Abstract
Background:
The new Contour®Plus ONE blood glucose monitoring system (BGMS) features an easy-to-use, wireless-enabled blood glucose meter that links to a smart mobile device via Bluetooth® connectivity and can sync with the Contour™ Diabetes app on a smartphone or tablet.
Methods:
The accuracy of the new BGMS was assessed in 2 studies according to ISO 15197:2013 criteria. In Study 1 (laboratory study), fingertip capillary blood samples from 100 subjects were tested in duplicate using 3 test strip lots. In Study 2 (clinical study), 134 subjects with type 1 or type 2 diabetes enrolled at 2 clinical sites. BGMS results and YSI analyzer (YSI) reference results were compared for fingertip blood obtained by untrained subjects’ self-testing and for study staff–obtained fingertip, subject palm, and venous results.
Results:
In Study 1, 99.0% (594/600) of combined results for all 3 test strip lots fulfilled ISO 15197:2013 Section 6.3 accuracy criteria. In Study 2, 99.2% (133/134) of subject-obtained capillary fingertip results, 99.2% (133/134) of study staff–obtained fingertip results, 99.2% (125/126) of subject-obtained palm results, and 100% (132/132) of study staff–obtained venous results met ISO 15197:2013 Section 8 accuracy criteria. Moreover, 95.5% (128/134) of subject-obtained fingertip self-test results were within ±10 mg/dl (±0.6 mmol/L) or ±10% of the YSI reference result. Questionnaire results showed that most subjects found the BGMS easy to use.
Conclusions:
The BGMS exceeded ISO 15197:2013 accuracy criteria both in the laboratory and in a clinical setting when used by untrained subjects with diabetes.
Keywords
Self-monitoring of blood glucose (SMBG) is described as an integral component of effective diabetes therapy by the American Diabetes Association (ADA) 1 and the American Association of Clinical Endocrinologists/American College of Endocrinology (AACE/ACE). 2 This technique allows people with diabetes to evaluate their own responses to prescribed therapy and to be aware of whether they are achieving their glycemic targets. 1 A recent consensus statement from AACE/ACE supports the concept of “meaningful monitoring,” which seeks to individualize SMBG and empower people with diabetes to manage their glucose levels and minimize the risk of hypoglycemia. 2 Up-to-date and accurate SMBG results can be useful for managing nutrition therapy and physical activity, preventing hypoglycemia, and adjusting medications.1,2 SMBG has been shown to be associated with improved outcomes in people with diabetes, including lowered glycated hemoglobin (HbA1c) values.1-5
Mobile apps and other emerging technologies are providing personalized, real-time, data-driven support to help people make decisions about their diabetes management.6,7 These technologies, including smartphone apps, patient monitoring devices, tablets, personal digital assistants, and other wireless devices, are helping to create a new digital universe of tools for more precise and consistent management of diabetes for patients and their care teams.6,7
The Contour®Plus ONE (Ascensia Diabetes Care, Parsippany, NJ, USA) is a new blood glucose monitoring system (BGMS) that features an easy-to-use, wireless-enabled blood glucose meter that links to a smart mobile device via Bluetooth® connectivity. The new BGMS is also designed to sync with the Contour™ Diabetes app, which is available on smartphone or tablet and provides multiple functions, including displays of trends and test results as compared with targets; testing reminders; graphs of test results over a day or a longer period of time; sharing of reports; and review of fasting, preprandial, postprandial, and overall results on a daily graph. The BGMS utilizes currently available Contour® Plus test strips (Ascensia Diabetes Care), which contain the flavin adenine dinucleotide-glucose dehydrogenase (FAD-GDH) enzyme and a proprietary electron mediator.
People with diabetes use blood glucose test results obtained from SMBG to make critical decisions about their treatment; it is therefore imperative that results are accurate to prevent nutritional and drug-dosing errors. 8 Guidelines provided by the International Organization for Standardization (ISO) are used to assess the accuracy of a BGMS. 9 The accuracy of the new BGMS was assessed in the laboratory and in clinical settings according to ISO 15197:2013 guidelines. In the first study, the analytical accuracy of the BGMS was examined in the laboratory based on ISO 15197:2013 Section 6.3 criteria. In the second study, the performance and ease of use of the new BGMS was assessed in a clinical setting by persons with diabetes according to ISO 15197:2013 Section 8 guidelines. The primary objective of the clinical study was to evaluate the performance of the system in the hands of subjects with diabetes using fingertip blood, based on ISO 15197:2013 Section 8 accuracy criteria. Additional objectives were to assess study staff–obtained fingertip, subject-obtained palm, and study staff–obtained venous results, and to obtain feedback from subjects on the ease of use of the BGMS and the User Guide instructions.
