Abstract

Keywords
Measurements with systems for self-monitoring of blood glucose (SMBG) can be affected by the blood sample’s hematocrit value.1-3
The International Organization for Standardization (ISO) standard ISO 15197:2013 4 is an established standard for analytical performance assessment of SMBG systems. ISO 15197:2013 was harmonized in the European Union as EN ISO 15197:2015 5 with no changes regarding its requirements for performance studies. According to ISO 15197:2013, clause 6.4.3, hematocrit effects are acceptable if for 3 different test strip lots the mean difference between the test sample at different hematocrit levels and the respective control sample with a hematocrit value of 42% ± 2% is ≤10 mg/dL and ≤10% for blood glucose (BG) concentrations <100 mg/dL and ≥100 mg/dL, respectively. Hematocrit influence shall be described in the instructions for use if the SMBG system exceeds these criteria.
In this study, hematocrit influence was assessed for Accu-Chek® Guide blood glucose system (Roche Diabetes Care GmbH, Mannheim, Germany) with one test strip lot based on testing procedures and acceptance criteria of ISO 15197:2013.
The study was conducted from May to June 2016 at the Institut für Diabetes-Technologie, Forschungs- und Entwicklungsgesellschaft mbH an der Universität Ulm, Germany in compliance with the German Medical Devices Act. The study was approved by the responsible Ethics Committee and exempted from approval by the German Federal Institute for Drugs and Medical Devices.
Meters and test strips were provided by the manufacturer. The system was tested on venous blood samples from 3 different subjects (≥18 years, type 1 diabetes or without diabetes).
The samples were distributed into three glucose concentration categories (30-50 mg/dL, 96-144 mg/dL and 280-420 mg/dL). Samples could be adjusted to achieve target glucose concentrations if required. For each concentration category, individual samples with seven different hematocrit levels were generated based on the system’s acceptable hematocrit range as indicated in the manufacturer’s labeling: 10%, 15%, 25%, 42%, 55%, 60%, 65% (±2% for the midlevel sample [42%] and ±3% for the other samples, except for the samples at the lower and the upper limit [within +3% and −3%, respectively]).
Ten consecutive measurements were performed on each individual sample (combination of glucose concentration and hematocrit value) by using 10 different meters. Laboratory measurements were performed in duplicate with a hexokinase method (Cobas Integra® 400 plus; Roche Instrument Center, Rotkreuz, Switzerland) before and after measurements with the SMBG system.
For each individual sample the bias between the mean glucose result measured with the SMBG system (mean of 10 measurements) and the mean result of the laboratory method was calculated. To assess the hematocrit influence for each glucose concentration, the difference between the bias at higher and lower hematocrit levels and the bias at the midlevel was calculated (normalized bias).
The system showed ≤10 mg/dL and ≤10% difference (normalized bias) between the bias at higher and lower hematocrit levels and the bias at the midlevel (42 ± 2%) at glucose concentrations <100 mg/dL and ≥100 mg/dL, respectively (Figure 1).
