Abstract

Keywords
In evaluating the quality of devices for self-monitoring of blood glucose, system accuracy is one major aspect to be considered. The international standard ISO 15197:2013 defines system accuracy as the agreement between blood glucose (BG) results obtained with a BG monitoring system (BGMS) and the respective comparison values. 1 In addition, procedures and criteria for the assessment of system accuracy are described in this standard. In this study, system accuracy of 2 BGMSs (GLUCOCARD™ G+ and GlucoMen® areo; A. Menarini Diagnostics, Florence, Italy) with 3 reagent system lots each was evaluated following ISO 15197:2013.
The study was performed in March and October 2014 at the Institut für Diabetes-Technologie Forschungs- und Entwicklungsgesellschaft mbH an der Universität Ulm, Ulm, Germany. The study was performed in compliance with the German Medical Devices Act and approved by the Ethics Committee and the notified authority.
GLUCOCARD™ G+ is a well-established BGMS available on the European market since 2007, while GlucoMen® areo is a novel system recently introduced in various European countries, with enhanced features like hematocrit compensation, postprandial reminder and near field communication (NFC) connectivity. Both systems were procured by the manufacturer. Capillary blood samples from at least 100 different subjects were obtained by skin puncture and BG concentrations were measured with the BGMS and the reference method using the hexokinase-based Cobas Integra® 400 plus (Roche Instrument Center, Rotkreuz, Switzerland). This method is traceable according to ISO 17511; 2 trueness and precision were verified during the test procedure.
Both BGMSs fulfilled the accuracy criteria of ISO 15197:2013 (Figure 1), which are (1) at least 95% of the results are within ±15 mg/dl of the reference glucose values at BG concentration <100 mg/dl and within ±15% at BG concentrations ≥100 mg/dl (for each individual test strip lot) and (2) at least 99% of the results of all 3 lots together are within zones A and B of the consensus error grid.