Methods
Laboratory Study: Analytical Accuracy (ISO 15197:2013 Section 6.3)
To evaluate accuracy, study staff–obtained fingertip capillary blood samples from 100 subjects were tested using 3 test strip lots; each sample was tested in duplicate (N = 600). Two sets of 6 blood glucose meters were alternated between subjects. After every 10 subjects, a new vial of test strips was used; a total of 11 vials per lot were included in the study. The distribution of blood glucose concentrations of the 100 blood samples conformed to the ISO 15197:2013 requirement. Most samples (n = 88) were tested fresh from the finger without modification. However, contrived samples at the low and high ends of the distribution were obtained by allowing 8 samples to glycolyze to a lower blood glucose concentration (<50 mg/dl [<2.8 mmol/L], n = 4; 50-80 mg/dl [2.8-4.4 mmol/L], n = 4) and supplementing 4 specimens with a concentrated glucose solution (20%) to increase the glucose level (>300-400 mg/dl [>16.7-22.2 mmol/L], n = 3; >400 mg/dl [>22.2 mmol/L], n = 1). To obtain reference values for comparison, capillary blood from the same fingertip lancing site was collected into a microcuvette and centrifuged to obtain plasma. Plasma samples were tested in duplicate on a YSI 2300 STAT Plus™ laboratory glucose analyzer (YSI; YSI Life Sciences, Inc., Yellow Springs, OH, USA). The fingertip blood was also collected into 2 microhematocrit capillary tubes (Drummond Scientific Company, Broomall, PA, USA) and centrifuged with a StatSpin I microcentrifuge (StatSpin, Inc., Norwood, MA, USA) to measure hematocrit. All results were obtained at 23 ± 5ºC.
The BGMS was evaluated for accuracy based on ISO 15197:2013 Section 6.3 criteria 9 (ie, ≥95% of results shall fall within ±15 mg/dl [±0.8 mmol/L] of the mean YSI reference result for samples with blood glucose concentrations <100 mg/dl [<5.6 mmol/L] and within ±15% for samples with blood glucose concentrations ≥100 mg/dl [≥5.6 mmol/L]). Regression analysis was performed to compare BGMS results with YSI reference results, and the Parkes-Consensus Error Grid analysis 10 was also used to evaluate the clinical accuracy of the BGMS. Finally, radar plots were constructed to compare BGMS results with YSI reference results.
Clinical Trial: User Performance Evaluation (ISO 15197:2013 Section 8)
This clinical trial was conducted at 2 sites and included both male and female subjects with type 1 or type 2 diabetes who were aged ≥18 years and had not used this BGMS previously. Before participating in capillary blood glucose testing, subjects stated that for ≥2 hours they had fasted, had not taken bolus insulin or oral diabetes medications, and had not vigorously exercised. Each subject’s participation consisted of 1 study visit, except for 1 subject who was enrolled on one day and returned to the site the next day for study testing due to an adverse event of hypoglycemia on the day of enrollment. The protocol, informed consent forms, and all study documents were approved by an Institutional Review Board prior to study start, and each subject completed the informed consent process before participating in the study.
During the single study visit, prior to blood glucose testing, subjects were provided with the BGMS and the instructional materials (User Guide and Quick Reference Guide) and were given time to review the materials to learn how to use the system; no additional training was provided. Each subject performed a fingertip test for blood glucose on the BGMS. Within 5 minutes of the subject fingertip test, site staff collected blood from a separate fingertip lancing for the capillary YSI reference assay. Subjects then performed a palm alternate site test for blood glucose on the BGMS. Immediately after the palm test, site staff performed a blood glucose test with the subject’s fingertip blood. Finally, site staff performed a venipuncture on each subject, and that blood was used to complete 1 meter test, a hematocrit measurement, and a venous YSI reference assay. Three test strip lots were used in the study, with each subject randomized to 1 lot. The test strip lots were provided to the investigator by the study sponsor. All BGMS results were compared with YSI reference results.
