Abstract
Background:
The ability of patients to improve glycemic control depends partly on their ability to interpret and act on blood glucose results. We investigated whether switching people with diabetes to blood glucose meters (BGMs) featuring a color range indicator (CRI) could improve glycemic control compared to remaining on their current BGM without color.
Methods:
163 adults with type 1 (T1D) or type 2 diabetes (T2D) and a hemoglobin A1c (A1c) of 7.5-11% were randomized to: One Touch Verio™ (Verio), OneTouch Verio Flex™ (Flex), or controls remaining on their current BGM. Diabetes nurses had standard conversations about diabetes management with all subjects at baseline. No changes in medication, insulin dosing, or SMBG frequency were recommended.
Results:
After 12 weeks, subjects who switched to Verio or Flex meters with CRI (n = 108) had a mean change in A1c 0.36% lower than controls (n = 55) (P = .017). A1c reductions were greatest in T1D subjects (n = 45), with a decrease of 0.50% (P = .004). T1D subjects using Verio meters (n = 25) contributed a 0.59% reduction compared to controls (P < .008), whereas T1D subjects using Flex meters (n = 20) had a clinical meaningful reduction in A1c of 0.40% without reaching statistical significance (P > .05). Verio and Flex users reported taking more action and easier understanding of diabetes management compared to previous BGMs.
Conclusions:
This study demonstrated that switching patients to BGMs featuring a CRI resulted in improvements in glycemic control compared to subjects using currently marketed BGMs that do not use a CRI.
Registration: Clinicaltrials.gov NCT02929654 https://clinicaltrials.gov/ct2/show/NCT02929654
We previously reported that blood glucose meters (BGMs) utilizing a color range indicator (CRI) to identify whether blood glucose (BG) results are low, high, or in range improved the ability of patients with type 1 (T1D) and type 2 (T2D) diabetes to interpret and classify BG readings.1,2 A follow up study examining decision-making found patients were more inclined to act on BG results, particularly patients with low numeracy, if the results appeared with color. 3 Others have observed that more appropriate decisions are made when BGMs display information that prompts users toward more favorable diabetes management decisions. 4 This is important given in a study of BG perceptions only 28% of patients considered results >235 mg/dl as high. 5 Another study found 56% of patients did not respond regularly to out-of-range BG readings with any self-care adjustments. 6 We previously reported hemoglobin A1c (A1c) reductions when patients used a BGM featuring a CRI in combination with health care professional (HCP) feedback via online diabetes management software, 7 or when patients received texts from their HCP to their smartphone based upon an app featuring this CRI. 8 A recent observational study also found that using a BGM with CRI improved diabetes self-management and reduced A1c levels. 9 Despite this past evidence, direct glycemic benefits of BGMs featuring CRI have not been proven in a randomized controlled trial. The current study investigated whether switching patients to BGMs featuring CRI using ColorSure Technology™ could improve glycemic control compared to patients that continued using their current BGM without CRI.
Materials and Methods
Materials
In the ACCENTS study (Assessment by Consumers of Color Enhanced BGMs and New Therapeutic Strategies) randomized subjects switched to meters with CRI and used OneTouch Verio™ or OneTouch Verio Flex™ 10 (LifeScan, Wayne, PA). Both meters use CRI to automatically indicate whether the BG result is low (blue), in range (green), or high (red) (Figure 1). Default range limits for CRI (low <70 mg/dl, in range 70-180 mg/dl, high >180 mg/dl) are preset, but can be customized. Verio also automatically identifies high and low BG patterns 11 and displays on-screen motivational and achievement-related messages based on glucose testing.12,13 Control subjects continued to use their own BGM (without color), which comprised a wide variety of BGMs available in the UK (Figure 1).
