Abstract
Aim:
The aim was to explore personal experiences and to determine the impact of impaired sleep on well-being and diabetes-related activities/decision making among a cohort of people living with T1D.
Method:
Adults with T1D over the age of 18 and parents/carers of children with T1D were invited to complete an online questionnaire about their quality and quantity of sleep. Questions included impact of sleep on diabetes-related decision making, effective calculation of bolus doses, important aspects of psychosocial functioning, and frequency of waking. Diasend download data were used to objectively determine frequency of nocturnal blood glucose testing in children.
Results:
A total of 258 parent/carer participants (n = 221 female, 85.6%) and 192 adults with T1D (n = 145, 75.5% female, age range 19 to 89 years) took part. In all, 239 parents/carers and 160 adults believed waking in the night has an impact on their usual daily functioning. Of these, 236 parents/carers and 151 (64%) adults reported the impact as negative. Chronic sleep interruption was associated with detrimental impact on mood, work, family relationships, ability to exercise regularly, ability to eat healthily, and happiness.
Conclusion:
Chronic sleep interruption is highly prevalent in adults with T1D and parents/carers of children with T1D with negative effects on daily functioning and well-being. Appropriate interventions are required to alleviate this burden of T1D, address modifiable risk factors for nocturnal hypoglycemia, and reduce the (perceived) need for nocturnal waking.
Many individuals living with type 1 diabetes (T1D), and parents of children with the condition routinely undertake nocturnal blood glucose testing to check for hypoglycemia, setting alarms to wake in the early hours of the morning, thus causing a regular interruption of their sleep. Fear of nocturnal hypoglycemia is especially prevalent amongst adults with T1D as well as parents of children with diabetes, representing a major challenge and potential barrier to achieving optimal glycemic control. 1 This is not surprising given that hypoglycemic events occurring at night are a common complication of insulin treatment. 2 Moreover, nocturnal hypoglycemia is associated to another cornerstone in the management of diabetes: exercise. Nocturnal hypoglycemia is common after afternoon exercise. 3
The consequences of chronic sleep interruption are often underappreciated in T1D care. 4 While it is established that sleep problems can impact negatively on an individual’s physical and mental well-being,4,5 the impact for parents of children with T1D can be similarly negative. Decreased sleep duration and/or impaired sleep quality directly influence glucose regulation—even a single night of partial sleep restriction in adults reduces insulin sensitivity by 14-21%, suggesting that exposure to chronic sleep restriction might contribute to insulin resistance in patients with T1D. 6 This is very relevant for T1D management as insulin resistance is known to be associated with an increased risk for microvascular and macrovascular complications in type 1 diabetes. 7 Thus, if T1D indeed causes disruption of sleep patterns, this may in turn impair glucose regulation, creating a vicious circle.
Irrespective of the impact of waking up to test blood glucose levels during the night, normal sleep architecture may also be disturbed in T1D after many years of living with the condition, with increased risk of next day depression and reduced quality of life. Daily functioning is also negatively impacted by chronic sleep interruption, leading to poor work and school performance and increasing the possibility of behavioral problems, impaired learning and suboptimal emotional regulation. 8 Yet several risk factors associated with nocturnal hypoglycemia are modifiable.
The aim of this mixed-methods study was to quantify the extent of chronic sleep interruption and to subjectively explore personal experiences of the impact of impaired sleep on well-being and diabetes-related activities/decision making among a cohort of people living with T1D and their parent/carers.
Methods and Participants
Adults with T1D over the age of 18 and parents/carers of children with T1D were invited to complete an online questionnaire about their quality and quantity of sleep. The survey was hosted on the secure University of Southampton isurvey site for 1 month following approval granted by local ethical review board. Each participant provided informed consent prior to study start. The questions were developed by the multidisciplinary research team including people with T1D, prior to piloting with potential participants. Minor revisions were made in line with feedback prior to “going live” in May 2015. To establish the extent of nocturnal blood glucose monitoring amongst a large clinic population, Diasend (medical information software) download data were analyzed for >200 days of glucose meter data for 5040 children.
