Abstract
Background:
We developed a system to suspend insulin pump delivery overnight when the glucose trend predicts hypoglycemia. This predictive low-glucose suspend (PLGS) system substantially reduces nocturnal hypoglycemia without an increase in morning ketosis. Evaluation of hypoglycemia risk factors that could potentially influence the efficacy of the system remains critical for understanding possible problems with the system and identifying patients that may have the greatest benefit when using the system.
Methods:
The at-home randomized trial consisted of 127 study participants with hemoglobin A1c (A1C) of ≤8.5% (mmol/mol) for patients aged 4-14 years and ≤8.0% for patient aged 15-45 years. Factors assessed included age, gender, A1C, diabetes duration, daily percentage basal insulin, total daily dose of insulin (units/kg-day), bedtime BG, bedtime snack, insulin on board, continuous glucose monitor (CGM) rate of change (ROC), day of the week, time system activated, daytime exercise intensity, and daytime CGM-measured hypoglycemia.
Results:
The PLGS system was effective in preventing hypoglycemia for each factor subgroup. There was no evidence that the PLGS system was more or less effective in preventing hypoglycemia in any one subgroup compared with the other subgroups based on that factor. In addition, the effect of the system on overnight hyperglycemia did not differ in subgroups.
Conclusions:
The PLGS system tested in this study effectively reduced hypoglycemia without a meaningful increase in hyperglycemia across a variety of factors.
An important goal of a closed loop system for individuals with type 1 diabetes is prevention of hypoglycemia, particularly overnight. We developed a system to suspend insulin pump delivery overnight when the glucose trend predicts hypoglycemia.1,2 The predictive low-glucose suspend (PLGS) system involves a continuous glucose monitor (CGM) device communicating with an insulin pump via a laptop computer that contains the hypoglycemia prediction algorithm. In an at-home randomized trial of 81 children aged 4-14 1 and 45 adults aged 15-45 2 with type 1 diabetes, we found that the PLGS system reduced the frequency of nocturnal hypoglycemia by 25% and 36%, respectively, compared with a control arm. Median time less than 70 mg/dL was reduced by more than 50%, prolonged nocturnal hypoglycemia (≤60 mg/dl for >2 hours) was reduced more than 3-fold, and there was no meaningful increase in hyperglycemia or ketosis. While the PLGS system greatly reduced hypoglycemia overall, it is possible subject characteristics or activities could influence the effectiveness of the PLGS system. During control nights, lower age, lower hemoglobin A1c (A1C) level, preceding hypoglycemia, and preceding exercise were associated with higher rates of nocturnal hypoglycemia. 3 We undertook the analyses reported herein to assess whether the efficacy of the PLGS system is related to the presence of these or other factors shown to be associated with nocturnal hypoglycemia.
Methods
The study was conducted at 3 clinical centers in the United States and Canada. The protocol was approved by each Institutional Review Board and informed consent and assent was obtained as appropriate. The study has been described elsewhere1,2 and listed on the ClinicalTrials.gov website (clinical trial registration numbers NCT01823341 and NCT01591681); key aspects of the study protocols are described below.
