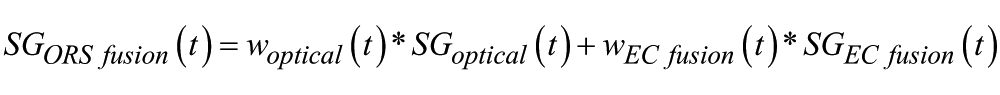Abstract
Background:
Orthogonal redundancy for glucose sensing (multiple sensing elements utilizing distinct methodologies) may enhance performance compared to nonredundant sensors, and to sensors with multiple elements utilizing the same technology (simple redundancy). We compared the performance of a prototype orthogonal redundant sensor (ORS) combining optical fluorescence and redundant electrochemical sensing via a single insertion platform to an electrochemical simple redundant sensor (SRS).
Methods:
Twenty-one adults with type 1 diabetes wore an ORS and an SRS concurrently for 7 days. Following sensor insertion, and on Day 4 with a standardized meal, frequent venous samples were collected for reference glucose measurement (laboratory [YSI] and meter) over 3 and 4 hours, respectively. Between study visits reference capillary blood glucose testing was undertaken. Sensor data were processed prospectively.
Results:
ORS mean absolute relative difference (MARD) was (mean ± SD) 10.5 ± 13.2% versus SRS 11.0 ± 10.4% (P = .34). ORS values in Clarke error grid zones A and A+B were 88.1% and 97.6%, respectively, versus SRS 86.4% and 97.8%, respectively (P = .23 and P = .84). ORS Day 1 MARD (10.7 ± 10.7%) was superior to SRS (16.5 ± 13.4%; P < .0001), and comparable to ORS MARD for the week. ORS sensor survival (time-averaged mean) was 92.1% versus SRS 74.4% (P = .10). ORS display time (96.0 ± 5.8%) was equivalent to SRS (95.6 ± 8.9%; P = .87).
Conclusions:
Combining simple and orthogonal sensor redundancy via a single insertion is feasible, with accuracy comparing favorably to current generation nonredundant sensors. Addition of an optical component potentially improves sensor reliability compared to electrochemical sensing alone. Further improvement in optical sensing performance is required prior to clinical application.
Keywords
For people with type 1 diabetes (T1D), matching insulin delivery to their varying insulin requirements is central to maximizing quality of life and minimizing diabetes complications.1,2 In contrast to glucose meters, 3 continuous glucose monitoring (CGM) systems measuring interstitial fluid (ISF) glucose provide near-continuous real-time glucose information. 4 CGM improves glycemia for adults with T1D5,6 and provides a vital component in artificial pancreas development. 7 However, CGM technology remains less accurate than the most accurate glucose meters, 8 with mean absolute relative differences (MARDs) of current generation CGM between 10 and 18%. 9 To replace fingerstick measurement, and to achieve sufficient functionality for use in an artificial pancreas system, CGM needs improved accuracy and reliability.
CGM technology refinements have increased accuracy, reliability and ease of use.10,11 However, configurations involving a single electrochemical sensor linked to a processing algorithm remain the basis of current commercially available CGM.9,12 Performance limitations of current sensors include sensor delay and drift, biofouling, tissue hypoxia, and mechanical factors affecting the sensor-tissue interface.10,11,13 Nonelectrochemical CGM modalities including optical technologies have been explored,10,11 though none are currently commercially available. One promising optical methodology is fluorescence-based sensing involving reversible competitive binding between a fluorophore-labeled glucose receptor and a glucose analog. 14 Relative to electrochemical sensing, optical fluorescence sensing has potential advantages including greater sensitivity to low glucose levels, less requirement to recalibrate in response to peri-sensor tissue reactions, and the option of noninvasive sensing.15,16 Potential disadvantages of fluorescence methodology are sensor damage due to foreign body responses and sensitivity to local pH affecting dye response.15,16
Sensor redundancy refers to multiple sensing elements measuring the same glucose output. This can involve separate sensors worn concurrently, 17 or multiple sensing elements on a single platform. 18 Simple redundancy refers to multiple sensors utilizing the same methodology, which to date has been via a glucose oxidase-based electrochemical approach.17,18 In contrast, orthogonal redundancy combines multiple glucose sensors utilizing different methods to quantify glucose. These different methods have independent failure modes, and local physiological conditions impact sensor performance differently.10,11,13 For example, electrochemical sensor accuracy is affected by differences between ISF and the peri-sensor wound fluid created by sensor insertion. Wound fluid produces a large amount of peroxide, which could potentially affect the reaction kinetics of the glucose oxidase-based chemistry. In contrast, in vitro studies have shown that peroxide formation has little effect on the optical sensing mechanism. Combining separate sensing elements utilizing glucose oxidase and optical fluorescence into a single platform fulfils orthogonal redundancy criteria. While studies evaluating simple or orthogonal redundancy are promising,18,19 a more robust solution may be a sensor incorporating both simple and orthogonal redundancy.
With redundancy, the information from multiple sensor inputs needs to be combined into a unified output. Using an intelligent algorithm to combine sensor data into a single fused glucose output may enhance sensor performance over simple signal averaging. The reliability of each sensor element can be calculated and weighted by the algorithm providing a robust and accurate glucose measurement. Therefore both the sensor properties and the data processing algorithm impact sensor performance.
We hypothesize that addition of a redundant orthogonal sensing component to simple electrochemical redundancy combined with an intelligent processing algorithm is feasible and may enhance glucose sensor reliability performance relative to simple redundancy alone. We compared the glucose sensing performance of a prototype orthogonal redundant sensor (ORS) integrating simple plus orthogonal redundancy via a single insertion to a prototype electrochemical simple redundant sensor (SRS) in adults with T1D.
Methods
This study, conducted from November 2014 to March 2015 at St Vincent’s Hospital Melbourne, Australia, was approved by the hospital ethics committee. Each participant provided written informed consent.
Investigational Sensor Design
The prototype ORS (Medtronic, Northridge, CA) integrates two distinct glucose sensing technologies, electrochemical and optical, onto a single sensing platform (Figure 1). Sensing elements are colocated within a single 21 gauge needle for insertion at 45° using a dedicated insertion device. The electrochemical sensing element (length 10 mm, width 0.5 mm) utilizes glucose oxidase methodology and comprises two independent working electrodes (WE1 and WE2) which estimate glucose levels independently, thereby providing simple redundancy. These two WEs are in a fold-over design, with sensing electrodes on both planar surfaces. The optical fluorescence sensing element (length 11 mm, width 0.25 mm) incorporates an assay surrounded by a glucose permeable membrane, allowing within-assay glucose to equilibrate with glucose in surrounding tissue (Figure 2). The assay incorporates a glucose analogue (dextran), and a glucose receptor (mannose binding lectin [MBL]) which is fluorophore-labeled to impart fluorescence. Equilibrium between MBL bound to glucose and dextran, respectively, determines fluorescence intensity. A nonglucose sensing macromolecule labeled with another fluorophore serves as an internal reference. The ratio of the assay and reference intensities is converted into a glucose concentration.
