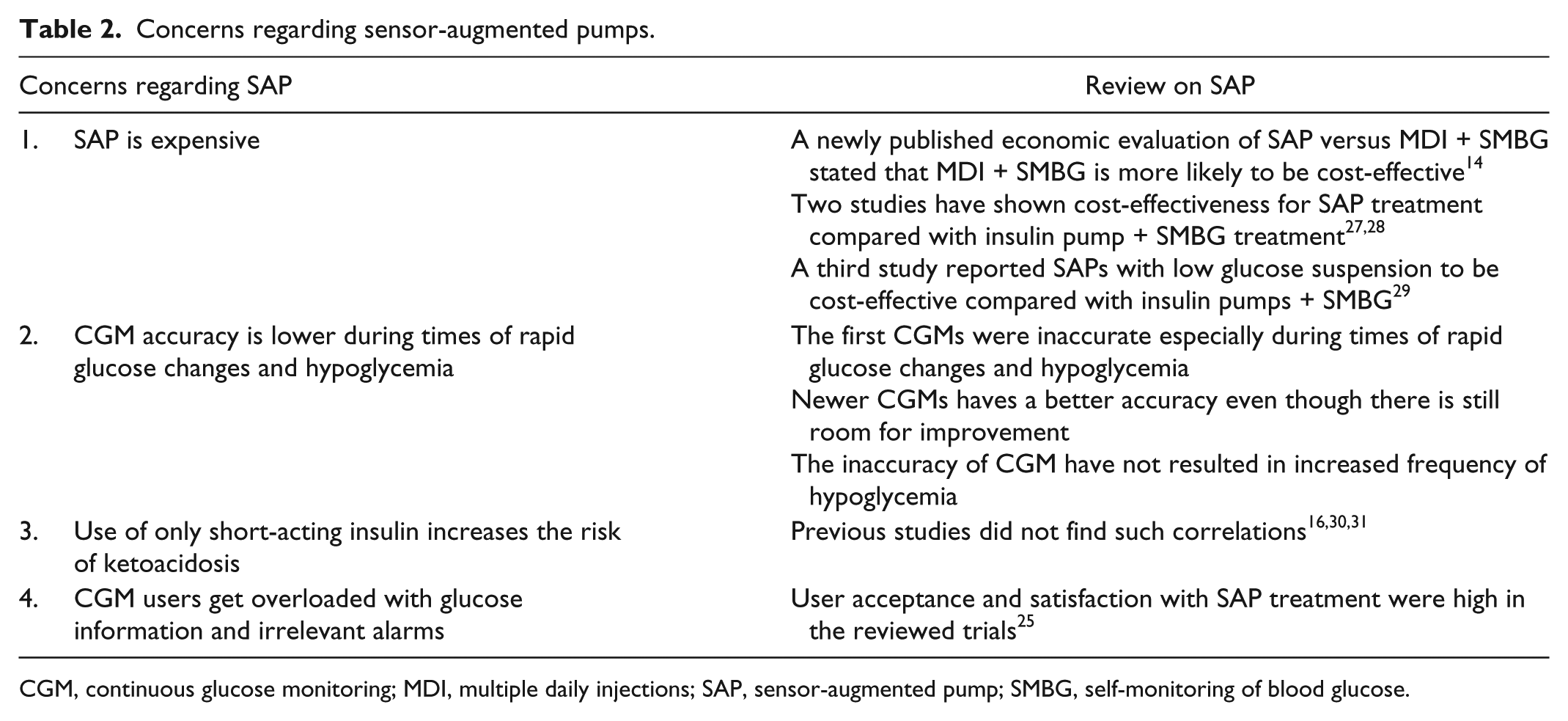
CGM, continuous glucose monitoring; MDI, multiple daily injections; SAP, sensor-augmented pump; SMBG, self-monitoring of blood glucose.