Advertising occurred via social media sites Twitter, Facebook, Glu, and DiabetesMine; patient advocacy groups including INPUT, Diabetes UK, and Diabetes Research and Wellness Foundation charities; and investigators’ health care professional networks. All eligible participants had access to a link to the online survey.The study was conducted in accordance with the Declaration of Helsinki.
Quantitative responses were analyzed using SPSS statistical software v.21, and free-text responses were analyzed using thematic coding and content analysis. Free-text data were analyzed using constant comparison methodology with 2 researchers independently analyzing and coding the text, before consensus was reached on key themes and findings.
Results
According to Diasend data, 99% of families (n = 4990) performed blood glucose testing during the night (defined as between midnight up to 6
Insulin Delivery Method Currently Used.

Frequency of Waking During the Night on Average.
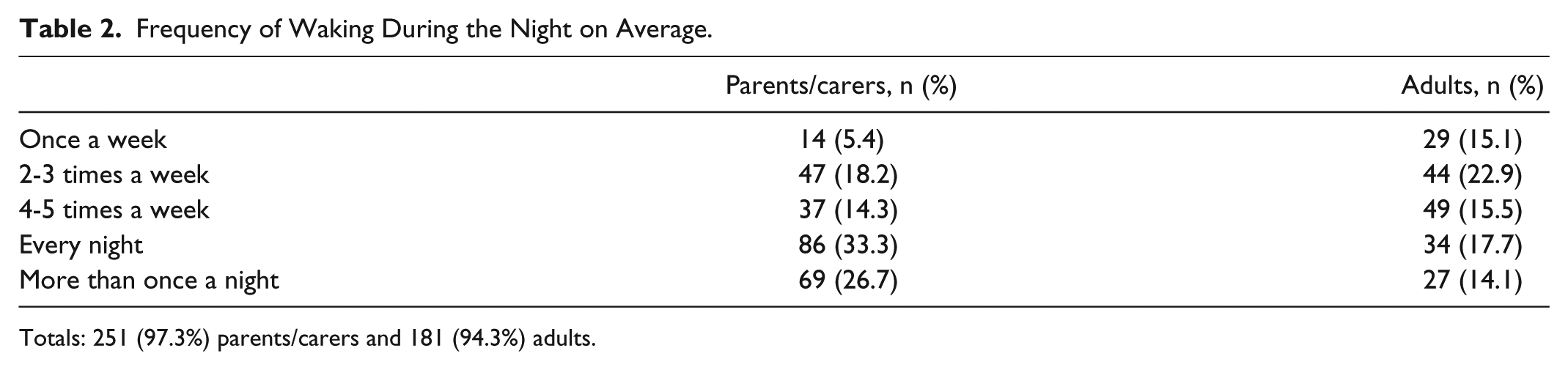
Totals: 251 (97.3%) parents/carers and 181 (94.3%) adults.
Reported Causes of Waking During the Night.
