Abstract
Background:
The aim was to compare changes in HbA1c and body weight after initiation of dapagliflozin or basal insulin supported oral therapy (BOT) in type 2 diabetes patients in primary care practices.
Methods:
Patients from 983 primary care practices who started dapagliflozin or BOT between December 2012 and July 2015 (index date, ID) were retrospectively analyzed (Disease Analyzer; Germany). Changes in HbA1c (%) and body weight (kg) were evaluated 90-270 days after ID. Propensity score (PS) matching (1:1) was used to adjust for differences in baseline clinical characteristics (180-0 days before ID: age, sex, health insurance, diabetologist care, glucose lowering therapy, HbA1c, body mass index) and duration (days) between start of therapies and last HbA1c or weight documentation after ID.
Results:
After PS matching, 766 dapagliflozin (mean ± SD; age: 63 ± 10 years; HbA1c: 8.9 ± 1.2%) and 766 BOT (age: 63 ± 10 years; HbA1c: 8.7 ± 1.1%) patients were included. HbA1c decreased by mean (SD) of 1.0% (1.3) in dapagliflozin and by 1.0% (1.4) in BOT patients after 90-270 days (HbA1c reduction; dapagliflozin vs BOT: –0.01%; P = .79). In 440 dapagliflozin users with available data, body weight (97.4 ± 19.9 kg) decreased by 3.1 (5.8) kg after 90-270 days, whereas no significant weight change was observed in 440 matched BOT patients (97.5 ± 19.9 kg) (weight reduction; dapagliflozin vs BOT: –3.0 kg; P < .05).
Conclusions:
Initiation of dapagliflozin therapy reduced HbA1c similar to basal insulin with the additional benefit of weight reduction in type 2 diabetes patients treated in general practices.
Sodium glucose co-transporter 2 (SGLT2), which is located in the proximal tubule of the kidney, is involved in the reabsorption of glucose. 1 Because the SGLT2 effects are independent of insulin, its inhibition should not be influenced by insulin resistance or secretion. 1 Thus, SGLT2 inhibitors have the potential to reduce hyperglycemia by stimulating urinary glucose excretion at any stage or duration of diabetes. 2 SGLT2 inhibitors are mentioned among the options for second-line therapy after metformin in the joint position statement on management of hyperglycemia in type 2 diabetes of the American Diabetes Association (ADA) and European Association for the Study of Diabetes (EASD). 3 Metformin is the first-line glucose-lowering drug therapy for type 2 diabetes. 3 If the HbA1c target is not achieved after 3 months, one of the six treatment options combined with metformin should be considered: a sulfonylurea, a thiazolidinedione, a dipeptidyl peptidase-4 inhibitor (DPP-4i), a glucagon-like peptide-1 receptor agonist (GLP-1RA), a SGLT2 inhibitor, or basal insulin. 3 SGLT2 inhibitors should also be considered as alternative first-line options in patients with contraindications to metformin or as an add-on to insulin therapy (dual or triple therapy). 3 Dapagliflozin was the first approved selective SGLT2 inhibitor. 4 In several randomized controlled trials, dapagliflozin monotherapy or combinations with metformin, sulfonylureas, dipeptidyl peptidase-4 (DPP-4) inhibitors, GLP-1 receptor agonists or insulin have demonstrated substantial reductions in HbA1c in all stages of type 2 diabetes in combination with favorable effects on body weight and a moderate lowering of blood pressure.5-15
Type 2 diabetes is a progressive disease, and many patients require insulin treatment to achieve and maintain good glycemic control. 16 Although insulin therapy is effective to lower HbA1c levels, insulin initiation is often considered as an unfavorable step by both patients and health care providers. 17 Known barriers for insulin therapy are hypoglycemia and weight gain. 16 These potential side effects often result in postponement of insulin therapy.16-18 The ADA/EASD position statement recommends initiation of basal insulin therapy as dual or triple treatment if type 2 diabetes patients did not achieve adequate glycemic control. 3 However, alternative treatment options are needed for patients with high HbA1c who also need to lose weight and to avoid hypoglycemia. Because glycemic improvements of SGLT2 inhibitors are often associated with weight reduction and a low risk of hypoglycemia, dapagliflozin might be considered as an alternative to insulin therapy.
The aim of this study was to investigate real-life treatment outcomes for HbA1c and body weight in type 2 diabetes patients initiating dapagliflozin or basal insulin supported oral therapy (BOT) using a large representative general practice database.
Methods
The German Disease Analyzer database (IMS HEALTH) was used for this retrospective observational study. Disease Analyzer contains anonymized longitudinal data on drug prescriptions, diagnoses, and basic medical and demographic data directly obtained from the computer system of a representative sample of general practitioners and internal medicine practices throughout Germany. 19 The analyzed database period for the current study was December 2012 to October 2015 (983 general and internal medicine practices). Patients with type 2 diabetes, who were initiated on dapagliflozin therapy or who started BOT during the study period (index date, ID) were included. The practice visit records were used to assemble data 180-0 days before and 90-270 days after ID, respectively.