The pump suspension system consisted of a MiniMed Paradigm® REAL-Time Veo™ System and Enlite™ glucose sensor (Medtronic Diabetes, Northridge, CA), in which the CGM and pump communicated with a bedside laptop computer which contained the hypoglycemia prediction algorithm developed by the investigators. The PLGS system used a Kalman filter to estimate the glucose level and ROC and suspended basal insulin delivery if glucose was predicted to fall below 80 mg/dl in the next 30 minutes. 4 Additional suspension/restart rules included a threshold suspend override once sensor glucose increased to 70 mg/dl, no suspension if CGM glucose concentration >230 mg/dl or if a pressure-induced sensor attenuation was suspected based on glucose ROC, 5 and restoration of basal insulin on the first CGM rise following a suspension. Audible alarms were set at 60 mg/dl. Additional details about the system have been published.4,5
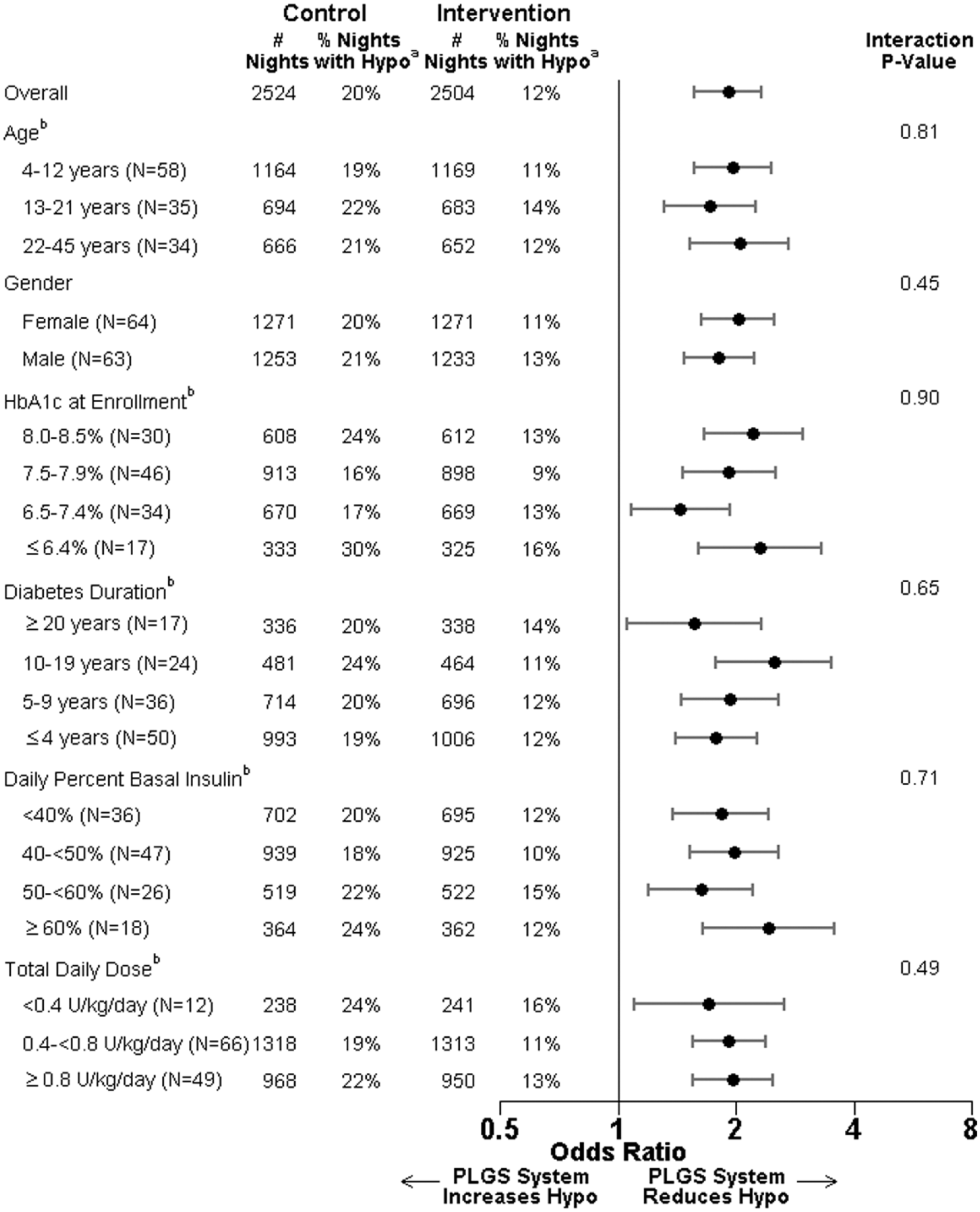
Major eligibility criteria included having type 1 diabetes with use of daily insulin therapy for ≥1 year and use of insulin infusion pump for ≥6 months. Patients aged 4-14 and 15-45 years old required an A1C level measured with a point-of-care device of ≤8.5% and ≤8.0%, respectively, at enrollment. Included patients also had a minimum amount of nocturnal hypoglycemia during a run-in phase. Additional criteria are listed in previously published articles.1,2 The 127 study participants included in this analysis ranged in age from 4 to 45 years; half were female and 94% were Caucasian. Supplemental Table S1 gives the participant characteristics at enrollment. Children aged 4-12, adolescents aged 13-21, and adults aged 22-45 (FDA-defined age groups) had a median duration of type 1 diabetes of 4, 8, and 22 years, respectively, and median A1C of 7.8%, 7.6%, and 6.6%, respectively.