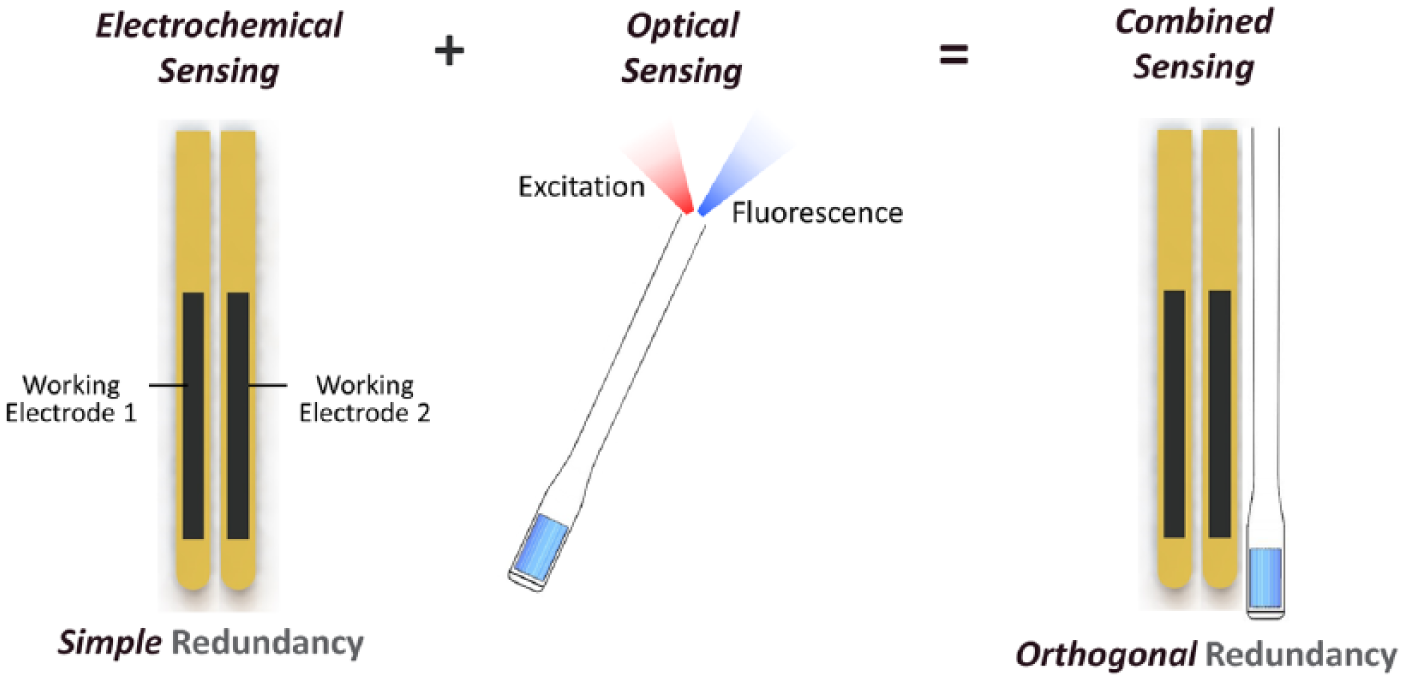
Orthogonal redundant sensing schematic representation showing the combination of redundant electrochemical and optical sensing to provide simple plus orthogonal redundancy.
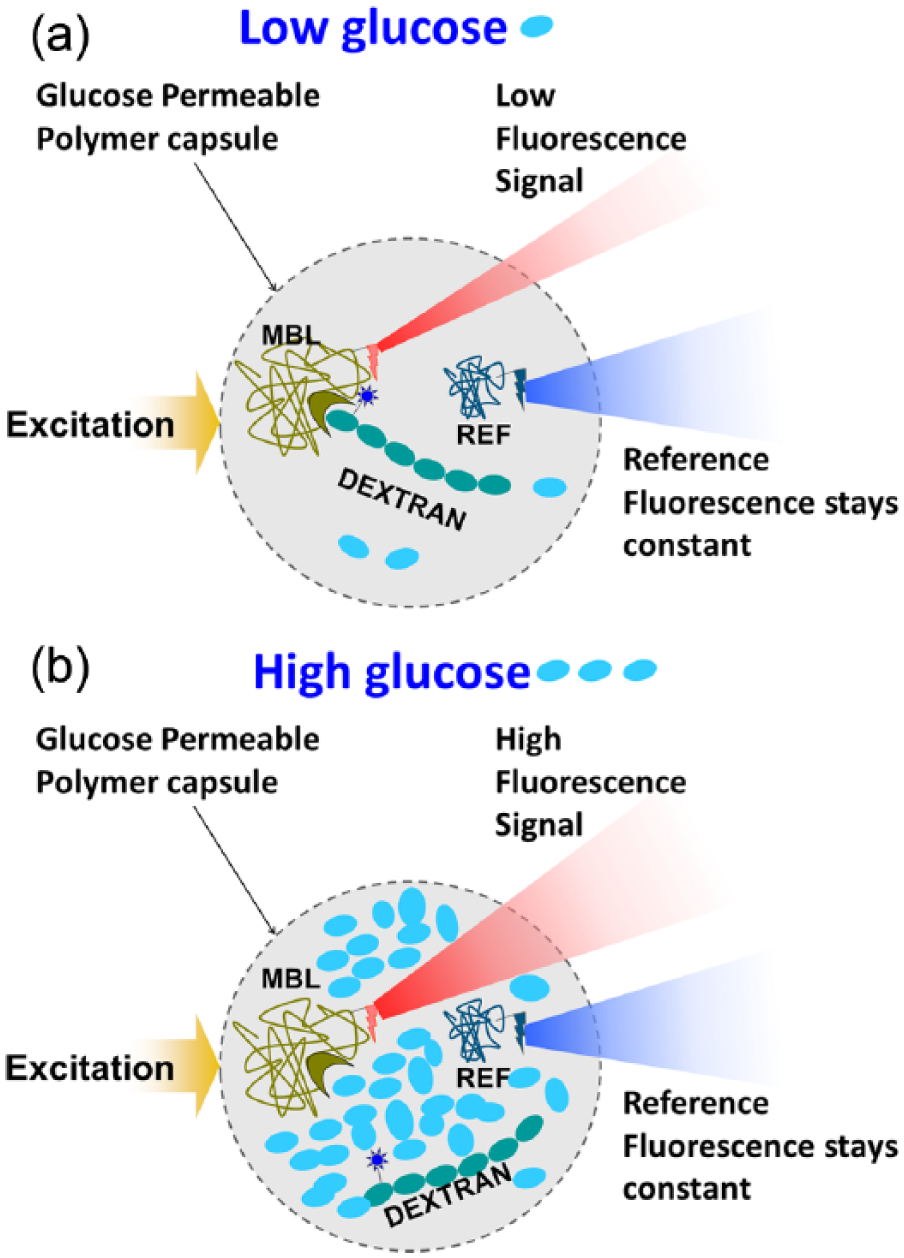
Optical sensing chemistry diagram demonstrating assay functionality in (a) low and (b) high glucose ranges.