Patient were included in the study if they (1) had ≥1 documented HbA1c 180-0 days before and 90-270 days after ID, (2) had ≥1 documentation of body weight 180-0 days before and 90-270 days after ID, and (3) had continuous dapagliflozin or BOT treatment between ID and the last HbA1c or body weight documentation 90-270 days after ID. BOT is defined as continuous prescription use of basal insulins over the whole study period in addition to oral glucose-lowering drugs.
In dapagliflozin users, patients with any insulin prescriptions between ID and the last documented HbA1c measurement (90-270 days after ID) were excluded. In the BOT cohort, all patients with prescriptions of SGLT2 inhibitors or other insulins (eg, rapid acting insulin, premixed insulin) between the ID and the last HbA1c (90-270 days after ID) were also excluded.
Propensity score 1:1 matching used to generate matched groups for dapagliflozin and BOT users. Matching was carried out for age (5-year groups), sex, last HbA1c or last body weight before ID, health insurance (private/statutory), glucose-lowering treatment (metformin, sulfonylureas, DPP-4 inhibitors, other) during 180-0 days before ID and time (days) between ID and last recorded HbA1c or weight 90-270 days after ID using a propensity score from logistic regression. The maximum allowed difference between propensity scores for matching within the matched pairs was 0.01.
Descriptive statistics were given and group differences (dapagliflozin vs BOT) after propensity score matching were assessed using paired t-tests or McNemar’s tests. Two sided tests were used and a P-value of < .05 was considered as statistically significant. Scatterplots were applied to visualize changes in body weight and changes in HbA1c after ID. All analyses were carried out following the German good practice recommendations of secondary data analysis 20 using SAS 9.3 (SAS Institute, Cary, NC, USA).
Results
Among 6297 patients with at least one dapagliflozin prescription during the study period, HbA1c values before and after ID were recorded in 2845 patients, of whom 1703 fulfilled the inclusion criteria (Table 1). BOT was started in 39 093 type 2 diabetes patients and 4502 were included. After propensity score matching, 766 in each group were analyzed. Data on body weight was less often recorded than HbA1c values. After applying the inclusion and exclusion criteria, 571 dapagliflozin and 1500 BOT users were eligible. Finally, 440 patients with dapagliflozin and 440 matched patients on BOT were included in the analysis of body weight changes (Table 1). The median time of last recorded HbA1c before (0-180 days: dapagliflozin: 19 days; BOT: 20 days; P > .05) and after (90-270 days: dapagliflozin: 200 days; BOT: 208 days; P > .05) index date were not different.
Patient Selection for Analyses of Changes in HbA1c and Body Weight in Type 2 Diabetes Patients Initiating Dapagliflozin or Basal Insulin Supported Oral Therapy (BOT) in Primary Care Practices in Germany (Disease Analyser).
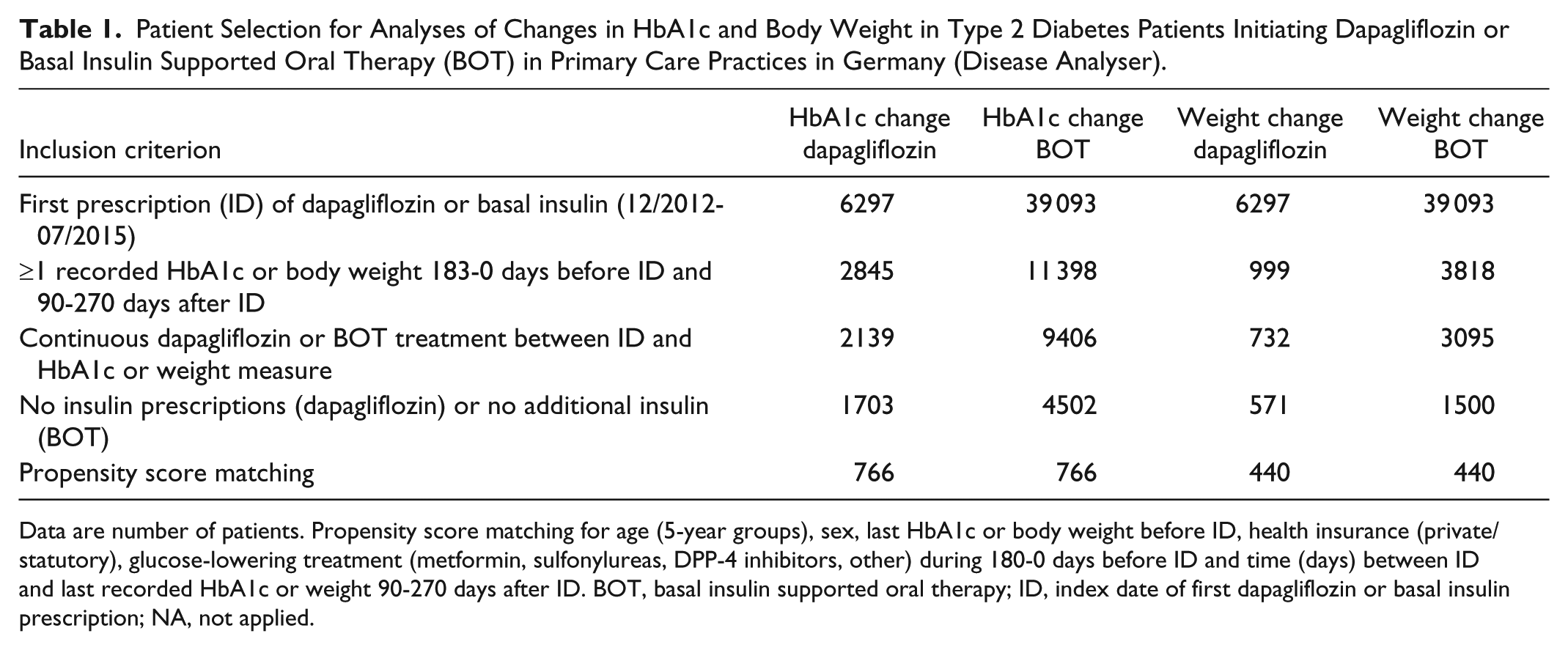
Data are number of patients. Propensity score matching for age (5-year groups), sex, last HbA1c or body weight before ID, health insurance (private/statutory), glucose-lowering treatment (metformin, sulfonylureas, DPP-4 inhibitors, other) during 180-0 days before ID and time (days) between ID and last recorded HbA1c or weight 90-270 days after ID. BOT, basal insulin supported oral therapy; ID, index date of first dapagliflozin or basal insulin prescription; NA, not applied.
