Abstract

The success of sensor-augmented pump (SAP) in patients with type 1 diabetes mellitus (T1D) is dependent on both health care providers (HCP; knowledge and experience) and patients (educational efforts and good training).
One of the available software for analyzing SAP is CareLink (Medtronic MiniMed, Inc, Northridge, CA). 1 A 14-day download consists of 9 different reports on 23 pages. Although the reports are well structured and organized, 2 HCP are finding difficulties in interpreting the data in their busy clinics.
We have developed a 5-step approach protocol in interpreting SAP using CareLink. The main objective of this pilot study was to assess the usability of the protocol. The main analysis is based on the Therapy Management Dashboard, where five segments are allocated: (1) basic statistics; (2) glucose and insulin overlay; (3) postprandial period; (4) basal/bolus insulin; (5) suspends and sensor. Possible changes are noted on a printed insulin settings page with evaluation of rationale/physiological insulin. Every segment is analyzed with correlation through others with step by step confirmation of possible change.
Thirty-four patients (F 18, M 16), age 15.3 ± 7.7 years with total of 128 CareLink downloads were analyzed on monthly basis in a period of 3 months.
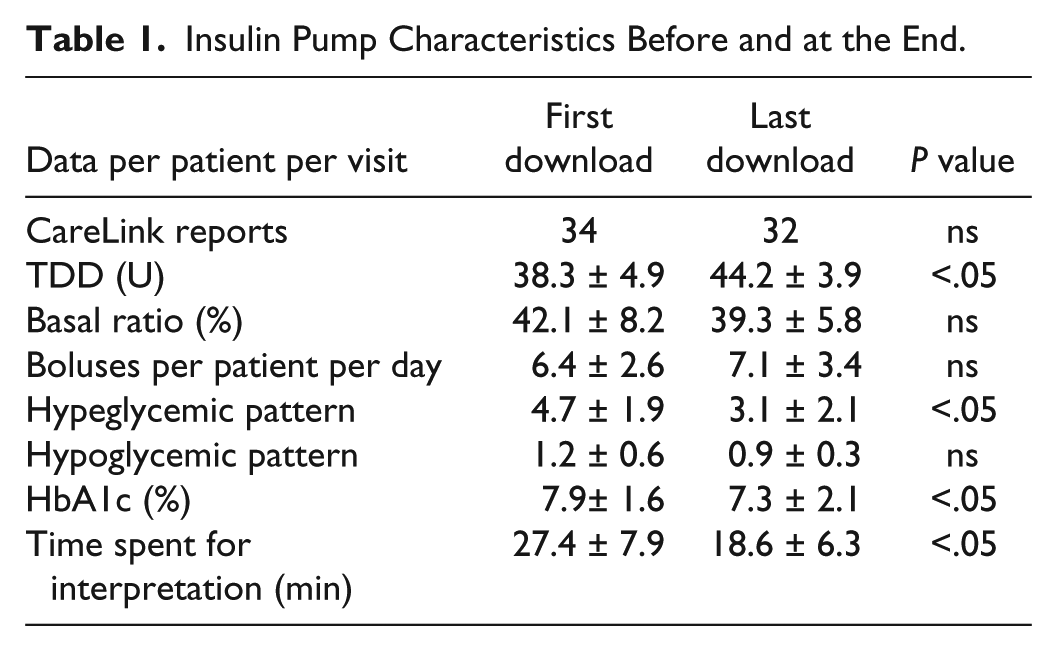
The patients profile matched our everyday clinical practice, similar to glucose control on patients using insulin pump reported in several studies.3,4 Our data showed increased insulin total daily dose (TDD), due to fine tuning with more bolus dose than the basal rates. The glucose control was improved (as seen in Table 1) with a decreased hyperglycemic and hypoglycemic patterns.
Insulin Pump Characteristics Before and at the End.
A 5-step approach protocol in interpreting SAP using CareLink is simple, convenient, and efficient and should be assessed on the multicenter level and with a wider population.
Footnotes
Abbreviations
CareLink, CareLink Therapy Management Software for Diabetes; HCP, health care providers; SAP, sensor-augmented pump; T1DM, type 1 diabetes mellitus; TDD, total daily dose.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