The ORS was connected to a glucose sensor recorder (GSR) which was uploaded following sensor explant. An algorithm analyses data from the two sensing elements, calculating a final fused sensor glucose (SG) output (Figure 3). The ORS algorithm has been designed using prior animal data, published clinical data, 14 and in-house electrochemical sensor data. The algorithm emulates real-time sensor use as data are all processed prospectively (ie, only preceding data are used by the algorithm). To calculate the fused SG the algorithm processes the redundant electrochemical and optical sensing elements in parallel.
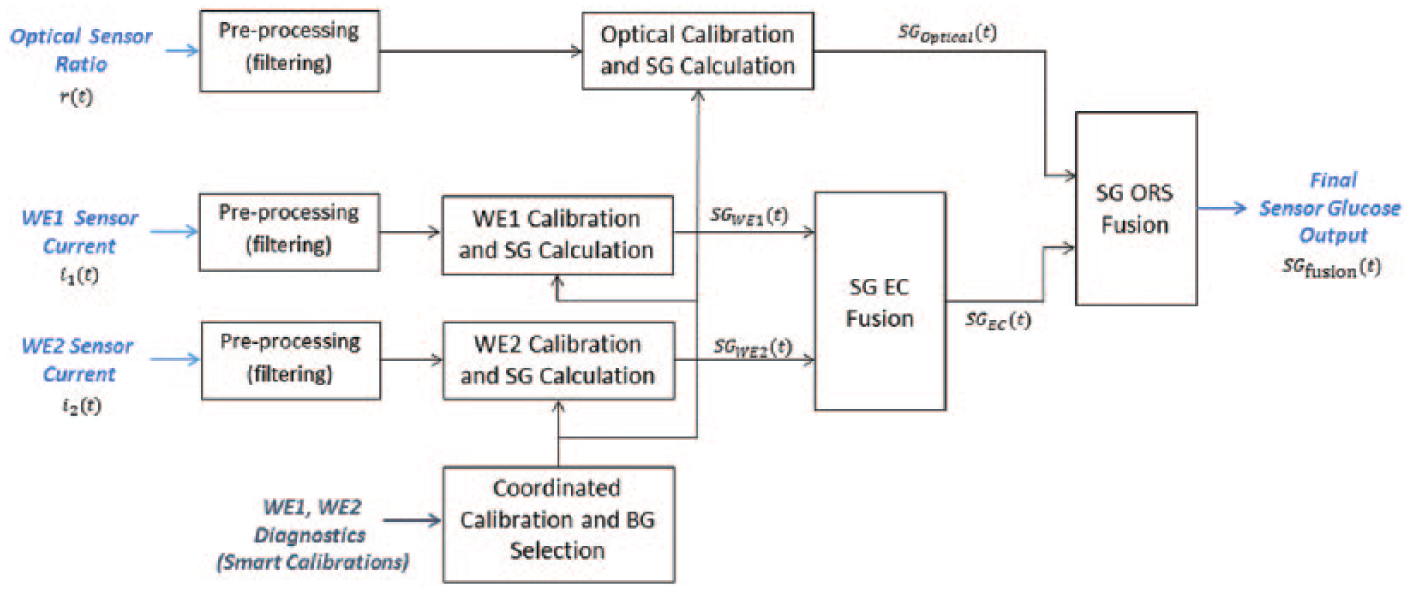
ORS signal processing algorithm schematic flow.
For electrochemical components independent currents from both WEs (
where
Concurrently, for the optical element, the optical ratio (
Four reference blood glucose values per day are used for calibration of both sensing elements. The final ORS SG fusion
where
Comparator Sensor Design
The comparator SRS is a single element (length 10 mm, width 0.5 mm) with two electrochemical WEs in a fold-over design, similar to the ORS electrochemical element, inserted via a single 26 gauge needle at 90° using a dedicated insertion device.
SG values are displayed only when predetermined trace characterization algorithms determine the ISF glucose signal quality is adequate. Sensor performance was calculated using a combination of plasma glucose (PG) and meter glucose (MG) reference values as detailed below.
Participants
Participants with T1D were aged 21 years and over, and had CGM experience. Those intolerant to adhesives, with a skin condition in the area of sensor placement, and pregnant females were excluded.
Experimental Protocol
Each participant wore an ORS and an SRS concurrently for 1 week (164-168 hours). Participants were masked to sensor data, and continued their usual diabetes care.
Visit 1: Sensor insertion
Following baseline clinical data collection, an ORS and an SRS were inserted into contralateral abdominal subcutaneous fat and connected to a GSR. An antecubital vein cannula was inserted for a frequent sampling test (FST) comprising 9 venous samples at standardized 15- and 30-minute intervals over 180 minutes. Following FST completion, participants departed the trial center to continue their usual activities. Participants were instructed to undertake fingerstick blood glucose testing at least 6 times per day using the study meter (described below).
Visit 2: Meal test
On Day 4 participants returned to the trial center fasting. An antecubital vein cannula was inserted for an FST comprising 17 venous samples at 15-minute intervals over 240 minutes. Sixty minutes after FST commencement participants consumed a standardized 65 g carbohydrate meal.
Visit 3
Participants returned to the trial center for sensor explants 164-168 hours postinsertion, followed by assessment of skin at sensor placement areas.
Reference Blood Glucose Measurement
Laboratory PG was quantified from FST blood using the glucose oxidase method (YSI 2300 STAT Plus™ glucose analyser; YSI Life Sciences, Yellow Springs, OH).