Accuracy of the BGMS was evaluated based on ISO 15197:2013 Section 8 accuracy criteria 9 (ie, within ±15 mg/dl [±0.8 mmol/L] or ±15% of the reference result for samples with blood glucose concentrations <100 mg/dl [<5.6 mmol/L] and ≥100 mg/dl [≥5.6 mmol/L], respectively). Regression analyses were performed to compare BGMS results with YSI reference results. Parkes-Consensus Error Grid analyses 10 were performed to evaluate results based on the clinical significance of differences between BGMS results and YSI reference values, and radar plots were constructed to compare BGMS results with YSI reference results.
Subjects also completed a questionnaire on the ease of use of the BGMS and the User Guide instructions; possible responses ranged between 1 (strongly disagree) and 5 (strongly agree). Subjects completed a second questionnaire on diabetes management behaviors; possible responses were 0 (no answer) and 1 (strongly disagree) to 5 (strongly agree). The questionnaires are provided in the Supplemental Material. Adverse events were monitored throughout the trial.
Results
Laboratory Study: Analytical Accuracy (ISO 15197:2013 Section 6.3)
In total, 600 BGMS results were obtained by testing 100 fingertip capillary blood samples in duplicate using 3 lots of test strips. The range of plasma blood glucose concentrations was 37 mg/dl (2.1 mmol/L) to 526 mg/dl (29.2 mmol/L). The hematocrit range was 34.5% to 56.0%, which was within the approved hematocrit range for this product.
Analysis of BGMS results based on ISO 15197:2013 Section 6.3 accuracy criteria showed that 99.0% (594/600) of combined results for all 3 test strip lots were within ±15 mg/dl (±0.8 mmol/L) or ±15% of the YSI reference results for samples with blood glucose concentrations <100 mg/dl (<5.6 mmol/L) or ≥100 mg/dl (≥5.6 mmol/L), respectively (Table 1). Furthermore, 96.3% (578/600) of combined results for all 3 test strip lots were within ±10 mg/dl (±0.6 mmol/L) or ±10% of the YSI reference results for samples with blood glucose concentrations <100 mg/dl (<5.6 mmol/L) or ≥100 mg/dl (≥5.6 mmol/L), respectively (Table 1).