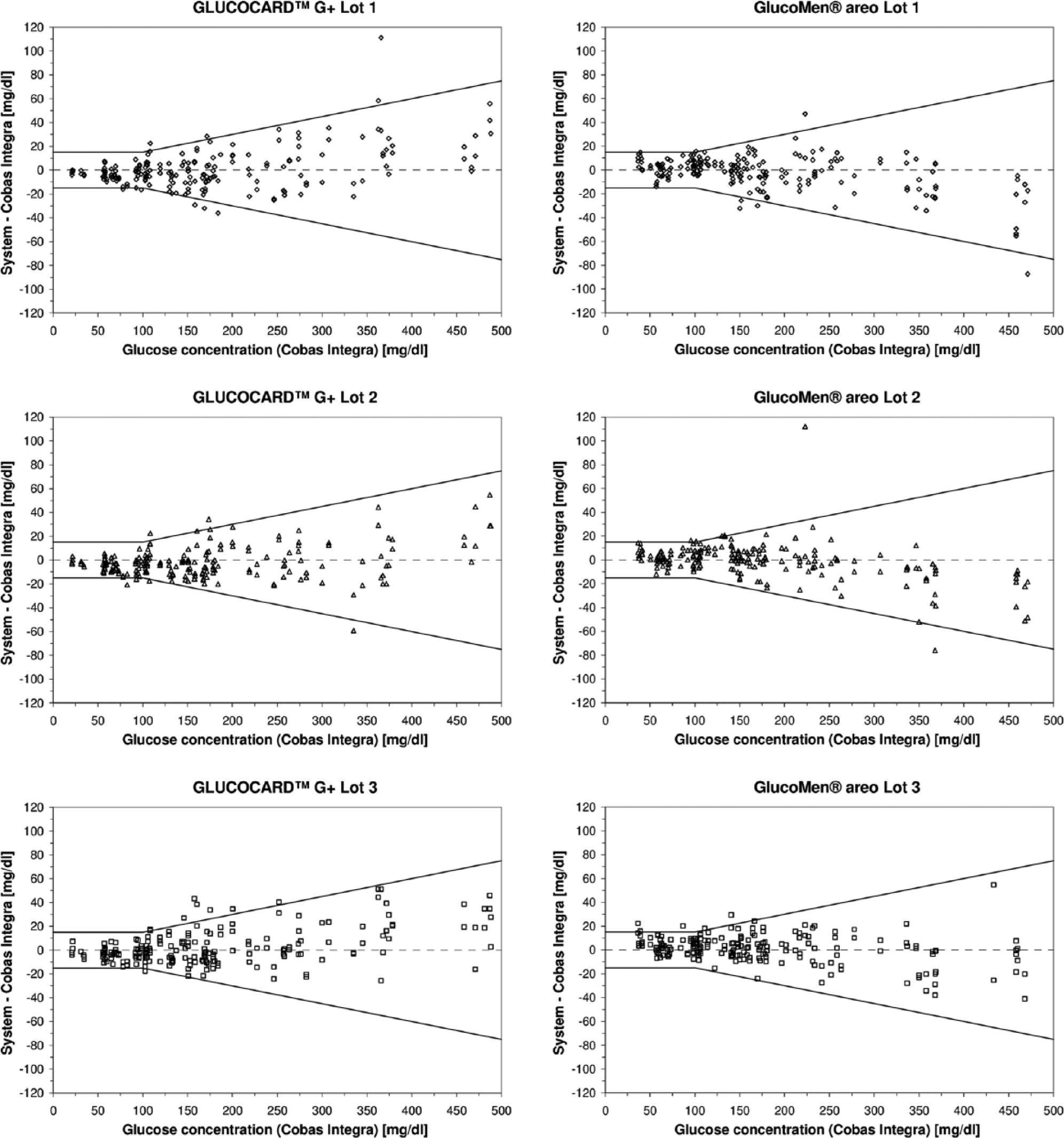
System accuracy of all 3 lots of the 2 systems. Absolute differences between the blood glucose concentration results measured with the systems and the respective comparison values measured with Cobas Integra (200 data, duplicate measurements on a sample with each test strip lot) are shown. Solid lines indicate the limits stipulated in ISO 15197:2013.
For BG values <100 mg/dl, GLUCOCARD™ G+ system showed 96.4% to 100% of results within ±15 mg/dl and 80.4% to 89.3% within ±10 mg/dl. GlucoMen® areo showed 94.6% to 100% within ±15 mg/dl and 82.1% to 85.7% within ±10 mg/dl. At BG concentrations ≥100 mg/dl, 93.8% to 96.5% of the values measured with GLUCOCARD™G+ system were within ±15% and 79.2% to 86.1% within ±10%. For GlucoMen® areo, 95.8% to 98.6% were within ±15% and 84.7% to 89.6% within ±10% in these BG concentrations. Considering all BG concentrations, 95% to 96.5% of GLUCOCARD™ G+ system results and 97% to 97.5% of GlucoMen® areo results were within the limits. 100% of the values were within zones A and B of the consensus error grid for both systems. The relative bias according to Bland and Altman 3 ranged from −3.2% to −0.8% for GLUCOCARD™ G+ and from +0.3% to +2.2% for GlucoMen® areo.
In this study, the 2 investigated BGMSs were shown to fulfill system accuracy criteria of ISO 15197:2013 when evaluated against a hexokinase-based reference method. The newer system, GlucoMen® areo, showed a superior level of accuracy.
Footnotes
Acknowledgements
The authors would like to thank the Institut für Diabetes-Technologie Forschungs- und Entwicklungsgesellschaft mbH an der Universität Ulm, Ulm, Germany for the performance of the study.
Abbreviations
BG, blood glucose; BGMS, blood glucose monitoring system; NFC, near field communication.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors are employees of A. Menarini Diagnostics, Florence, Italy.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by A. Menarini Diagnostics, Florence, Italy.
