Abstract

Keywords
The international standard ISO 15197 describes system requirements for blood glucose monitoring systems (BGMS). The implementation of a new version of this standard in 2013, which was harmonized in the EU as EN ISO 15197:2015, aimed to further improve the quality of available systems by, for example, setting stricter accuracy criteria.1,2
This study was conducted to assess conformity of a new version of the Accu-Chek® Active (Roche Diabetes Care GmbH, Mannheim, Germany) to system accuracy requirements of ISO 15197:2013.
The improved BGMS does not require coding with a lot-specific code chip anymore which will eliminate a possible source of improper use of codechips. 3 The BGMS comprises the meter and test strips and is labeled for self-testing by people with diabetes as well as for use by health care professionals.
The study was conducted at the Institut für Diabetes-Technologie Forschungs- und Entwicklungsgesellschaft mbH an der Universität Ulm, Ulm, Germany in July 2016 and was performed in compliance with the Good Clinical Practice guidelines and regulatory requirements were fulfilled.
Meters, test strips and control solution were provided by Roche Diabetes Care GmbH. Systems were set and maintained according to the manufacturer’s instructions and daily control measurements were performed.
Based on procedures of ISO 15197:2013, capillary blood samples from the fingertips of at least 100 different subjects with a defined distribution of blood glucose (BG) concentrations were measured with 3 different lots of the test strips of the BGMS and a hexokinase-based comparison method (Cobas Integra® 400 plus; Roche Instrument Center, Rotkreuz, CH). Comparison measurements were performed at the study site; the method is ISO 17511 traceable according to the manufacturer. 4
Data were analyzed at the study site. Differences between results of the BGMS and the comparison measurements were calculated and the number of values within the system accuracy limits of ISO 15197:2013 was determined. In addition, the relative bias was calculated according to Bland and Altman. 5