Following a run-in phase, the randomized trial consisted of 42 nights with at least 4 hours of sensor glucose data, during which approximately half the nights were intervention nights (PLGS algorithm active) and half control (PLGS algorithm inactive) nights. The order of intervention versus control nights was randomized nightly and blinded to the participant. At the time of system activation each night, the participant entered the level of exercise intensity for that day (none, low, medium, or high) and whether or not a bedtime snack was consumed. CGM device, pump downloads, and blood glucose meter downloads provided data used in analysis for insulin delivery and occurrence of hypoglycemia during both day and night as well as capillary glucose levels when the system was activated each night and turned off in the morning.
Statistical Methods
The time period for outcome assessment each night was from activation until deactivation the following morning. Randomized nights with at least 6 hours of CGM data were included in the analysis. A hypoglycemia night was defined as having at least 30 minutes of CGM values below 60 mg/dl. Both subject-level and night-level factors were assessed for possible influence on the efficacy of the system in preventing hypoglycemia. Subject-level factors included age, gender, A1C at enrollment, diabetes duration, daily percentage basal insulin, and total daily dose of insulin (units/kg-day). Night-level factors included bedtime BG, bedtime snack, insulin on board at activation, CGM rate of change (ROC) before activation, day of the week, time system activated, daytime exercise intensity preceding nighttime system use, and daytime CGM-measured hypoglycemia preceding nighttime system use. A previous article found the system was less effective in the first 3 hours; 2 a secondary analysis was performed to assess factors influencing the effectiveness of the system to prevent hypoglycemia in the first 3 hours.
To assess preceding daytime CGM-measured hypoglycemia, time below 60 mg/dl was measured from noon until 8pm. Nights activated between 8
The percentage of control nights and intervention nights with hypoglycemia was tabulated for each factor, giving equal weight to each night. An interaction effect was tested to assess if the PLGS system was more or less effective preventing hypoglycemia within the subgroups for a given factor. A repeated-measures regression model with random subject effects and spatial power autocorrelation structure was fitted to test for the interaction effect. Factors with a P value < .01 were considered significant to adjust for multiple hypothesis testing. A linear relationship was fitted for continuous factors. A 99% confidence interval of the odds ratio was calculated for each subgroup. Odds ratios greater than 1 indicated hypoglycemia was more likely to occur on control nights than intervention nights. All P values are 2-tailed and analyses were performed using SAS 9.4.
Results
Nocturnal hypoglycemia with at least 30 minutes of CGM values below 60 mg/dl occurred on 515 of 2524 (20%) control nights versus 299 of 2504 (12%) intervention nights (odds ratio 1.91 [99% CI 1.57-2.32]; P value < .001).