Summary of BGMS Results From the Laboratory Study (ISO 15197:2013 Section 6.3).
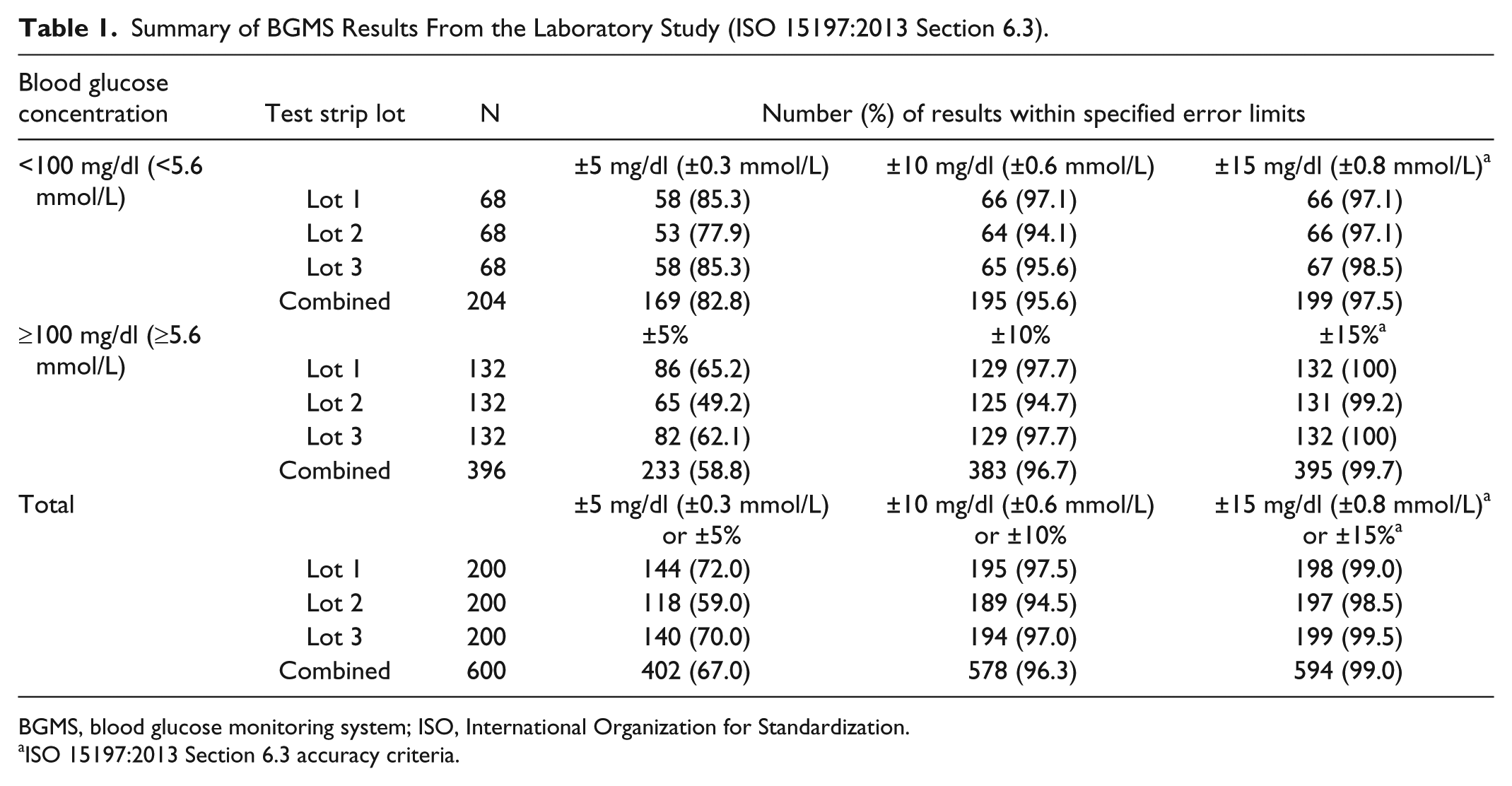
BGMS, blood glucose monitoring system; ISO, International Organization for Standardization.
ISO 15197:2013 Section 6.3 accuracy criteria.
An ad hoc analysis was conducted to determine the smallest error range that contained ≥95% of meter inaccuracies (ie, differences between meter results and YSI reference results), with the determination being that 95% of meter results had inaccuracies that were within 9.4 mg/dl (0.5 mmol/L) or 9.4% of the YSI reference result. A plot of the differences of BGMS results from YSI reference results is shown in Figure 1A. Regression analysis demonstrated a high degree of agreement between BGMS results and YSI reference results overall (y = 0.97x – 1.4 mg/dl [−0.1 mmol/L]; R2 = 0.9911) and by individual lot (Lot 1: y = 0.98x – 2.2 mg/dl [−0.1 mmol/L]; R2 = 0.9907; Lot 2: y = 0.96x – 0.9 mg/dl [−0.1 mmol/L]; R2 = 0.9908; Lot 3: y = 0.97x – 1.1 mg/dl [−0.1 mmol/L]; R2 = 0.9921). By Parkes-Consensus Error Grid analysis (Figure 1B), 99.8% (599/600) of all results were within Zone A, and the remaining 0.2% (1/600) were within Zone B, satisfying the ISO 15197:2013 Section 6.3 accuracy criteria (ie, ≥99% of results shall fall within the combination of Zones A and B).
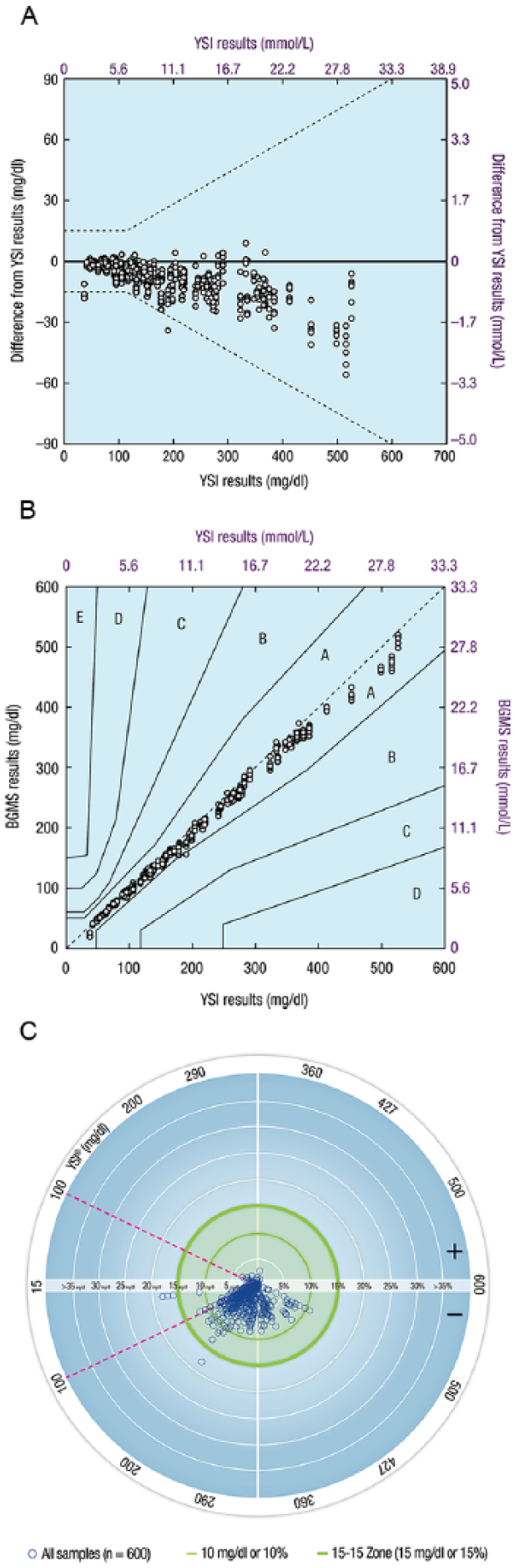
Graphical representations of BGMS results from the laboratory study (ISO 15197:2013 Section 6.3). (A) Differences of BGMS results from YSI reference results.a (B) Parkes-Consensus Error Grid analysis of BGMS results compared with YSI reference results. (C) Radar plot of BGMS results compared with YSI reference results.b
A radar plot comparing BGMS results to YSI reference results is shown in Figure 1C. A radar plot is a new way to plot the differences between BGMS values and reference instrument values. The outer green circle (bolder line) represents ±15 mg/dl (±0.8 mmol/L) or ±15% error for samples with YSI blood glucose concentrations <100 mg/dl (<5.6 mmol/L) or ≥100 mg/dl (≥5.6 mmol/L), respectively. Points within this circle satisfy ISO 15197:2013 Section 6.3 accuracy criteria.
