Abstract
Inpatient hyperglycemia, in patients with and without a history of diabetes, is associated with increased risk of complications, mortality, and longer hospital stay in medicine and surgical patients. Bedside capillary point of care testing is widely recommended as the preferred method for glucose monitoring and for guiding glycemic management of individual patients; however, the accuracy of most handheld glucose meters is far from optimal. Recent studies in the hospital setting have reported that the use of continuous glucose monitoring (CGM) can provide real-time information about glucose concentration, direction, and rate of change over a period of several days. Because it provides glucose values every 5-10 minutes 24 hours a day, CGM may have an advantage over point of care testing with respect to reducing the incidence of severe hypoglycemia in acute care. Real-time CGM technology may facilitate glycemic control and to reduce hypoglycemia in insulin-treated patients. Recent guidelines, however, have recommended deferring the use of CGM in the adult hospital setting until further data on accuracy and safety become available. In this study, we review the advantages and disadvantages of the use of real-time CGM in the management of dysglycemia in the hospital setting.
Inpatient dysglycemia—hyperglycemia, hypoglycemia, and increased glycemic variability—is common and is associated with increased risk of hospital complications and mortality. Several observational studies and intervention trials have reported a prevalence of hyperglycemia (> 140 mg/dl) in ~ 80% of critically ill and cardiac surgery patients1,2 and in 38%-46% of non–critically ill hospitalized patients.3,4 The development of hyperglycemia, in patients with and without a history of diabetes, is associated with increased risk of complications, mortality, and longer hospital stay.3,5 Like hyperglycemia, inpatient hypoglycemia has been associated with higher rates of hospital complications, longer hospital stay, higher health care resource utilization, and hospital mortality, creating a J-shaped relationship between glucose levels and mortality rates.6-9 A glucose level < 2.8 mmol/L (50 mg/dL) was reported to be associated with a mortality rate of 22.2%, compared to 2.3% in patients without hypoglycemia. 10 Increasing evidence from in vitro and human studies has indicated that oscillating glucose levels may be more dangerous than stable hyperglycemia, particularly in activating the pathways involved in the pathogenesis of diabetes complications. Glucose fluctuations result in increased oxidative stress 11 and vascular inflammation, 12 which can lead to increased cardiovascular events. Mendez et al 13 recently reported that increased glycemic variability during hospitalization is independently associated with longer hospital stay and increased mortality in non–critically ill patients.
Bedside capillary point of care (POC) testing is recommended as the preferred method for glucose monitoring and for guiding glycemic management of individual patients. 14 POC testing is usually performed before meals and at bedtime for patients who are eating15,16 or every 4-6 hours for patients who are not eating or receiving continuous enteral nutrition. Among critically ill patients in the ICU, more frequent glucose monitoring is indicated in patients treated with continuous intravenous insulin infusion17,18 and in patients with frequent episodes of hypoglycemia.15,19 Unfortunately, the accuracy of most handheld glucose meters is far from optimal. 20 The Food and Drug Administration (FDA) allows for a variance of 20% for meter measurements and 10% for central laboratory results as compared to the true blood glucose level.16,21 This accepted variance can lead to the omission of important clinical information or to the use of inappropriate therapies. Furthermore, patient factors such as pH changes, oxygenation status, tissue perfusion, varying hemoglobin concentrations, and other interfering hematologic factors in acutely ill patients can affect meter measurements.21-23
The use of continuous glucose monitoring (CGM) has evolved from being a research tool to serving as a device useful for outpatient clinical care in patients with type 1 and type 2 diabetes. Designed to successfully improve glucose control without the addition of medication in ambulatory patients, CGM provides information about glucose concentration, direction, and rate of change over a period of several days. Because it provides glucose values every 5-10 minutes 24 hours a day, CGM may have an advantage over POC testing with respect to reducing the incidence of severe hypoglycemia in acute care.24,25 Two types of CGM device are commercially available: retrospective and real-time CGM. Retrospective CGM is a Holter-type device that measures interstitial glucose levels and stores the information over a period up to 7 days to facilitate insulin adjustment, recognition of daily blood glucose (BG) fluctuations, and prevention of hypoglycemia. 26 Its retrospective nature, however, represents a significant limitation, because patients are unable to react to BG changes before they reach abnormal ranges.27-32 In contrast, real-time CGM technology provides current BG estimates and direction and magnitude of glucose trends, thus allowing patients to take necessary actions to reduce glycemic excursions outside a target range. 26 Real-time CGM technology has been shown to facilitate glycemic control and to reduce hypoglycemia in insulin-treated patients.26,33-35 Recent guidelines, however, have recommended deferring the use of CGM in the adult hospital setting until further data on accuracy and safety become available. In this study, we review the advantages and disadvantages of the use of real-time CGM in the management of dysglycemia in the hospital setting.