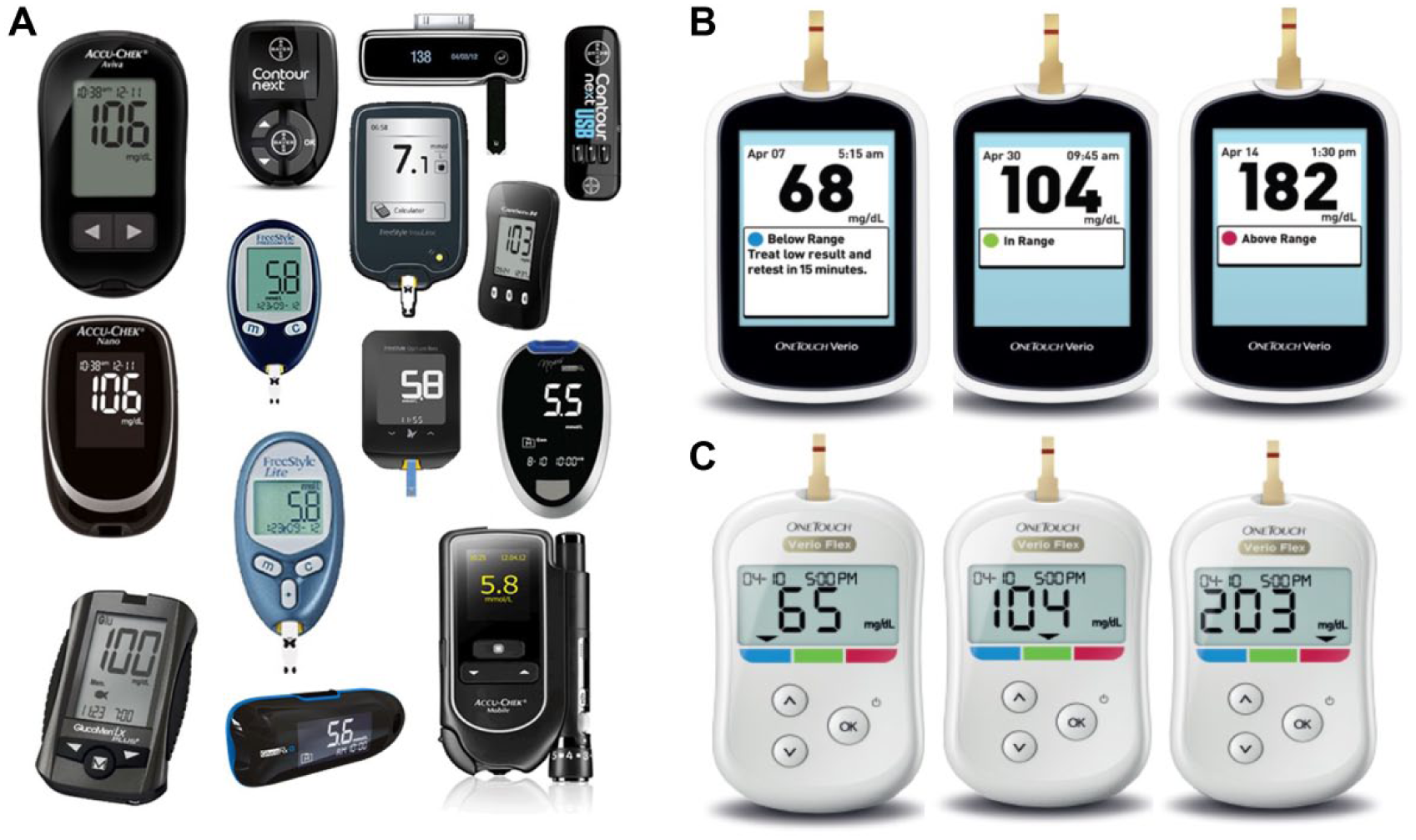
Blood glucose meters (BGMs) at baseline. A total of 163 subjects were randomized to either (A) continue to use their current blood glucose meter for the next 12 weeks (representative BGMs shown) or switch to (B) OneTouch Verio or (C) OneTouch Verio Flex meter for the next 12 weeks. Each of these meters feature color range indicators (ColorSure Technology) to identify whether blood glucose results are low, high, or in range. Within each study site, subjects were stratified based on baseline A1c (7.5 to <9.0% or ≥9.0 to 11.0%) and diabetes type (T1DM or T2DM) before being randomized to group A, B, or C.
Methods
This parallel, three-arm, open label, randomized controlled study was conducted between October 2016 and June 2017 at three sites in the United Kingdom: Highland Diabetes Institute (Inverness), Edinburgh Royal Infirmary, and Heartlands Hospital (Birmingham). Ethics approval and subject informed consent were obtained before study initiation. Subjects were between age 18 to 70 years; diagnosed with T1 or T2D for ≥ 3 months; current A1c 7.5 to 11.0%; and performing self-monitoring of blood glucose (SMBG). Exclusion criteria included subjects using insulin pumps or continuous glucose monitoring systems; currently (or previously) using a LifeScan BGM featuring a CRI or a BGM with pump connectivity (eg, Accu-Chek Aviva Combo or Contour Next Link). The primary endpoint was A1c change from baseline in subjects using BGMs with CRI (Verio or Flex with Verio test strip lot: 4085326) compared to subjects continuing to use their current BGM without CRI. Subjects using BGMs with CRI also provided feedback on their perceptions and experiences using the BGM.
Visit 1 (Screening)
Informed consent, collection of demographic, medical history information, and evaluation of entrance criteria. A venous blood draw was performed to establish baseline A1c.
Visit 2 (Baseline)
One week ± 3 days after baseline, eligible subjects were randomized to switch to Verio or Flex or continued using their current meter. Prebaseline self-monitoring data were downloaded from all subjects to establish an SMBG baseline. In addition, subjects completed a validated subjective numeracy tool. 14 Control subjects were advised to continue diabetes management based on insights provided by their current BGM and advice of their HCP but were not retrained on the usage or features of their existing BGM. Subjects randomized to Verio or Flex were trained on features of their assigned meter (including CRI and pattern messaging) in addition to the same advice given to the control group.
Visit 3 (12 weeks)
Venous blood was drawn for A1c measurement and SMBG data downloaded from each BGM. All subjects completed surveys on their perceptions and experiences of the meter they used.