MG was measured by a Contour® Next Link glucose meter (Bayer, Tarrytown, NY) using the glucose oxidase method with venous blood during the FSTs and capillary blood for the remainder. This meter meets the ISO 15197:2013 standard. 20
Sensor Data Analysis
Descriptive data analyses for aggregate and individual sensor performance were undertaken. ORS data were also analyzed by sensing modality. As the study tested sensor performance, recorder, connection and calibration failures are excluded.
Accuracy, compared to reference PG and MG values, was assessed by (1) mean absolute relative difference (MARD); (2) proportion of points meeting ISO 15197:2013 standard; 20 (3) Clarke error grid analysis (EGA); 21 and (4) proportion of points within 20 mg/dL and 40 mg/dL of reference for glucose ≤80 mg/dL, and within 20% and 40% of reference for glucose >80 mg/dL (known as 20/20 and 40/40 agreement, respectively). Within the low glucose range (40-80 mg/dL), accuracy was assessed by mean absolute difference. Reliability was assessed by display time (the proportion of time the algorithm displayed a glucose value), and lifetime of functioning sensor (presented as time-averaged mean survival, and by Kaplan–Meier plot). Premature sensor loss was defined as functionality <164 hours. Comparison testing was performed via 2-tailed unpaired t-test for continuous data with normal distribution, log-rank test for survival distribution, and chi-square test (or Fisher’s exact test when samples sizes were small) for categorical data. Statistical significance was taken at P < .05.
Sensor insertion site appearance was assessed visually postexplant.
Results
All participants (9 males, 12 females) completed the protocol. Their clinical characteristics (mean ± SD) were age 54 ± 13 years, body mass index 27.5 ± 5.7 kg/m2, diabetes duration 29 ± 13 years, HbA1c 7.3 ± 0.8% (56 ± 8 mmol/mol), total daily insulin 61 ± 49 units. Sensor data were not recorded for n = 1 ORS (connection failure) and n = 2 SRS (recorder failures).
Study Glucose Meter Performance
There were 522 study meter glucose values with paired PG. These MG points had a MARD of 3.9% compared to PG reference. EGA showed 100% (98.9%) of MG points were in zones A+B (A), and 98.3% were within ISO 2013 boundaries, thereby meeting the ISO standard.
Sensor Accuracy
ORS and SRS performance data benchmarked against 1,314 and 944 paired MG evaluation points, respectively, are summarized in Table 1. Overall MARD (mean ± SD) for ORS was 10.5 ± 13.2% versus SRS 11.0 ± 10.4% (P = .34). ORS MARD during the 24 hours postinsertion (10.7 ± 10.7%) was superior to SRS (16.5 ± 13.4%; P < .0001). The 20/20 agreement, 40/40 agreement, and proportion within ISO boundaries did not differ significantly between ORS and SRS. There was no significant difference between ORS and SRS performance in high or low glucose ranges. The proportions of ORS values within Clarke EGA zone A and zones A+B were 88.1% and 97.6%, respectively, versus SRS 86.4% and 97.8%, respectively (P = .23 and P = .84; Figures 4a and 4b).
Sensor Performance Data (Versus MG).
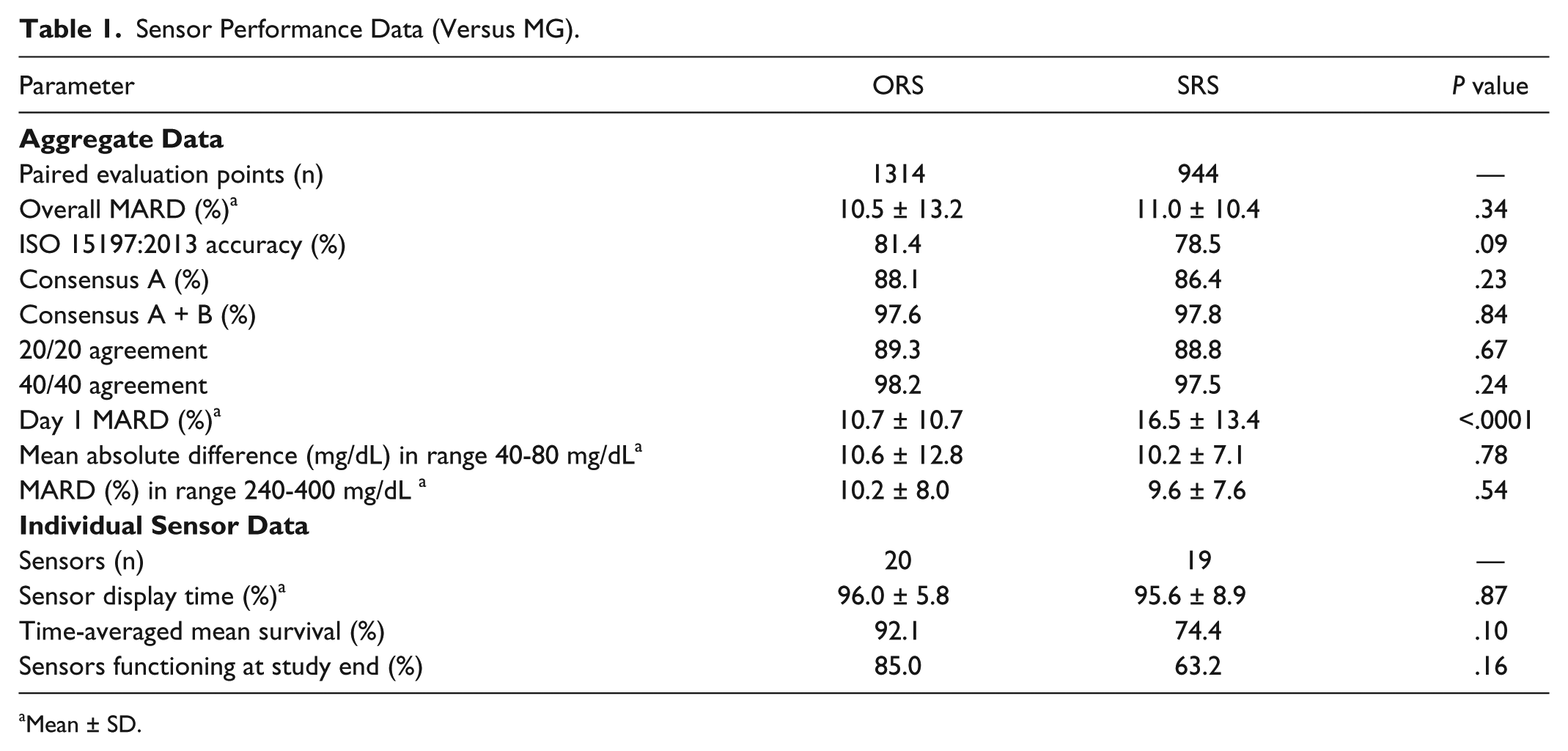
Mean ± SD.