History of BG Monitoring
Significant progress has been made since James Herrick reported in 1901 that the examination of urine for glucose was the crucial test for the diagnosis of diabetes mellitus in ambulatory patients as well as in patients with infections, cardiovascular disorders, and diabetic coma. 36 During the following decades, several investigators pioneered laboratory methods for quantitative BG estimations in the laboratory for the diagnosis and critical care management of diabetes. 37 In 1965, an Ames research team headed by Clemens developed Dextrostix, the first BG test strip, which was a paper reagent strip that used the glucose oxidase/peroxidase reaction. 20 Shortly thereafter, Boehringer Mannheim developed the Chemstrip glucose strip. A large drop of blood (50-100 μL) was applied to the reagent pad, and the pad color was visually assessed against a color chart to determine a semiquantitative BG value. Although these test strips were designed for use in doctors’ offices, they were widely used in clinics, surgery departments, and hospital wards to monitor glycemic control. Dextrostix, the first quantitative glucose device to use reflectance meters, became commercially available in 1970. 37 Despite its high cost, large size, and weight (1.2 kg) and the need to use the wash-and-blot technique to remove red blood cells, it became used in emergency departments, because it provided rapid results and a few studies reported good correlation with laboratory reference methods. Several reflectance meters requiring a smaller blood volume (20-30 ul), which were more accurate and could be removed simply by wiping with a cotton ball, were developed in subsequent years. Dextrometer, the first battery-operated meter with a digital display was launched in 1980. Since then, a large number of smaller, lightweight, portable, nonwipe technique, battery-operated, digital reflectance meters have become available for glucose monitoring. Glucose meters for self-monitoring of blood glucose (SMBG) have been effective in improving diabetes control and represent an essential tool for the management of ambulatory and hospitalized patients with diabetes and hyperglycemia.
The concept of the biosensor for measuring glucose levels was first proposed in 1962 by Clark and Lyons of the Children’s Hospital of Cincinnati. 38 The first CGM prototypes, which became available for research purposes in the early 1970s, extrapolated blood through a double-lumen catheter used for the development of glucose sensor-controlled insulin infusion systems.39,40
In 1977, Miles Laboratories produced the Biostator, a relatively large bedside unit. The Biostator incorporates an in-line venous cannula to measure glucose and calculates the correct insulin and dextrose infusion rate on the basis of this measurement. However, the device has serious limitations in clinical practice: it needs constant supervision, is bulky, and requires the continuous withdrawing and discarding of venous blood to measure glucose levels ex vivo using an oxidase-containing membrane. 41 In the past 2 decades, considerable technological progress resulted in the regulatory approval of different continuous and semicontinuous monitors.
