Abstract
Importance
Opening the cochlear‒carotid recesses through the endoscope-assisted modified translabyrinthine approach to treat lesions in the petrous apex has been demonstrated to preserve the cochlea and hearing. The objective parameters of recesses and their relationships with vital structures were fundamental to the effective application of this technique.
Objective
Herein, we present a quantitative analysis of cochlear‒carotid recesses on high-resolution computed tomography (HRCT) images and discuss their implications for hearing preservation in the surgical management of massive petrous bone cholesteatomas (PBCs).
Design
Observational study.
Participants
Twenty samples (40 sides) obtained between April and June 2021 radiographically via HRCT.
Methods
HRCT images of 40 ears were acquired at 0.60 mm thickness and processed via Mimics. The intrinsic structures of the temporal bone were reconstructed in a 3D view. The morphological data were precisely measured on multiplanar reconstructed images. In addition, clinical implications were demonstrated in one patient with massive temporal bone cholesteatoma.
Results
A 3D model of the cochlear‒carotid recesses was reconstructed. The recesses were essentially divided into 2 parts, namely, the triangle anterior and superior to the cochlea (TASC) and the triangle anterior and inferior to the cochlea (TAIC). The size of the cochlear‒carotid recesses varied widely among the samples. The TASC was accessible in 80% of the samples via a conventional ear endoscope with a diameter of 3 mm, while the applicability of the TAIC was 62.5%. We further discovered that the low position of the trigeminal nerve impression and the small distance between the cochlea and the jugular bulb suggested narrow recesses. The thickness of the compact bone shell of the cochlear lumen acted as a safe drilling divider.
Conclusions
HRCT and 3D reconstruction clearly displayed cochlear‒carotid recesses. For patients with massive PBC, the use of an endoscope to open recesses is feasible in the majority of cases and increases the possibility of preserving the structure and function of the cochlea. Individualized preoperative measurements of cochlear‒carotid recesses via HRCT could help surgeons perform safe and accurate dissection.

Introduction
Petrous bone cholesteatoma (PBC) is a type of epidermoid cyst located in the petrous portion of the temporal bone. 1 PBC is a nonneoplastic but locally aggressive lesion that can result in severe functional damage (hearing loss, facial paralysis, vertigo, etc.) if left untreated.2,3 Radical resection is the standard treatment for PBC, and it takes priority over hearing preservation.4,5 However, with the development of new surgical pathways and the assistance of endoscopy, the likelihood of preserving structures and function has increased. 6
However, for the subtypes of PBC involving the apical area defined in accordance with the Sanna classification, especially the massive type, the transotic (TO) approach and modified transcochlear (MTC) approach are regularly employed.4,5,7,8 In these 2 methods, the cochlea is sacrificed to gain room to access the lesion area; thus, hearing preservation is practically impossible. In the era of auditory implantation, sacrificing the cochlea certainly limits the possibility of future hearing rehabilitation.
In a previous study, through the endoscope-assisted modified translabyrinthine (MTL) approach, opening the cochlear‒carotid recesses to treat lesions in the petrous apex without removing the cochlea was feasible. 9 The cochlear‒carotid recesses are located anteriorly to the cochlea and are divided into 2 spaces, namely, the triangle anterior and superior to the cochlea (TASC) and the triangle anterior and inferior to the cochlea (TAIC). Moreover, studies have shown that the MTL approach is a promising hearing preservation strategy for the existence of the utriculoendolymphatic valve.7,10,11 Therefore, hearing preservation in the management of massive PBC is promising.
However, the exact parameters of cochlear‒carotid recesses and their relationships with vital structures remain unknown. In this study, we performed a quantitative analysis of the cochlear‒carotid recesses based on high-resolution computed tomography (HRCT) images and discussed the implications of this anatomical information in guiding the choice of surgical approach and minimizing the occurrence of complications
Methods and Materials
Patients and Scanning Techniques
We randomly studied 20 samples (40 sides) obtained between April and June 2021 radiographically via HRCT. The original 0.6 mm slice DICOM data were processed with the independent software Mimics Medical 21.0 (Materialize Belgium). Among these samples, 10 were from males. The average age was 41.75 years, with a range of 26 to 59 years. None of these samples had any lesions around the lateral skull base, and the scanning was performed for reasons unrelated to skull base surgery. Subjects who had ever had chronic middle ear diseases were also excluded.