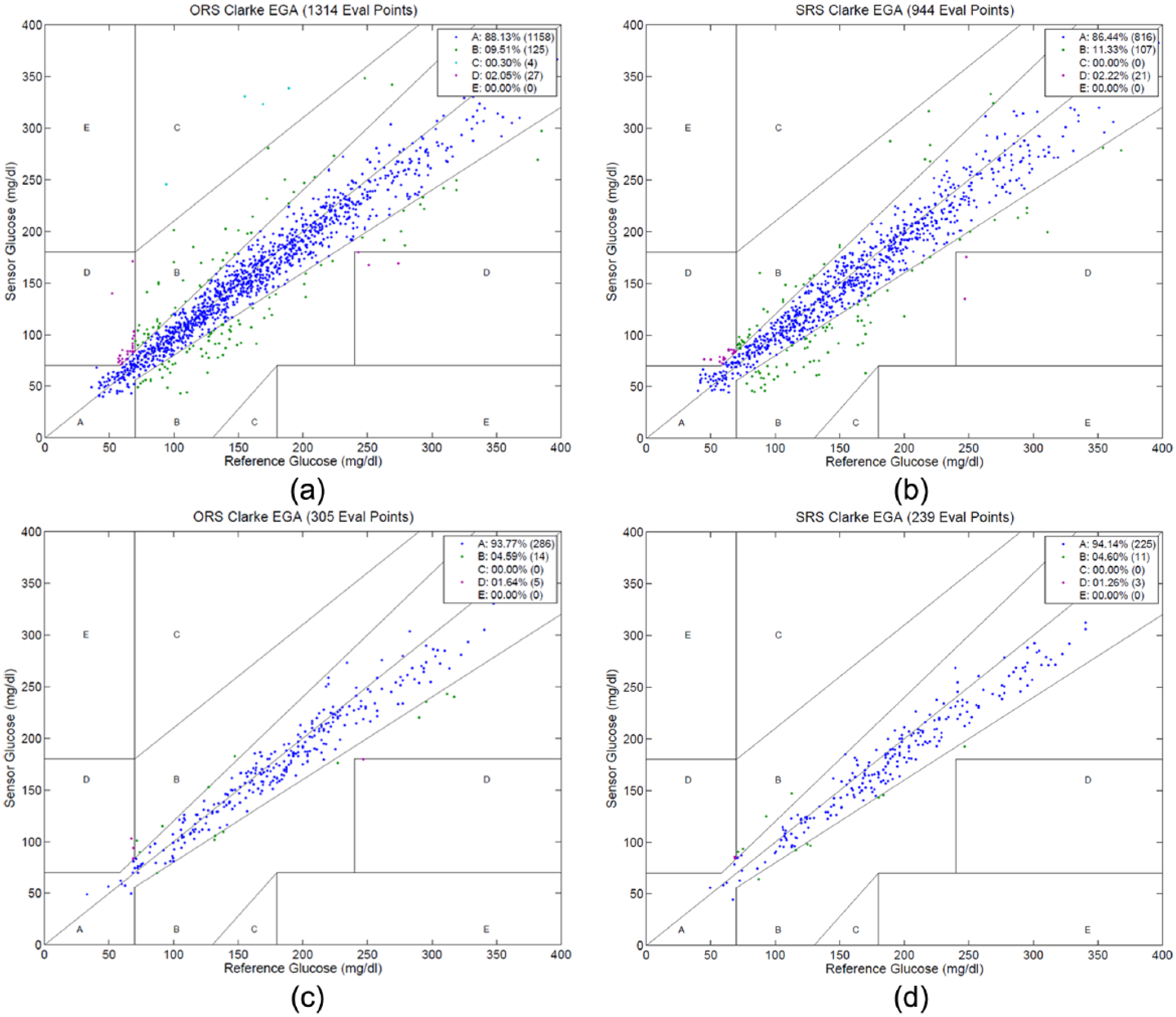
Clarke error grid analyses for (a) ORS overall versus MG; (b) SRS overall versus MG; (c) ORS Day 4 FST versus PG; (d) SRS Day 4 FST versus PG.
The meal FST ORS and SRS performance data benchmarked against 305 and 239 paired PG evaluation points, respectively, are summarized in Table 2. Surviving sensor MARD for ORS (8.1 ± 7.3%) was not significantly different to SRS (8.6 ± 6.4%; P = .46). Meal FST EGA zone A and zones A+B (Figures 4c and 4d), 20/20 agreement, 40/40 agreement, and proportion within ISO boundaries did not differ significantly between ORS and SRS. There was no significant difference between ORS and SRS Day 4 FST performance in high or low glucose ranges.
Sensor Performance Data for Day 4 FST (Versus PG).
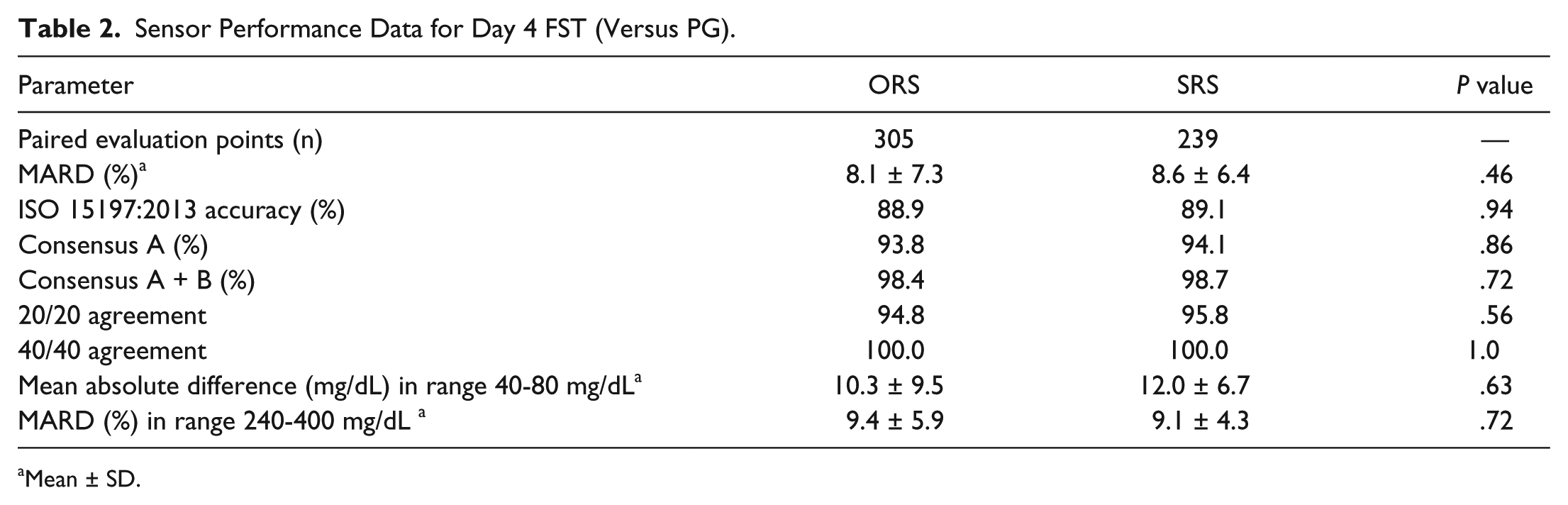
Mean ± SD.
