Abstract
Continuous standardized verification of the accuracy of blood glucose meter systems for self-monitoring after their introduction into the market is an important clinically tool to assure reliable performance of subsequently released lots of strips. Moreover, such published verification studies permit comparison of different blood glucose monitoring systems and, thus, are increasingly involved in the process of evidence-based purchase decision making.
Continuous standardized verification of the accuracy of blood glucose meter systems for self-monitoring after their introduction into the market is an important clinically tool to assure reliable performance of subsequently released lots of strips. 1 Moreover, such published verification studies permit comparison of different blood glucose monitoring systems and, thus, are increasingly involved in the process of evidence-based purchase decision making. ISO 15197:2013, 2 for which mandatory compliance is recommended for SMBG systems by 2016, 3 has tighter accuracy requirements than ISO 15197:2003, 4 and outlines current minimum accuracy standards necessary in Europe for CE marking.
In the present study, a postmarketing evaluation of the CE-marked VivaChek™Ino SMBG system was performed in accordance with ISO 15197:2013 protocols and requirements for system accuracy. The glucose monitoring system is manufactured and distributed by VivaChek Laboratories, Inc (Wilmington, DE, USA). Two glucose monitoring systems (serial numbers 10101A0005669 and 10101A0005666) and strips from 3 lots (1100080, 1100081, and 1100082 with expiry dates in February 2017, respectively) were supplied by the manufacturer for the study that was conducted from June 30 to July 17, 2015, at the Institute of Diabetes “Gerhardt Katsch,” Karlsburg, Germany. The study was approved by the Ethics Committee of the University of Greifswald (registration no. BB 081/15).
Ear lobe capillary blood samples were drawn from 104 subjects for duplicate glucose determination using the glucometer and the glucose-oxidase-based YSI2300 STAT PLUS (YSI Incorporated, Yellow Springs, OH, USA) plasma glucose reference method. Trueness and precision of the comparison assay were verified using a range of YSI bioanalytical standards and controls. Samples had hematocrit values between 20% and 70% and after examination of glucose concentration ranges using the YSI, 100 subjects were included in the analysis of accuracy.
Table 1 demonstrates performance in relation to the minimum accuracy requirements of ISO 15197:2013 where for each of the 3 lots of strips at least 95% of results must fall within ±15 mg/dl of the comparison measurement results at blood glucose concentrations <100 mg/dl and within ±15% at concentrations ≥100 mg/dl. The standard also requires that at least 99% of individual results fall within consensus error grid zones A and B 5 when clinical accuracy is evaluated with 3 test strip lots. The relative bias ranged from −6.58% to −4.93% and the mean absolute relative difference from 3.83% to 5.08%.
Analytical and Clinical Accuracy of the Glucometer With 3 Lots of Strips.
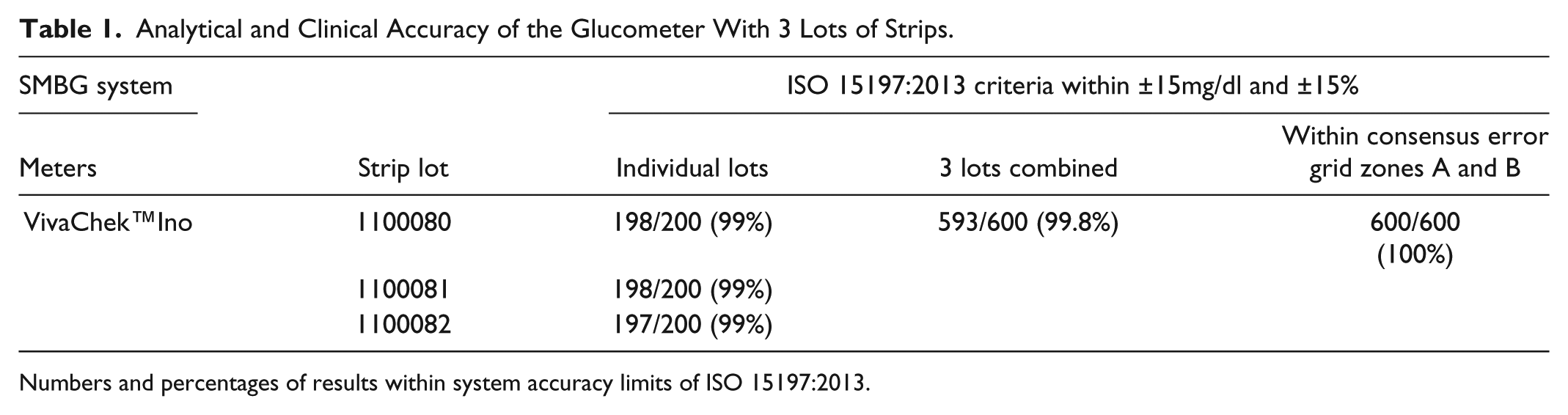
Numbers and percentages of results within system accuracy limits of ISO 15197:2013.
In conclusion, the present study demonstrates that the VivaChek™Ino SMBG system fulfils and even exceeds the minimum analytical and clinical accuracy requirement of ISO 15197:2013.
Footnotes
Acknowledgements
VivaChek Laboratories, Inc was permitted to review and comment on the manuscript, but final decision on content was retained by the authors.
Abbreviations
CE, Conformité Européene; ISO, International Organization for Standardization; SMBG, self-monitoring of blood glucose.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors are employees of the Institut für Diabetes, Karlsburg, Germany, which carries out studies evaluating blood glucose meter systems on behalf of various companies.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by VivaChek Laboratories, Inc, 913 N Market St, Ste 200, Wilmington, DE 19081, USA.
