Abstract
Background:
The glycemic response and its relation to postoperative complications following gynecologic laparotomies is unknown, although these surgeries carry a substantial risk for postoperative morbidity. Therefore, our objective was to assess the prevalence of perioperative hyperglycemia and glucose variability in women undergoing a gynecologic laparotomy.
Methods:
In this prospective cohort study, capillary glucose was measured every hour during the perioperative period. The primary outcome measures were the proportion of patients with postoperative hyperglycemia (glucose >180 mg d l-1) and the glucose variability in the intra- and postoperative period. Postoperative complications were assessed as secondary outcome measure.
Results:
We included 150 women undergoing a gynecologic laparotomy. Perioperative hyperglycemia occurred in 33 patients without diabetes (23.4%) and in 8 patients with diabetes (89%). Glucose variability was significantly higher (mean absolute glucose change [MAG] 11 mg dl-1 hr-1 [IQR 8-18]) in the intraoperative compared to the postoperative period (MAG 10 mg dl-1 hr-1 [IQR 3-16], P = .03). Neither hyperglycemia nor glucose variability was associated with postoperative complications.
Conclusions:
Hyperglycemia and glucose variability seem to be a minor problem during gynecologic laparotomy. Based on the current data, we would not advocate standardized glucose measurements in every patient without diabetes undergoing gynecologic laparotomy.
Gynecologic laparotomies can be classified as major abdominal surgery and carry a substantial risk of postoperative morbidity. 1 However, the glycemic response to major gynecologic surgery is not known, contrary to many other types of surgery.2-5 In the perioperative period, plasma glucose can increase due to surgical stress, so-called stress hyperglycemia. 6 The occurrence of stress hyperglycemia is related to excessive production of cortisol and catecholamine’s, leading to increased gluconeogenesis and peripheral insulin resistance. 6 This might contribute to complications like an increased risk of wound infection, activation of coagulation and longer hospital stay,3,5,7 although not in all surgical populations. 8 In addition to the type of surgery, several patient characteristics, such as increased age and a higher body mass index, put this patient population at risk for the development of perioperative hyperglycemia. 9
Retrospective studies have suggested that increased perioperative glucose variability in combination with hyperglycemia is more detrimental than hyperglycemia alone,3,10-13 and glucose variability is associated with an higher risk of mortality in critically ill patients.11,12 However, all studies on glucose variability are retrospective in nature, with significant bias due to the number of glucose measurements performed. Until now, no prospective studies have described the role of glucose variability in the perioperative period.
Only when the glycemic response and glucose variability during gynecologic laparotomies are known, can their relevance in the perioperative setting of gynecologic laparotomies be assessed. We therefore prospectively investigated the prevalence of perioperative hyperglycemia and glucose variability in the intra- and postoperative period of major gynecologic laparotomies.
Material and Methods
This prospective observational cohort study was conducted in the Academic Medical Centre in Amsterdam. Patients were included between November 1, 2011, and July 31, 2013. The study protocol NL37308.018.11 was approved by the Medical Ethics Committee of the Academic Medical Centre. Written informed consent was obtained from every patient the day before surgery.
Patients aged 18-85 years, with or without a history of diabetes (DM), scheduled for major gynecological surgery lasting >150 minutes were included in this study. Exclusion criteria were early termination of surgery (and therefore with only 1 intraoperative glucose sample available) and admission to the intensive care unit postoperatively. We included patients scheduled for abdominal hysterectomy for uterine fibroids or endometrium cancer, primary or secondary debulking surgery for ovarian cancer or Wertheim Okabayashi radical hysterectomy for cervical cancer.
Glucose samples were taken from an intravenous or arterial line every 60 minutes during surgery. Measurements started 30 minutes prior to surgery and continued until the end of the recovery period on the postanesthesia care unit. On postoperative days 1, 2, and 3, a fasting capillary glucose was measured on the ward. All glucose samples were measured using the Accu-Chek Inform (Roche Diagnostics Corporation, Indianapolis, IN, USA). A plasma glucose > 180 mg dl-1 was treated with a bolus of intravenous insulin at the discretion of the attending anesthesiologist.