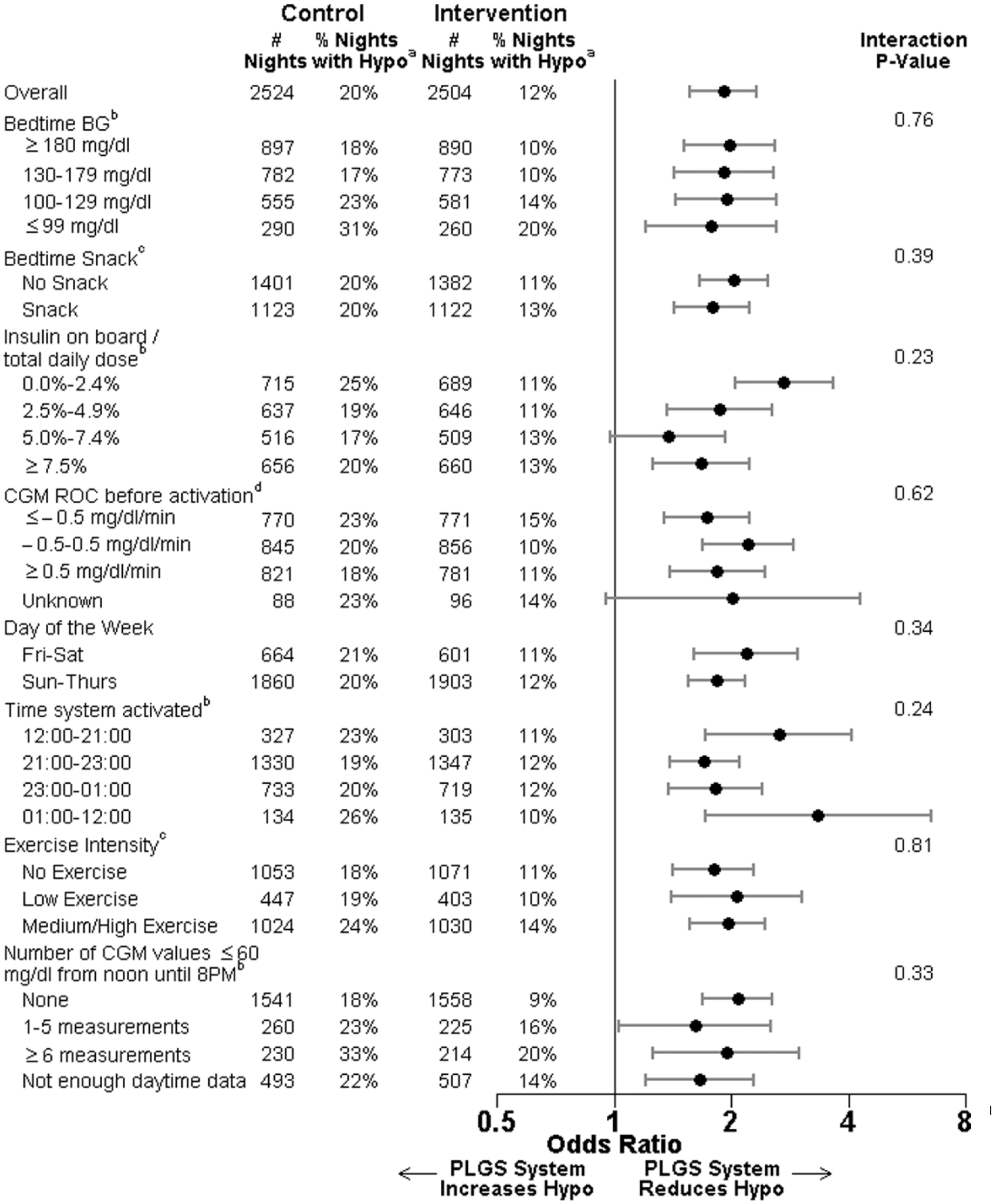
The PLGS system was effective in preventing hypoglycemia for each patient-level factor subgroup (Table 1) and each night-level factor subgroup (Table 2). There was not strong evidence in any subgroup that the PLGS system was more or less effective in preventing hypoglycemia than in other subgroups based on that factor (all interaction P values > .01).
Patient-Level Factors Influence on the PLGS System to Prevent Hypoglycemia.