The baseline characteristics of dapagliflozin and BOT users for the HbA1c analysis are shown in Table 2. Before matching, dapagliflozin users were younger and comprised more men than the BOT cohort (P < .05). About a quarter of the patients with dapagliflozin were treated by diabetologists (BOT: 14%; P < .05). There was also a higher prevalence of privately insured patients in dapagliflozin users (P < .05). Finally, a higher prevalence of metformin prescriptions were observed in the dapagliflozin cohort (P < .05). The mean time (days) between ID and last documented HbA1c was similar in both cohorts. After matching, no differences in the above mentioned and other characteristics (baseline HbA1c, glucose-lowering therapy) were found (Table 2).
Baseline Characteristics of Type 2 Diabetes Patients Initiating Dapagliflozin or Basal Insulin Supported Oral Therapy (BOT) in Primary Care Practices in Germany (Disease Analyser): Analysis of Change in HbA1c.
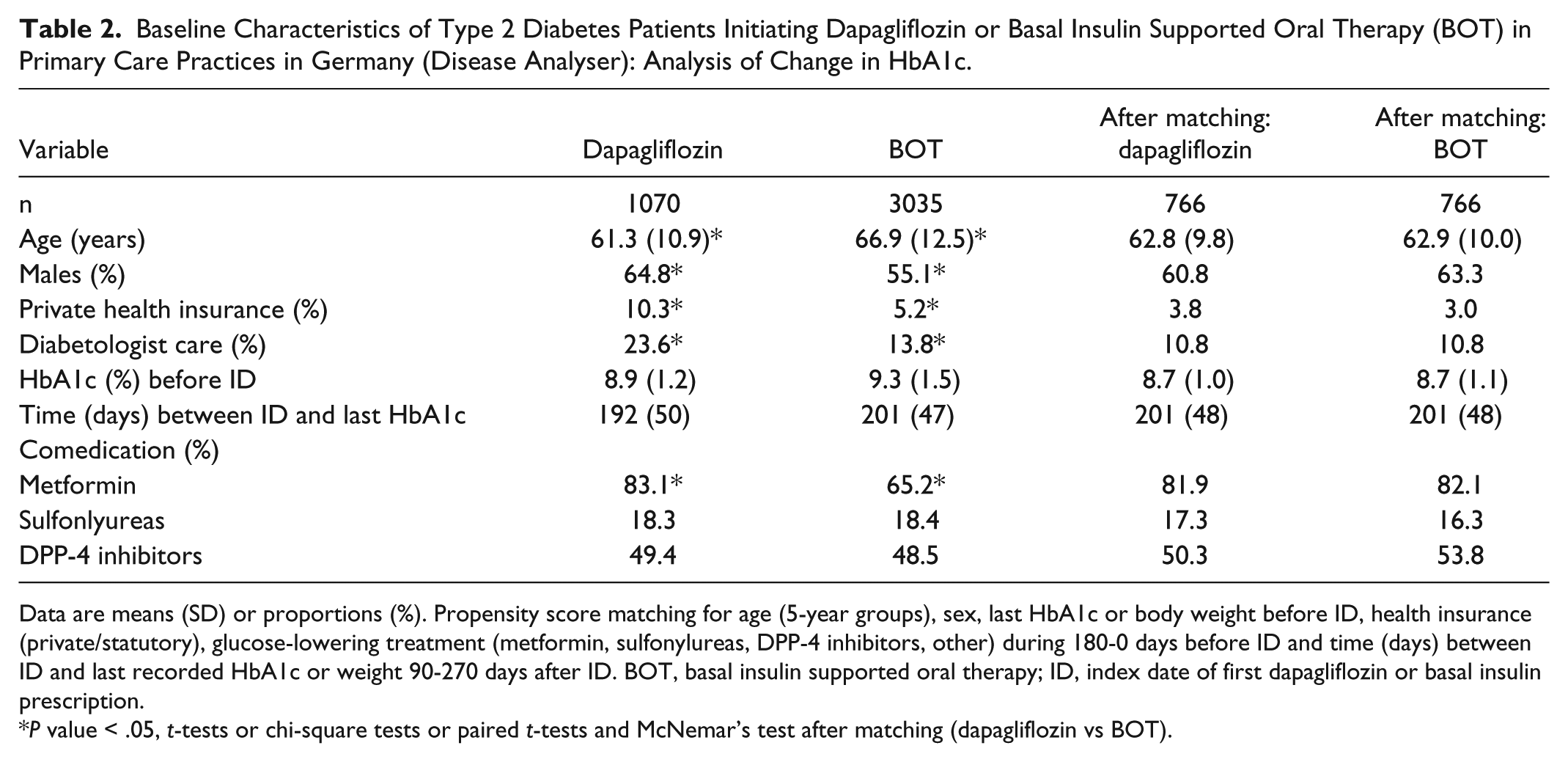
Data are means (SD) or proportions (%). Propensity score matching for age (5-year groups), sex, last HbA1c or body weight before ID, health insurance (private/statutory), glucose-lowering treatment (metformin, sulfonylureas, DPP-4 inhibitors, other) during 180-0 days before ID and time (days) between ID and last recorded HbA1c or weight 90-270 days after ID. BOT, basal insulin supported oral therapy; ID, index date of first dapagliflozin or basal insulin prescription.
