Abstract

For people with diabetes, self-monitoring of blood glucose (SMBG) is an essential instrument in the maintenance of glycemic control. Minimum accuracy criteria for the evaluation of SMBG systems are described in the International Organization for Standardization (ISO) standard 15197:2003. 1 A revised version of this standard (ISO 15197:2013), 2 for which mandatory compliance is recommended in 2016, defines more stringent requirements for system accuracy: at least 95% of the system’s results shall fall within ±15 mg/dl of the average values measured with the comparison method for blood glucose (BG) concentrations <100 mg/dl and within ±15% for BG concentrations ≥100 mg/dl. Furthermore, 99% of the results shall be within zones A and B of the consensus error grid. Assessment of conformity with this standard is usually performed by manufacturers to obtain a Conformité Européene (CE) mark for their system in the European Union.
In this study, system accuracy of a new version of the Accu-Chek® Active (Roche Diagnostics GmbH, Mannheim, Germany) was evaluated with 3 different reagent system lots following testing procedures and applying accuracy criteria of ISO 15197:2013.
The new generation of the SMBG system, which is based on photometric measurement technology, has recently been introduced in many countries worldwide. Compared to its predecessor, the system has a new design and additional features like a postprandial reminder and underdose detection with redose option. Furthermore, the reagent system contains a new enzyme.
The study was performed between March and April 2014 in compliance with the German Medical Devices Act at the Institut für Diabetes-Technologie Forschungs- und Entwicklungsgesellschaft mbH an der Universität Ulm, Ulm, Germany. The study protocol was approved by the Ethics Committee and the Federal Institute for Drugs and Medical Devices.
Meter and reagent system lots were purchased from a Polish pharmacy since they are not available in Germany. Measurements were performed using capillary blood samples from 100 different subjects following testing procedures of ISO 15197:2013. Comparison measurements were performed by a hexokinase laboratory method (cobas® 6000 c501, Roche Diagnostics GmbH, Mannheim, Germany) from deproteinized capillary whole blood. This method is traceable to National Institute of Standards and Technology (NIST) standard reference material.
