Abstract

Measuring hemoglobin A1c level in blood is one of the methods for diagnosing and monitoring diabetes.1,2 Nondiabetic A1c values usually vary in the 4.0-5.7% range, and diabetic A1c values vary above 6.5%.
3
(A1c values are expressed according to National Glycohemoglobin Standardization Program [NGSP] conventions.) However, unusually low (≤5%) or high (≥17%) A1c values have also been reported, although many of these are falsely influenced by medications or various rare hemoglobin traits.4,5 Therefore, validating the entire analytical range of the instrument is important to accurately address unusual cases. This range for the majority of A1c analyzers is usually 3.0-18.0%. Ideally, whole blood samples should be used as the verification material. However, rarity at extreme A1c values, impurity, and instability are major drawbacks for whole blood to be used as verification material. An alternative verification material based on stabilized blood with relevant analytical properties could better serve this purpose. Since hemoglobin A1c is an intracellular protein, its measurement involves lysis of red blood cells. Therefore, the presence of hemoglobin A1c within an intact cell is an important property for such reference material. In this article, the interinstrument correlation of such a reference material, Streck’s A1c-Cellular® Linearity calibration verification kit, is evaluated. This kit contains 5 levels that can be used in the same manner as a patient sample. To evaluate the interinstrument correlation, the A1c values for each instrument are averaged from 6 runs, across 4 lots, with data collected from 2 laboratories. The average A1c values for levels 1-5 are approximately 3.5%, 6.8%, 10.4%, 14.1%, and 17.9%. The coefficients of determination (
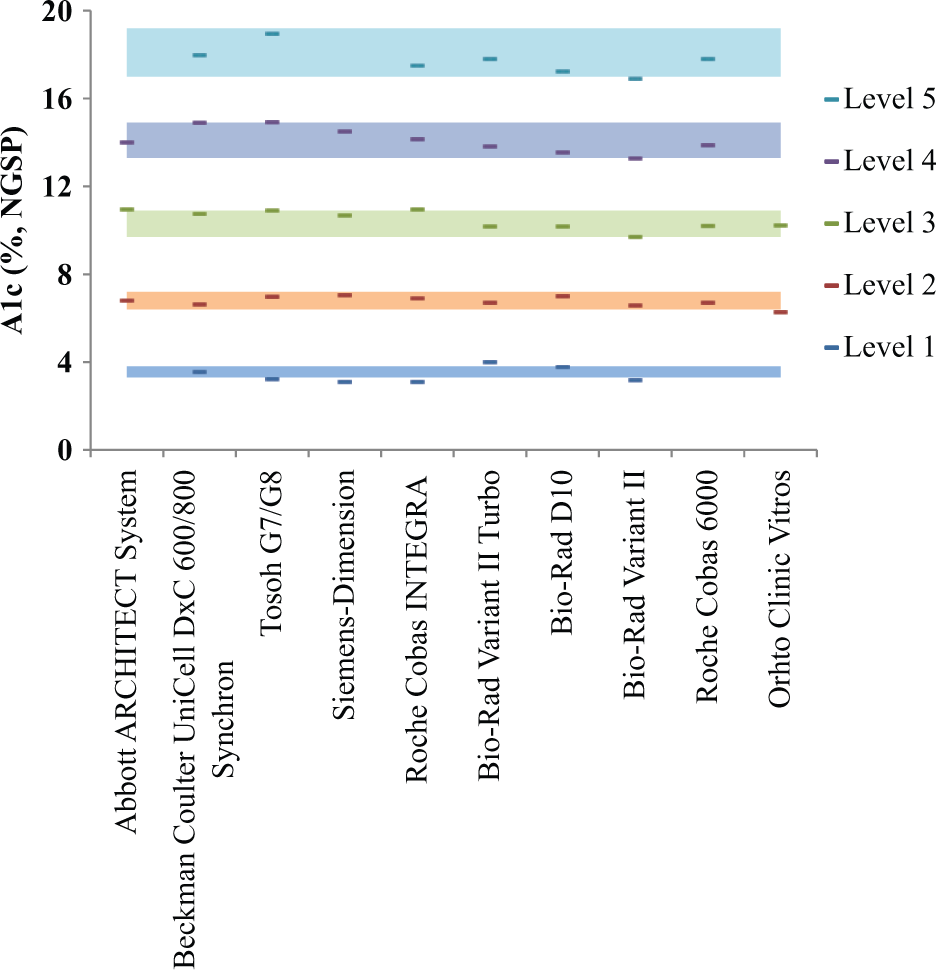
Interinstrument correlation of the 5-level A1c Cellular Linearity calibration verification kit. The A1c values are expressed in National Glycohemoglobin Standardization Program units. The shaded area represents the ±6% of the respective target A1c values for each level. The A1c values for each instrument are averaged from at least 6 runs, across 4 lots with data collected from 2 laboratories. Data obtained outside the instrument analytical ranges were excluded from the analysis.
In conclusion, the A1c values of the 5-level A1c Cellular Linearity calibration verification kit across several analyzers are within CAP-accepted guideline and NGSP certification. In addition, presence of hemoglobin A1c within intact red cells allows this reference material to test all analytical steps.
Footnotes
Acknowledgements
The authors acknowledge Gary Krzyzanowski and Justyna Tesmer, Streck Inc.
Abbreviation
NGSP, National Glycohemoglobin Standardization Program.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Kausik Das, Stephanie M. Wigginton, and Joel M. Lechner are employees of Streck Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research is funded by Streck Inc.