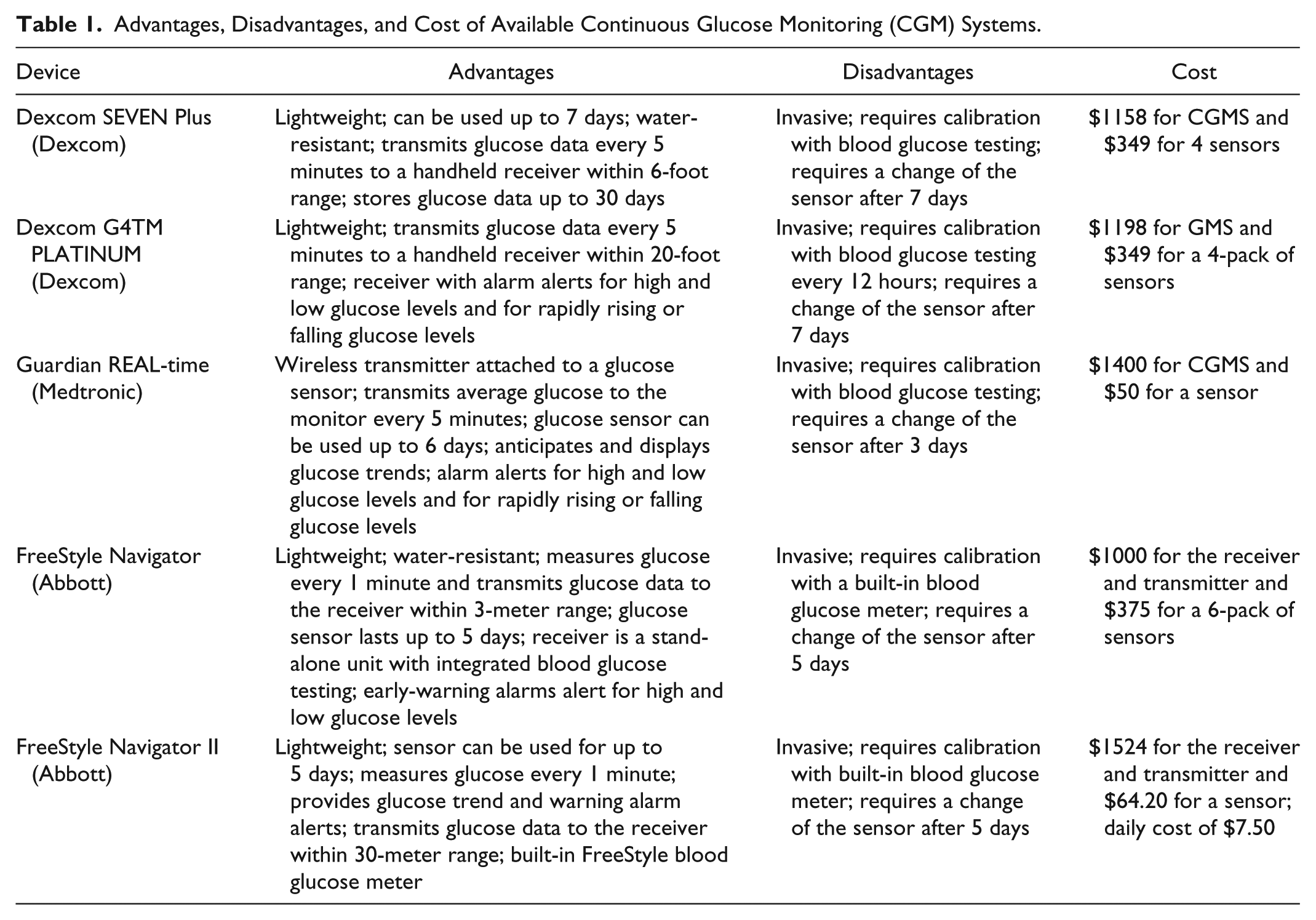
The first CGM device made available in the United States was the GlucoWatch biographer (no longer in use), approved in 1999 by the FDA. This device was worn like a wristwatch and provided glucose measurements every 10 minutes via transdermal extraction of interstitial fluid by reverse iontophoresis, a process by which a device extracts glucose samples from fluids in the body by applying extremely low electric currents to intact skin.42,43 Several limitations, including skipped readings, skin irritation, and inaccuracy, kept the device from receiving clinical acceptance. Other semi-invasive continuous glucose monitors, consisting of a micropump (with a flow rate of 15-100 mL/min) and a biosensor coupled with a microdialysis system, followed. Most CGM devices continue to employ subcutaneous sensors to determine the glucose concentration in interstitial fluid. Therefore, they require frequent calibration by fingerstick tests and cannot be used for more than a few days. Some of the recently developed CGM devices are wireless; their sensors are inserted into the subcutaneous tissue of the abdomen or upper arm. Important features of available CGM devices are listed in Table 1.
