Abstract
Background:
To assess the occurrence of white coat adherence, defined as an increase in adherence to treatment regimens prior to a study appointment, in adolescents with type 1 diabetes (T1D) using insulin pumps and participating in a randomized adherence intervention trial.
Methods:
Blood glucose monitoring (BGM) readings, carbohydrate inputs, and insulin boluses delivered were downloaded from the insulin pumps of adolescents, aged 10-18 years, at 3 consecutive T1D study visits. Linear mixed models were used to analyze patterns of BGM, carbohydrate inputs, and insulin boluses delivered in patients who had 40 consecutive days of data stored in their insulin pumps prior to the study visit.
Results:
Stratified linear mixed models revealed that adolescents randomized to the Tailored Feedback Intervention group increased their blood glucose monitoring (P < .01), carbohydrate inputs (P < .0001), and insulin bolusing (P < .0001) prior to study appointments. In contrast, white coat adherence did not occur in adolescents randomized to the Treatment as Usual group (Ps > .42).
Conclusions:
White coat adherence may occur in adolescents participating in clinical trials. Meter and insulin pump data downloads representing the 1- to 2-week period prior to a study visit are likely to overestimate actual adherence during the time frame between study visits.
Keywords
White coat adherence, the tendency to increase adherence behaviors prior to an appointment with a medical provider, has been well-documented in adults with epilepsy, HIV, and dermatology conditions.1-3 Pediatric studies have also shown that white coat adherence occurs in children <12 years of age.3-6 Specifically, children with type 1 diabetes (T1D) and their parents increased the frequency of their blood glucose monitoring (BGM), carbohydrate inputs, and insulin bolusing prior to their visit with their medical provider; however, white coat adherence did not occur for adolescents with T1D. 6 This overall pattern of increased medication use in the case of insulin, and other aspects of increased T1D care (eg, BGM and carbohydrate inputs) that precedes the T1D appointment raises interesting questions about the reinforcing role that the appointment has on adherence, especially because better T1D adherence is associated with better glycemic control. 7 Moreover, if participants engage in white coat adherence during observational studies, 6 then additional questions are raised as to whether pediatric participants engage in it during clinical trials. Although white coat adherence does not appear to be a concern in the usual care of adolescents with T1D, we do not know if it may occur when adolescents are asked to join clinical trials. Consequently, the primary aim of the current study was to examine whether randomizing adolescents with T1D who use insulin pumps to an adherence intervention would increase their frequency of BGM, carbohydrate inputs, and insulin boluses prior to their study visit, which coincided with their routine T1D clinic appointment.
Methods
Participants (N = 75) were part of a larger randomized clinical trial aimed at providing intervention for 1 year to improve insulin pump adherence (BGM, carbohydrate inputs, insulin bolusing). Participants were eligible for the larger study if they were between the ages of 10 and 18 years, had a T1D diagnosis for ≥1 year, and used any model of a Medtronic insulin pump ≥6. Participants were recruited from 2 independent private practice pediatric endocrinology offices from 2012 to 2015. In addition, there were no exclusion criteria imposed on eligible participants regarding adherence or glycemic control status. For the purposes of the analyses in the current study, participants were included if their insulin pump contained ≥28 days of data prior to a study visit. This study was approved by the Florida State University Institutional Review Board and data collection is ongoing.
Study Design
This study used downloaded Medtronic insulin pump data from the first 3 study visits (ie, study visit 1/baseline, study visit 2, study visit 3) of a larger ongoing clinical trial in which all study visits coincided with the participant’s routine T1D clinic appointments. Participants provided consent and assent at study visit 1. At that time, the participant’s Medtronic insulin pump was downloaded which included no more than 90 days of data prior to study enrollment. These data served as a baseline measure of the participant’s insulin pump adherence behaviors. Participants were then randomized to either Tailored Feedback (intervention) or Treatment as Usual (control).
Adolescents randomized to Tailored Feedback received 2 standardized (ie, the same content was provided although the data differed for each participant), user-friendly reports (designed specifically for this study), and individualized recommendations based on their Medtronic insulin pump data downloaded at study visit 1, and then again 3 months later at study visit 2. The Tailored Feedback intervention was delivered either by a doctoral-level clinical psychologist or an advanced clinical psychology doctoral student. Participants in the Treatment as Usual group received standard care, which included feedback and recommendations from their T1D provider. Because downloaded pump data represents adherence behaviors prior to a study visit, downloaded pump data at study visit 1 served as a baseline indicator of adherence prior to randomization and downloaded pump data from study visits 2 and 3 served as indicators of the impact of Tailored Feedback versus Treatment as Usual.