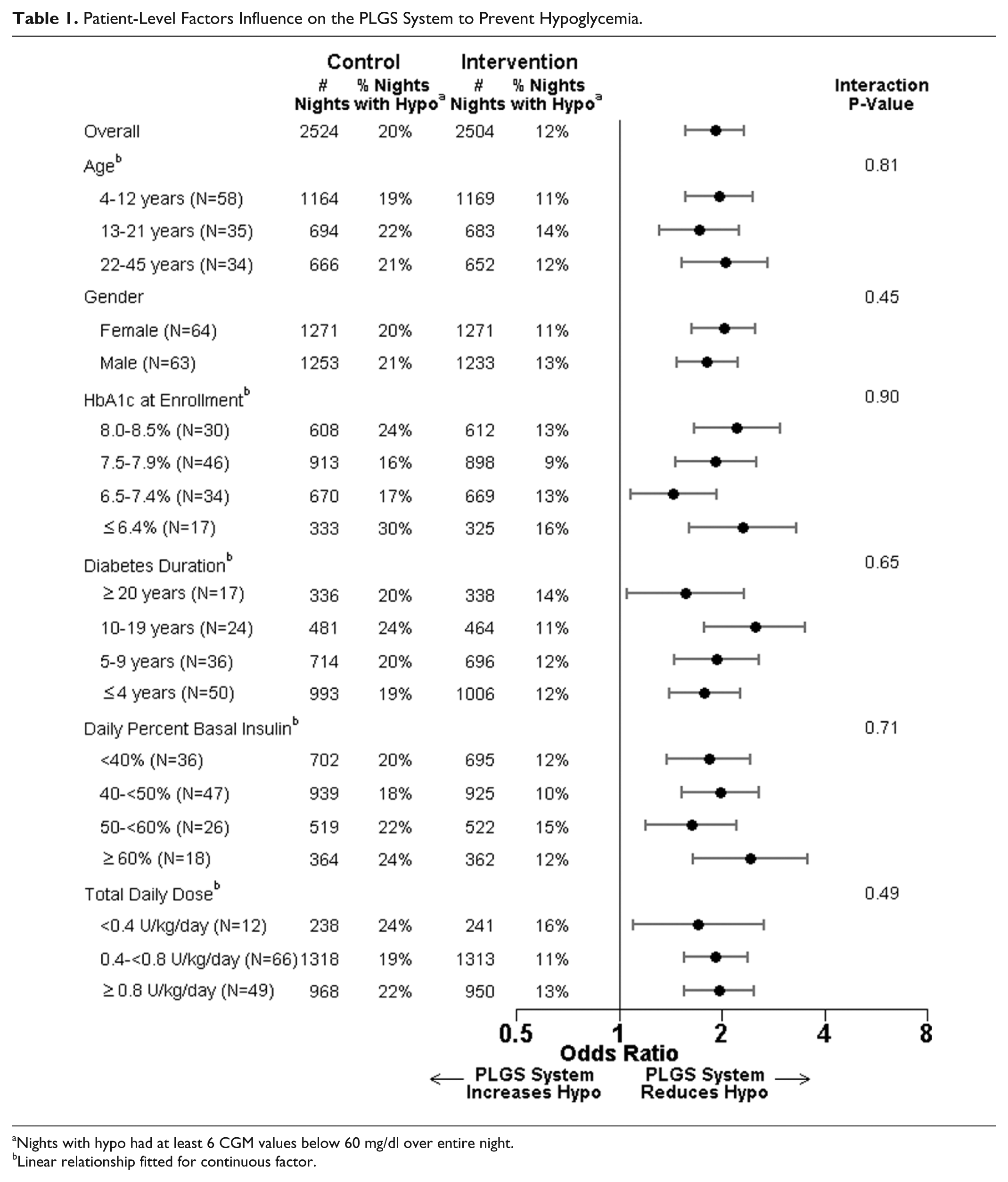
Nights with hypo had at least 6 CGM values below 60 mg/dl over entire night.
Linear relationship fitted for continuous factor.
Night-Level Factors Influence on the PLGS System to Prevent Hypoglycemia.