The separate performances of the ORS electrochemical and optical components benchmarked against MG are presented in Table 3 to demonstrate the relative contributions of each sensing modality. Of note, the ORS design is intended to provide a single fused SG output as per Figure 3. The ORS electrochemical component had superior accuracy to the ORS optical component with MARD 9.4 ± 9.5% versus 32.7 ± 38.7%.
ORS Performance Data by Sensing Modality (Versus MG).
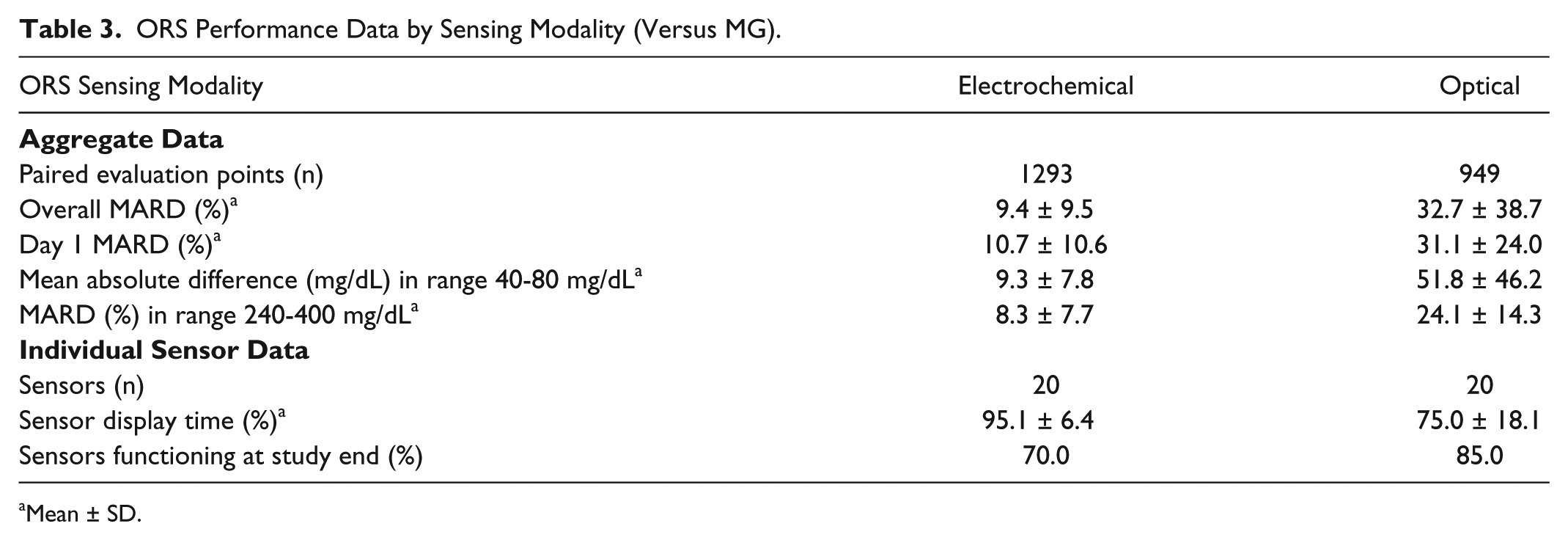
Mean ± SD.
MARD distribution for individual sensors favored ORS (10.6 ± 2.0%) over SRS (12.0 ± 4.2%), though did not reach statistical significance (P = .18; Figure 5).
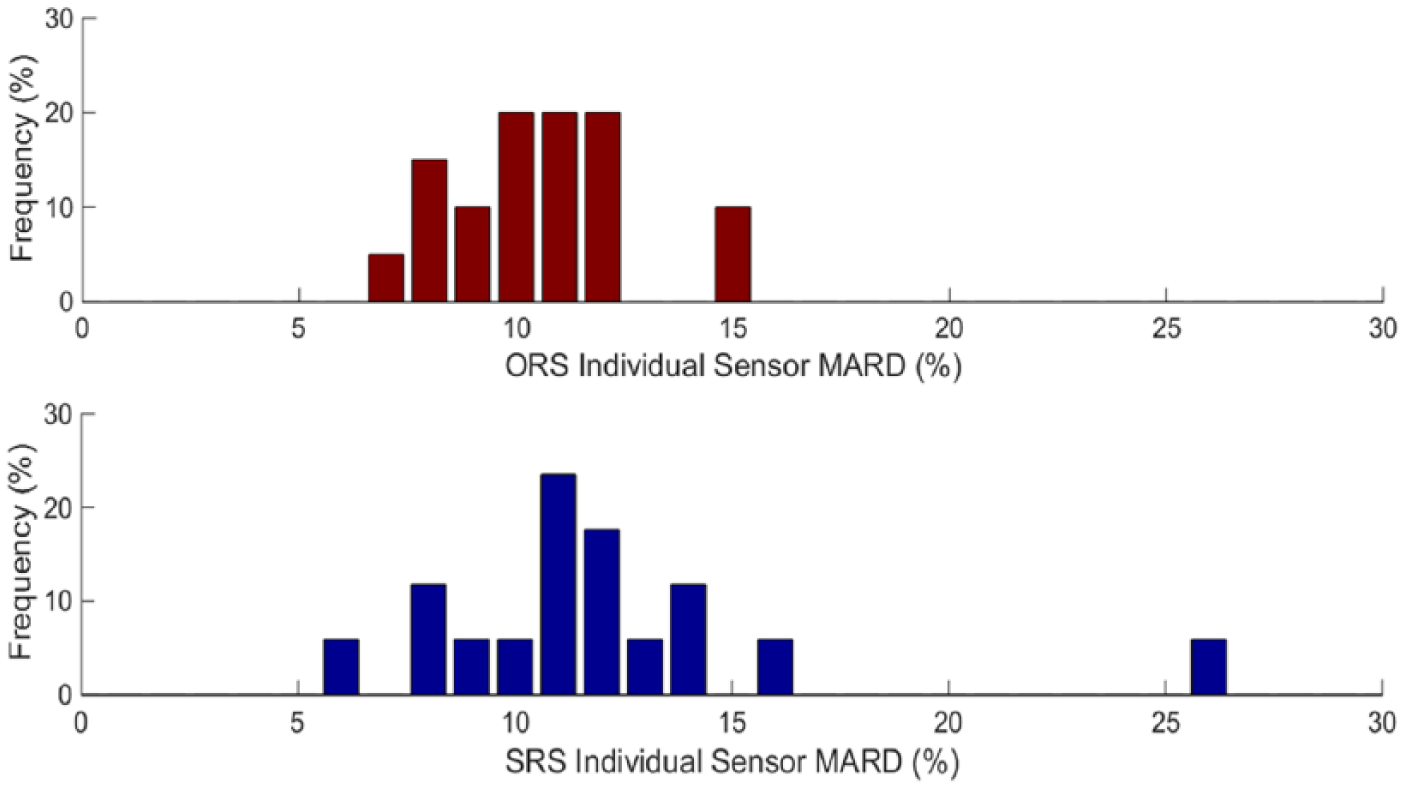
Frequency distribution plot for individual sensor MARD.