Goals for providing the Tailored Feedback report included increasing the frequency of BGM, carbohydrate inputs, and insulin bolusing and increasing the frequency of correcting high (151-249 mg/dL) or very high (≥250 mg/dL) blood glucoses. As part of the Tailored Feedback intervention, participants were provided with summary statistics about each insulin pump adherence behavior. For example, the percentage of time a participant bolused after a very high blood glucose was provided in writing. All summary statistics were reviewed with the participant and written recommendations were provided. Participants and their parents received a copy of the Tailored Feedback report as part of study visits 1 and 2.
Insulin Pump Downloads
The Medtronic insulin pump typically stores 2-3 months of data including BGM readings (either wirelessly transmitted from compatible monitors or manually entered), carbohydrate inputs (as estimated by the patient and/or parent and manually entered), and insulin bolus doses delivered. Data were downloaded from participants’ Medtronic insulin pumps during 3 study visits that occurred at the time of the participant’s routine T1D clinic visit, scheduled approximately 3 months apart.
Glycemic Control
Hemoglobin A1C (A1C) representing the average glucose level during the past 2.5 to 3 months was obtained at each study visit using a Siemens Healthcare Diagnostics DCA Vantage (reference range 4.2-6.5%, Malvern, PA, USA ), which is National Glycohemoglobin Standardization Program certified as having documented traceability to the Diabetes Control and Complications Trial Reference Method.
Statistical Analyses
Since the focus of the current study was to examine white coat adherence, downloaded Medtronic insulin pump data for the 40 days prior to a study visit were selected for analysis because 40 days represent the approximate midpoint between study visits. Each participant’s data file was cleaned to identify and correct technological errors (eg, duplication of data, data included for dates from the previous study visit). 1 Descriptive statistics including means, standard deviations, and ranges were conducted for participant characteristics (eg, T1D duration, sex), A1C, and insulin pump variables (BGM, carbohydrate inputs, and boluses delivered).
Linear mixed models were used to examine white coat adherence. Linear mixed models have several advantages over other approaches including (1) ability to model individual change across time, (2) different number of observations per participant is allowable, and (3) time can be continuous rather than a fixed set of points (eg, no need to segment the data into blocks of time such as 2 weeks, 1 month, 2 months). Importantly, linear mixed models adjust within participant dependence in designs with repeated measures in which data from the same participant is intraindividually related. SAS Version 9.2 (Cary, NC, USA) was used for all analyses.
Separate linear mixed models using the 40 consecutive days of data prior to 2 T1D study visits (ie, study visits 2 and 3) were used to test the effect of day (ie, white coat adherence) on the frequency of 3 insulin pump adherence behaviors: BGM, carbohydrate inputs, and insulin bolusing.
Variables included in each model were (1) participant characteristics including age, T1D duration, sex, and duration of insulin pump use; (2) baseline adherence frequency, obtained at study visit 1, reflecting the average number of BGM readings per day, carbohydrate inputs per day, and insulin boluses per day delivered prior to study entry; (3) visit (to test for the main effect of time across study visits on insulin pump adherence behaviors); (4) day, reflecting the effect of the number of days prior to a study visit (ranging from 40 days prior to 1 day prior to a study visit) on insulin pump adherence behaviors; (5) treatment group (ie, Tailored Feedback or Treatment as Usual); and (6) day × treatment group interaction. Models were trimmed by deleting nonsignificant variables. Of particular interest was whether day—reflecting white coat adherence—occurred in this adolescent sample of insulin pump users and whether white coat adherence occurred more often in those randomized to Tailored Feedback than in those in the Treatment as Usual group.
Results
Patient Characteristics
At study visit 1, participants ranged in age from 10 to 18 years (mean = 14.8 ± 2.3 years; 53% female; 86.7% Caucasian; 35.1% public insurance) with a T1D duration of 1.1-14.1 years (mean = 6.65 ± 3.5 years) and insulin pump use duration of 0.60-13.0 years (mean = 4.4 ± 3.2 years). Average A1C at study visit 1 was 8.8% (206 mmol/mol) ± 1.5 (range = 6.6-14%). Participant characteristics in this study were representative of each private practice site and the sample as a whole.