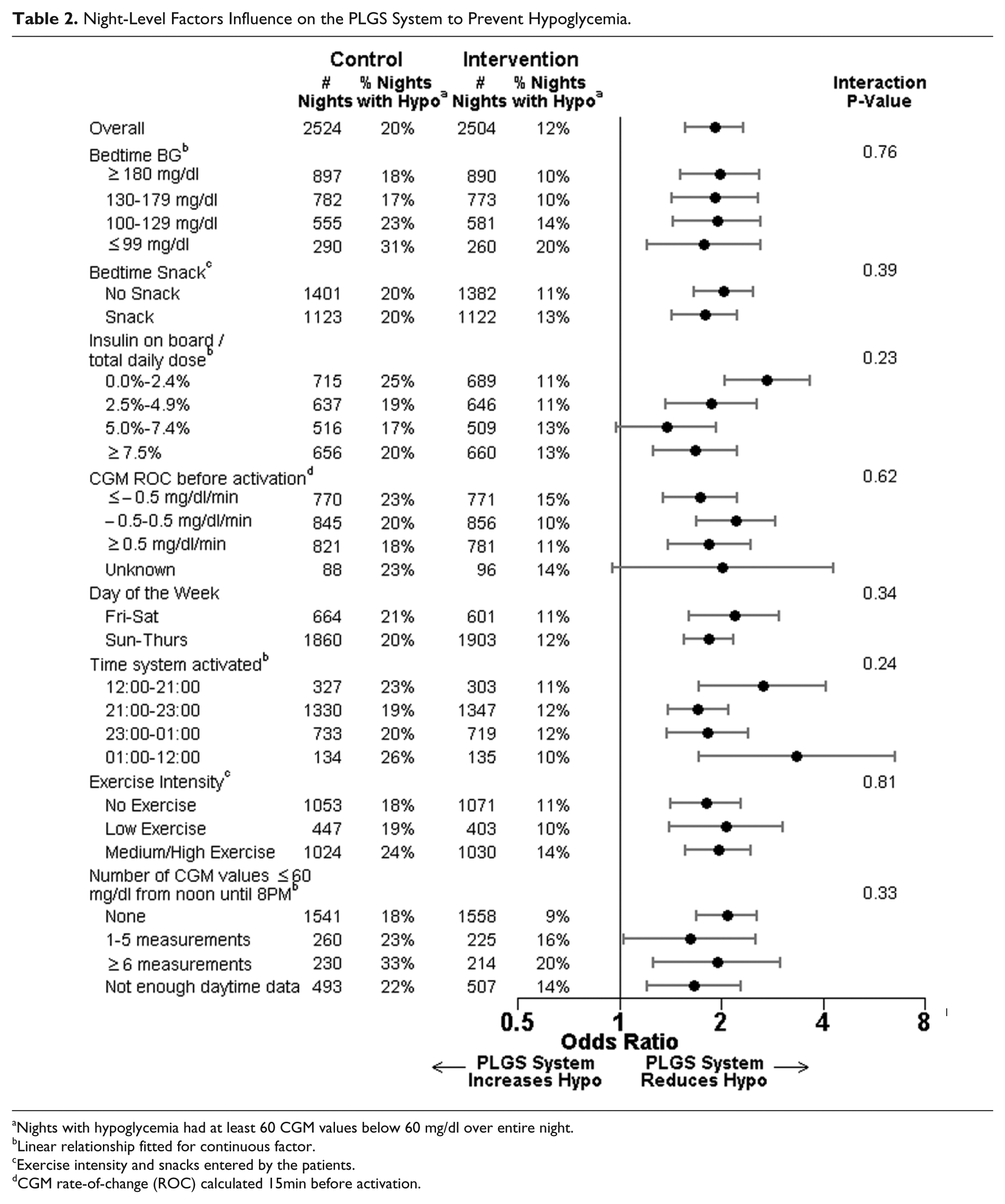
Nights with hypoglycemia had at least 60 CGM values below 60 mg/dl over entire night.
Linear relationship fitted for continuous factor.
Exercise intensity and snacks entered by the patients.
CGM rate-of-change (ROC) calculated 15min before activation.
Supplemental Table S2 provides the nocturnal hypoglycemia rates stratified by age. Nocturnal hypoglycemia rates during the first 3 hours on intervention and control nights were similar (Supplemental Table S3). There was a substantial benefit of the PLGS system after 3 hours of system use. Nocturnal hyperglycemia with at least 30 minutes of CGM values above 250 mg/dl was similar on control and intervention nights (19% vs 21%, respectively) with no evidence any factor subgroup had higher rates of hyperglycemia on intervention nights compared with control nights.