Advantages, Disadvantages, and Cost of Available Continuous Glucose Monitoring (CGM) Systems.
Glucose Monitoring in the Non–Critical Care Setting
Recent guidelines recommend the use of POC testing as the preferred method for guiding the inpatient glycemic management of individual patients. 44 Matching the timing of POC testing with nutritional intake and the diabetes medication regimen in the hospital setting is consistent with recommendations for the outpatient setting. POC testing is usually performed 4 times a day: before meals and at bedtime for patients who are eating15,16 and every 4-6 hours for patients who are NPO or receiving continuous enteral nutrition. More frequent glucose monitoring is indicated in patients treated with continuous intravenous insulin infusion,17,18 in patients with frequent episodes of hypoglycemia,15,19 and after a medication change that could alter glycemic control, such as corticosteroid use or abrupt discontinuation of enteral or parenteral nutrition.45-47
Even though quality control programs are essential to meeting FDA requirements and to maintaining the accuracy and reliability of BG testing, 16 a 20% variance between BG measurements derived from POC testing and the laboratory value is considered acceptable.16,21 Recent reports have advocated improvement or tightening of POC meter accuracy standards. 21 However, capillary BG values continue to vary between POC meters, especially at high or low hemoglobin levels, at low tissue perfusion, and in the presence of some extraneous substances.20,48 Furthermore, POC testing involves potential inaccuracies due to issues inherent to the technology, variability between different batches of test strips, varying hemoglobin concentrations, and other interfering hematologic factors in acutely ill patients.21-23 A study of 5 commonly used glucose meters conducted by the Centers for Disease Control determined that mean differences from a central laboratory method were as high as 32% and found a coefficient of variation of 6%-11% in tests performed by individual trained medical technologists. 21