Clinical Trial: User Performance Evaluation (ISO 15197:2013 Section 8)
Subjects
A total of 134 subjects enrolled in the clinical trial and all subjects completed the trial. Mean (range) age was 54 (18-77) years; 47% of subjects had type 1 diabetes, 52% had type 2 diabetes, and 1% had diabetes of unknown type (Table 2). The ratio of female to male subjects was balanced (52% female; 48% male). Most subjects (75%) reported that they self-test their blood glucose ≥2 times daily.
Subject Demographic and Baseline Characteristics (Clinical Study).

SMBG, self-monitoring of blood glucose.
Percentages may not total 100% because of rounding.
Subject-obtained capillary fingertip results were analyzed from 134 subjects for self-testing. Eight subjects did not have evaluable subject-obtained palm test results because the YSI blood glucose value was <70 mg/dL (n = 4) or the palm test was not performed (n = 4). Two subjects did not have evaluable study staff–obtained venous blood tests due to unsuccessful venipuncture.
The blood glucose concentrations of the subjects’ blood samples, as measured by YSI, ranged from 44.3 mg/dl (2.5 mmol/L) to 474.5 mg/dl (26.3 mmol/L) for capillary blood and 42.9 mg/dl (2.4 mmol/L) to 463.0 mg/dl (25.7 mmol/L) for venous blood. Hematocrit measurements were performed in duplicate for each subject; average hematocrit values ranged from 33% to 56%, with a mean of 43%.
Accuracy
Evaluation of subject-obtained capillary fingertip results showed that 99.2% (133/134) of results met the ISO 15197:2013 Section 8 accuracy criteria (Table 3). In addition, 95.5% (128/134) of subject-obtained capillary fingertip results were within ±10 mg/dl (±0.6 mmol/L) or ±10% of the YSI reference results. For other endpoints, 99.2% (133/134) of study staff–obtained fingertip results, 99.2% (125/126) of subject-obtained palm results, and 100% (132/132) of study staff–obtained venous results met ISO 15197:2013 Section 8 accuracy criteria (Table 3).
Summary of BGMS Accuracy in the Clinical Study (ISO 15197:2013 Section 8).