P value < .05, t-tests or chi-square tests or paired t-tests and McNemar’s test after matching (dapagliflozin vs BOT).
The mean age of dapagliflozin users included in the body weight analysis was also lower compared to the BOT cohort before matching (Table 3). There were also differences with respect to sex and private health insurance between the two groups. Baseline body weight and prescription prevalence for metformin were both significantly higher among dapagliflozin users than in the BOT cohort (P < .05). The mean time between the ID and the last recorded body weight was similar in both groups. BOT users were significantly more often in diabetologist care than the dapagliflozin cohort (P < .05). After matching there were no significant differences for any of the baseline clinical characteristics (Table 3). The mean daily dose of basal insulin in the BOT group was 25.8 (SD: 17.6) units.
Baseline Characteristics of Type 2 Diabetes Patients Initiating Dapagliflozin or Basal Insulin Supported Oral Therapy (BOT) in Primary Care Practices in Germany (Disease Analyser): Analysis of Change in Body Weight.
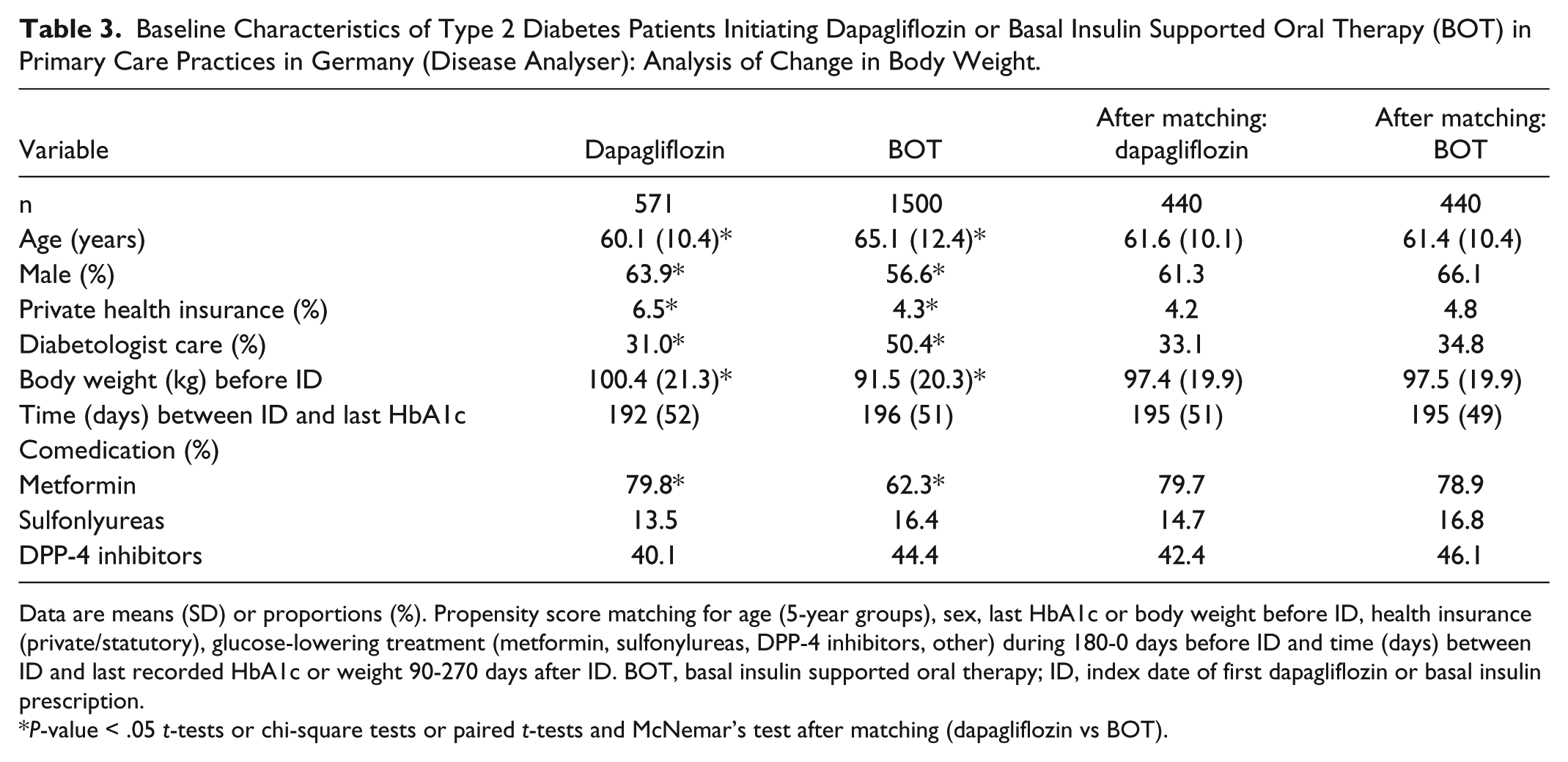
Data are means (SD) or proportions (%). Propensity score matching for age (5-year groups), sex, last HbA1c or body weight before ID, health insurance (private/statutory), glucose-lowering treatment (metformin, sulfonylureas, DPP-4 inhibitors, other) during 180-0 days before ID and time (days) between ID and last recorded HbA1c or weight 90-270 days after ID. BOT, basal insulin supported oral therapy; ID, index date of first dapagliflozin or basal insulin prescription.