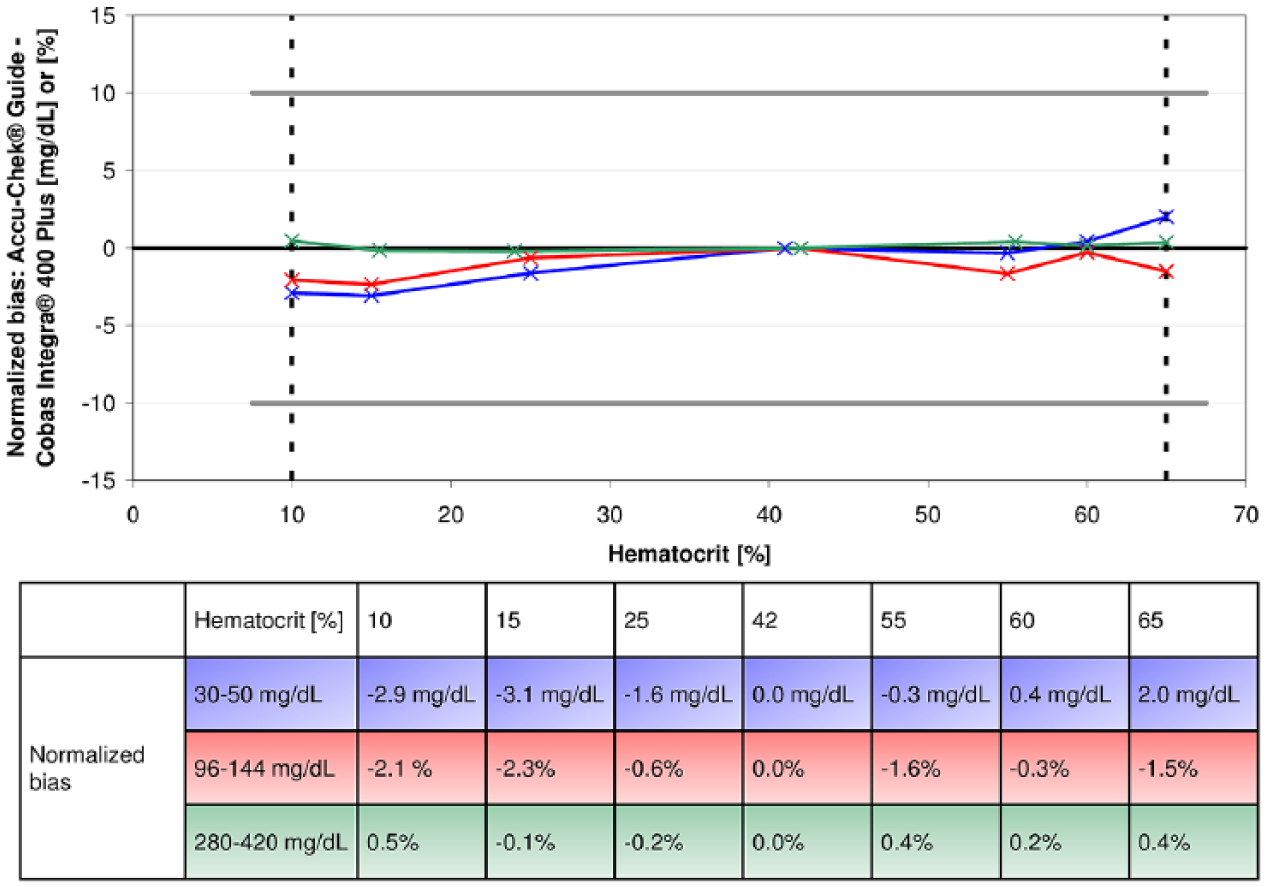
The difference between the bias at higher and lower hematocrit levels and the bias at the midlevel (42 ± 2%) was calculated (normalized bias) for three glucose concentration categories (blue: 30-50 mg/dL, red: 96-144 mg/dL, green: 280-420 mg/dL). The bias was calculated by using a hexokinase laboratory method. Lines connecting individual data points are provided for a simplified visualization and do not represent measured data. The system was tested within the labeled hematocrit range (10%-65%; indicated by dashed lines). Solid gray lines show ≤10 mg/dL and ≤10% limits at glucose concentrations <100 mg/dL and at glucose concentrations ≥100 mg/dL, respectively.
The system fulfilled ISO 15197:2013 acceptance criteria with the investigated test strip lot within the hematocrit range given in the manufacturer’s labeling.
Footnotes
Acknowledgements
We would like to thank the study personnel and other employees of the Institut für Diabetes-Technologie, Forschungs- und Entwicklungsgesellschaft mbH an der Universität Ulm, Ulm, Germany (IDT) who conducted the study and helped prepare the manuscript.
Abbreviations
BG, blood glucose; ISO, International Organization for Standardization; SMBG, self-monitoring of blood glucose.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: GF is general manager of the IDT, which carries out clinical studies on the evaluation of BG meters and medical devices for diabetes therapy on its own initiative and on behalf of various companies. GF/IDT have received speakers’ honoraria or consulting fees from Abbott, Ascensia, Bayer, LifeScan, Menarini Diagnostics, Novo Nordisk, Roche, Sanofi, Sensile, and Ypsomed. All authors are employees of the IDT.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Roche Diabetes Care GmbH, Mannheim, Germany.