Conclusion
Several factors have been associated with the frequency of nocturnal hypoglycemia including lower age, 3 lower hemoglobin A1c level, 6 occurrence of prior hypoglycemia6,7 or an exercise session,8,9 no bedtime snack, 10 and lower bedtime BG. 11 A reasonable hypothesis is that a PLGS system would be more effective on nights with higher risk of hypoglycemia, but there was no evidence this was true for the PLGS system we tested; the system was equally beneficial irrespective of the patient’s activity or characteristics.
There is little research devoted to factors influencing the efficacy of closed-loop systems in preventing hypoglycemia. High insulin on board has been shown to affect the ability of glucagon to prevent hypoglycemia. 12 In a preliminary study, 4 we found changing the PLGS algorithm to a longer projection horizon with slower restoration of basal insulin following suspension led to worse performance in preventing hypoglycemia.
The PLGS system was ineffective in reducing hypoglycemia in the first 3 hours after activation. We speculated that high insulin on board contributed to the inability of the PLGS system to reduce early hypoglycemia, but the ineffectiveness of the system did not differ for any factor subgroup. This could be attributed to the low rates of early hypoglycemia on control nights (only 8% of control nights had hypoglycemia in the first 3 hours), making it difficult to reduce the nocturnal hypoglycemia rate further. Many studies utilize a warm-up period13,14 or have patients wear the system continuously. 15 Further research is warranted to assess whether continuously worn systems are more effective in the early part of the night.
A limitation of the study was that exercise intensity and bedtime snack were self-reported. A guideline for exercise intensity was given to subjects, but exercise duration, snack size, and snack composition were not reported. Three-way interactions were not assessed, so it is possible that a combination of 2 factors could influence the efficacy of the PLGS system. The study consisted of patients aged 4 to 45 years old with an A1C ≤8.5%, and the results may not be generalizable outside this domain. In addition, the findings may not generalize to closed-loop or other PLGS systems. Sensor inaccuracies potentially diluted the treatment effect, but would not affect the probability of a type 1 error. A major strength of the study is the large sample size. The data consisted of 5028 randomized nights from 127 patients thereby making it possible to assess the efficacy and safety of the system under a variety of conditions. The novel randomization scheme where the treatment assignment (control or intervention) was randomized each night minimized bias that could be attributed to a patient being aware of the treatment assignment or changing their diabetes management.
The PLGS system safely and effectively reduced hypoglycemia for all patient-level and night-level factor subgroups. The ability of the PLGS system to reduce hypoglycemia without increasing overnight hyperglycemia under a variety of conditions illustrates the system is robust and comprehensive.
Footnotes
Acknowledgements
We would like to recognize the efforts of the participants and their families and thank them. We also would like to recognize Martin Cantwell, BSC, Medtronic MiniMed, Inc, Northridge, CA, Werner Sauer and Denny Figuerres, Jaeb Center for Health Research, Tampa, FL, for their significant engineering contributions.
Abbreviations
A1C, hemoglobin A1c; CGM, continuous glucose monitor; PLGS, predictive low-glucose suspend system; ROC, rate of change.