P-value < .05 t-tests or chi-square tests or paired t-tests and McNemar’s test after matching (dapagliflozin vs BOT).
After starting dapagliflozin, mean (SD) HbA1c was lowered by 0.8% (1.3) in the whole cohort after 90-270 days (BOT: –1.0% [1.8]) (Table 4). In subgroup analyses, including patients with inadequate glycemic control (HbA1c >7.5%) at baseline, larger HbA1c reductions were observed in both cohorts (Table 4). After propensity score matching, mean HbA1c decline was 1.0 (1.3) in dapagliflozin users, which was equal to the HbA1c change in the BOT cohort (HbA1c difference dapagliflozin vs BOT: –0.01%; P = .79).
Changes in HbA1c (%) and Body Weight (90) of Type 2 Diabetes Patients After 90-270 Days Initiating Dapagliflozin or Basal Insulin Supported Oral Therapy (BOT) in Primary Care Practices.
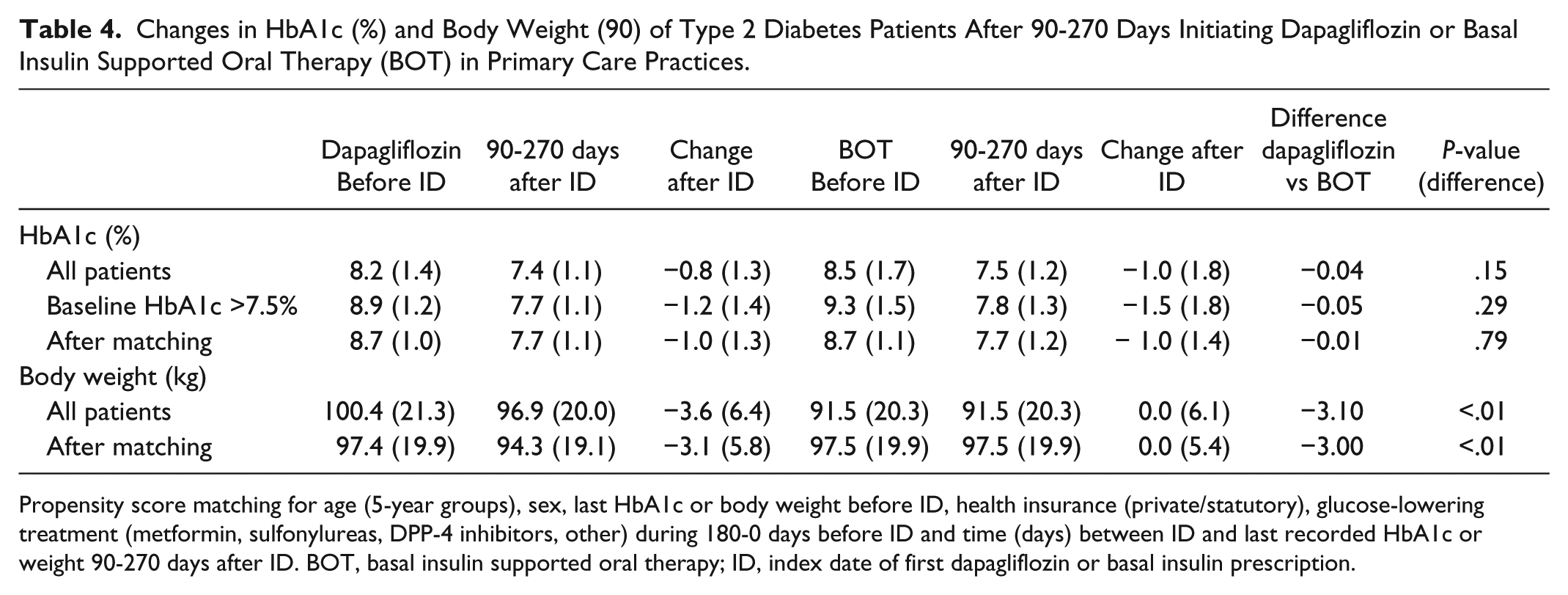
Propensity score matching for age (5-year groups), sex, last HbA1c or body weight before ID, health insurance (private/statutory), glucose-lowering treatment (metformin, sulfonylureas, DPP-4 inhibitors, other) during 180-0 days before ID and time (days) between ID and last recorded HbA1c or weight 90-270 days after ID. BOT, basal insulin supported oral therapy; ID, index date of first dapagliflozin or basal insulin prescription.