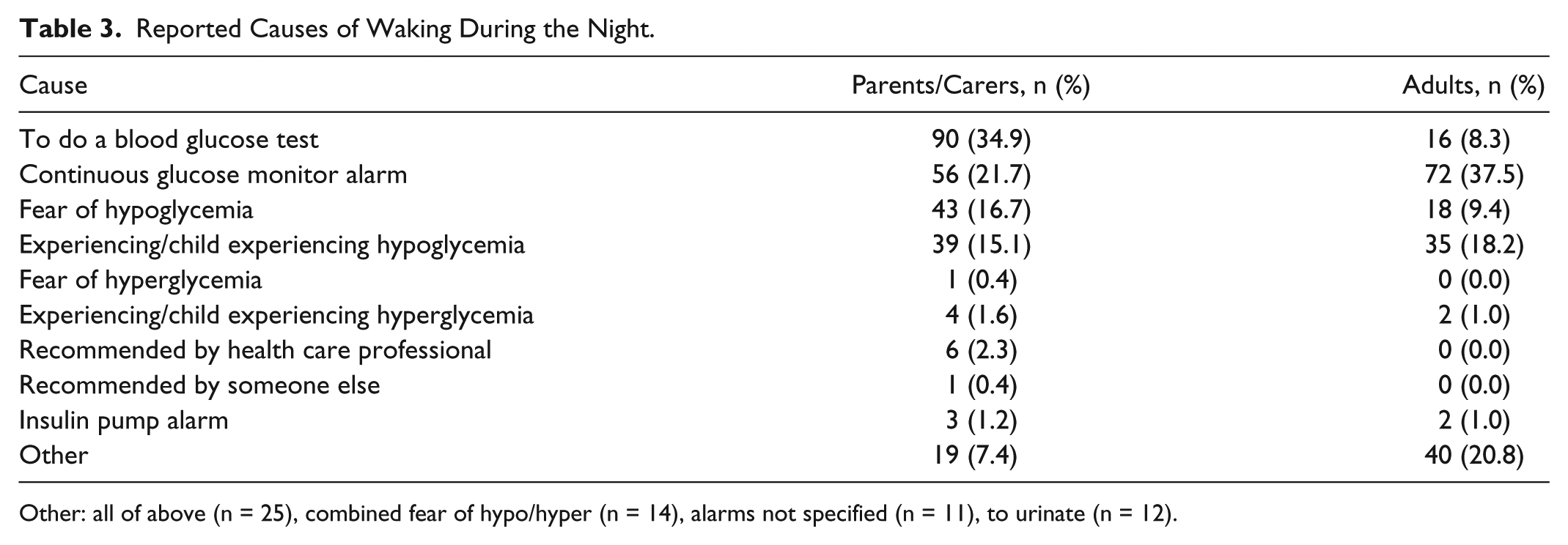
Other: all of above (n = 25), combined fear of hypo/hyper (n = 14), alarms not specified (n = 11), to urinate (n = 12).
If the CGM alarms, How Often Does a “False” Alarm Wake You in the Night?
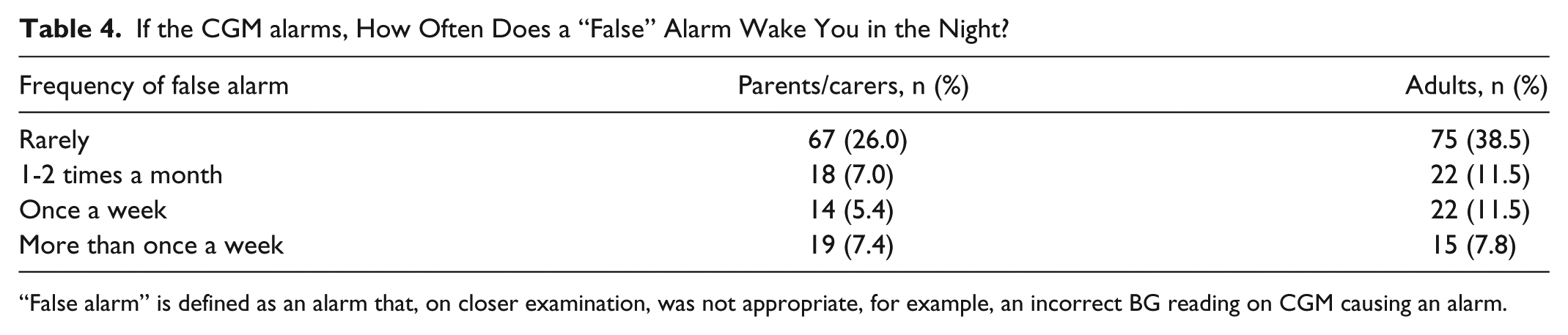
“False alarm” is defined as an alarm that, on closer examination, was not appropriate, for example, an incorrect BG reading on CGM causing an alarm.
Parents/Carers: Impact of Waking During the Night.