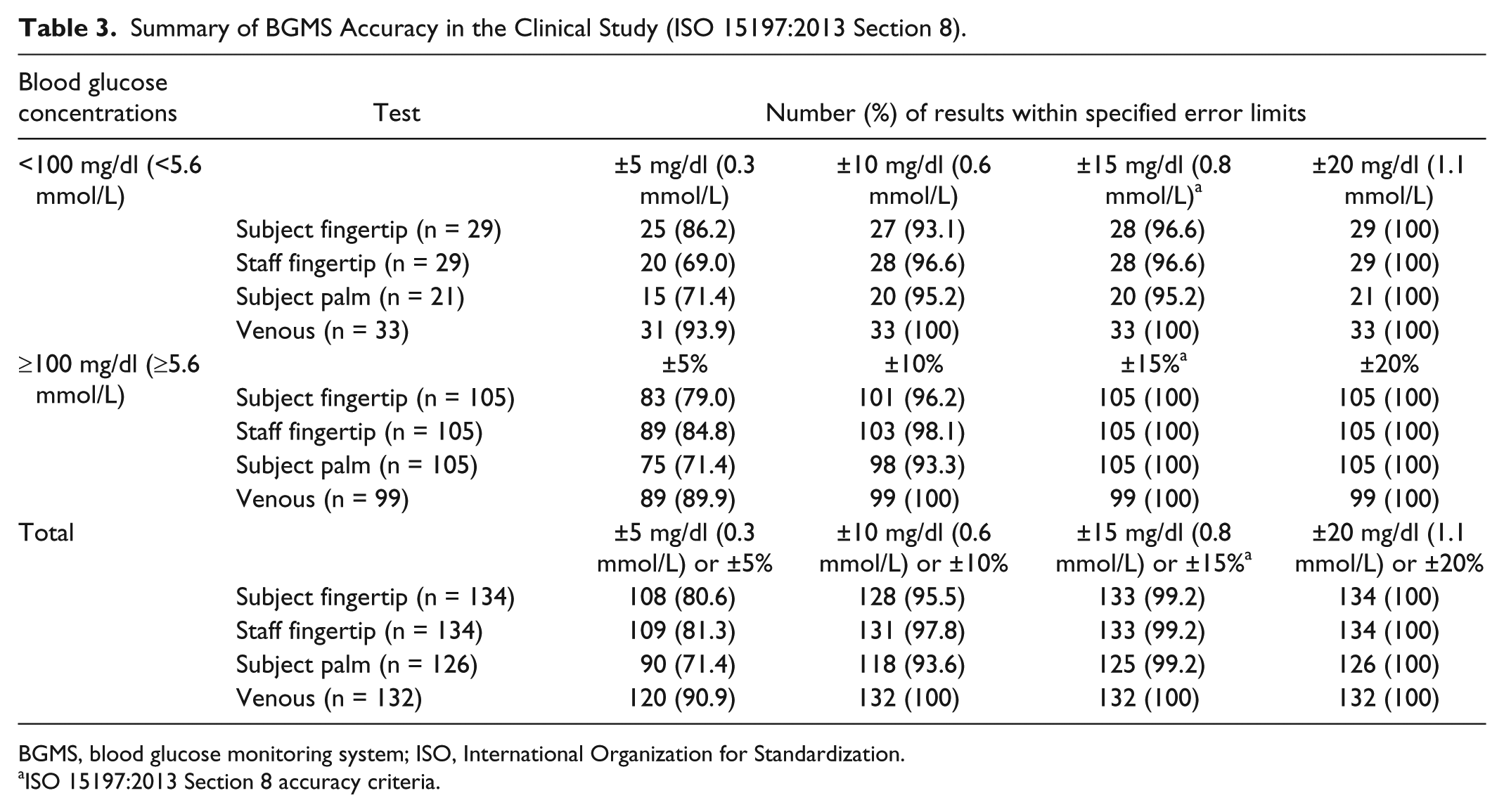
BGMS, blood glucose monitoring system; ISO, International Organization for Standardization.
ISO 15197:2013 Section 8 accuracy criteria.
An ad hoc analysis was conducted to determine the smallest error range that contained ≥95% of meter inaccuracies (ie, differences between meter results and YSI reference results). Overall, 95% of results had inaccuracies that were within 8.5 mg/dl (0.5 mmol/L) or 8.5% of the YSI reference results for subject-obtained fingertip blood tests, within 8.6 mg/dl (0.5 mmol/L) or 8.6% of the YSI reference results for study staff–obtained fingertip blood tests, within 10.4 mg/dl (0.6 mmol/L) or 10.4% of the YSI reference results for subject-obtained palm blood tests, and within 6.3 mg/dl (0.3 mmol/L) or 6.3% of the YSI reference results for study staff–obtained venous blood tests.