Randomization and Statistical Analyses
Subjects were randomized to Verio, Flex, or control groups within each investigational site using a stratified block randomization design with two levels: baseline A1c (7.5 to <9.0% or 9.0 to 11.0%) and diabetes type (T1 or T2D). Sample size was estimated to achieve 80% power at 5% significance to detect a 0.3% decrease in A1c in the combined Verio + Flex groups versus controls. Analysis of covariance was used to assess mean change in A1c from baseline corrected for any difference in starting A1c. Correlations between numeracy score and A1c were assessed using Pearson correlation coefficient. Minitab 17.0 and SPSS V21.0 were used for all analyses.
Results
Subjects
Baseline characteristics of all 163 subjects are shown in Table 1. Control (n = 55) and Verio + Flex subjects (n = 108) had similar characteristics including mean A1c (8.6% and 8.8%, respectively) and mean duration of diabetes (17.5 years and 17.8 years, respectively). A total of 56% reported performing self-monitoring ≥ 3 times per day and 92% at least once per day. In all, 83% were on some form of insulin with 49% on basal/bolus insulin therapy. Of the 95 subjects with T2D, 72% were on some form of insulin therapy. The three hospital-based UK National Health Service sites generally care for complex or difficult to manage people with diabetes who usually have been referred from general practice. Therefore, these sites typically have a higher proportion of more intensively managed patients (eg, multiple daily insulin injections) than encountered in general practice.
Baseline Subject Demographics.
Blood Glucose Monitoring Devices
At baseline, subjects were using a wide variety of BGMs with similar numbers of each brand in each study group (Figure 2). Of the meters used at baseline, Roche (36%) and Abbott (24%) accounted for the highest proportion with more recent manufacturers also represented, for example, GlucoRX (10%) and Spirit (7%). LifeScan BGMs were underrepresented since enrollment excluded subjects currently using a LifeScan BGM featuring a CRI. A protocol amendment was implemented shortly after study initiation to exclude subjects using the Roche Combo/Expert BGM since this BGM includes a CRI, albeit different from Verio or Flex. Three control subjects enrolled before this amendment continued using this Roche BGM for the study duration and were included in data analyses.
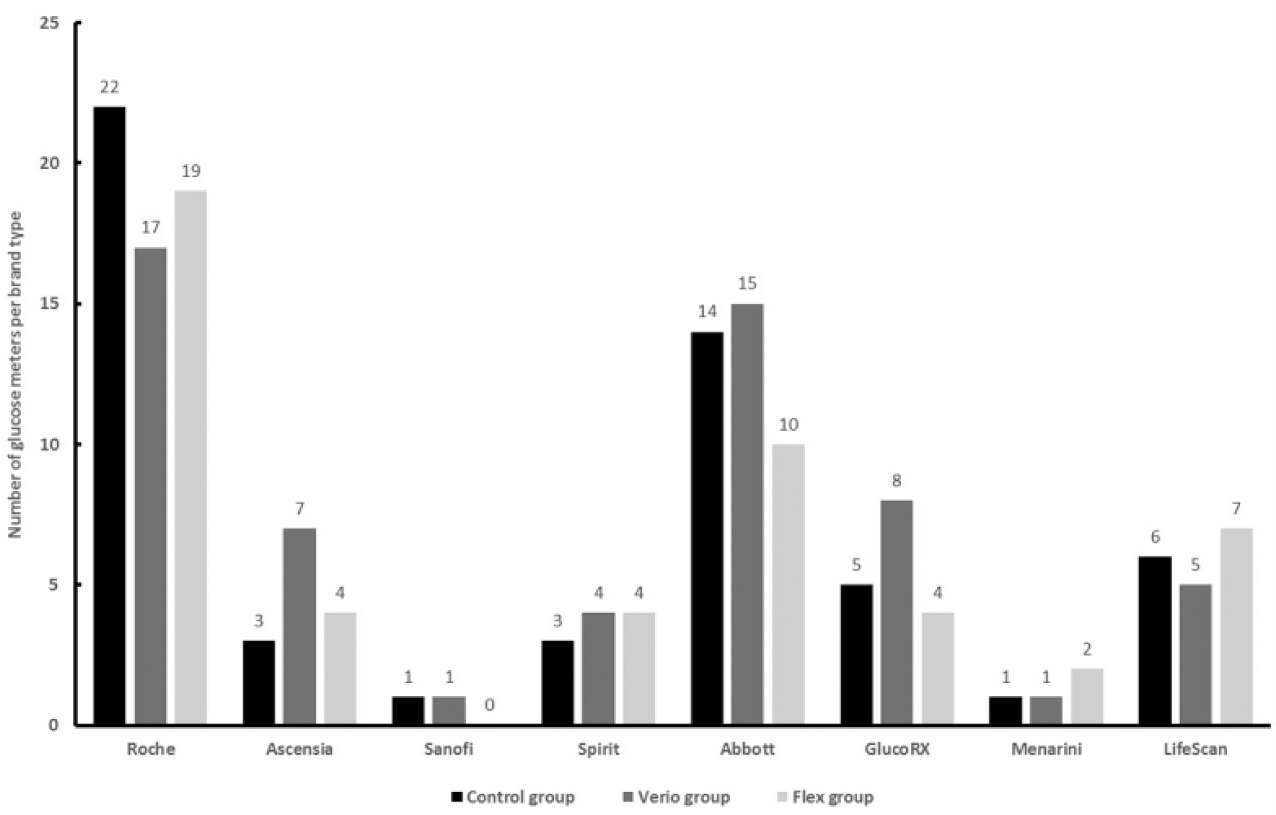
Blood glucose meter (BGM) brands at baseline. Meter brands at baseline are shown for all 163 subjects. Subjects randomized to the control group continued to use the same baseline meter for the next 12 weeks. Subjects randomized to the Verio or Flex group stopped using their baseline BGM and immediately switched to using a OneTouch Verio or OneTouch Verio Flex meter for the next 12 weeks. Subjects were excluded if they had previously used a OneTouch Verio, OneTouch Verio Flex, or OneTouch Select Plus meter. Subjects could be randomized if they had previously used LifeScan branded meter that did not feature color range indicators (eg, OneTouch Ultra).