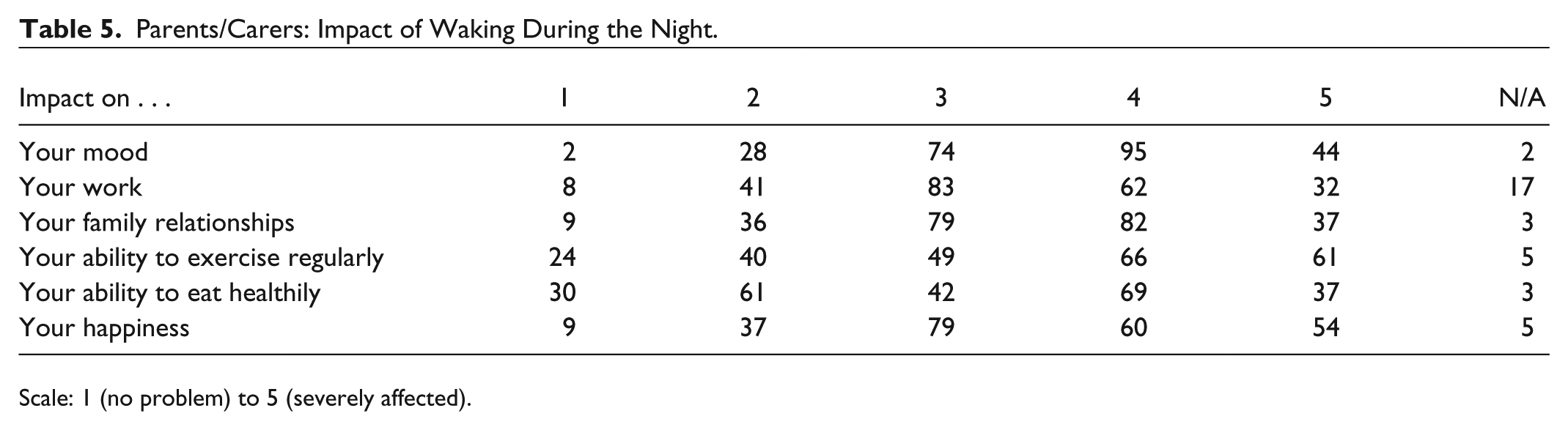
Scale: 1 (no problem) to 5 (severely affected).
Adults With T1D: Impact of Waking During the Night.
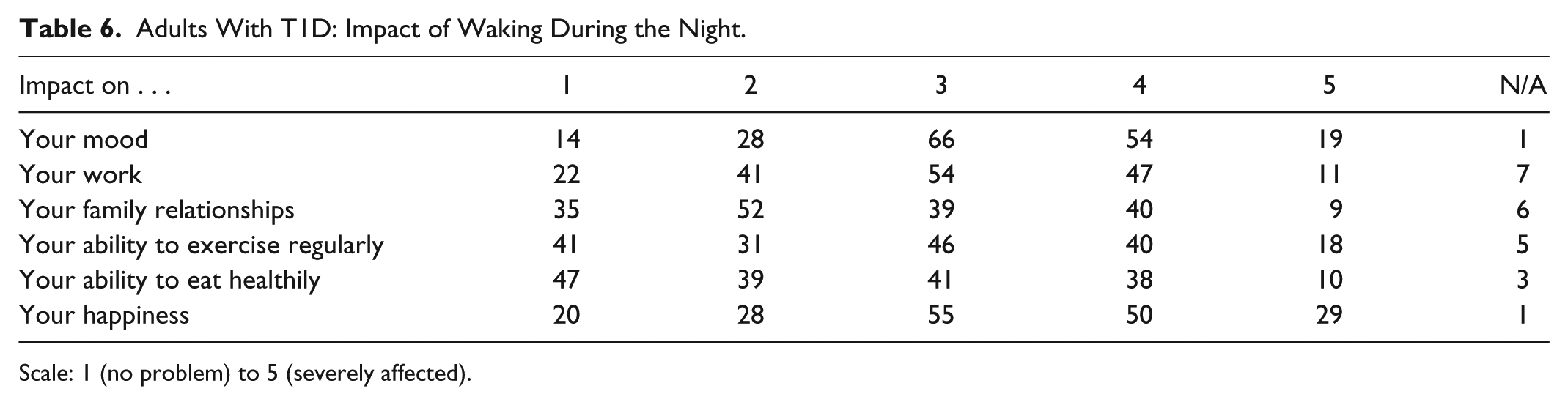
Scale: 1 (no problem) to 5 (severely affected).