At BG concentrations <100 mg/dl (52 values), 100% (Lots 1, 2, and 3) of BGMS results fell within ±15 mg/dl of the comparison values. At BG concentrations ≥100 mg/dl (148 values), 98% (Lot 1), 99.3% (Lot 2), and 100% (Lot 3) of BGMS results were within ±15%. For all BG concentrations (28 mg/dl-451 mg/dl), 98.5% (Lot 1), 99.5% (Lot 2), and 100% (Lot 3) were within the respective limits. Furthermore, 100% of the results were in consensus error grid zone A (see Figure 1). The relative bias was -5.4% (Lot 1), -2.3% (Lot 2), and -1.6% (Lot 3).
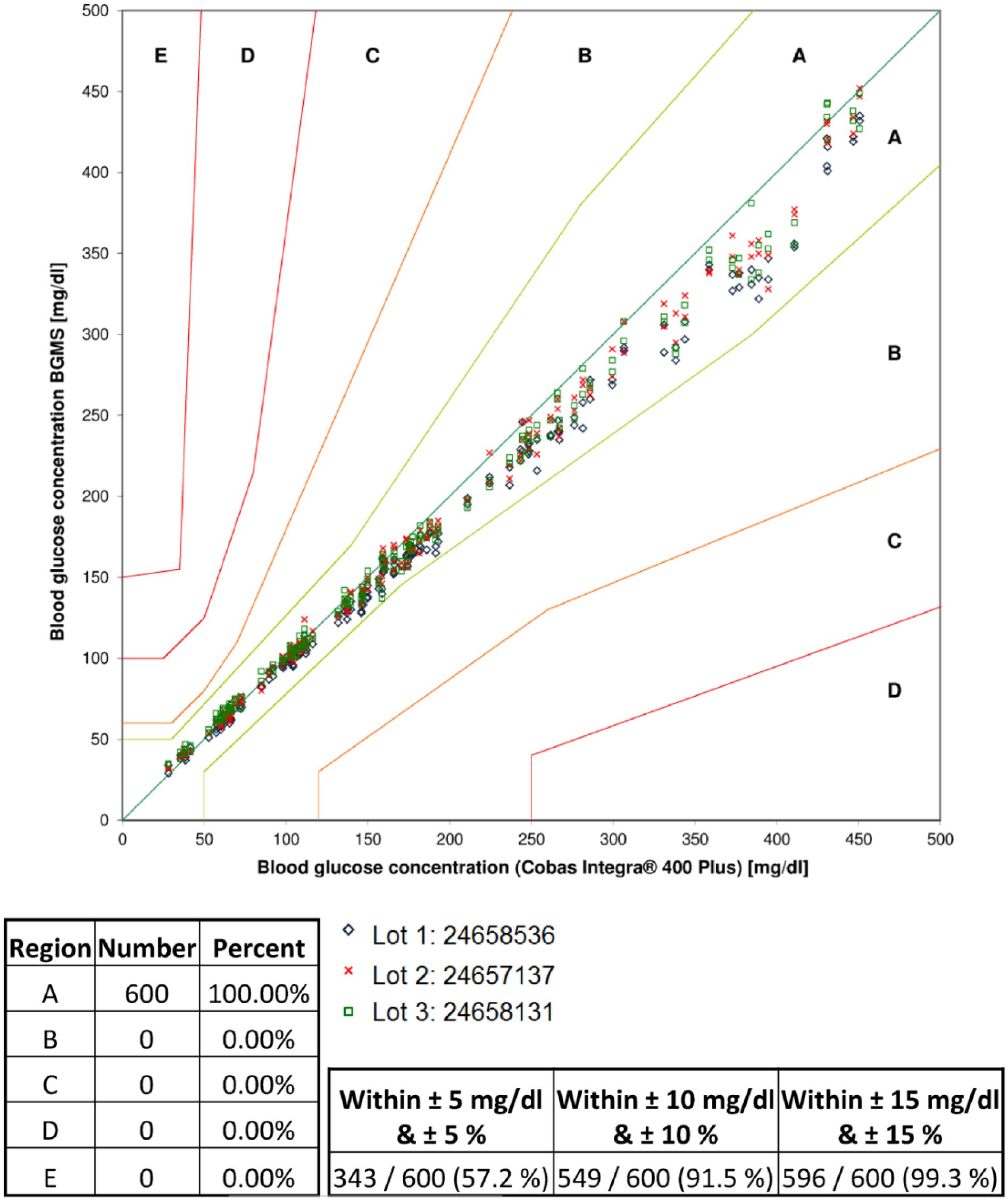
Consensus error grid for the BGMS with three reagent system lots. The error grid is a tool to evaluate the clinical relevance of system accuracy. The error grid shows different zones. Zone A: no affection of clinical decisions; zone B: clinical decisions may be altered but without any clinical outcome; zone C: clinical decisions will be altered and are likely to affect the clinical outcome; zone D: clinical decisions will be altered and could result in a significant medical risk; zone E: clinical decisions will be altered and could have potential dangerous consequences.1,2
This study confirmed that the BGMS had at the introduction on the market a high level of accuracy and fulfilled with three manufacturer-provided lots the system accuracy requirements of ISO 15197:2013.
Footnotes
Acknowledgements
The authors would like to thank the laboratory staff of the Institut für Diabetes-Technologie Forschungs- und Entwicklungsgesellschaft mbH an der Universität Ulm, Ulm, Germany for the performance of the study.
Abbreviations
BG, blood glucose; BGMS, blood glucose monitoring system; ISO, International Organization for Standardization.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: UK, AB, NJ, and CH are employees of the Institut für Diabetes-Technologie Forschungs- und Entwicklungsgesellschaft mbH an der Universität Ulm, Ulm, Germany (IDT). GF is general manager of the IDT, which carries out clinical studies on the evaluation of BG meters and medical devices for diabetes therapy on its own initiative and on behalf of various companies. GF/IDT have received speakers’ honoraria or consulting fees from Abbott, Ascensia, Bayer, Berlin-Chemie, Becton-Dickinson, Dexcom, LifeScan, Menarini Diagnostics, Novo Nordisk, Roche, Sanofi, Sensile and Ypsomed.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded and the writing of the manuscript was supported by Roche Diabetes Care GmbH, Mannheim, Germany.
