Abstract

Keywords
The fast diffusion of technologies in diabetes (insulin pumps; continuous glucose monitoring, CGM; sensor-augmented pumps, SAP) requires a large organizational effort. Uncertainties about using these instruments could lead to acute diabetes-related complications (ie, severe hypoglycemia or hyperglycemia)1-3 with high risk of patient’s failure. 4 In our diabetes center (Niguarda Hospital, Milan, Italy), since 2012 we have called in technical staff from companies in the field, setting up a “technological desk” (TD) to back up the clinical work with medical and nursing staff. Actually, the TD reaches 80% of all the subjects with T1D we care for (almost 1500). Formal contracts describing the tasks (eg, first application/replacement of devices; training for advanced pump functions; discharge of devices-recorded data) were drawn up between the hospital and the companies. All activities are supervised by the diabetologist. The companies provide their service free of charge.
Our proposal of a technological path is illustrated in Figure 1. Whenever a clinical indication to CSII is identified an appointment at the TD is scheduled. An expert technician of the company explains how to prepare the pump for use, perform the infusion set change, set and change the basal rate, and select the boluses (at first only normal bolus). Patients are invited to perform self-monitoring of blood glucose (SMBG) at home according to an individual plan. An emergency alternative insulin regimen is established. The patient is advised to contact the technician for every technical problem. In a second meeting (after 7-10 days) at the TD, SMBG data are discussed and insulin therapy is eventually modified by the diabetologist. Patients are asked about any technical problems and the portability of the system. Furthermore, advanced functions (special boluses, temporary and alternative basal) are explained. A new visit is scheduled after 20-30 days. If a SAP therapy is needed, then the patient is educated to the use of the sensor. If the system has a low glucose suspend (LGS) or a predictive LGS function, then this is not activated initially but in a new visit after 7-10 days.
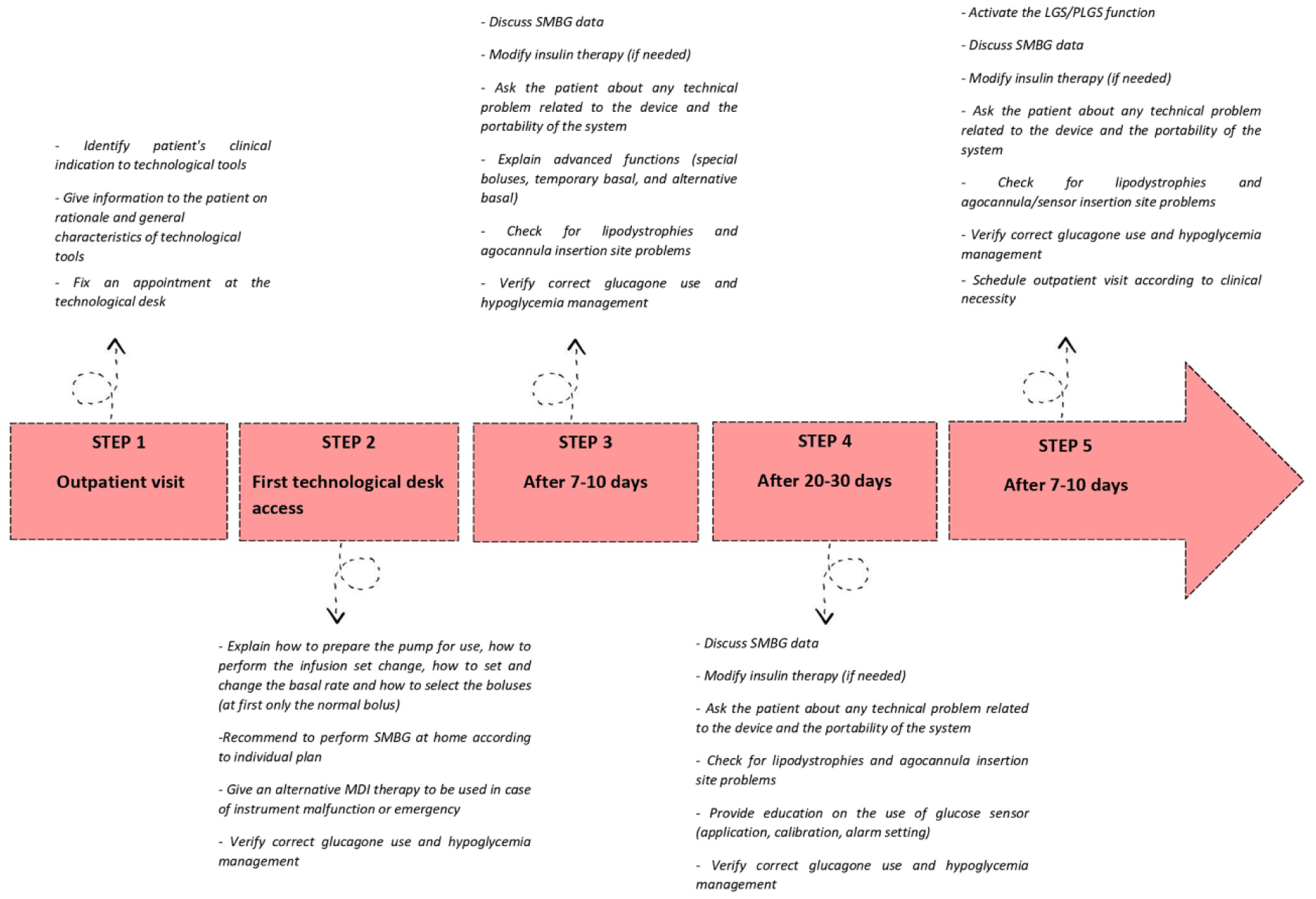
Technological path description.