Baseline characteristics, including a history of DM and surgical and anesthetic details, were noted. Postoperative complications were assessed for the first 90 days after surgery. The assessment was done by retrospective chart review and a short questionnaire by telephone 90 days postoperatively. If chart review was unavailable and patients did not answer their phone on 3 different dates and times, they were considered lost to follow-up.
The primary outcome was the glycemic response to the laparotomy. We used several parameters to describe this response: the prevalence of intra- and postoperative hyperglycemia, defined as plasma glucose > 180 mg dl-1 (the advised cutoff value of the American Diabetes Association [ADA] for treatment of hyperglycemia); 14 the intra- and postoperative median glucose value; and the intra- and postoperative glucose variability.
We chose mean absolute glucose change (MAG) and the coefficient of variation (%CV) to describe variability, as both have been associated with mortality in studies including critically ill patients.11,15,16 Furthermore, as many studies in this field report mean glucose with SD, this is also reported.
To assess the clinical relevance of these glucose parameters in the perioperative period, we assessed the relation of hyperglycemia, MAG or %CV with postoperative complications and length of hospital stay. Previous reports on the relation of perioperative glucose parameters and outcome in abdominal and vascular surgery included 150 to 211 evaluable patients.5,8 However, as the type of surgery is a major determinant of outcome, power calculation based on these studies might not be reliable for gynecological laparotomies. Therefore, these associations were assessed as secondary outcomes.
The occurrence of postoperative complications was a composite outcome of death, readmission, wound bleeding, postoperative infection, delirium, thromboembolic complications (such as deep vein thrombosis, lung embolism, stroke, and myocardial infarction) and other complications. Postoperative infection was defined as wound, pulmonary, or cystitis/urinary tract infection for which antibiotic treatment was started.
Statistical Analyses
We aimed to prospectively include 200 women within a period of 2 years, as a feasible cohort size for descriptive analyses on perioperative glucose parameters.5,8 We planned per protocol analyses including every patient with at least 2 intraoperative glucose measurements available. Taking into account the dropout rate of 25% due to early termination of surgery, 150 women were available for further analyses.
For every intra- and postoperative hour, we calculated the median glucose with interquartile range (IQR). The Wilcoxon signed rank test was used to compare the intra- and postoperative measurements with the baseline measurement in all women with at least 2 glucose measurements available. The proportion of women with hyperglycemia was calculated for the intra- and postoperative period for women with and without DM.
The MAG (mg dl-1 hr-1) was calculated by the sum of the absolute glucose difference between samples divided by the time over which the samples were taken. 11 The %CV was calculated by dividing the SD by the mean glucose times 100. The differences between intra- and postoperative median glucose, intra- and postoperative MAG, intra- and postoperative %CV, and intra- and postoperative SD were compared with the Wilcoxon signed-rank test, as these data were not normally distributed. Subgroup analyses were performed for women who received dexamethasone during surgery.
The intra- and postoperative MAG and the intra- and postoperative %CV were divided into quartiles of equal group size for further analyses. We used multivariate logistic regression analyses to assess whether intra- or postoperative hyperglycemia; intra- or postoperative MAG and intra- or postoperative %CV were associated with postoperative complications. Multivariate linear regression analyses were performed to determine the relation of hyperglycemia, MAG, or %CV with length of stay, also analyzed separately as intra- and postoperative variables. These analyses were adjusted for age, body mass index, ASA classification, dexamethasone use, DM, and duration of surgery. P values for all the multivariate regression analyses were corrected for multiple testing using the Benjamini and Hochberg method. 17 A P value < .05 was considered significant.
Patients with only 1 glucose measurement during surgery were excluded from the analyses. Statistical analyses were done using SPSS version 20.0 (SPSS Inc, Chicago, IL, USA).