Changes in Glycemic Control (A1c)
A1c at baseline and 12 weeks are shown in Figure 3. After 12 weeks, the primary endpoint was met, namely A1c was significantly reduced by 0.36% in Verio + Flex subjects compared to control, in whom A1c remained unchanged (P = .017). This 0.36% improvement in A1c represents both a statistically and clinically meaningful reduction in A1c The relative contribution of the Verio meter to this reduction was greater than that of the Flex meter group. Mean A1c in subjects using Verio meters (n = 58) was reduced by 0.42% compared to controls (P = .018), whereas mean A1c reduction in subjects using Flex meters (n = 50) of 0.21% was not statistically significant (P > .05).
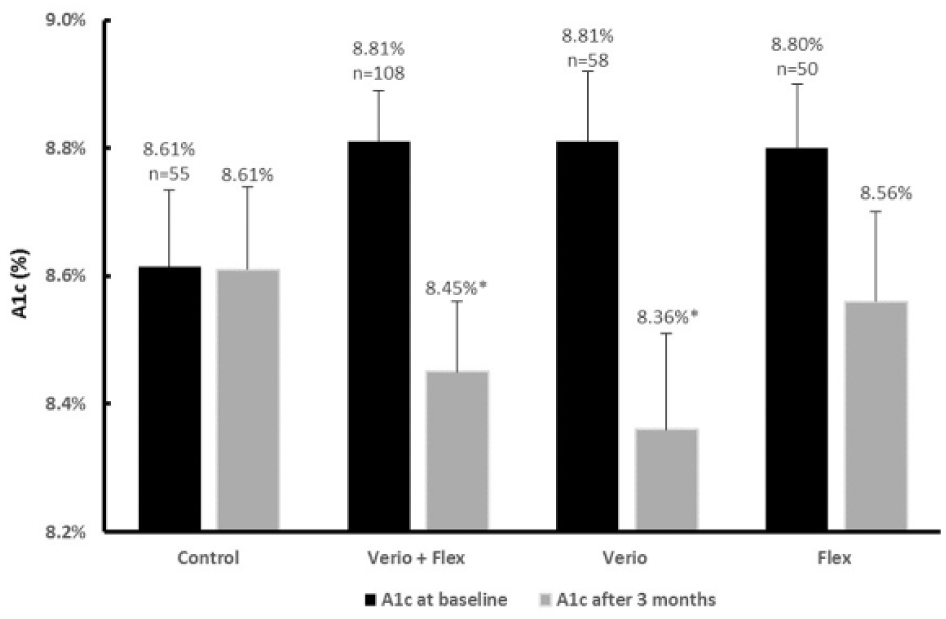
Change from baseline in hemoglobin A1c after 12 weeks of home SMBG in subjects with T1 or T2D. Verio, OneTouch Verio blood glucose meter; Flex, OneTouch Verio Flex blood glucose meter; Verio + Flex, combined data from both meter groups. Data shown are mean changes ± standard error of the mean. *Reductions in hemoglobin A1c in subjects using Verio + Flex (P = .017) or Verio meters (P = .018) were statistically significantly different from changes in control.
A key finding of the study was clinically meaningful reductions in A1c observed in T1D subjects using either Verio or Flex meters. After 12 weeks, subjects with T1D who switched to Verio or Flex meters (n = 45) had a mean A1c reduction of 0.50% compared to controls (n = 23) (P = .004) (Figure 4). Again, the relative contribution of this decline was greater in the Verio meter group compared to the Flex meter group. Subjects with T1D using the Verio meters (n = 25) had a mean A1c reduction of 0.59% compared to controls (n = 23) (P = .008), whereas subjects with T1D using Flex meters (n = 20) had A1c reduced by 0.40% compared to controls (n = 23) (P > .05) (Figure 4). Although this difference was not statistically significant we suggest that this represents a clinically significant improvement in glycemic control. Subjects with T2D using Verio or Flex meters (n = 63), Verio (n = 33), or Flex (n = 30) had reductions in mean A1c of 0.21%, 0.31%, and 0.09%, respectively, compared to controls (n = 32), although none of these differences were statistically significant (P > .05). Finally, clinically meaningful improvements in A1c in subjects using Verio or Flex were not correlated with significant changes in average glucose or in the number of glucose results in specific glycemic ranges (low, in range, or high).