Table 1 provides descriptive statistics for data downloaded from insulin pumps that preceded each of the T1D study visits. The following were calculated for each child: mean blood glucose level (mg/dL), mean number of BGM readings per day, mean carbohydrates consumed (grams) on each occasion, mean number of carbohydrate inputs per day, mean insulin delivered (units) on each occasion, and mean number of insulin boluses delivered per day. The 90 days of data before study visit 1 and all available data in the interval between visit 1 and visit 2, and then visit 2 and visit 3 were used to calculate these means.
Insulin Pump Data Downloaded at 3 Separate Study Visits.
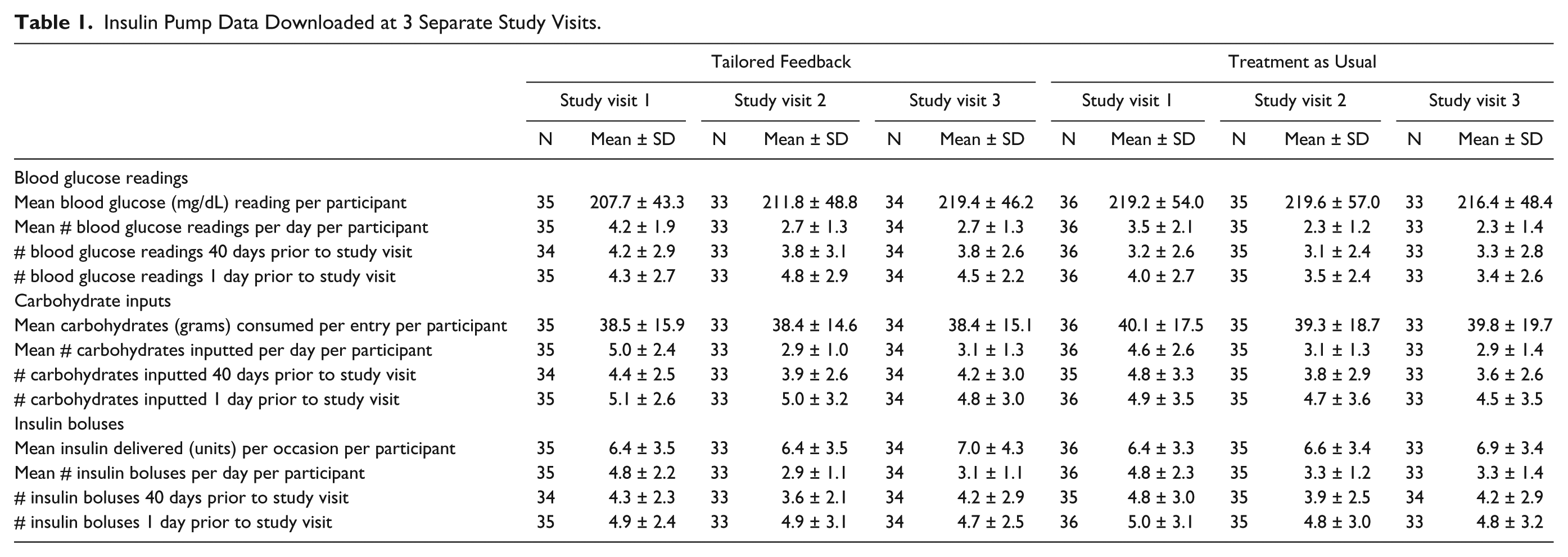
Also depicted in Table 1 is the white coat adherence effect. The average number of BGM readings, carbohydrate inputs, and insulin boluses delivered at 40 days and 1 day prior to the T1D study appointment are provided as well as standard deviations and ranges. These days were chosen as representative days with day 40 as the approximate midpoint between T1D study appointments, and day 1 the day immediately before the appointment. The average number of BGM readings, carbohydrate inputs, and insulin boluses delivered increased from 40 days to 1 day prior to the T1D study visit.