All 3 tested lots of the system comply with accuracy criteria described in ISO 15197:2013 (Figure 1).
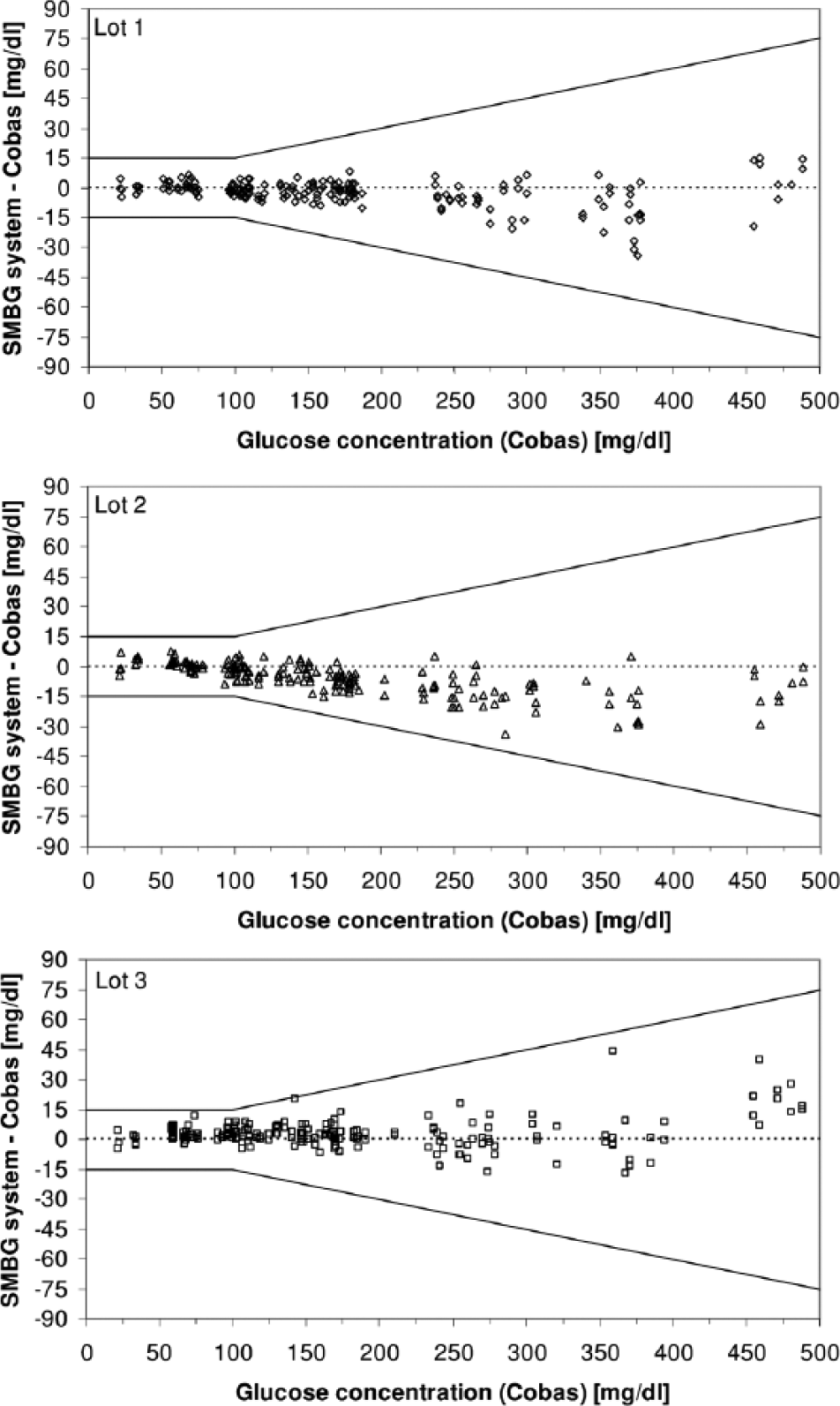
Difference plots for the 3 evaluated reagent system lots. Absolute differences between the system’s measurement results and the mean comparison measurement results (200 data, duplicate measurements on a sample with each lot) are displayed. Lines indicate criteria of ISO 15197:2013: ±15 mg/dl / ±15% for glucose concentrations <100 mg/dl / ≥100 mg/dl.
For BG concentrations <100 mg/dl, 100% of the results were within ±15 mg/dl, 98.1% to 100% within ±10 mg/dl, and 80.8% to 94.2% within ±5 mg/dl of the comparison method measurement results. For BG concentrations ≥100 mg/dl, 100% of the results were within ±15%, 98.6% to 100% within ±10%, and 64.2% to 89.9% within ±5%. In addition, 100% of the measured values were within consensus error grid zone A. The relative bias according to Bland and Altman 3 ranged from –2.4% to 1.6%.
For the predecessors of the investigated system compliance with accuracy criteria of ISO 15197:2003 has been reported previously. 4 The advanced system evaluated in this study even showed high accuracy when the criteria of ISO 15197:2013 were applied with only minor variations between the 3 evaluated reagent system lots.
Footnotes
Acknowledgements
We would like to thank Volker Lodwig and Andrew Hattemer for their valuable input.
Abbreviations
BG, blood glucose; CE, Conformité Européene; ISO, International Organization for Standardization; NIST, National Institute of Standards and Technology; SMBG, self-monitoring of blood glucose.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors are employees of the Institut für Diabetes-Technologie Forschungs- und Entwicklungsgesellschaft mbH an der Universität Ulm (IDT), Ulm, Germany. GF is general manager of the IDT, which carries out studies evaluating BG meters and medical devices for diabetes therapy on behalf of various companies. GF/IDT received speakers’ honoraria or consulting fees from Abbott, Bayer, Berlin-Chemie, Becton-Dickinson, Dexcom, Menarini Diagnostics, Roche Diagnostics, Sanofi, and Ypsomed.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a grant from Roche Diagnostics GmbH, Mannheim, Germany.