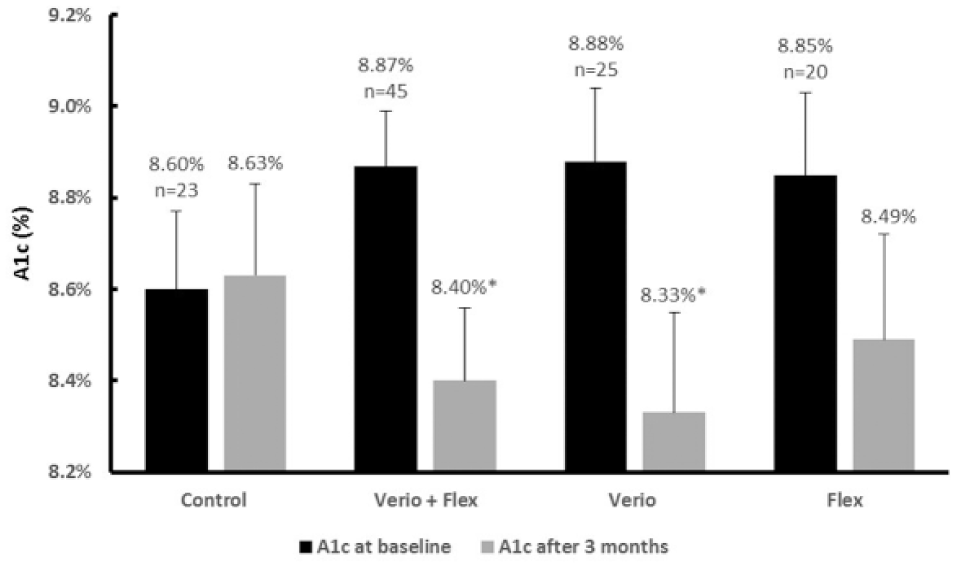
Change from baseline in Hemoglobin A1c after 12 weeks of home SMBG in subjects with T1D. Verio, OneTouch Verio blood glucose meter; Flex, OneTouch Verio Flex blood glucose meter; Verio + Flex, combined data from both meter groups. Data shown are mean changes ± standard error of the mean. *Reductions in hemoglobin A1c in T1D subjects using Verio + Flex (P < .004) or Verio meters (P < .008) were statistically significantly different from changes in control.
Self-Monitoring of Blood Glucose Frequency
Control subjects performed SMBG on average 1.8 times per day (T2D 1.6, T1D 3.2); Verio subjects tested 2.3 times per day (T2D 1.6, T1D 3.4); and Flex subjects tested 1.7 times per day (T2D 1.4, T1D 3.5). There was no statistical difference among groups or change in test frequency over time (P > .05).
Subject Numeracy and Changes in Glycemic Control (A1c)
The median (and range) of subjective numeracy at baseline on a scale of 8 to 48 (48 = highest numeracy) was Verio + Flex, 33 (10 to 48), Verio, 35 (13 to 47), Flex, 31 (10 to 48), and control, 28 (12 to 48). Median scores were not statistically different among groups. The median score across all subjects was 32. There was no correlation between A1c improvement and baseline numeracy across all subjects or comparing control to Verio + Flex subjects. There was also no association between A1c improvement in any group when comparing subjects above or below median numeracy.
Subject Perceptions of Verio and Flex BGMs
Based on survey questions completed at the end of the study (Table 2), the use of blue, green and red colors to indicate low, in range, or high BG results, respectively, in Verio and Flex meters was intuitive and easy to understand. The use of green for in range (92%), red for high (91%) and blue for low (75%) were all felt by the subjects to be intuitive. 92% of the subjects agreed that the CRI was helpful for identifying low and high BG results and helped them treat low (76%) or high (75%) BG results. 82% of subjects would recommend the Verio or Flex BGMs and 80% said that they would switch to a BGM with this CRI.
Subject Responses to Survey Statements (Percentages).