Key Negative Themes.
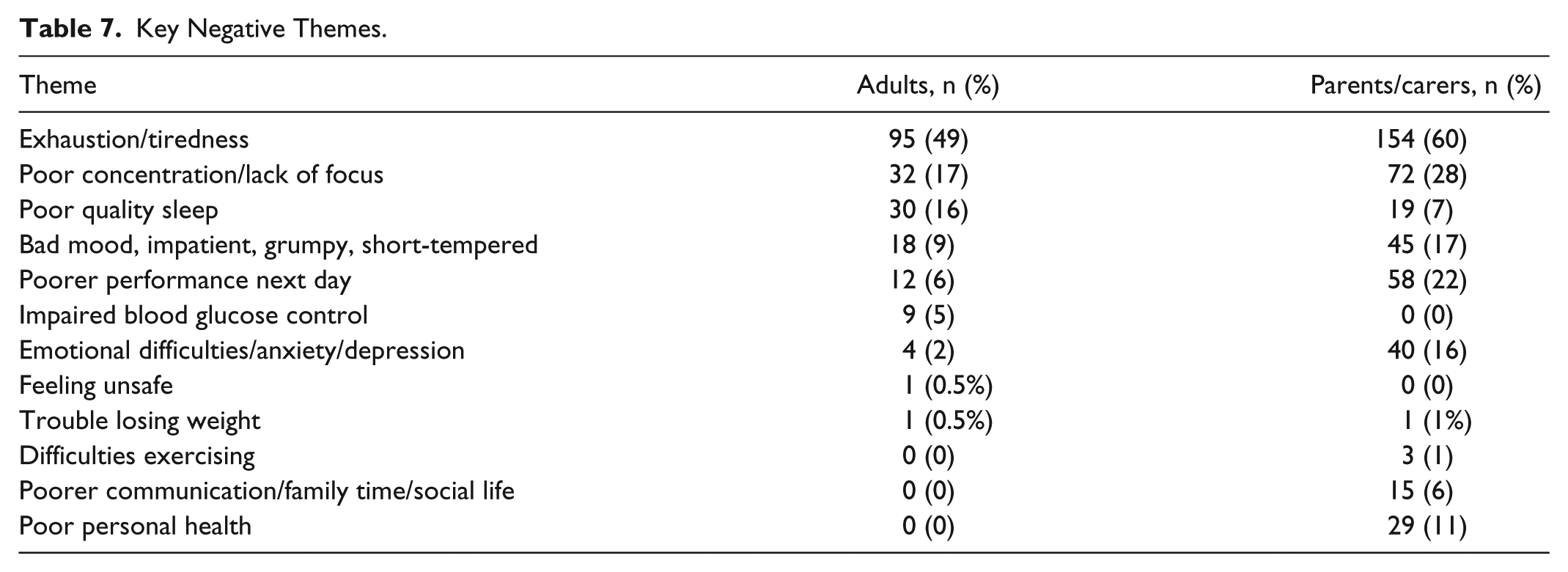
A total of 239 parents/carers and 160 adults believed waking in the night impacted their usual daily functioning. Of these, 236 parents/carers and 151 adults reported the impact as negative.
Only 19% of parents/carers and 12% of adults reported that waking during the night impacted on their ability to effectively calculate a bolus dose. Furthermore, 35% of parents/carers and 33% of adults said it affected their ability to make diabetes-related decisions. This is inconsistent however with the responses to questions that delved more deeply into such impact as detailed in Tables 5 and 6.