Linear Mixed Models
In the first set of analyses, 3 separate linear mixed models were used to examine the effect of day on the frequency of BGM, carbohydrate inputs, and insulin boluses delivered, including any day × treatment group interaction. The main effect of baseline adherence frequency (obtained at study visit 1) was significant in all 3 models (all Ps < .0001). The main effect of visit was significant for carbohydrate inputs (P < .001) and insulin boluses delivered (P < .0001), but not BGM. Of primary interest was the day × treatment group interaction, which was significant for carbohydrate inputs (P < .01) and insulin boluses delivered (P < .0001) and approached significance for BGM (P = .10). Consequently, stratified linear mixed models were used to test the effect of day (ie, white coat adherence) on frequency of BGM, carbohydrate inputs, and insulin boluses delivered for the Tailored Feedback vs Treatment as Usual groups separately.
Table 2 demonstrates that after controlling for visit and baseline adherence frequency, participants in the Tailored Feedback group demonstrated white coat adherence prior to their T1D study visits for BGM, carbohydrate inputs, and insulin boluses delivered (Ps < .01, < .0001, and < .0001, respectively), but those in the Treatment as Usual group did not (Ps > .42).
Participants in Tailored Feedback Demonstrate White Coat Adherence in the 40 Days Prior to T1D Study Visits.
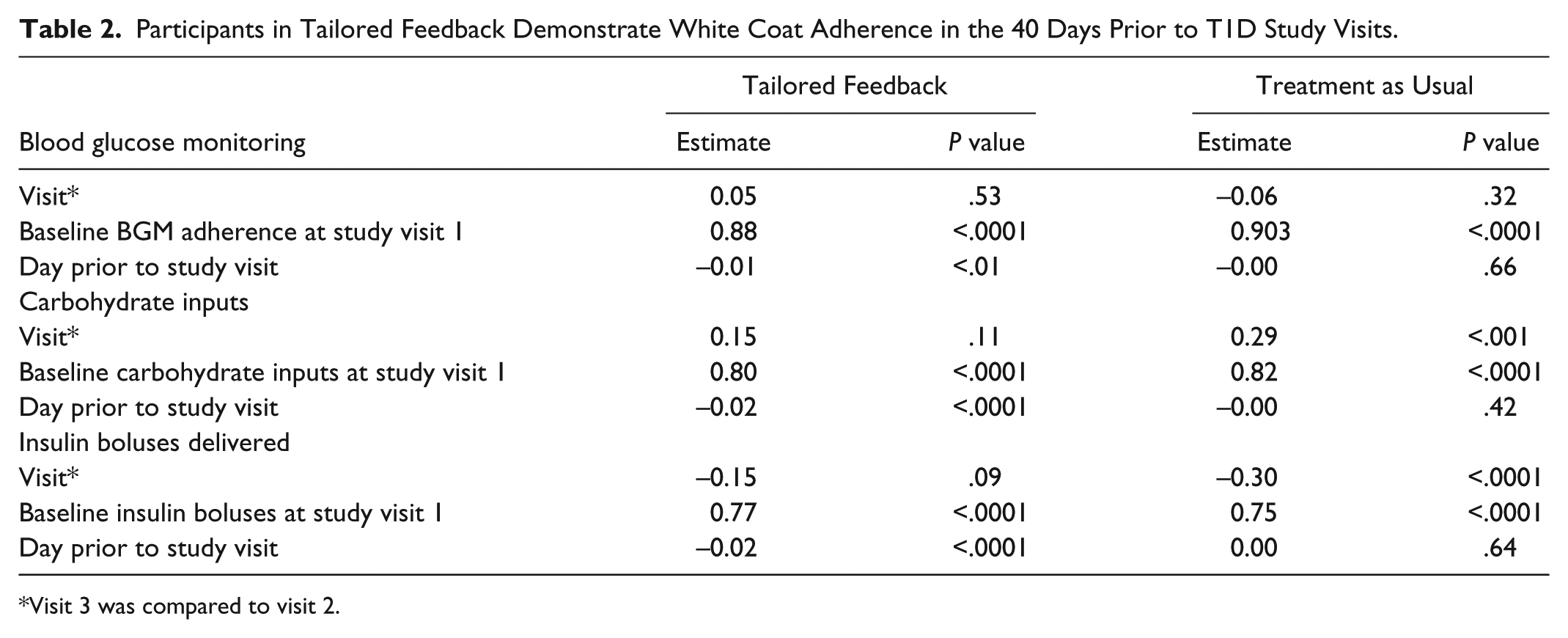
Visit 3 was compared to visit 2.