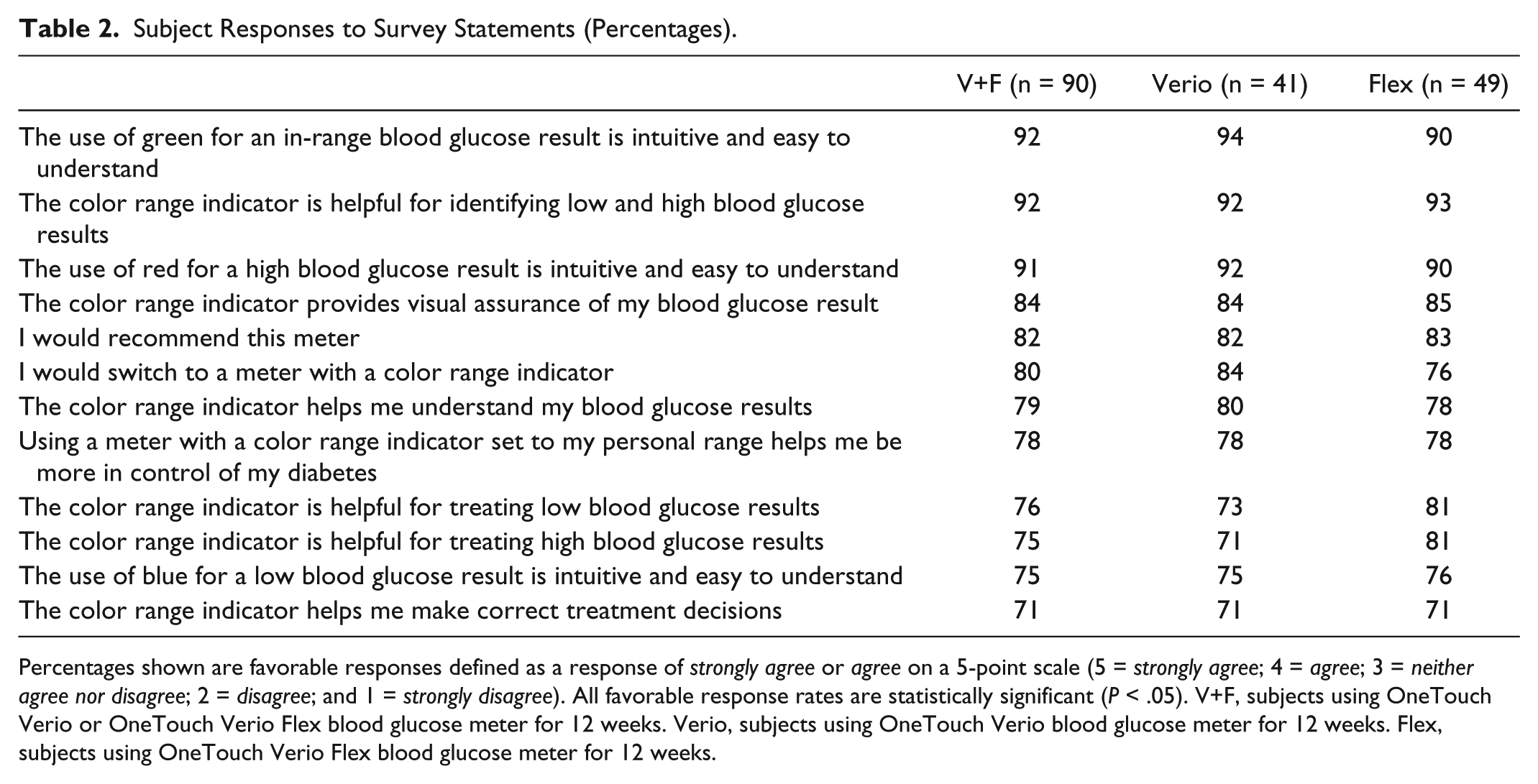
Percentages shown are favorable responses defined as a response of strongly agree or agree on a 5-point scale (5 = strongly agree; 4 = agree; 3 = neither agree nor disagree; 2 = disagree; and 1 = strongly disagree). All favorable response rates are statistically significant (P < .05). V+F, subjects using OneTouch Verio or OneTouch Verio Flex blood glucose meter for 12 weeks. Verio, subjects using OneTouch Verio blood glucose meter for 12 weeks. Flex, subjects using OneTouch Verio Flex blood glucose meter for 12 weeks.
Based on additional survey questions completed at the end of the study (Figure 5), subjects felt the Verio meter made it easier to spot glucose patterns (79%), prompted them to take more action (75%), and made it easier to understand diabetes control (86%) than their previous meter. Subjects agreed the Flex meter with its color range feature made it easier to understand diabetes control (72%), prompted them to take more action (70%), and helped them get more from BG testing (74%) than their previous meter (Figure 5).