In patients with recorded measurement 90-270 days after initiating dapagliflozin, the mean (SD) weight loss was 3.6 (6.4) kg in the whole sample (Table 4). After matching, a weight decline of 3.1 (5.8) kg was observed in the dapagliflozin cohort. There was no significant change in body weight after starting BOT therapy (matched analysis: weight difference dapagliflozin vs BOT: –3.0 kg; P < .01) (Table 4).
A scatterplot representing the relationship between the changes in HbA1c (%) and body weight (kg) is shown in Figure 1. Of patients receiving dapagliflozin, 57% responded simultaneously to the two-item end point of combined HbA1c and body weight reduction. Furthermore, 16% of dapagliflozin patients achieved a HbA1c reduction despite no decrease in body weight (Figure 1). Only 8% had unfavorable outcomes with both increased HbA1c and body weight after initiating dapagliflozin treatment.
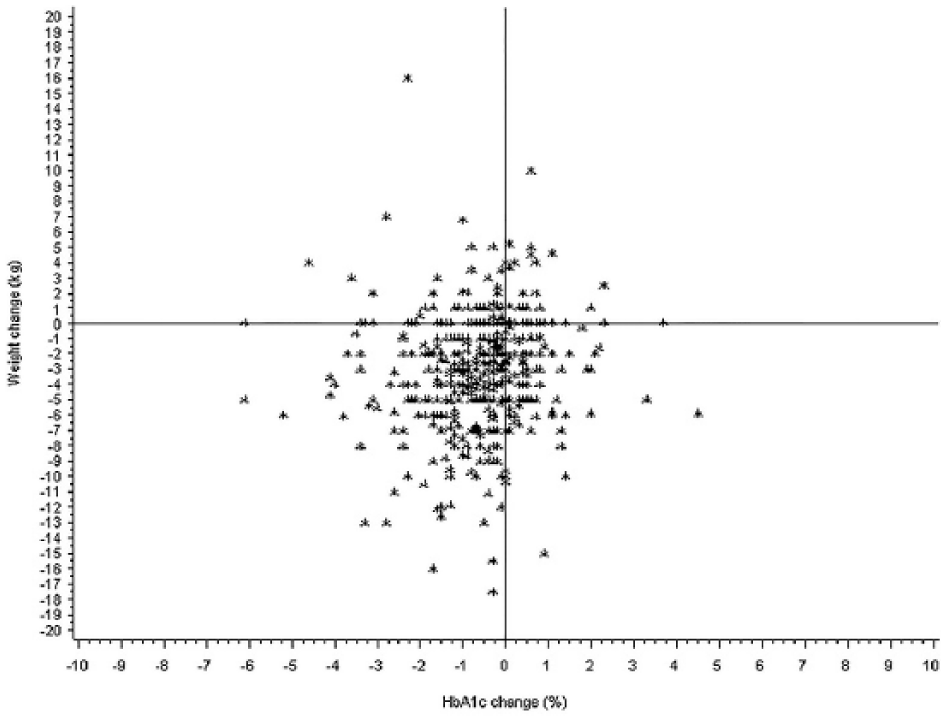
Scatterplot representing the relationship between the change of HbA1c (%) and change of body weight (kg) after 90-270 days in type 2 diabetes patients initiating dapagliflozin in primary care. n = 465 patients with known HbA1c and weight values, taken from the population prior to matching.
Figure 2 shows the scatterplot for HbA1c and body weight changes 90-270 days after start of BOT. Only 30% of the BOT cohort showed a combined decline of HbA1c and body weight. Furthermore, 40% of the BOT users had a HbA1c decrease without an accompanying decrease in body weight. Finally, 18% showed both an increase in HbA1c and body weight following initiation of BOT (Figure 2).
