Abstract
In 2 articles of the present issue, Bendini et al report about performance results obtained with 2 blood glucose monitoring systems of the Contour Next platform. Using several analysis methods, the authors demonstrate a very high accuracy, which meets all actual regulatory performance criteria. With consistent MARD results < 5% under daily routine conditions, this meter platform is finally fulfilling the accuracy request as set forth by the American Diabetes Association already in the late 1980s. This meter platform is representative for the successful effort of the device manufacturers who were consequently improving the analytical performance of blood glucose meters during the Past 2 decades, starting with an MARD of 12-15% at the end of the past century and reaching an excellent accuracy < 5% today.
In 2 articles published in the current issue,1,2 Bendini et al report about the performance of the Contour Next USB and the Contour XT in large postmarketing studies in Spain. When employing a modified ISO protocol and using a hexokinase reference method, they observed mean absolute relative deviations (MARD) of 3.5 to 5.0% when analyzing different ranges, including the hypoglycemic range. While this approach is not entirely in line with the current ISO 15197 protocol, 3 it is still an impressive result. In any case, it is a representative proof for the continuous efforts of the device and strip developers and manufacturers over the past 2 decades to improve the performance of their systems.
When in the late 1980s, the ADA defined the desire for self-monitoring blood glucose meters (SMBG) to not deviate more than 5% from the laboratory methods, this was an unreachable target far beyond the capability of any of the existing technology platforms. By searching our own data archive, I was able to visualize the evolution of the Bayer blood glucose meters during the past decades as we experienced it with study results obtained in our laboratories, for example, while performing comparative tests of the commercially available blood glucose meters as requested by different sponsors. The MARDs, observed for the Bayer meters in the course of time and versus different reference methods, are displayed in Figure 1. With all limitation of comparing studies with different protocols, one can clearly see the improvement in technology performance over time.
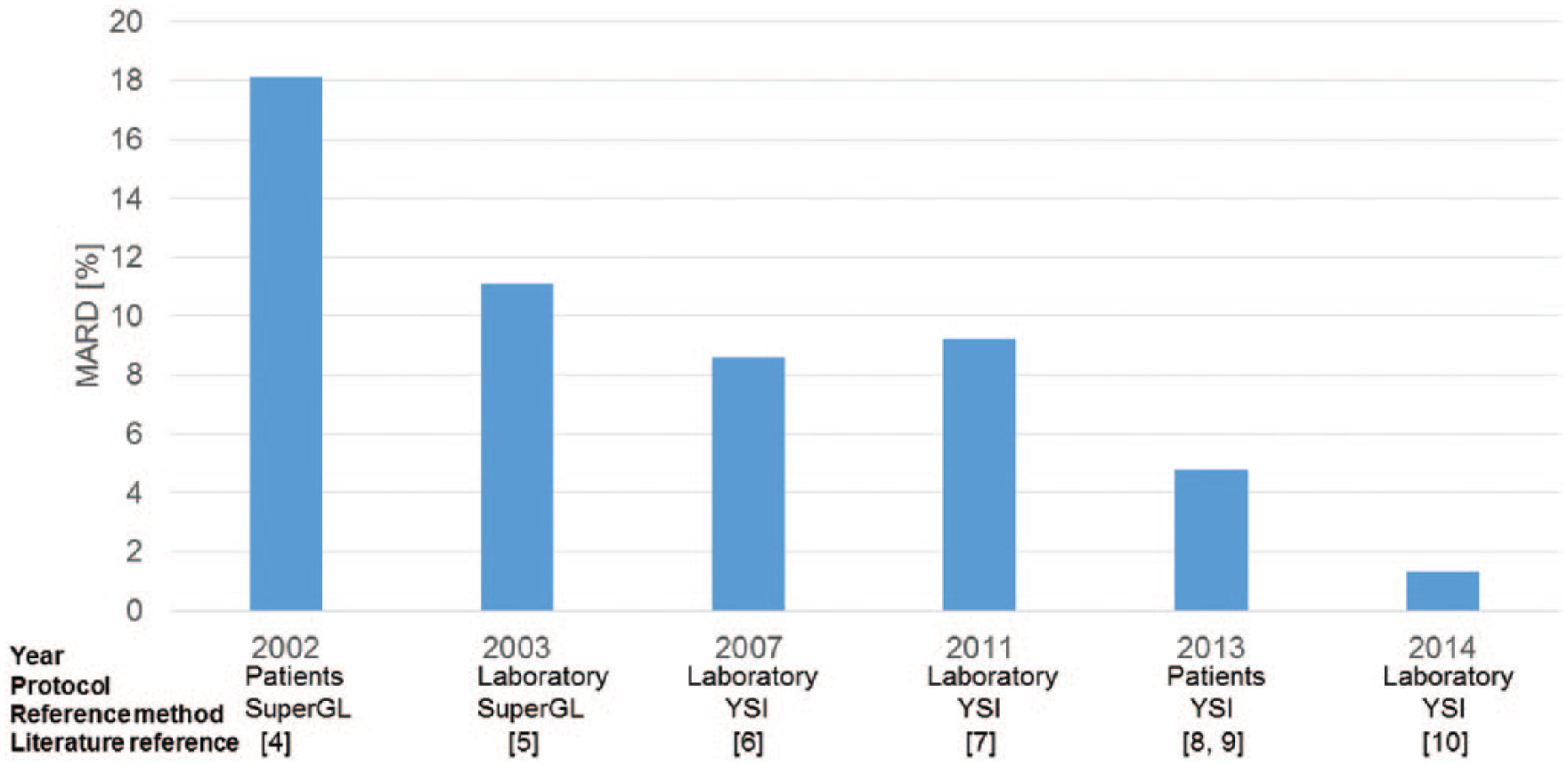
MARD results as assessed in the authors laboratory with meters from the Bayer company by means of different laboratory and patient protocols and in comparison to different reference methods over a period of 15 years.
Our survey started in 2002, when we measured an MARD of 18% with Glukometer Elite in a patient setting using a Glucose Oxidase point-of-care method (SuperGL, Müller Gerätebau, Freital, Germany) as the reference test. 4 A laboratory comparison resulted in an MARD of 11.1% in comparison to the same reference method 1 year later. 5 The following device, Ascensia Contour, showed MARD values against our YSI (Yellow Springs Analyzer) reference device of 8.6% and 6.2%, respectively, in our laboratory setting (reference: YSI).6,7 We saw a major further accuracy improvement with the introduction of the Contour XT (4.8% in patients8,9) and finally our first study with Contour Next, the same device and strip platform tested by Bendini et al,1,2 resulted in an MARD 1.3% with samples processed in our laboratory. 10