One concern about SAP treatment is that the accuracy of CGMs has been found to be lower during times of rapid glucose changes and hypoglycemia. 25 This could potentially give the user a false security feeling. The concern is, however, not supported by the reviewed trials in which there were similar or lower frequencies of severe hypoglycemia in the SAP treated groups.
Also the cost of SAP treatment is of concern. The direct extra cost of SAP treatment compared with insulin pump treatment with SMBG, includes the cost of sensors and transmitters as well as patient and provider training in device use and interpretation of data. The direct savings count a reduction in number of SMBG, which is significantly lower using SAP. Indirect savings include treatment of diabetes late complications, treatment of hypoglycemia and reduced ability to work. On top of that come other consequences of SAP use, which are equally important but much harder to estimate. Examples are children missing fewer school days during SAP 9 (and probably their parents will have less time away from work), reductions in mild hypoglycemia which also may reduce sick days and hypoglycemia associated anxiety among individuals with T1D and their relatives. Some, but not all, of these cost changes can be included in health economic models. Accordingly, we believe that current health economic models may underestimate the benefits of SAP therapy. Models are complex and dependent on the structure of a given health care system and model outcomes should be evaluated with that in mind. A newly published economic evaluation of SAP versus MDI plus SMBG stated that the latter is more likely to be cost-effective. 26 The evidence base underlying the evaluation was, however, poor. Two studies from Sweden and Australia, respectively, have shown SAP treatment to be cost-effective compared with insulin pump plus SMBG treatment.27,28 A third study from the United Kingdom, reported SAPs with low glucose suspension to be cost-effective in patients with poor metabolic control compared with insulin pumps plus SMBG. 29 We are not aware of any studies evaluating the cost-effectiveness of predicted low glucose managements systems. Despite the rather convincing evidence of cost-effectiveness of SAP therapy over insulin pump alone therapy, there is still little or no insurance coverage of CGMs by some insurance carriers and national health care systems. A theoretical disadvantage of automated suspension of short-acting insulin for up to 2 hours is the risk of ketoacidosis. Previous studies did not find such correlations.16,30,31
Finally, other potential disadvantages include lack of treatment adherence related to psychosocial factors such as the stress of CGM alarms and glucose information overload. 32 In spite of this, user acceptance and satisfaction of SAP treatment were high in the reviewed trials.
Limitations of This Review
One obvious limitation of this review is the lack of studies with hypoglycemia parameters as primary endpoint. Most studies reviewed had glycated hemoglobin as primary endpoint and hypoglycemia as secondary endpoint and were not powered to detect differences in hypoglycemia. Even though mild hypoglycemia greatly affects quality of life among individuals with T1D, none of the studies we reviewed had mild hypoglycemia as the primary endpoint.
A potential bias when comparing these studies is that there are different definitions of
Different inclusion criteria were used in the studies we reviewed. Some studies excluded subjects with impaired awareness of hypoglycemia, which further compromised the present review to compare all the studies.
The studies referenced in this review, all investigated SAPs produced by Medtronic. We did not find any studies of SAP systems by other manufacturers. Theoretically, studies of different SAP systems could show slightly different results due to differences in ease of use and accuracy, however, we do not believe that SAP brand significantly affects the overall conclusions of this technology review. The first generation of Medtronic sensors (Sof-Sensor) could sometimes cause pain upon insertion and was not as accurate as the sensors produced today by Medtronic and other companies. Discomfort and inaccuracy may have affected treatment adherence and thereby the results of early studies with Medtronic’s SAP system.
The studies included in this review may not only reflect the isolated effect of SAP treatment but also the effects of the trial setting and the human factors. It is difficult—if not impossible—to offset this source of bias. For instance, along with the introduction of SAPs, study subjects often received training in different aspects of general T1D management such as carbohydrate counting, which in itself could influence study outcomes. 35 Furthermore, subjects often get more attention from health care professionals than individuals attending routine clinical practice. We cannot rule out that the reduction in hypoglycemia in individuals using SAP treatment was obtained by change in behavior rather than the system itself.