Taken together, the results revealed the following: (1) participants who had a higher frequency of BGM, carbohydrate inputs, and insulin boluses delivered at baseline, continued to engage in higher frequencies of each following randomization; and (2) frequency of BGM, carbohydrate inputs, and insulin boluses delivered increased the closer the study visit approached for those in the Tailored Feedback group, but not for those in the Treatment as Usual group.
Discussion
This study adds to a growing body of literature using objectively downloaded data from insulin pumps that demonstrate that white coat adherence occurs in pediatric T1D across multiple study visits that coincide with the routine T1D clinic visit.4,6 Previous studies have not shown that adolescents with T1D engage in white coat adherence.4,6 Adolescents randomized to Treatment as Usual in the current study behaved in a similar fashion, exhibiting no evidence of white coat adherence. In contrast, adolescents randomized to the intervention, which focused on improving specific insulin pump behaviors, demonstrated white coat adherence. One possible explanation is that the T1D study visit and contact with the T1D team may serve as a “trigger” or prompt, resulting in increased insulin pump adherence when adolescents know they are in a clinical trial to test an adherence intervention.5,8 In this context, adolescents may be motivated to gain their T1D study team’s approval (or avoid their disapproval) by appearing more adherent. 5
White coat adherence may result in benefits to patients with T1D such as providing more data to the T1D provider and the receipt of positive reinforcement (eg, praise, encouragement) from members of the T1D team. 6 However, this additional increase in adherence behaviors prior to a study or clinic visit may give the T1D provider—and in the case of clinical trials—study personnel and investigators, the impression that adherence is much higher than is actually the case. In an effort to reduce the amount of data to interpret, T1D care providers and clinical trial investigators routinely download BGM, continuous glucose monitoring, and insulin pump data for the 2 weeks prior to the T1D study visit, (not the entire amount of data contained in pumps) which is the industry software standard default.9-11 This means that T1D providers and clinical trial investigators are using only the data from the time period when white coat adherence is highest, resulting in the acquisition of an inaccurate picture of adherence behaviors immediately preceding the appointment.
The present study has a number of strengths that expand on past research on the occurrence of white coat adherence. This study focused on adolescents with T1D, replicating another of our studies in which an independent sample of participants, including adolescents, engaged in insulin pump white coat adherence. 6 By using objectively downloaded insulin pump data, this study also demonstrates the stability of white coat adherence across time. Finally, this is the first study to consider that white coat adherence may occur in pediatric clinical trials. However, the results also need to be evaluated in the context of several limitations. Although downloaded data are the closest approximation available to actual adherence, technology is limited in that carbohydrates must still be manually entered. The data contained in insulin pumps are not without challenges, and not all pumps hold the same amount of data. Finally, participation was restricted to participants who used Medtronic insulin pumps because in the region where this study took place, the vast majority of children chose Medtronic pumps over other pumps. In addition, the data contained in different insulin pumps were not comparable. Although we suspect that white coat adherence is not specific to Medtronic insulin pump users, additional research is needed to evaluate it in non-Medtronic insulin pump users.
Nevertheless, the results of the current study are highly relevant, both clinically and scientifically, as members of T1D teams need to be aware that white coat adherence occurs in adolescents when they are randomized to an adherence intervention as part of a clinical trial. It is unknown whether white coat adherence would occur in adolescents who are participating in other types of randomized controlled trials such as drug trials. However, clinical trials involving adults with HIV and hypertension have shown white coat adherence.12,13 One study found that increasing nursing visits in a hypertension clinical trial decreased the occurrence of white coat adherence suggesting that more frequent visits with providers may reduce the occurrence of white coat adherence. 12 Therefore, it is recommended that in clinical trials in which objective data are measured, the possibility that white coat adherence is occurring in the treatment group should be examined in the analytic approach. In clinical trials in which adherence cannot be objectively measured, the occurrence of white coat adherence should be indicated as a possibility and the lack of objective data as a limitation.
Footnotes
Acknowledgements
We are grateful to the adolescents and their parents who participated in this study.
Abbreviations
BGM, blood glucose monitoring; T1D, type 1 diabetes.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Institute of Diabetes and Digestive and Kidney Disease (DK091558) and American Diabetes Association (1-12-JF-15)