3D Reconstruction
To clearly represent the cochlear‒carotid recesses and the surrounding anatomic relationship, we reconstructed the TASC and TAIC in a 3D view. The 3D models were built from HRCT via Mimics’ masking and calculating 3D functions. The structures included the internal carotid artery (ICA), the internal jugular vein, the lumen of the cochlea, the facial nerve, and the border of the middle cranial fossa.
Measurements of the Multiplanar Reconstructed Images
Slices across TASC and TAIC were acquired via the multiplanar reconstruction function of Mimics. The slice across the TASC was cut perpendicular to the horizontal segment of the ICA. The slice across the TAIC was cut in the sagittal plane (Figure 1). Based on these slices, the following measurements were considered: the minimum cross-sectional area of the TASC and TAIC; the maximum cylinder diameter that could be accommodated within the TASC and TAIC; the angle between the jugular bulb (JB) and the horizontal segment of internal carotid artery (HICA); and the thickness of the compact bone that covered the cochlear lumen in different directions.
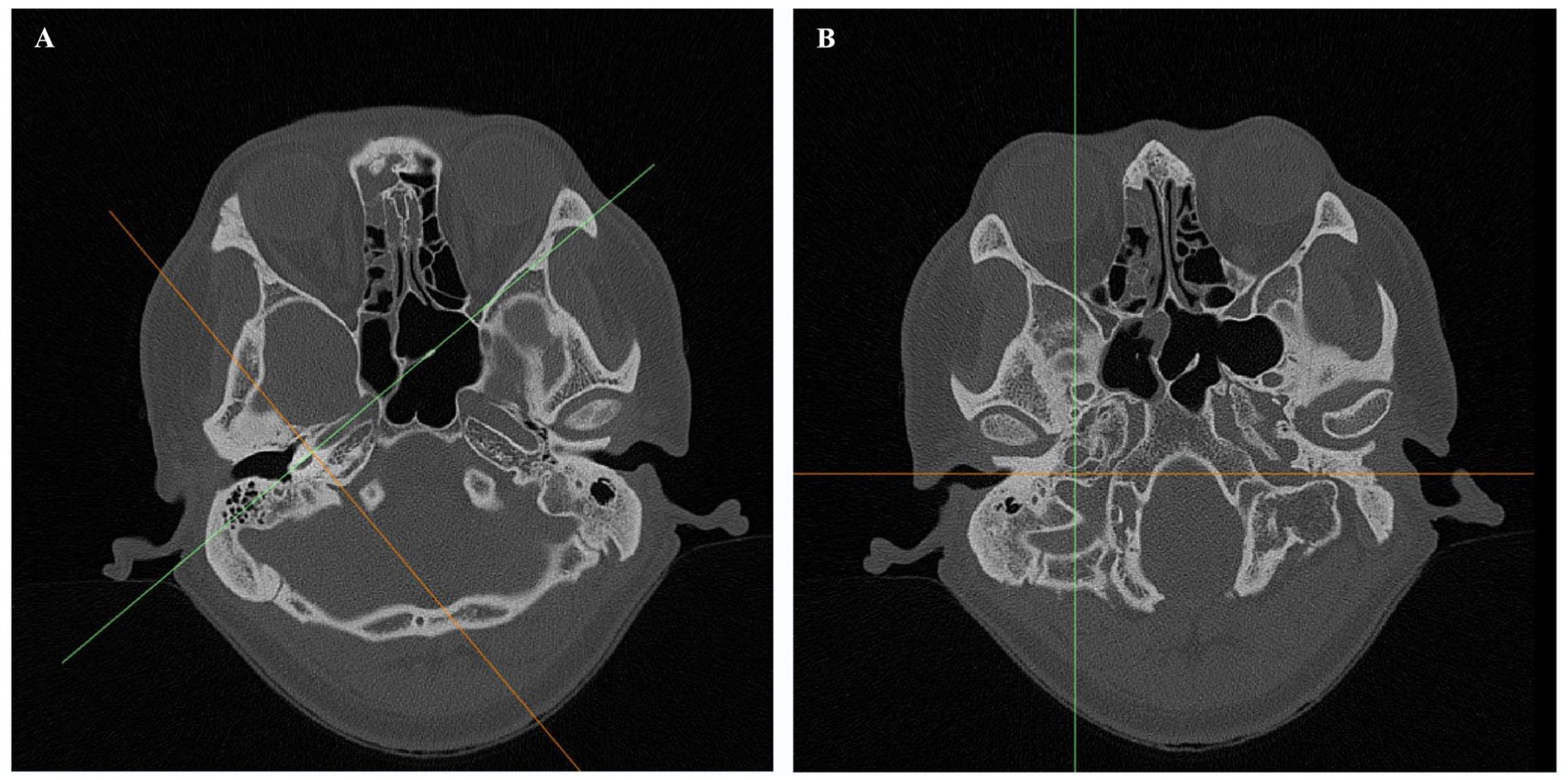
Axial images showing the projection of multiplanar reconstruction. The green line indicates the cross-section, and the yellow line represents the cut direction. (A) The slice across the TASC cuts along a direction perpendicular to the horizontal segment of the ICA. (B) A slice across a TAIC cut in the sagittal plane. ICA, Internal carotid artery; TASC, Triangle anterior and superior to the cochlea; TAIC, Triangle anterior and inferior to the cochlea.
Statistical Analysis