A plot of the differences of subject-obtained capillary fingertip results from YSI reference results is shown in Figure 2A. Regression analysis demonstrated a strong correlation between BGMS results and YSI reference results. The adjusted R2 value was 0.9848 for subject-obtained capillary fingertip results. The adjusted R2 values for study staff–obtained fingertip, subject-obtained palm, and study staff–obtained venous results were 0.9864, 0.9697, and 0.9940, respectively.
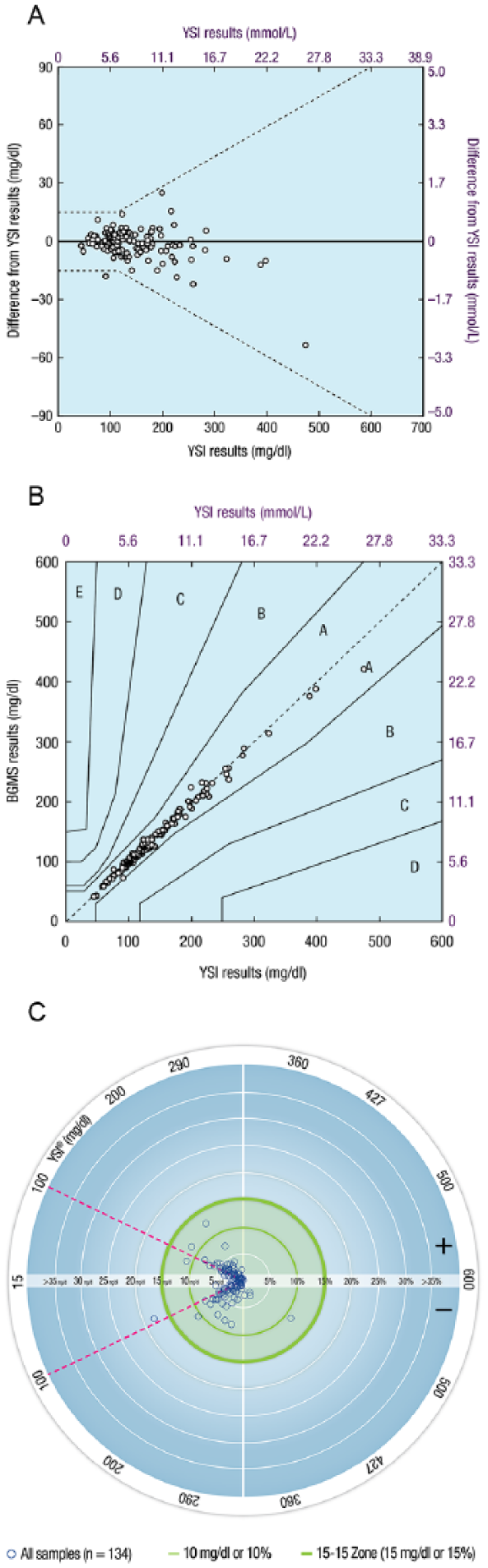
Graphical representations of BGMS results from the clinical study (ISO 15197:2013 Section 8). (A) Differences of subject-obtained capillary fingertip results from YSI reference results.a (B) Parkes-Consensus Error Grid analysis of subject-obtained capillary fingertip results compared with YSI reference results. (C) Radar plot of subject-obtained capillary fingertip results compared with YSI reference results.b
Parkes-Consensus Error Grid analysis demonstrated that 100% (134/134) of subject-obtained capillary fingertip results were within Zone A (Figure 2B). All results (100%) were also within Zone A for study staff–obtained fingertip (134/134), subject-obtained palm (126/126), and study staff–obtained venous (132/132) testing. A radar plot comparing subject-obtained capillary fingertip results to YSI reference results is shown in Figure 2C.
Subject questionnaires
Ease-of-use questionnaire responses demonstrated that most subjects “strongly agree,” “agree,” or are “neutral” that it is easy to do a fingerstick blood test with this meter (99.2%), the meter display is easy to see and read (98.5%), it is easy to understand the test results (100%), the instructions are easy to understand (96.3%), the instructions clearly explain how to run a test (97.8%), and the instructions clearly explain what to do if an error message is displayed by the meter (99.2%).