Specific education on CHO counting, glucagon use, and hypoglycemia management is provided. At every visit the presence of lipodystrophies and cannula/sensor insertion site are checked.
In a sample of consecutive subjects with T1D (37.5% males, mean age 38±11 years) who started our technological path for a SAP therapy with Predictive Low-Glucose Suspend (PLGS) (Medtronic MiniMed 640G insulin pumps with Enlite glucose sensor Northridge, CA), after a median follow-up of 8 months (IQR: 6-11) of technology use, HbA1c levels (61.1 ± 12.9 mmol/mol vs 54.7 ± 10.6 mmol/mol or 7.7 ± 3.3% vs 7.2 ± 3.1%, P = .009) significantly decreased compared to baseline. Patients participated in the Italian Clinical Service Projects, a national medical care project aiming to improve the quality of diagnostic and therapeutic strategies. No hospitalization for severe hypoglycemia or diabetic ketoacidosis and no technical problems linked to the devices were recorded.
Our TD is unique in Italy, in providing the presence of a service provided by private companies in public health settings, which is a novelty. The public-private integration allows us to exchange prescriptive appropriateness with practical technical support. Our experience allows transparency and correctness in the relationship with companies in the sector.
Footnotes
Acknowledgements
The authors very much appreciate the support received by Ester Tartaglione (Medtronic Regional Clinical Center EMEA & Americas) for the statistical analysis.
Abbreviations
CGM, continuous glucose monitoring; LGS, low glucose suspend; PLGS, predictive low-glucose suspend; SAP, sensor-augmented pump; SMBG, self-monitoring of blood glucose; TD, technological desk, T1D, type 1 diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: BP declares consulting fees from Roche, Healthware group, Johnson & Johnson, and LO.LI.pharma. OD declares fees for serving on advisory boards from Eli Lilly, Boehringer, Astra Zeneca, Sanofi; lecture fees from Novo Nordisk, travel support from Astra Zeneca, Eli Lilly, Boehringer, Novo Nordisk, Sanofi; consulting fees from Novo Nordisk; and clinical trial support to her institution from Eli Lilly, Novonordisk, Sanofi. MB declares consulting fees from Medtronic, Roche, and Sanofi. FB, IG, GP, and AP declare no conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