Sensor Reliability
There was a trend toward less premature sensor loss (time-averaged mean survival) with ORS (92.1%) than SRS (74.4%; P = .10; Figure 6). The proportion of sensors functioning at study-end for ORS was 85.0% versus SRS 63.2% (P = .16). Sensor display time was not different for ORS versus SRS. The ORS electrochemical component contributed the majority of the ORS display time.
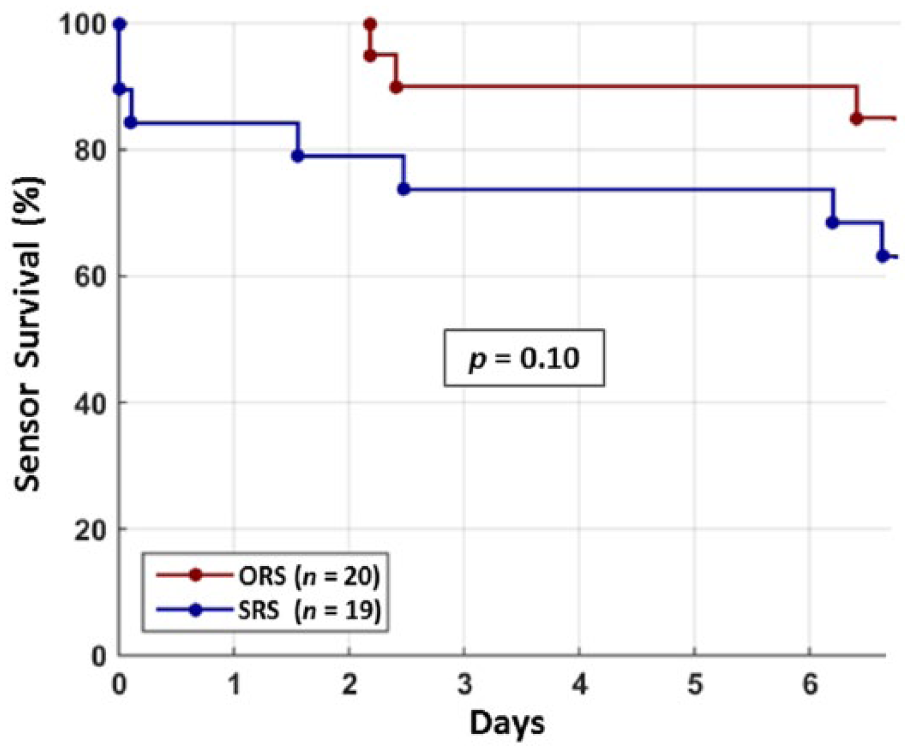
Kaplan–Meier plot for sensor lifetime.
Sensor Insertion Site Complications
Qualitative data indicated all sensors were well-tolerated, with no evident sensor insertion site irritation or infection following explant.
Discussion
This is the first human study to demonstrate the feasibility of a clinically acceptable single insertion platform that combines a redundant electrochemical sensor with an optical fluorescent sensor. The ORS output in our study represents data processed by an intelligent algorithm assessing signal quality from each of three components (optical, WE1, WE2), weighted by quality and stability to provide a fused output, thereby providing true orthogonal redundancy. ORS accuracy compares favorably to current nonredundant sensors. 9
Previous reports of optical fluorescence glucose sensing predominantly involve in vitro studies, animal studies, and inpatient platforms such as intravascular sensing.22-24 This study represents a progression to a clinically applicable in vivo subcutaneous fluorescent sensing platform. The accuracy of the optical sensing component overall, in high and low glucose range (MARDs 32.7%, 24.1%, and mean absolute difference 51.8 mg/dL, respectively; Table 3) was considerably inferior to the accuracy of the both the ORS electrochemical component and to the SRS (overall MARDs 9.4% and 11.0%, respectively), and to current-generation nonredundant electrochemical sensors. 9 Limitations of subcutaneous fluorescent sensing include foreign body response-related sensor damage, and local pH or tissue oxygen tension affecting dye response. 15 Subcutaneous fluorescence sensors are not yet viable as a stand-alone clinical platform, and are most clinically applicable to be incorporated as an adjunct, that is, as a redundant component in combination with a more mature method for measuring glucose. With further development fluorescent sensor technology autonomy may become possible.15,16
The goal of redundancy in glucose sensing is improved reliability and accuracy. Early approaches to sensor redundancy involved separate insertions of identical nonredundant sensors with nonintelligent processing algorithms (signal averaging, principal component analysis). 17 Patient acceptance and adherence may be compromised by multiple sensor insertion systems. A recent study utilizing simple electrochemical redundancy via single insertion of a dual-electrode sensor with intelligent algorithm processing showed improved accuracy and reliability compared to current generation nonredundant systems. 18 Although simple redundancy improves accuracy, each electrochemical electrode is subject to the same potential interferences, including local tissue reactions necessitating frequent calibration, acetaminophen use and ascorbic acid supplementation.10,11 An alternative sensing methodology may not be as vulnerable to these factors. An optical sensing component in an orthogonal configuration may address these limitations. Colocation of sensing elements, even with differentiated sensing modalities, may remain subject to site-specific failure modes common to multiple modalities, such as tissue trauma at the insertion site or insertion-related thrombus.