SAP effectiveness is highly dependent on human factors. For instance, SAPs with low glucose suspension cannot compensate for large errors made by the user, such as the individual not counting carbohydrates in a meal correctly and therefore taking an excessive insulin bolus. Furthermore, “human factors” refers not only to the user of the system, but also to the persons responsible for adjustment of insulin pump settings including limits for low glucose suspension.
Conclusion and Future Considerations
Summarizing the available knowledge of SAP treatment in relation to hypoglycemia is like trying to explain the aroma of hot chocolate with whipped cream. The thick brown chocolate is enriched by the light white cream, and yet both chocolate and cream taste delicious separately. Likewise, SAP reduces hypoglycemia but insulin pumps and CGMs also reduce hypoglycemia when used separately.36,37 It remains unclear whether the insulin pump, the CGM, the accompanying training or another mechanism holds the main responsibility for the positive effects observed. Irrespectively, there is evidence that SAP treatment reduces moderate and severe episodes of hypoglycemia compared with multiple daily injections plus SMBG. There is some evidence that SAPs both with and without automated suspension reduce the frequency of severe hypoglycemic events compared with insulin pumps without CGM. More studies are needed with hypoglycemia parameters as primary endpoint to further clarify the role of SAP treatment in hypoglycemia prevention. Each of the clinical trials described is unique with respect to population, hypoglycemia assessment and study design. As the subjects in these studies received special attention and education, it is not possible to apply the results to the average person with T1D in everyday life. However, some important and valuable messages can still be taken from these studies.
In the planning of future clinical studies that should clarify the role of SAP in hypoglycemia prevention, we encourage researchers to make hypoglycemia the primary study focus and endpoint (and power it as such) instead of the so often used glycated hemoglobin. There are, however, many different measures of hypoglycemia and to complicate matters even further, some measures do not have uniformly accepted definitions. It would ease interpretation and comparisons of studies if the same endpoints were applied, which in turn would strengthen the argumentation for need of CGM reimbursement. We suggest taking advantage of the many data collected by CGM and use time spent in hypoglycemia as primary endpoint. This may not reflect all aspects of hypoglycemia, but it is a measure that is easy to understand for most and it is easy to obtain from CGM readings. Secondary study endpoints should include hypoglycemia-related psychosocial measures, for example, the Hypoglycemia Fear Survey, and registration of sick days caused by low blood glucose values. Sick days include absence from school or work for the person with T1D, but sometimes also for relatives who have to stay home to provide care for the diseased. Furthermore, a clinically relevant cohort should be recruited for such studies, that is, people who do actually experience hypoglycemia on a regular basis.
It is our experience that the demand from individuals with T1D for new technology that will ease their diabetes management is enormous. We are convinced that the increased complexity of insulin dosing software for pumps has eased daily insulin management by reducing the burden of hypoglycemia and taking over some of the daily dosing decisions. Still, we acknowledge the limitations of the current SAP systems and look forward to the next generations which are likely to benefit from faster acting insulin, CGMs with better accuracy, and the option of dosing insulin when hyperglycemia is predicted or detected.
SAP with automated insulin suspension is a step closer to the artificial pancreas, which combine insulin pumps, CGMs, and control algorithms for insulin dosing to deliver insulin in a semi- or fully automated manner.38,39 Until these systems are ready, SAPs available today may mitigate the physiological and psychological burden of hypoglycemia in individuals with T1D without compromising metabolic control.
Footnotes
Abbreviations
CGM, continuous glucose monitor; SAP, sensor-augmented pump; SMBG, self-monitoring of blood glucose; T1D, type 1 diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: IS has received speaking honorarium from Rubin Medical and Roche Diabetes Care. KN has received speaker honorarium from Medtronic, Rubin Medical and Roche Diabetes Care and is a member of advisory boards for Medtronic. SS is a member of Roche Diabetes Care advisory board and works as an advisor for Unomedical a/s.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by research grants from the Danish Diabetes Academy supported by the Novo Nordisk Foundation.