Positive Impact of Waking During the Night
A small number of adults reported positive aspects of nocturnal waking to be the ability to quickly make blood glucose corrections (n = 11) and therefore feel healthier or less unwell the next day. Parents/carers reported positives in more dramatic terms, for example, “diabetic child is living,” “I go back to sleep knowing my kids are alive,” and “I’d rather be tired than my son going into a coma because blood dropped too low.” Parents also reported the reassurance of their child being in a “safe” range of blood glucose level (n = 5).
Negative Impact of Waking During the Night
Free-text responses about the negative impact of chronic sleep disturbance were provided by 147 adults and 243 parents/carers provided.
Free-text comments from adult comments included:
“Tired, irritable groggy, easily distracted at work, less patience, less energy”
“It is difficult to function properly”
“I never feel rested when I wake up”
“More prone to being grumpy, short-tempered, hard to concentrate and more likely to feel depressed”
Parent/Carer comments included:
“Exhaustion. It is torture”
“Feel unmotivated and tired most days while pushing on to do the daily things that need accomplished”
“Feel tired often . . . affects relationship with family and friends as not enough energy. One of my friends said ‘it has lost a part of me’”
“I am never truly rested since my daughter’s diagnosis 3 years ago”
Impact of Sleep Disturbance on Bolus Calculations
In all, 12 adults and 35 parents/carers reported difficulties in concentration when making calculations, that is:
Adults:
“feels like your brain isn’t working properly”
“makes it harder to think about mathematical equations”
“not fully conscious so not thinking 100% clearly”
Parents/carers:
“it’s hard to make good decisions when you are exhausted”
“brain does not function, even though you want it to”
“the other day I gave the wrong dose”
“I have bolused her for a correction and food in the middle of the night out of habit . . . she should only be getting a correction”
Impact of Sleep Disturbance on Diabetes-Related Decisions
A total of 41 parents/carers and 18 adults reported poor concentration and diabetes-related decisions to be more difficult, with mistakes or “wrong” decisions being a consequence, that is:
Parents/carers:
“I’ll be more likely to over-correct a low, then she’ll jack up too high”
“I choose to have my daughters at a higher BG than I would typically want them to be”
“When I reach the point of utter exhaustion I will let high numbers go uncorrected if it means I get a few more minutes of sleep”
“sometimes I’m so confused and fuzzy, I have a hard time concentrating”
Adults:
“Because of being tired . . . I feel that I might not be making the best diabetes choices during the night”
“Exhaustion impedes all decisions”
“More likely to rage bolus”
“Taking too little or too much insulin or over-writing a low”
“Once I injected extra insulin and it was too much”
Discussion
The data show that waking during the night as a direct consequence of living with T1D is highly prevalent. In all, 99% of parents/carers wake to some extent, with over half, and a third of adults, waking at least once every night or more than once a night. This is consistent with previous literature and highlights a major burden of T1D on the ability to have a normal sleep pattern. 4 Furthermore, increased time in deep sleep is associated with less risk of hypoglycemia at night as well as improved HbA1c levels. 9 Here also, the majority of participants believed this had a negative impact on their usual daily functioning.
While most participants were using insulin pump therapy, use of continuous glucose monitoring was low amongst children compared with adult participants. Rates of CGM use are still higher amongst participants than in the general T1D population where reported usage is around 7%. 10 Therefore, even with access to modern technology, sleep interruption remains common and may even be augmented by the devices themselves. 11 Interestingly, there were no specific difference between CGM users and non-CGM users in terms of frequency of waking during the night nor reported impact on psychosocial functioning.