The overall ORS performance largely reflected performance of its glucose oxidase-based electrochemical redundant sensing component, which while itself a prototype utilizes an established glucose-measuring method. 25 The current ORS configuration had an overall MARD of 10.5% versus meter, and meal FST MARD 8.1% versus PG. This compares favorably to an independent assessment of three nonredundant electrochemical CGMs on the market (MARDs versus PG: Dexcom G4 Platinum 10.8%, Abbott Freestyle Navigator 12.3%, and Medtronic Enlite 17.9%). 9 ORS Day 1 accuracy (MARD 10.7%) was equivalent to ORS accuracy over the full week. This is notable as performance during Day 1 postinsertion is a recognized challenge in sensor design,26,27 which may relate to time until full sensor hydration. Within the ORS processing algorithm, early postinsertion challenges may be mitigated by ORS algorithm diagnostics fusing the two electrochemical signals, thereby deemphasizing a sensing element which is not yet stabilized. ORS display time (96.0%) and time-averaged mean survival (92.1%) were inferior to commercially available products, 9 likely relating to the structural configuration of the prototype, which requires refinement.
In this study ORS and SRS sensors had similar overall accuracy (MARDs 10.5% and 11.0%, respectively). However, the SRS had inferior Day 1 accuracy, and an unexpectedly high early failure rate (Figure 6). The Day 1 accuracy and early failure rate are related as the processing algorithm suspends sensor function if the signal quality fails to meet predetermined benchmarks, therefore only surviving sensors contribute to subsequent accuracy metrics. The difference in Day 1 MARD (calculated using all available data points following insertion until 24 hours postinsertion) and early survival could not be accounted for by the ORS optical signal as the fluorescence component requires ~10 hours to initialize, compared to 1 hour for the electrochemical component. It may be that structural differences in the ORS and the SRS contributed to the early postinsertion performance observations. The dual-filament (optical and electrochemical) ORS may have greater structural integrity than the single-filament SRS, and different sensor insertion angles (45° for ORS versus 90° for SRS) and length (11 mm versus 10 mm) may have conferred structural protection against trauma and stress forces upon insertion. These factors may have impacted initial sensor performance. Reassuringly the individual sensor MARD frequency distribution (Figure 5) demonstrates no poorly performing ORS outliers. The outlier SRS could clinically compromise artificial pancreas application in which insulin is being both delivered and withheld based on sensor input.
We note that the performance of the comparator SRS herein compared unfavorably with the performance of another similar prototype. 18 These two sensor prototypes represent discrete iterations with manufacturing differences in the chemistry stack leading to different performance parameters. In addition, the sensor-recorder connection in the SRS may not have been sufficiently robust in this redundant electrochemical prototype to reproduce results reported elsewhere.9,18 Nevertheless, a comparison of survival of the optical versus electrochemical components of ORS, despite limitations in accuracy, suggests greater durability of the optical component. We propose that a potential benefit of incorporating optical technology into a redundant design is improved reliability. From a clinical perspective this may enable artificial pancreas systems to remain in closed-loop mode a greater proportion of the time.
In addition to ORS performance findings, this study provided mechanical learnings for further development. For example, suboptimal connections contributed to relatively high ORS MARDs through increased noise and reduced signal stability, and led to one ORS GSR not recording data. The 21 gauge ORS needle is less favorable for wearers than the 26 gauge SRS needle; there may be potential to down-size the ORS needle in the future, though miniaturized prototypes are yet to undergo clinical testing. The ORS requirement of 4 blood glucose calibrations per day is a limitation compared to commercially available sensors, and this will be addressed in future iterations.
The ORS involves multiple novel technologies successfully combined into a single platform for this proof-of-concept study. However, further refinement of optical fluorescence glucose sensing is required to improve accuracy of the optical component for this technology to become clinically viable. The next ORS iteration (undergoing rodent studies) incorporates improved optical chemistry providing a more robust and predictable optical sensor, thereby increasing its contribution to fused data. In detail, this improved sensing methodology follows the Singaram approach utilizing fluorescence for glucose detection via a boronic acid derivative and a fluorescent dye. 28 The two-component system is entrapped in a hydrogel matrix; when glucose molecules are present it changes the charge on the boronic acid derivative, making it less susceptible to ionic binding to the dye thereby reducing the fluorescent transfer. Improved synthetic-based fluorescence chemistry would provide greater hypoglycemia range sensitivity (following Michaelis–Menten kinetics), a faster equilibration assay increasing optical Day 1 contribution, and a more robust device design with greater signal intensity. The redundant electrochemical component of the ORS is also undergoing refinements aimed at improving its structural characteristics. The comparison in this study was designed to explore how simple plus orthogonal redundancy compared to simple redundancy alone. However, the prototype simple redundant sensor used for comparison required further development. Once the ORS configuration is finalized a formal comparison to the best commercially available sensor is needed.
Conclusions
This is the first report demonstrating the feasibility of a novel glucose sensor combining redundant electrochemical sensing with fluorescent sensing via a single insertion platform (providing simple and orthogonal redundancy). Redundant sensor technology is evolving, and we expect the next iteration to be more robust and accurate. Evolution will include development and refinement of the optical sensing component to address the limitations noted in this study.
Footnotes
Acknowledgements
Thank you to our enthusiastic volunteer research participants. We gratefully acknowledge the assistance of Judith Gooley, Andrea Varsavsky, and Marion Jamieson.
Abbreviations
CGM, continuous glucose monitoring; EGA, error grid analysis; FST, frequent sampling test; GSR, glucose sensor recorder; ISF, interstitial fluid; MARD, mean absolute relative difference; MBL, mannose binding lectin; MG, meter glucose; ORS, orthogonal redundant sensor; PG, plasma glucose; SG, sensor glucose; SRS, simple redundant sensor; T1D, type 1 diabetes; WE, working electrode.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Medtronic loaned study equipment and provided technical support for its use. SAM, GMW, and RJM have received grant support for investigator-initiated studies from Medtronic. TTD, AB, SA, and RVS are employees of Medtronic. AJJ and DNO have received honoraria and research support from Medtronic.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from JDRF International, the Leona M. and Harry B. Helmsley Charitable Trust, and the St Vincent’s Hospital Melbourne Research Endowment Fund.