Results
A total of 328 patients were eligible for the study, 150 were analyzed (Figure 1). Postoperatively, 2 women had to be admitted to the intensive care unit and their postoperative glucose values were excluded from the postoperative analyses. Twelve had incomplete follow-up data. The patient characteristics are displayed in Table 1. No pregnant women were included in this study. Of patients, 92.7% were Caucasian, 3.4% of African American origin, 0.7% Chinese, and 3.2% or other ethnicities. Perioperative data are shown in Table 2. Three patients with DM were insulin-dependent and received a perioperative infusion of glucose 5% with 8 units of insulin. Two patients with DM received an insulin bolus (2 and 5 units) during surgery, 5 patients (2 patients with DM) received an insulin bolus in the immediate postoperative period. No additional sources of glucose were administered in the perioperative period.
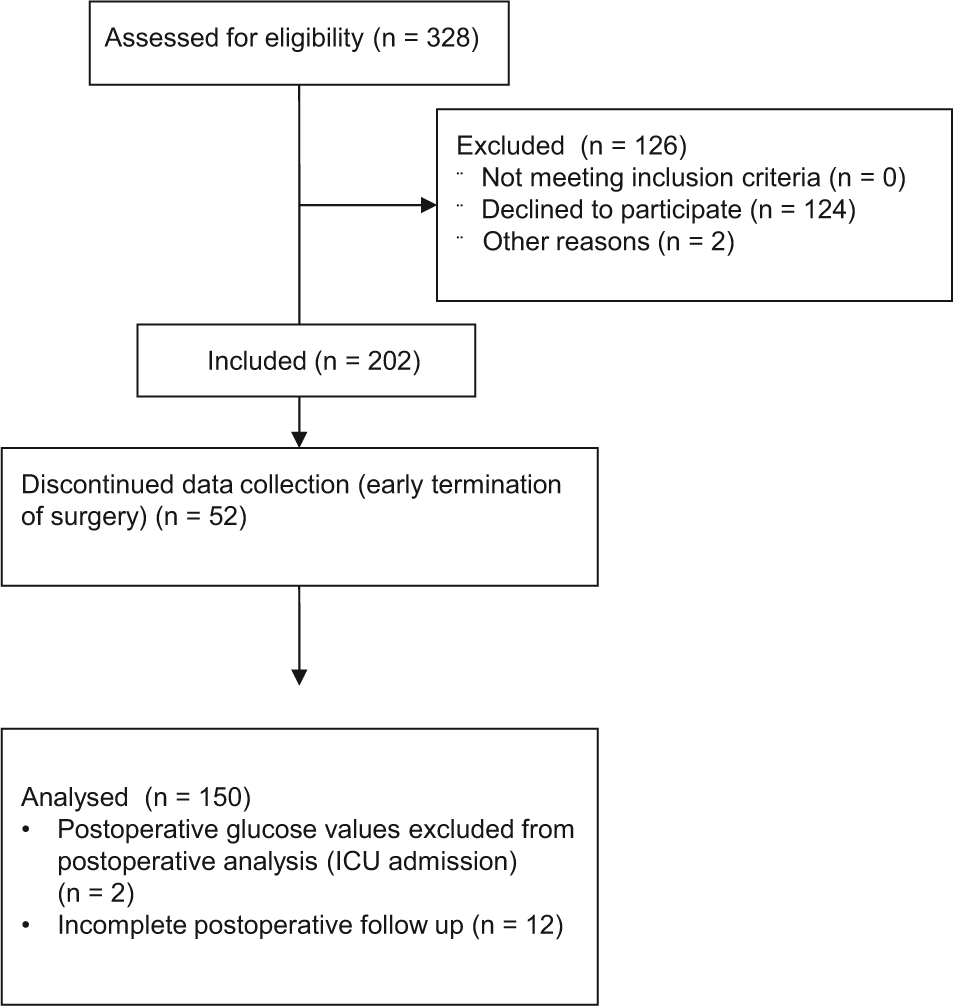
Consort flow diagram for the PAVAS study.
Patient Characteristics (N = 150).