Use of CGM Devices in the Hospital Setting
Comparison studies on the efficacy and clinical benefits of CGM devices and SMBG have produced mixed results, in part due to small sample sizes and variability across studies, including differences in age of subjects, in type of CGM utilized (retrospective versus real time), and in duration of follow-up.49,50 In a recent meta-analysis of ambulatory patients with diabetes, we reported that CGM is associated with significantly better incremental benefit in HbA1c reduction compared to SMBG. 51 Greater benefits of CGM relative to SMBG were observed in studies of more than 12 weeks of intervention and in subjects > 25 years of age. In addition, CGM resulted in less time spent in hypoglycemic ranges than did SMBG, but there was no difference in the frequency of hypoglycemia. 51
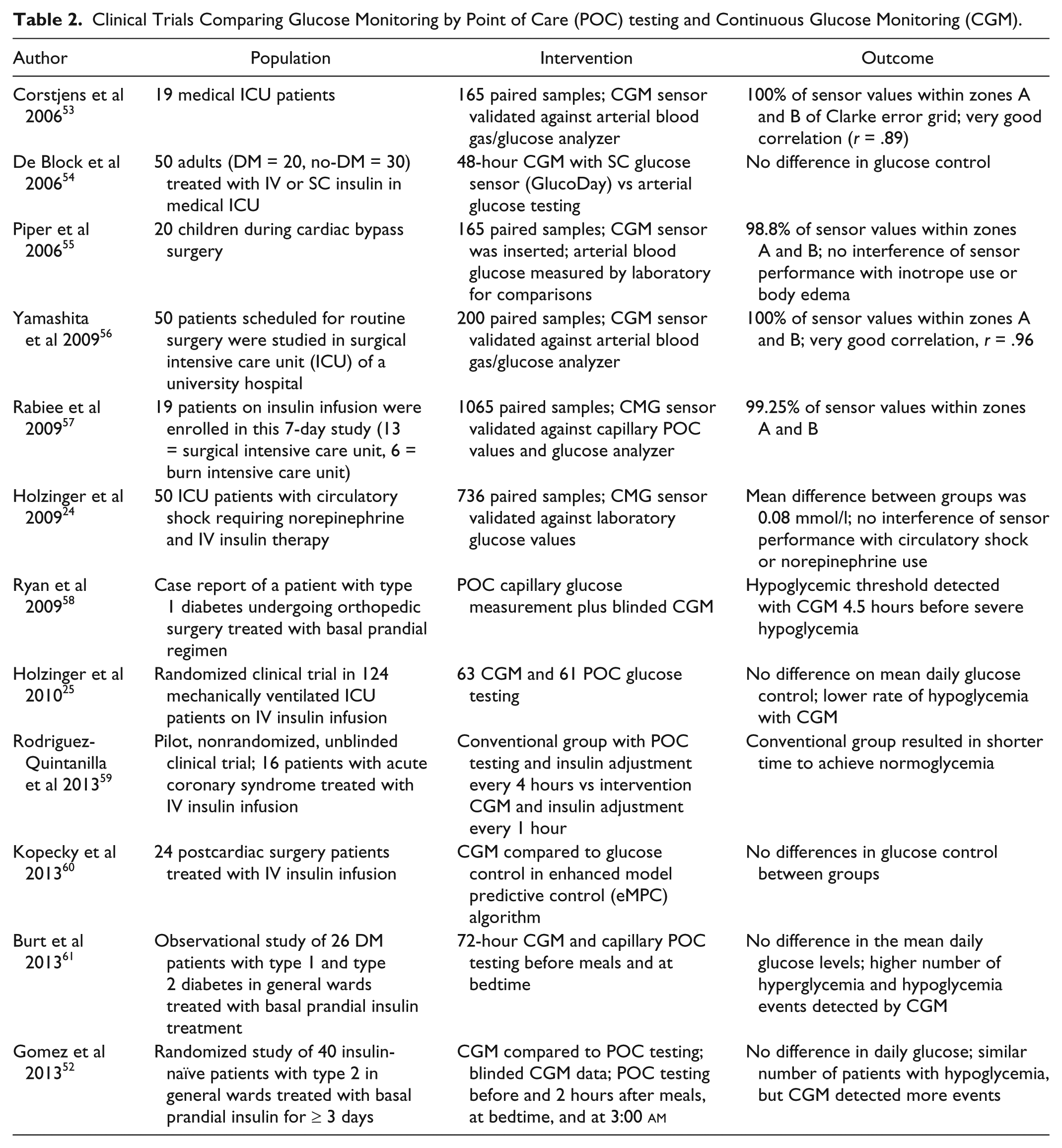
Recent studies have suggested that continuous BG monitoring devices may be helpful in reducing the incidence of severe hypoglycemia in acute care24,25 (Table 1). In some small clinical trials, CGM devices have been shown to improve hypoglycemia and hyperglycemia detection when compared with POC capillary glucose measurements (Table 2). The majority of ICU studies have reported good correlation between glucose values measured by interstitial CGM and arterial/capillary and laboratory measurements, with most CGM measurements falling within zones A and B of the Clarke error grid. Recent studies in non-ICU wards comparing CGM interstitial glucose testing with capillary POC testing reported no differences in mean daily glucose, but CGM testing detected a higher rate of postprandial hyperglycemic excursions and of hypoglycemic events (Table 2).
Clinical Trials Comparing Glucose Monitoring by Point of Care (POC) testing and Continuous Glucose Monitoring (CGM).