The most commonly reported cause of waking for parents/carers was to conduct self-monitoring of blood glucose (SMBG), whereas for adults it was to respond to a CGM. Rates of “false” alarms were reported to be relatively low, however 19.8% of parents/carers and 30.8% of adults experienced waking due to a false alarm at least once a month. Experience, or fear of, hypoglycemia were common, reported by 32% of parents and 28% of adults. This is again consistent with previous studies where fear of hypoglycemia particularly is pervasive. 12 Furthermore, hypoglycemia unawareness affects around 25% of people with T1D and contributes to both fear and associated behaviors, often resulting in reliance on family members to detect and treat severe hypo episodes, 13 suggesting an urgent and extensive need for information and support for family members.
De Zoysa et al DAFNE-HART 14 pilot study data show an intervention based on motivational interviewing and cognitive behavior techniques was effective, however participant numbers were small and data somewhat lacking on the sustainability of any improvement. Several risk factors to reduce the risk of nocturnal hypoglycemia are modifiable, including awareness of the impact of HbA1c level, exercise, bedtime blood glucose level, and prior daytime hypoglycemic events. 15 It may be that greater awareness of these risk factors may help patients and providers decrease the risk of hypoglycemia at night. Other nocturnal hypoglycemia avoidance strategies include the use of insulin pumps with low glucose suspend features, 16 considering different insulin formulations, use of insulin bolus calculators, 17 and in the near future use of overnight automated closed-loop systems. 18 For others using medications to help with diabetes related complications, choice of prescription medicine can impact sleep quality and architecture. 19
A minority of participants reported aspects of nocturnal testing to be positive for example adults reported the ability to quickly make blood glucose corrections while parents/carers reported reassurance as a key positive. Dramatically parents also described that testing during the night enabled them to know their child was alive and in a “safe” range. It could be argued that this reassurance is somewhat false, however, resulting from a pervasive and maladaptive fear. Perception and understanding of risk, in a culturally risk averse population, requires greater clarification in terms of how risk is perceived and the implications for parents in terms of potential societal judgment and disapproval. 20
Interestingly, the majority of participants said that waking during the night did not impact on their ability to effectively calculate an insulin bolus dose or make diabetes-related decisions the next day. This is inconsistent with the free-text responses where the majority of participants reported exhaustion/tiredness as a negative impact of chronic sleep interruption and poor concentration/lack of focus. Chronic sleep disturbance is associated with poorer cognitive function, reduced ability for rational thought, poorer well-being, psychological stress, poor physical health and heightened sensitivity.21,22 Improved sleep, by comparison, is associated with child and family functioning.
Both parents/carers and adult participants commonly reported the negative impact on important aspects of psychosocial functioning. These included mood, work, family relationships, exercise, healthy eating, and happiness. Free-text responses similarly reported bad mood, impatience and short temper, emotional difficulties, increased anxiety, and depression. Each of these is associated with poorer health (of carers) and suboptimal glycemic control.
Limitations of the current study include the online study design of the survey and the self-select nature of participation; thus survey participants may not be representative of the wider T1D population. As data was obtained from 2 different datasets, it was not possible to draw direct comparisons across them. Furthermore, the self-report data, while providing valuable information on the impact of chronic sleep interruption, may lead to a rather crude estimate of the sleep characteristics per se, which require further investigation. Strengths however include the online study design as it enabled us to reach a wide range of participants from the United Kingdom and United States and larger numbers of participants than would have been possible with local paper survey. Additional strengths include the mixed-methods approach, enabling participants to provide greater detail about the impact of chronic sleep disturbance on a range of aspects of their daily lives and functioning.
Conclusions
Chronic sleep interruption is highly prevalent in adults with T1D and parents/carers of children with T1D with negative effects on daily functioning and well-being. Further research on sleep characteristics and development of appropriate interventions are required. Modifiable risk factors for nocturnal hypoglycemia should also be targeted to reduce the (perceived) need for nocturnal waking.
Footnotes
Abbreviations
BG, blood glucose; CGM, continuous glucose monitoring; DAFNE, dose adjustment for normal eating; SMBG, self-monitoring of blood glucose; T1D, type 1 diabetes; UK, United Kingdom; USA, United States of America.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