Diabetes management questionnaire responses demonstrated that the majority of subjects responded “strongly agree,” “agree,” or “neutral” that accuracy is important: (1) to help with their ability to manage their diabetes (100%), (2) to help with understanding how food or exercise affects low blood sugars (100%), (3) to help with preventing low blood sugars (100%), and (4) to help with using their results to gain better control of their diabetes (100%). In addition, 80.0% of subjects responded “strongly agree,” “agree,” or “neutral” that they use their current meters because their insurance companies cover the strips, and 86.5% of subjects responded “strongly agree,” “agree,” or “neutral” that they preferred the meter used in this study to their regular meters.
Safety
There were 4 mild, anticipated, non–device-related adverse events, all classified as hypoglycemia. All adverse events resolved prior to the subjects leaving the testing site.
Discussion
SMBG has been shown to lead to improved outcomes for persons with diabetes; thus, it is an important part of the self-management of diabetes.1,3-5 Mobile apps can also help people manage their diabetes by providing useful information to guide their diabetes management decisions and by providing valuable data to their health care providers.6,7 Treatment modification recommendations have been shown to be more effective in improving glycemic control when supplied by physicians with access to patient-provided SMBG data compared with recommendations initiated by physicians without SMBG data. 4 Various clinical benefits of using mobile apps for diabetes, including reductions in HbA1c levels, have been demonstrated in some studies; however, additional research is needed to provide further support for these benefits.11-14
Both the laboratory study and the clinical study described previously demonstrated the accuracy of the new BGMS that has been developed for use with currently available test strips that contain the FAD-GDH enzyme and a proprietary mediator. The BGMS was designed to wirelessly interact with the Contour™ Diabetes app on a smartphone or tablet.
The BGMS exceeded ISO 15197:2013 accuracy criteria in the laboratory study (Section 6.3) and in the clinical study when used by untrained subjects (Section 8). In the laboratory study, 99% of results with the BGMS met ISO 15197:2013 Section 6.3 accuracy criteria, and 96.3% of results were within 10 mg/dl or 10% of the YSI reference values. In the clinical study, ISO 15197:2013 Section 8 accuracy criteria were met by 99.2% of capillary fingertip, 99.2% of palm, and 100% of venous blood samples. Accuracy was similar whether the system was used by subjects who had never used this BGMS previously or by study staff.
In the clinical study, the BGMS also demonstrated ease of use in the hands of untrained subjects with diabetes. The majority of subjects rated the system as easy to use with respect to performing a fingertip blood test. The majority of subjects also felt that the meter display is easy to see and read and that the test results and user instructions are easy to understand. All subjects agreed on the importance of BGMS accuracy to help with managing their diabetes, understanding how food or exercise affects low blood sugars, preventing low blood sugars, and using their results to gain better control of their diabetes.
Conclusions
In conclusion, the analytical and clinical accuracy of the new BGMS as well as its ease of use in persons with diabetes were demonstrated by the results of these studies.
Footnotes
Abbreviations
AACE/ACE, American Association of Clinical Endocrinologists/American College of Endocrinology; ADA, American Diabetes Association; BGMS, blood glucose monitoring system; FAD-GDH, flavin adenine dinucleotide-glucose dehydrogenase; HbA1c, glycated hemoglobin; ISO, International Organization for Standardization; SMBG, self-monitoring of blood glucose; YSI, YSI analyzer.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TSB has received research support from Abbott, ACON, Ascensia Diabetes Care, Bristol-Myers Squibb, Dexcom, GlaxoSmithKline, Halozyme, Insulet, Janssen, Lexicon, LifeScan, Lilly, Medtronic, Merck, Novo Nordisk, Orexigen, and Sanofi; has received consulting honoraria from Ascensia Diabetes Care, AstraZeneca, Becton Dickinson, Lilly, Medtronic, Novo Nordisk, and Sanofi; and has received speaking honoraria from Abbott, Insulet, Novo Nordisk, and Sanofi. JFW, SP, MEWW, BH, and RM are full-time employees of Ascensia Diabetes Care. MC has nothing to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: These studies were supported by Bayer HealthCare, the predecessor-in-interest of Ascensia Diabetes Care, Parsippany, NJ, USA. Medical writing assistance was provided by Allison Michaelis, PhD, of MedErgy, and was funded in part by Ascensia Diabetes Care and in part by Bayer HealthCare as Ascensia’s predecessor-in-interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