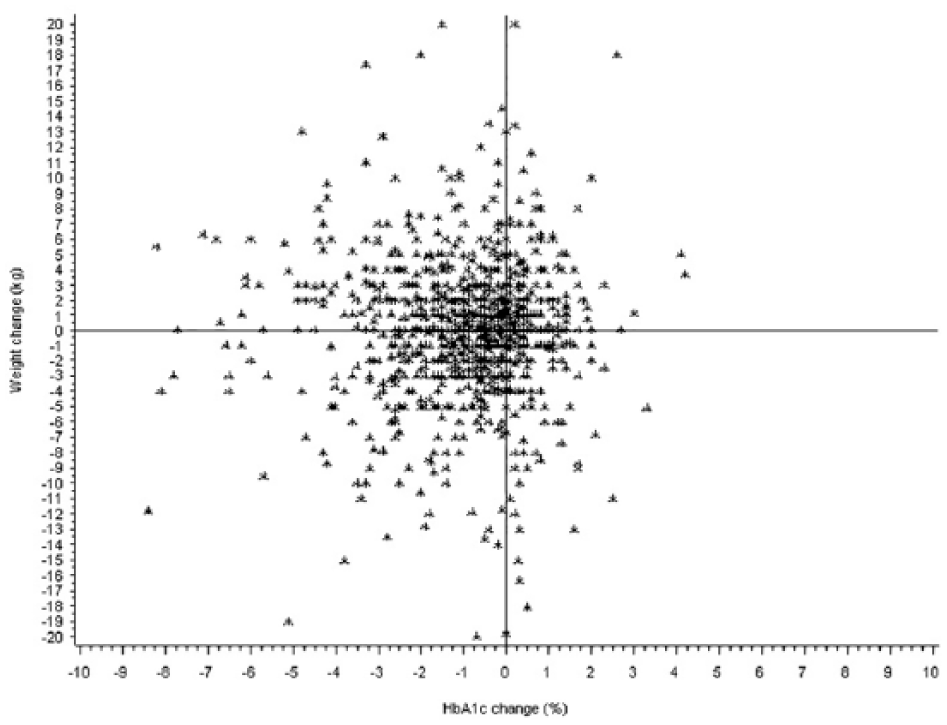
Scatterplot representing the relationship between the change of HbA1c (%) and change of weight (kg) after 90-270 days in type 2 diabetes patients initiating basal insulin supported oral therapy in primary care. n = 1045 patients with known HbA1c and weight values, taken from the population prior to matching.
Discussion
The present real-world study evaluated the short-term changes in HbA1c and body weight in type 2 diabetes patients initiating dapagliflozin predominantly after metformin alone or together with sulphonylurea or DPP-4 inhibitors compared to basal insulin supported oral treatment with a matched antidiabetic background therapy in primary care practices. Overall, dapagliflozin therapy effectively reduced HbA1c (–1.0%) and body weight (–3.1 kg) at 90-270 days. Furthermore, HbA1c reductions were larger in patients with poor glycemic control. These real-world treatment effects are comparable to results of randomized clinical trials.5-15 There was a comparable decline in HbA1c following BOT in propensity score matched patients (–1.0%), however, the high baseline body weight of the BOT cohort was maintained after 90-270 days. Therefore, initiation of dapagliflozin therapy reduced HbA1c as much as basal insulin with the additional benefit of weight reduction in type 2 diabetes patients treated in general practices.
The HbA1c and body weight reduction after initiating dapagliflozin treatment observed in our study was comparable to a recent large retrospective general practice study from the UK. 21 The slightly larger decline in HbA1c (0.89-1.16%) and in body weight (2.6-4.6 kg) in the primary care study (UK) may be due to the higher baseline values for both variables compared to our German sample. 21 It has been shown that a higher body mass index and HbA1c at baseline was related with greater weight loss and larger decline of the glycemic marker. 21 The mean HbA1c reduction after initiating BOT found in the present study was also comparable to results from previous real-life clinical practice data.22,23 In a previous analysis of the Disease Analyzer database, an average HbA1c reduction of −1.1% within 6 months after start of insulin therapy was observed. 22 In another retrospective database analysis from the UK with high mean baseline HbA1c of 9.6%, a mean change in HbA1c of −1.3% was achieved after initiation of insulin therapy, including 52% with BOT (23).
In the absence of a head-to-head comparison between SGLT2 inhibitors and basal insulins from randomized controlled trials, the present real-world study indicates that dapagliflozin is an alternative to insulin therapy in type 2 diabetes patients inadequately controlled with metformin alone or together with either sulphonylureas or DPP-4 inhibitors.
Before the era of DPP-4 and SGLT2 inhibitors, early initiation of insulin was the only possibility to adequately improve glycemic control after metformin or metformin plus sulphonylurea use. This fact together with the results of the ORIGIN trial, which showed that basal insulin treatment (insulin glargine) was safe in persons with type 2 diabetes with preexisting cardiovascular events, lead to the paradigm of early insulin use in type 2 diabetes patients. 24 What was good back then is still present today, particular in Germany. Despite the availability of newer options of combination therapies such as metformin and SGLT2 inhibitors or oral triple therapies consisting of metformin, DPP-4 inhibitor and SGLT2 inhibitor, which does have no intrinsic risk of hypoglycemia, in Germany early insulin initiation is highly used by physicians due to no budget limitation. However, high insulin use in Germany does not result in substantial improved glycemic control. 25 Quicker therapy escalation up to an oral triple combination using dapagliflozin might help to improve glycemic targets further and additionally reduce body weight and blood pressure. Clinical studies on the combination of metformin and dapagliflozin 26 as well as oral triple combinations with metformin, saxagliptin, and dapagliflozin versus placebo 14 showed sustained reductions in HbA1c over 2 years and 1 year, respectively. However, the long-term effectiveness of SGLT2 inhibitors compared to insulin therapy in real-world patients need to be investigated further.