Authors’ Note
The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Diabetes and Digestive and Kidney Diseases or the National Institutes of Health. In Home Closed Loop Study Group: Clinical Centers: Listed with clinical center name, city, and state; personnel are listed as PI for principal investigator, (I) for coinvestigator, (C) for coordinator, and (O) for other personnel: Division of Pediatric Endocrinology and Diabetes, Stanford University, Stanford, CA: Bruce A. Buckingham, MD (PI); Darrell M. Wilson, MD (I); Trang Ly, MD (I); Tandy Aye, MD (I); Paula Clinton, RD (C); Kimberly Caswell, RD (C); Jennifer Block, RD (C); Breanne P. Harris (O); Barbara Davis Center for Childhood Diabetes, University of Colorado, Denver, CO: H. Peter Chase, MD (PI); David M. Maahs, MD, PhD (I); Robert Slover, MD (I); Paul Wadwa, MD (I); Dena Gottesman (C); Laurel Messer, RN, CDE (C); Emily Westfall, BA (O); Hannah Goettle, BA (C); Jaime Realsen (C); St. Joseph’s Health Care, London, ON: Irene Hramiak, MD, FRCP (PI); Terri Paul, MD, MSc, FRCPC (I); Marsha Driscoll, BScN, RN, CDE (C); Sue Tereschyn, RN, CDE, CCRA (O); Children’s Hospital, London Health Sciences Centre, London, ON: Cheril Clarson, MD (PI); Robert Stein, MD (I); Patricia Gallego, MD (I); Margaret Watson, RD (C); Keira Evans (O); Rensselaer Polytechnic Institute, Troy, NY: B. Wayne Bequette, PhD (PI); Fraser Cameron, PhD (I); JDRF Canadian Clinical Trial Network: Olivia Lou, PhD (O). Coordinating Center: Jaeb Center for Health Research, Tampa, FL: Roy W. Beck, MD, PhD (PI); John Lum, MS; Craig Kollman, PhD; Dan Raghinaru, MS; Judy Sibayan, MPH, Nelly M. Njeru; Denny Figuerres; Carlos Murphy; Werner Sauer; Jennifer Lott. Data and Safety Monitoring Board: John C. Pickup, BM, DPhil (chair), Irl Hirsch, MD; Howard Wolpert, MD.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Continuous glucose monitors and sensors were purchased at a bulk discount price from Medtronic MiniMed, Inc (Northridge, CA). Bayer HealthCare, LLC, Diabetes Care provided blood glucose meters, test strips, control solution, and lancets as product support through an investigator sponsored research grant. Home ketone meters and test strips were provided by Abbott Diabetes Care, Inc. The companies had no involvement in the design, conduct, or analysis of the trial or the manuscript preparation. PMC has no disclosures. BAB reports grants from NIDDK, during the conduct of the study; grants, personal fees, and nonfinancial support from Medtronic MiniMed, Inc, personal fees from Sanofi, personal fees from Tandem, personal fees from Novo-Nordisk, personal fees from Animas, outside the submitted work. In addition, BAB reports a Kalman filter based hypoglycemia prevention algorithm patent is pending. DMM reports grants from American Diabetes Association- Medtronic MiniMed, Inc. IH reports grants from JDRF-Federal Development funding during the conduct of the study, as well as grants, personal fees, and nonfinancial support from Abbott, from AstraZeneca/Bristol Myers Squibb; personal fees and nonfinancial support from Boehringer Ingelheim, grants, personal fees, and nonfinancial support from Eli Lilly, grants, personal fees, and nonfinancial support from Janssen-Ortho/Johnson & Johnson (JNJ), personal fees and nonfinancial support from Medtronic MiniMed, Inc, grants, personal fees, nonfinancial support, and other from Merck, grants, personal fees, and nonfinancial support from Novo Nordisk, grants from Pfizer, grants, personal fees, and nonfinancial support from Sanofi Aventis, outside the submitted work; DMW reports research supplies support from LifeScan, Inc and Medtronic MiniMed, Inc and a Kalman filter based hypoglycemia prevention algorithm patent pending. T. Aye has no disclosures. PC has no disclosures. HPC reports grants from Dexcom during the conduct of the study, as well as a Kalman filter based hypoglycemia prevention algorithm patent is pending. LM has no disclosures. RWB reports grants from NIH and from JDRF during the conduct of the study. CK reports consultant fees from Medtronic MiniMed, Inc. RWB reports grants from NIH and from JDRF during the conduct of the study. JWL has no disclosures.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project described was supported by the National Institute of Diabetes and Digestive and Kidney Diseases (R01DK085591), grants from JDRF (22-2013-266), and the JDRF Canadian Clinical Trial Network (CCTN), which is a public-private partnership including JDRF International, JDRF-Canada (JDRF-C), and the Federal Economic Development Agency for Southern Ontario (FedDev Ontario); and is supported by JDRF 80-2010-585.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.