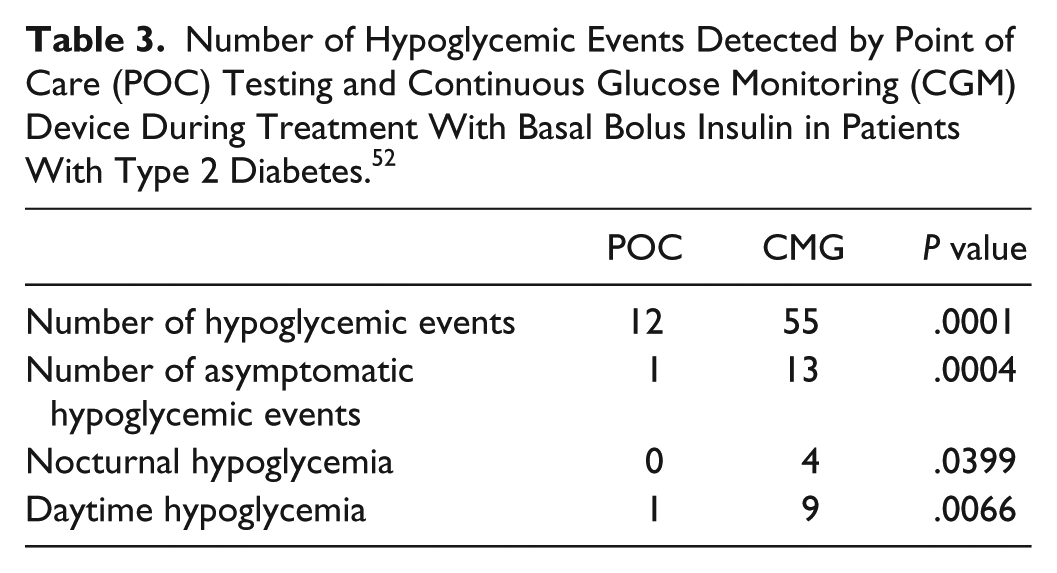
In a recent prospective study, we compared inpatient glycemic control by CGM (Sofsensor iPro 2, Medtronic) and capillary POC testing in general medicine patients with type 2 diabetes treated with a basal bolus insulin regimen for ≥ 3 days.
52
Both patients and hospital staff were blinded to the CGM data. POC testing measurements were performed before meals, 2 hours after meals, at bedtime, and at 3:00
Number of Hypoglycemic Events Detected by Point of Care (POC) Testing and Continuous Glucose Monitoring (CGM) Device During Treatment With Basal Bolus Insulin in Patients With Type 2 Diabetes. 52
In summary, although CGM technology is promising, few studies have tested its use in the acute care setting. Therefore, the Endocrine Society advises against its use because extensive evidence regarding CGM’s safety and efficacy is lacking. 5 This expert committee considered that maintaining direct access to the blood on a continuous basis for an extended period of time is impractical due to lack of reliability in terms of accuracy and the precision of the various systems, and due to the high cost of the use of CGM devices in the general inpatient (non-ICU) setting. In addition, recent guidelines on inpatient glycemic control recommended against intensive inpatient glucose control for the majority of non-ICU patients.14,44 Departing from historic intensive insulin regimes with recent guidelines no longer recommending tight glucose targets may also reduce the need to use CGM devices in non–critically ill patients.
Footnotes
Abbreviations
BG, blood glucose; CGM, continuous glucose monitoring; DM, diabetes mellitus; FDA, Food and Drug Administration; ICU, intensive care unit; POC, point of care; SMBG, self-monitoring blood glucose.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AMG has participated in clinical research studies for Medtronic, Sanofi, Novartis, and Abbott Pharmaceuticals. She has received lecture honoraria from Medtronic, Novo Nordisk, Elli Lilly, Sanofi, Novartis, and Boeringher Ingelheim. GEU has received unrestricted research support for inpatient studies (at Emory University) from Sanofi, Merck, Novo Nordisk, Boehringer Ingelheim, Eli Lilly, and Endo Barrier, and has received consulting fees and/or honoraria for membership in advisory boards from Sanofi, Merck, and Boehringer Ingelheim.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: GEU is supported in part by research grants from the American Diabetes Association (1-14-LLY-36), PHS grant UL1 RR025008 from the Clinical Translational Science Award Program (M01 RR-00039), and grants from the National Institute of Health and the National Center for Research Resources.