Dapagliflozin also has a favorable adverse event profile. The main known adverse effects of SGLT2 inhibitors are an increased risk of genital fungal infections and an increase in bacterial urinary tract infections. 2 Further rarely reported adverse events are volume depletion in particularly in the elderly and diabetic ketoacidosis. 2
Type 2 diabetes treatment guidelines recommend that a patient-centered approach should guide the selection of glucose-lowering agents. 3 Efficacy, costs, adverse events, body weight effects, risk of hypoglycemia, and patient preferences should be considered. 3 SGLT2 inhibitors as a new class of oral antihyperglycemic agents show promise as a treatment option given their favorable effects on weight and low risk of hypoglycemia. In particular, patients desiring weight loss may be more reasonable candidates for treatment with dapagliflozin than with insulin. Both real-world and clinical trial data showed that initiation and intensification of insulin therapy was associated with substantial weight gain. 16 In the present study, no short-term change in body weight was observed in BOT users, but previous primary care database studies have shown weight increases of 0.9 to 4.4 kg at 0.5 to 3 years after insulin initiation. 16 Although the risk of hypoglycemia related to basal insulin treatment is low compared to other insulin regimes, the incidence of hypoglycemia increases when the HbA1c levels decline after onset of therapy. 16 Furthermore, SGLT2 inhibitors are oral formulations that may be preferred by some patients due to an easier administration compared to insulin. On the other hand, basal insulin therapy may be a better option in patients with chronic kidney disease, because based on their glucose-lowering mechanism SGLT2 inhibitors are less effective in patients with renal impairment. 16
Finally, the SGLT2 inhibitor empagliflozin appears to reduce the risk of cardiovascular events and mortality in high-risk type 2 diabetes patients within secondary prevention. 27 Various factors beyond glycemic control may contribute to these beneficial effects, including weight decline, blood pressure lowering and renal hemodynamic effects. 28 Other drugs of the same class including dapagliflozin are currently investigated for cardiovascular outcomes not only in secondary prevention such as empagliflozin, but in both primary and secondary prevention. A recent meta-analysis of cardiovascular events from 21 phase 2b and 3 dapagliflozin clinical trials showed no increased risk of major cardiovascular events, but trended toward favorable results for dapagliflozin over active comparators or placebo. 29
It is noteworthy that an increased cardiovascular risk related to insulin therapy has been recently shown in observational studies.30,31 In a recently presented propensity score matched real world study comparing 2047 new users of dapagliflozin to 4094 new users of insulin in the national Swedish Prescribed Drug Register and followed in National Patient- and Cause of Death Registers, dapagliflozin was significantly associated with lowering the risk of all-cause mortality and fatal and non-fatal cardiovascular disease by 56% and 49%, respectively. 32 Thus, the use of dapagliflozin should be considered especially in type 2 diabetes patients with increased cardiovascular risk rather than insulin.
The present study provides a picture of prescriptions and diagnoses in primary care. Because it used primary care records, a number of limitations should be mentioned. First, no valid information regarding onset of diabetes was provided. Furthermore, assessment of co-morbidities relied on ICD codes filled in by primary care physicians only. Finally, measurements of HbA1c and body mass index values were not standardized and were not available in all patients initiating dapagliflozin or BOT. The strength of the study is the large nationwide database and the unbiased assessment of prescriptions and outcomes. The relatively small sample sizes could limit the statistical power. However, this potential drawback is outweighed by the control of confounders due to the propensity score matching.
In conclusion, this first observational study in primary care patients with type 2 diabetes found that HbA1c reduction in new dapagliflozin and basal insulin users (BOT) was comparable with the additional benefit of weight reduction in patients treated with the SGLT2 inhibitor.
Footnotes
Acknowledgements
We thank Dr Orm Proske and Katja Rohwedder for their help in proofreading this article.
Abbreviations
ADA, American Diabetes Association; ATC, Anatomical Therapeutic Chemical (ATC) Classification System; BMI, body mass index; CHD, coronary heart disease; DPP4i, dipeptidyl peptidase-4 inhibitors; EASD, European Association for the Study of Diabetes; GLP-1, glucagon-like peptide-1; GP, general practitioner; HbA1c, hemoglobin A1c; ICD, International Classification of Diseases; PAD, peripheral arterial disease; SD, standard deviation; SGLT2, sodium dependent glucose transporter; T2DM, type 2 diabetes mellitus.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: KK is an employee of IMS Health. RR, SB, and MFS are employees of AstraZeneca GmbH, Germany. The authors report no other conflicts of interest in this work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: AstraZeneca GmbH provided medical writing support for this study.