All analyses were performed via SPSS Statistics 22.0 (IBM). Continuous variables are presented as the mean ± Standard deviation. Student’s t test or Mann-Whitney U test was used for comparisons of differences between 2 groups. A P value <.05 at the confidence interval of 95% was considered significant.
Results
3D Reconstructed Images of TASC and TAIC
Through 3D reconstruction, TASC and TAIC can be clearly distinguished. The space enclosed by the HICA, the cochlea, and the skull base of the middle cranial fossa is the TASC, whose cross-section was parallel to the direction of the HICA. The space surrounded by the ICA, the cochlea, and the JB is the TAIC, whose cross-section was almost in the sagittal plane (Figure 2).

Reconstructed 3D model of the right TASC and TAIC. The space enclosed by the horizontal ICA, the cochlea, and the skull base of the middle cranial fossa is the TASC. The TAIC is surrounded by the ICA, the cochlea, and the jugular bulb. This figure is a view from the right side (T top, L left, P posterior). ICA, Internal carotid artery; TASC, Triangle anterior and superior to the cochlea; TAIC, Triangle anterior and inferior to the cochlea.
Quantification of Morphometric Data Around the TASC
By multiplanar reconstruction, the TASC (Figure 3A) could be presented intuitively on the computed tomography (CT) image. The minimum cross-sectional area of TASC was 54.53 ± 36.842 mm2. The maximum cylinder diameter that could be accommodated within the TASC, that is, the maximum allowable diameter of the endoscope that could be placed in the TASC, was 4.97 ± 1.716 mm. Among all 40 samples, 20.0% (n = 8) were less than 3 mm, which meant that the TASCs of these samples were too small to fit the conventional ear endoscope.
