Abstract

Keywords
In the European Union, the CE (Conformité Européenne) mark is required prior to market approval of medical devices, including systems for self-monitoring of blood glucose (SMBG). For SMBG systems, evidence of sufficient measurement quality has to be provided, for example from studies following ISO 15197.1,2 This is a 1-time process, and postmarket release evaluations or independent evaluations are not mandatory, despite studies repeatedly showing test strip lots of available systems not adhering with ISO 15197 accuracy criteria.3 -6
In 2 studies, performed in April and May 2013 and in October 2014, the SMBG systems GlucoCheck neo (aktivmed GmbH, Germany; equivalent to GluNeo®, Infopia Ltd, Korea) and Element™ (Infopia Ltd, Korea) were investigated with 3 lots of test strips to assess system accuracy with respect to the criteria of ISO 15197:2003 and its revision ISO 15197:2013.1,2 The devices and test strips were provided by aktivmed and Infopia, respectively.
After approval of the protocol by the ethics committee and the competent authority, the studies were performed at the Institut für Diabetes-Technologie Forschungs- und Entwicklungsgesellschaft mbH an der Universität Ulm, Ulm, Germany in compliance with the German Medical Devices Act (MPG). For each reagent system lot 100 capillary blood samples from different adult subjects with type 1 or type 2 diabetes or no diabetes were measured in duplicate, resulting in 200 measurements for each test strip lot, following procedures described in ISO 15197. Cobas® c111 (Roche Instrument Center, Switzerland) and Cobas Integra® 400 Plus (Roche Instrument Center), both hexokinase-based laboratory analyzers, were used for comparison measurements in capillary plasma. Trueness and precision of both laboratory analyzers were confirmed by regular internal and external quality control measurements.
Results of the system accuracy evaluation are provided in Table 1.
System Accuracy Results for GlucoCheck neo and Element.
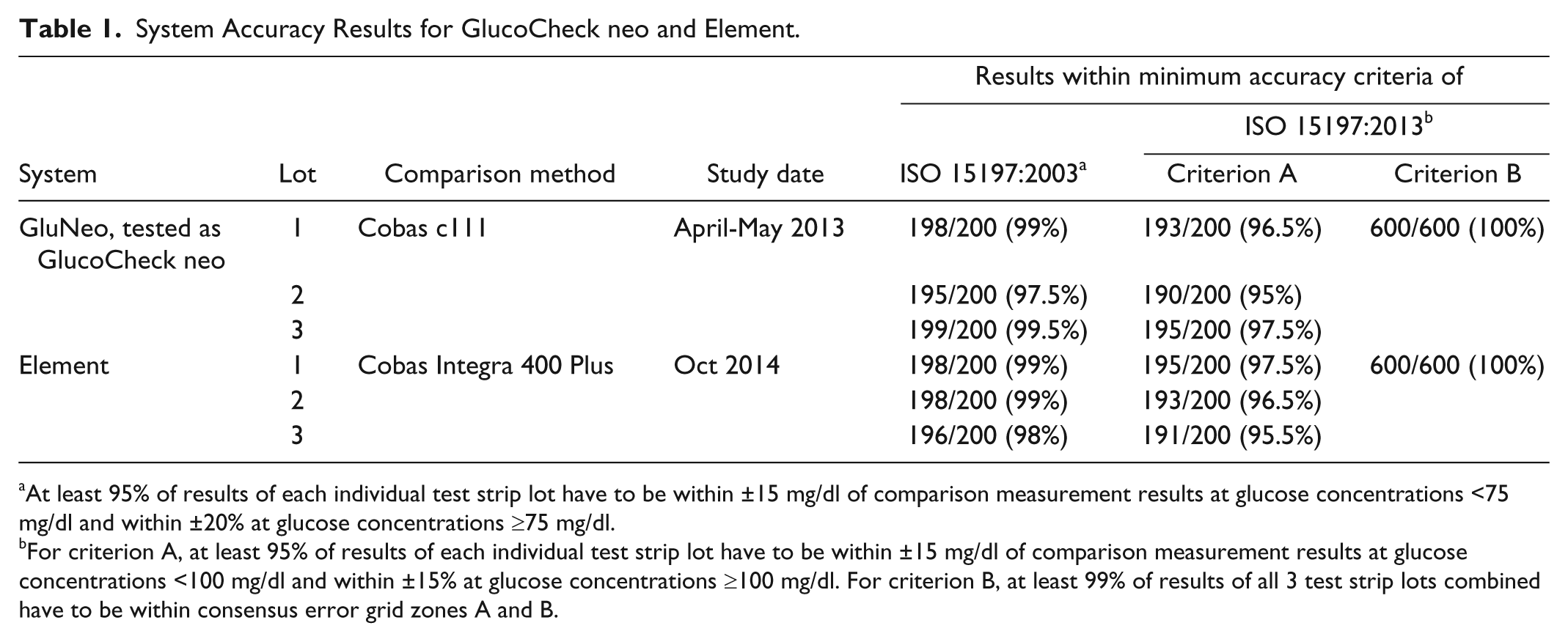
At least 95% of results of each individual test strip lot have to be within ±15 mg/dl of comparison measurement results at glucose concentrations <75 mg/dl and within ±20% at glucose concentrations ≥75 mg/dl.
For criterion A, at least 95% of results of each individual test strip lot have to be within ±15 mg/dl of comparison measurement results at glucose concentrations <100 mg/dl and within ±15% at glucose concentrations ≥100 mg/dl. For criterion B, at least 99% of results of all 3 test strip lots combined have to be within consensus error grid zones A and B.
ISO 15197:2003 minimum system accuracy criteria were fulfilled by both systems with all 3 tested lots with 97.5% to 99.5% (minimum requirement: 95% of results of each lot within ±15 mg/dl of comparison results at glucose concentrations <75 mg/dl and within ±20% at glucose concentrations ≥75 mg/dl). Both systems also fulfilled the ISO 15197:2013 minimum system accuracy criteria with 95% to 97.5% for criterion A (minimum requirement: 95% of results of each lot within ±15 mg/dl of comparison results at glucose concentrations <100 mg/dl and within ±15% at glucose concentrations ≥100 mg/dl) and with 100% for criterion B (minimum requirement: 99% of results of all 3 lots within consensus error grid zones A and B).
In conclusion, this evaluation after market release showed that both SMBG systems fulfilled the system accuracy requirements of ISO 15197:2003 and ISO 15197:2013 with all 3 evaluated test strip lots. Regular and independent evaluations after market release are a helpful tool to ensure continuing adherence of available test strips to regulatory requirements.
Footnotes
Acknowledgements
The authors would like to thank the Institut für Diabetes-Technologie Forschungs- und Entwicklungsgesellschaft mbH an der Universität Ulm, Ulm, Germany for the performance of the studies.
Abbreviations
CE, Conformité Européenne; MPG, German Medical Devices Act; SMBG, self-monitoring of blood glucose.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PH is employed by Neon Diagnostics Ltd.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The studies about GluNeo (tested as GlucoCheck Neo) and Element were funded by aktivmed GmbH, Germany, and Infopia Ltd, Korea, respectively. Medical writing was supported by Neon Diagnostics Ltd, United Kingdom.