Perioperative Data (N = 150).
During surgery, glucose increased significantly when compared to the preoperative fasting level. Intraoperative glucose values were significantly higher than preoperative values (P < .001, Figure 2). In the postoperative period glucose was relatively stable, and the postoperative glucose values did not significantly differ from the first postoperative glucose value (P > .05, Figure 2).
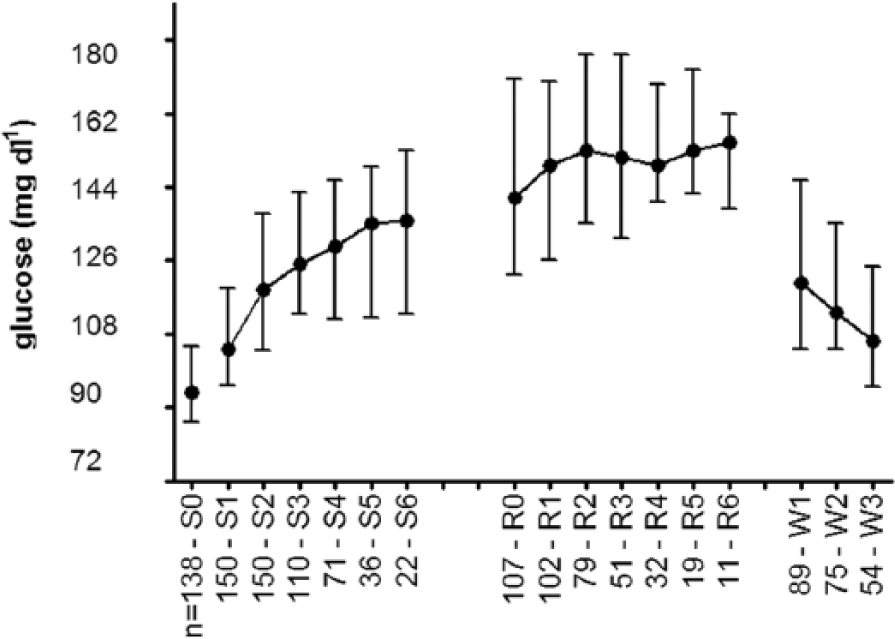
Perioperative median glucose with IQR. n, number of patients; S0, preoperative; S1-S6, hours intraoperative; R0-R6, hours of postoperative recovery period; W1-W3, fasting glucose day 1-3 on the ward.
In total 41 patients (27.3%) developed perioperative hyperglycemia. This occurred in 33 patients without DM (23.4%); 5 patients from this population developed hyperglycemia intraoperatively, 25 patients developed hyperglycemia postoperatively, and 3 patients developed hyperglycemia on the ward. In contrast, perioperative hyperglycemia occurred in 8 of the 9 patients with DM (89%), of whom 5 developed hyperglycemia intraoperatively, 2 postoperatively and 1 on the ward.
The postoperative median glucose (151 mg dl-1, IQR 131-169) was significantly higher compared to the intraoperative median glucose (114 mg dl-1, IQR 101-131, P < .001). On postoperative day 3, glucose values (106 mg dl-1, IQR 95-124) had not yet returned to the preoperative fasting level (94 mg dl-1, IQR 86-104, P < .039) and 3.8% of the women still had a glucose > 180 mg dl-1. Median’s and mean glucose values are displayed in Table 2.
As dexamethasone, given as an antiemetic during anesthesia, might influence glucose levels, we investigated glucose values in women with and without dexamethasone injection. The mean dose was 4.2 mg (SD 1.0). There were no significant differences in median postoperative glucose values between patients who did receive dexamethasone (n = 117, glucose 155 mg dl-1 [131-171] and who did not receive dexamethasone (n = 33, glucose 142 mg dl-1 [131-157], P = .221).