One reason, why the accuracy of the Contour Next platform in comparison to other devices has finally reached such a high level, may be seen in the elimination of hematocrit interference by means of a mathematical correction algorithm. The efficacy of employing algorithms similar to dynamic electrochemistry to correct for hematocrit interference is dependent on the determination of specific equation factors derived from laboratory experiments. Improving these factors by performing more laboratory experiments is one possibility to improve the final device accuracy. Already in 2011, the previous Contour meter and strip showed a hematocrit interference factor, which was the third best in the benchmark comparison by our standardized laboratory protocol. 7 In 2015, a similar result of 6.1% put Contour Next into the pole position with respect to hematocrit interference elimination amongst the other meters tested (up to 10% meaning no interference). 10 Other technology advances bringing the meters and strips to such high accuracy standards included, but were not limited to change of strip and electrode design, change of electrode materials, change in the strip chemistry, and improvement of the mathematical algorithms and in the device software.
So about 30 years after the formulation of a wish for an accuracy < 5% for blood glucose meters for patient self-testing by ADA, this request is finally fulfilled. Coming so close to the laboratory accuracy level will have an impact on future accuracy tests. It means that the choice of the reference method and the design of benchmark testing protocols may have a stronger input on future accuracy test results with such accurate meters than the analytical system accuracy of the device systems.
In conclusion, the blood glucose meter and strip platform described in the articles by Bendini et al in this journal1,2 are representations of the highest possible level of clinical accuracy that can be achieved with patient self-test devices today. Whether further improving current blood glucose meters and strips with respect to accuracy is necessary or even possible remains speculative. In any case, prescribers and health care cost carriers should not only be driven by economic reasons when selecting the meter systems for the patients. Having reached almost laboratory quality with patient self-test meters, they may moving forward rather be interested in the opportunity to eliminate potential clinical problems introduced by cheaper but less accurate blood glucose monitoring systems.
Footnotes
Abbreviations
MARD, mean absolute relative deviations; SMBG, self-monitoring blood glucose meters.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: During the last five years AP has received speaker and consultancy fees, research grants, and travel support from the following organizations, which are related to the topic of this article: AstraPharma, Abbott, Bayer, Becton Dickinson, CellNovo, Diabetes Technology Society, LifeScan, NovaBiomedical, A. Menarini Diagnostics, Roche Diagnostics, Sanofi Diabetes.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