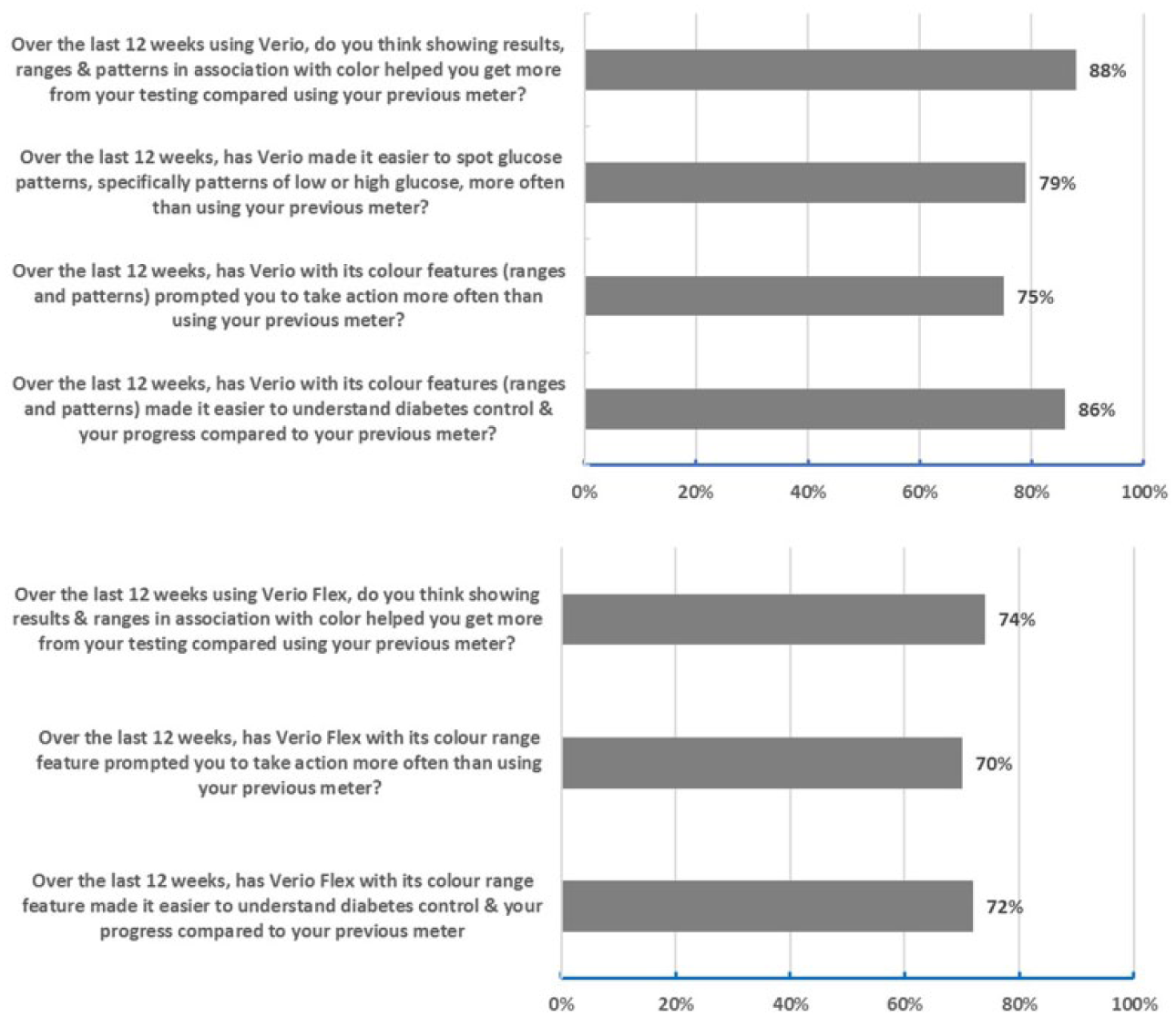
Subject responses to survey statements. Percentages shown are favorable responses defined as a response of strongly agree or agree on a 5-point scale (5 = strongly agree, 4 = agree, 3 = neither agree nor disagree, 2 = disagree, and 1 = strongly disagree). All favorable response rates are statistically significant (P < .05).
Subject Feedback on Insights and Actions Using Verio and Flex BGMs
At the final site visit, subjects were asked to rank four statements describing how Verio or Flex assisted them with their diabetes during the study. Flex subjects rated “simply knowing when results were in ranges” (52%), “being able to take immediate action” (22%), “prompting them to consider why a result was low, in range, or high” (20%) or “making more informed decisions” (6%) as most important. When Flex subjects were asked to reflect on what they had done or noticed using this BGM, “spotting when I was running high more often than I thought” (41%), “adjusting my insulin more frequently” (26%), “feeling on track” (17%), or “spotting how food affected glucose levels” (15%) were regarded as most important.
Similar views were provided by Verio subjects with “showing me when results were in specific ranges” (48%), “pointing out low or high patterns to consider why they had happened” (25%), “easily viewing past patterns, averages or results” (16%), and “positive on-screen messages” (11%) rated as most important. When Verio subjects were asked to reflect on what they had done or noticed using this BGM, “being alerted about specific times I had patterns” (40%), “adjusting my insulin more frequently” (29%), “spotting how food affected glucose levels” (23%), and “feeling on track” (14%) were regarded as most important.
Safety and Tolerability
A total of 69 adverse events and 1 serious adverse event were reported from 163 subjects during the 12-week study. None were related to use of any BGM.
Discussion
There has been little evidence directly comparing effects of different BGMs in a randomized controlled trial. In this study, improved glycemic control was demonstrated when subjects switched to BGMs featuring a CRI compared to subjects who continued using BGMs without color. Additional features in the Verio meter (range based messaging and pattern detection) complimented the key feature of the CRI and potentially contributed to the greater improvement in glycemic control than observed using the Flex meter. In 2007, patients using the OneTouch® Ultra Smart® BGM, with advanced on-meter trend graphs and electronic logbooks, achieved a modest A1c difference compared to conventional BGMs. 15 However, other BGMs at that time generally displayed only a blood glucose result. Today most BGMs have more sophisticated and broadly similar onboard features. Instead of comparing effects of different BGMs, recent studies focus on structured testing16,17 using a single BGM, comparing basic to advanced features on the same BGM, 18 investigating BGMs along with a diabetes app 19 or comparing a relatively simple BGM to advanced glucose sensing technology. 20 Therefore, there remains a gap in our understanding of the value a BGM can bring to outcomes achieved by patients because outcomes are often masked by introduction of other interventions including diabetes apps, complex test regimens, or additional HCP encounters. This is also important given that many patients may not wish to use diabetes apps or computer software. Our study suggests that switching patients to BGMs that feature a CRI can deliver clinically meaningful changes in glycemic control over a short period of time.