The MAG decreased from 11 mg dl-1 hr-1 (IQR 8-18) intraoperatively to 10 mg dl-1 hr-1 (IQR 3-16) after surgery (P = .030; Figure 3). The %CV decreased form 13.3% (IQR 9.4-19.5) to 8.3% (IQR 4.2-14.0, P < .001). The SD decreased from 16 mg dl-1 (IQR 11-23) intraoperatively to 13 mg dl-1 (IQR 6-22) postoperatively (P = .053).
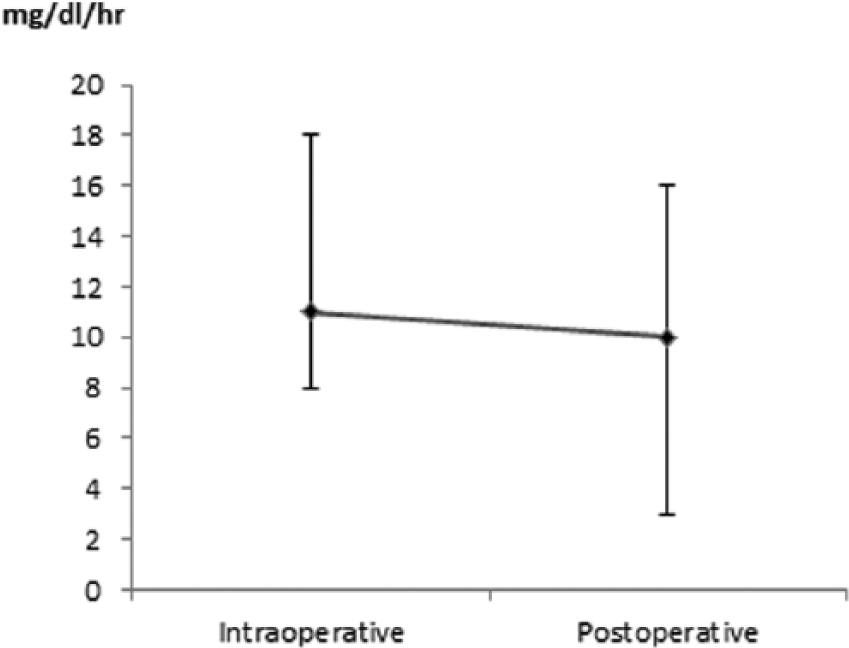
Intra- and postoperative glucose variability. Expressed as mean absolute glucose change per hour (MAG).
Postoperative complications are shown in Table 3; 18.1% of the patients had a postoperative infection; 9.3% of the women were diagnosed with a thromboembolic event. None of the patients were newly diagnosed with DM postoperatively.
Postoperative Complications.

After correction for multiple testing, intra- or postoperative hyperglycemia, intra- or postoperative MAG, and intra- or postoperative %CV were not associated with our composite end point of any postoperative complication nor with length of hospital stay in the multivariate regression analyses.
Discussion
This study shows a prevalence of perioperative hyperglycemia of 23.4%, with significantly higher glucose variability in the intraoperative period compared to the postoperative period in gynecologic laparotomies. Glucose increased significantly intraoperatively and was relatively stable in the postoperative period, which subsequently resulted in a decrease of glucose variability in the postoperative period. No relation could be established between postoperative hyperglycemia or glucose variability and postoperative complications.
The observed increase in glucose concentration during and after surgery has been described previously2,3 and is reflected in the prevalence of hyperglycemia. Our findings are comparable to those of retrospective cohort studies, where the perioperative prevalence of hyperglycemia (glucose > 180 mg dl-1) ranges from 17.2% to 32.2%.18,19 In these studies, the majority of hyperglycemic episodes occurred in the immediate postoperative phase. Thus, when strictly looking at glucose, the immediate postoperative phase would be the most sensible period to measure glucose when screening for stress hyperglycemia. However, 77% of the patients without DM did not develop hyperglycemia, most likely due to the low comorbidity rate, thus not requiring treatment according to the ADA guideline.
