Abstract

Dear Editor,
When glycemic control remains inadequate with metformin monotherapy, a sulfonylurea derivative (SU) can be added.1,2 Previous studies using a continuous glucose measurement system (CGMS) demonstrated that elderly patients with type 2 diabetes mellitus (T2DM) using SUs have unrecognized hypoglycemic events.3,4 Unfortunately, neither an appropriate control group (patients without diabetes or patients using metformin monotherapy) was used nor differences in hypoglycemia rate between different SUs were investigated. Therefore, this pilot study aimed to investigate the prevalence of (a)symptomatic hypoglycemia in frail elderly T2DM patients treated with metformin and a sulfonylurea, explicitly gliclazide and glimepiride. We included primary-care treated patients ≥ 70 years with T2DM, HbA1c < 58 mmol/mol (7.5%), and a Groningen Frailty Indicator (GFI) score ≥ 4. Patients used metformin monotherapy or metformin combined with gliclazide or glimepiride. A blinded CGMS (iPro2, Medtronic, Northridge, CA, USA) was worn for 5 consecutive days and patients were instructed to recorded symptoms of hypoglycemia. Primary outcome was the total number of hypoglycemic events (glucose levels < 3.0 mmol/L) persisting for at least 15 minutes with or without symptoms. The trial was approved by the medical ethics committee of the Isala hospital (Zwolle, Netherlands), registered in the local Dutch register (number: NTR 4952) and funded by the non-commercial Hein Hogerzeil Foundation.
A total of 23 participants (47% male, 76 [72, 81] years old, with a GFI of 5 [4, 6], median diabetes duration of 9 [5, 11] years, and mean HbA1c 49 ± 4 mmol/mol [6.6 (0.4) %]) were monitored for a median period of 97 hours (range: 86 hours 15 minutes to 117 hours 25 minutes). Besides a lower GFI in the metformin and gliclazide group, there were no differences between groups. Five patients (22%) experienced a total of 15 events with a glucose level < 3.0 mmol/L recorded with CGMS. Eight patients (35%) experienced a total of 25 events with a glucose level < 3.5 mmol/L. None of these patients reported experiencing symptoms of hypoglycemia, all these patients used a SU, and there was no difference between gliclazide and glimepiride (see Table 1). On average, patients had significantly lower HbA1c levels (50 [3.4] vs 46 [4.1] mmol/mol (6.7 [0.3] vs 6.4 [0.4]%)) and higher GFI scores (4 [4, 5] vs 6 [4, 8]) if they experienced 1 or more hypoglycemic event(s) with a glucose level < 3.5 mmol/L than if they experienced no event. Although this pilot-study has (obvious) limitations and the clinical relevance of asymptomatic hypoglycemic events is unclear at present, we conclude that this study confirms the presence of hypoglycemia (<3.0 mmol/L) in well-controlled frail elderly T2DM patients using SUs and no events in patients treated with metformin only. All these events are asymptomatic and there are no relevant differences in frequency between gliclazide and glimepiride.
Frequency and Distribution of Hypoglycemia.
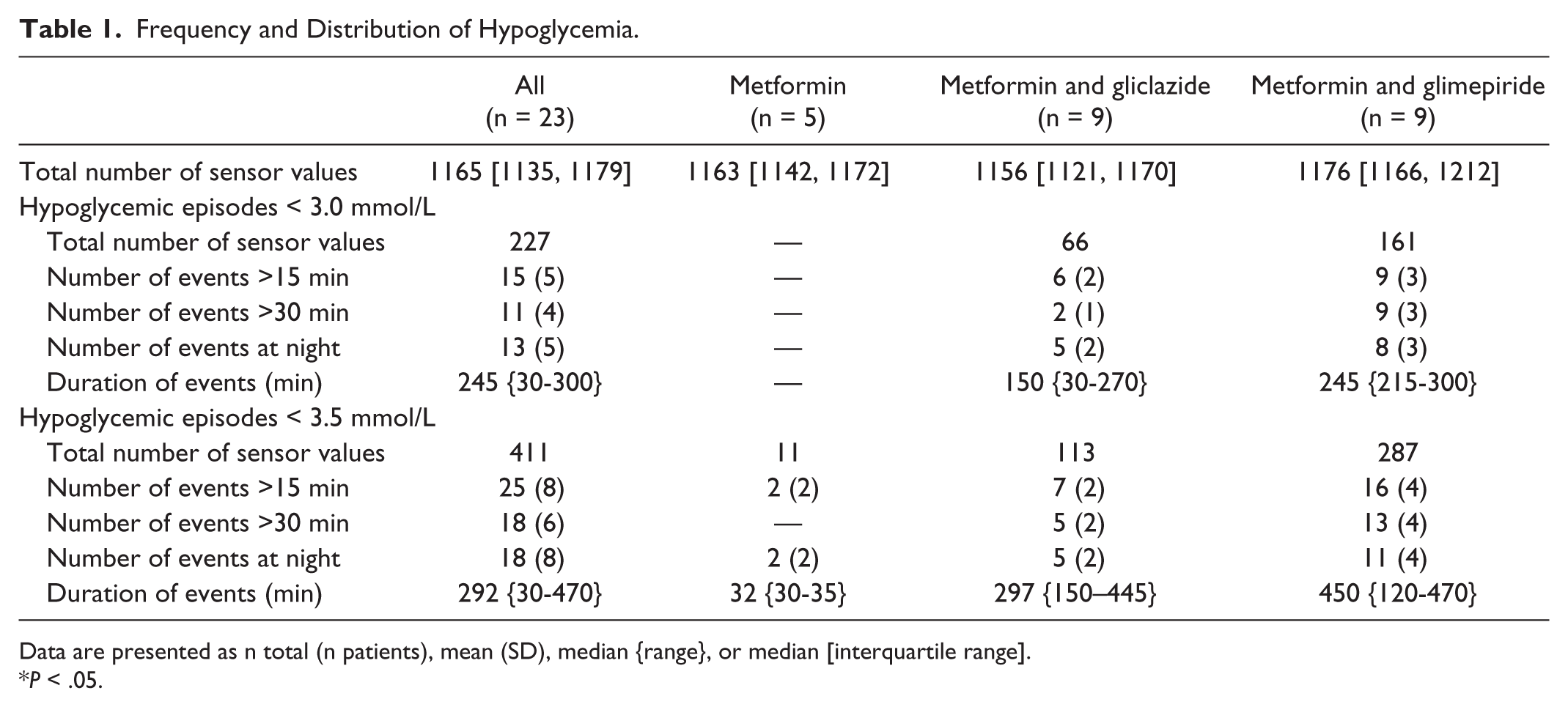
Data are presented as n total (n patients), mean (SD), median {range}, or median [interquartile range].
Footnotes
Abbreviations
CGMS, continuous glucose monitoring system; GFI, Groningen Frailty Indicator; SU, sulfonylurea derivative; T2DM, type 2 diabetes mellitus.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received funding from the Hein Hogerzeil foundation.