Our study population reflected the usage of a full spectrum of modern BGMs marketed in the UK covering 8 manufacturing brands and 27 BGM models. Importantly, similar numbers of brands (eg, Roche, Abbot, and Ascensia) and models were used across each group at baseline. This ensured a level playing field in terms of previous exposure to BGM features (eg, BG averages, low/high BG alerts, testing or medication reminders, customizable audible alarms, ketone measurement and alerts, insulin tracking, or bolus calculation). This also ensured that insights, knowledge and skills derived from prior BGM experiences were relatively similar across subjects before randomization and would be less likely to account for any differences in outcomes among groups. It is worth noting that some subjects who switched to the relatively simple Flex meter may have actually experienced a reduction in the number of meter features compared to their previous BGM. Despite this, subjects using Flex still exhibited positive improvements in glycemic control. As previously demonstrated, these positive trends may be due to patients better interpreting1,2 and more likely to act on results and insights derived from the CRI. 3 In keeping with these findings, Flex subjects in the current study agreed that CRI made it easier to understand diabetes control, prompted more action, and helped them get more from testing than their previous BGM.
Subjects switching to Verio meters exhibited pronounced improvements in glycemic control compared to controls. The strong improvement in glycemic control may be due to additional features (eg, pattern detection tools, motivational and achievement messages) on the Verio meter in addition to CRI. The pattern alert tool on Verio highlights potential opportunities for improving glycemic control in patients who self-adjust insulin. 12 Verio has also been shown to improve diabetes management practices in insulin-using patients. 9 In our study, nearly all subjects using the Verio meter agreed spotting patterns was easier and prompted them to take more action compared to their previous BGM. To improve glycemic control, meter features need to contribute to altered patient behavior and these patient viewpoints indicate that behavioral changes may be occurring and may account for some of the observed improvement in glycemic control. Subjects suggested that color was an important attribute which simplified understanding (eg, glucose ranges) or analysis (eg, patterns) of the results or elicited tangible changes in diabetes management (eg, adjusting my insulin more frequently).
Poor numeracy has been shown to impact improvements in glycemic control in patients with both T1 and T2D. 21 Similar subjective numeracy scores in the control, Verio, and Flex groups suggested that the improved A1c in intervention subjects could not be explained by higher numeracy. In addition, subjects with lower subjective numeracy in the CRI groups had A1c improvements similar to subjects with higher subjective numeracy indicating that CRI can be beneficial in low numeracy individuals.
It is known that increased levels of self-monitoring are associated with improvements in A1c. 22 However, in our study the clinically meaningful improvements in glycemic control observed using meters with a CRI were achieved despite no increase in self-monitoring.
Subjects with T1D using meters with CRI exhibited greater glycemic reductions compared to the reductions measured in subjects with T2D. Possibly this is because people with T1D can act upon BGM insights by making insulin dose adjustments after considering information from a CRI or pattern alerts. However, about 70% of the T2D subjects in this study were also insulin-users. Perhaps subjects with T2D did not exhibit similar glycemic reductions as those with T1D since most of these subjects were on fixed dose insulins (premix or basal) with the remainder of the T2D subjects being on a stable regimen of oral therapies with limited facility to make changes therapeutically in response to BGM insights. We postulate that patients on fixed doses might be more inclined in real world conditions to contact their HCP and either be advised to make behavioral or pharmacologic changes.
Conclusion
In conclusion, this randomized controlled study demonstrated that switching patients to OneTouch Verio or OneTouch Verio Flex BGMs featuring color range indicators was associated with improvements in A1c compared to subjects using a wide selection of marketed BGMs without color. We speculate that the beneficial insights and actions expressed by subjects using Verio or Flex meters translated into behavioral changes that resulted in improvements in glycemic control.
Footnotes
Acknowledgements
The authors would like to thank Laura Ritchie for leading the study setup, site conduct, and data entry activities with assistance from individual site leads Barry Irvine and Ghazala Rafiq. In addition, special thanks to diabetes study nurses Charlotte Ferris and Cheryl Davis (Birmingham, Heartlands Hospital); Alison Sudworth, Catherine Somerville, and Beena Poulose (Edinburgh Royal Infirmary); and Fiona Barrett and Ian Shread (Inverness, Highland Diabetes Institute) for all patient interactions.
Abbreviations
ACCENTS, Assessment by Consumers of Color Enhanced BGMs and New Therapeutic Strategies; A1C, hemoglobin A1C; AHA, antihyperglycemic agents (oral and/or GLP-1 agonists); BG, blood glucose; BGM, blood glucose monitor; CRI, color range indicator; HCP, health care professional; SMBG, self-monitoring of blood glucose; T1DM, type 1 diabetes; T2DM, type 2 diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MG is an employee of LifeScan Scotland, Ltd. LBK and BLL are employees of LifeScan Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by LifeScan, Inc.