In 23% of patients, relevant postoperative hyperglycemia will occur, which requires attention and treatment from the anesthesiologist. One could argue that when glucose is only measured on indication, clinically relevant hyperglycemia could be missed. However, we do not think that it should be pursued to perform standardized glucose measurements in patients without DM undergoing a gynecologic laparotomy. As described above, the prevalence of clinical relevant hyperglycemia in our prospective data (23%) is comparable to 17-32% found in retrospective data,18,19 thus missing clinically relevant hyperglycemia seems to be a minor problem. In other words, most cases of relevant hyperglycemia will be found when glucose is only measured with routine samples or on indication.
We found no significant effect of dexamethasone on postoperative glucose values. This is comparable with a previous study. 20 We suppose that the stress response to a gynecologic laparotomy might have had a greater effect on glucose than the small steroid load of a single dexamethasone dose and the treating gynecologist should not withhold dexamethasone to prevent hyperglycemia in the perioperative period.
Contrary to previous studies,3,5 we did not find an association between hyperglycemia and postoperative complications, which might be due to the relatively low complication rate; 18.1% had a postoperative complication, whereas previous studies described complication rates of 36-58%.3,5 In the past decade, a lot of effort has been made to improve postoperative outcomes via various strategies, 21 which might have contributed to the low postoperative complication rate we found.
Up to now the association of glucose variability and complications was consistently observed in critically ill patients,11,22,23 but was ambivalently present in patients admitted to surgical and general wards.24,25 When looking at perioperative glucose variability, the quartiles of the MAG in our study were consistent with those found previously in critically ill patients. 11 However, in the latter study the MAG was associated with mortality in critically ill patients, whereas we did not find an association of increased MAG with postoperative complications, nor with mortality. Furthermore, in our study, the median %CV was 13% intraoperatively and 8% postoperatively. This is well below the threshold of 30%, above which the association with mortality in critically ill patients was seen. 16 Only 6 (4%) patients experienced a %CV of > 30% in our study. This could, indicate that glucose variability was a relatively minor problem in the perioperative period in this patient population. In addition, in comparison with the prolonged duration of increased glucose variability in previous ICU studies, short-term glucose variability is probably of little clinical significance in the perioperative period of a gynecologic laparotomy.
This study has several limitations. First, our study population consisted of fairly young women, with little comorbidity except for their diagnosed malignancy. Second, the proportion of women with diabetes mellitus was very small (n = 9). Thus, one should be cautious to extrapolate our findings to surgical patients with DM. However, excluding the women with DM from the analyses did not significantly change the results. Furthermore, loss to follow-up is a frequently encountered problem in prospective observational cohort studies; however, this was limited to only 12 patients in our study. In 52 patients the tumor was inoperable and surgery was terminated prematurely. Our aim was to assess the glycemic response and glucose variability of gynecologic laparotomies lasting more than 150 minutes. Due to the very short duration (< 60 minutes) and less invasive character of the operation, only 1 glucose measurement was available in these early finished surgical procedures. Therefore, these patients were subsequently excluded, but this might have introduced selection bias. Finally, due to the much lower complication rate after gynecological laparotomy (18%) compared to for example a pancreaticoduodenectomy (58%), 3 this study could have been underpowered to allow a definite statement about the association of hyperglycemia or glucose variability with postoperative complications. With a much larger sample size, one might find an association between glucose regulation and postoperative complications. However, the clinical relevance of the outcome of such a major cohort study would be questionable.
Conclusions
The data presented here, for the first time prospectively collected on glucose variability during the perioperative period, show that the glycemic response and glucose variability seem to be small and probably of minor clinical significance in the perioperative period of gynecologic laparotomies. Thus, the usefulness of additional standardized glucose measurements in women without diabetes undergoing gynecologic laparotomies can be discussed. Evidently, we do suggest standardized glucose measurements in women with diabetes undergoing gynecologic laparotomies.
Footnotes
Abbreviations
ADA, American Diabetes Association; %CV, coefficient of variation; DM, diabetes mellitus; ICU, intensive care; IQR, interquartile range; MAG, mean absolute glucose change; SD, standard deviation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