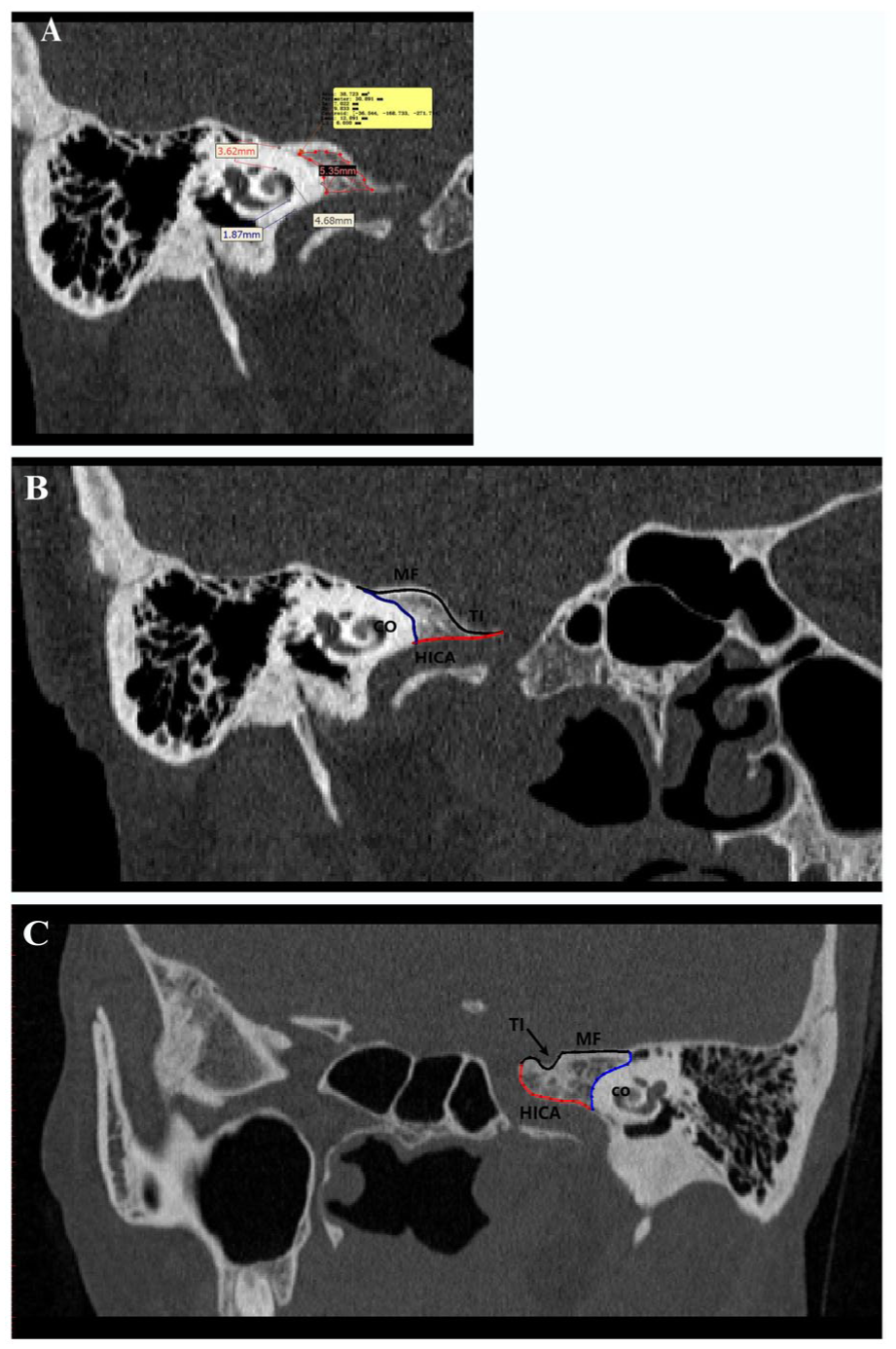
(A) Morphological data from TASC. (B) An example of a trigeminal nerve impression clings to the bone wall of the HICA. (C) An example of a trigeminal nerve impression does not cling to the bone wall of the HICA. TASC, Triangle anterior and superior to the cochlea; HICA, Horizontal segment of the internal carotid artery.
Moreover, a set of other morphometric data that are important for safe exposure to TASC was obtained from multiplanar reconstructed (MPR) HRCT images. The thickness of the compact bone, which acts as a divider between the TASC and the cochlear lumen, was 4.26 ± 0.968 mm. The thicknesses of the compact bone that separates the cochlear lumen from the dura of the middle cranial fossa and the posterior genu of the ICA were 2.93 ± 0.634 mm and 1.78 ± 0.735 mm, respectively (Table 1).
The Morphological Data of TASC.
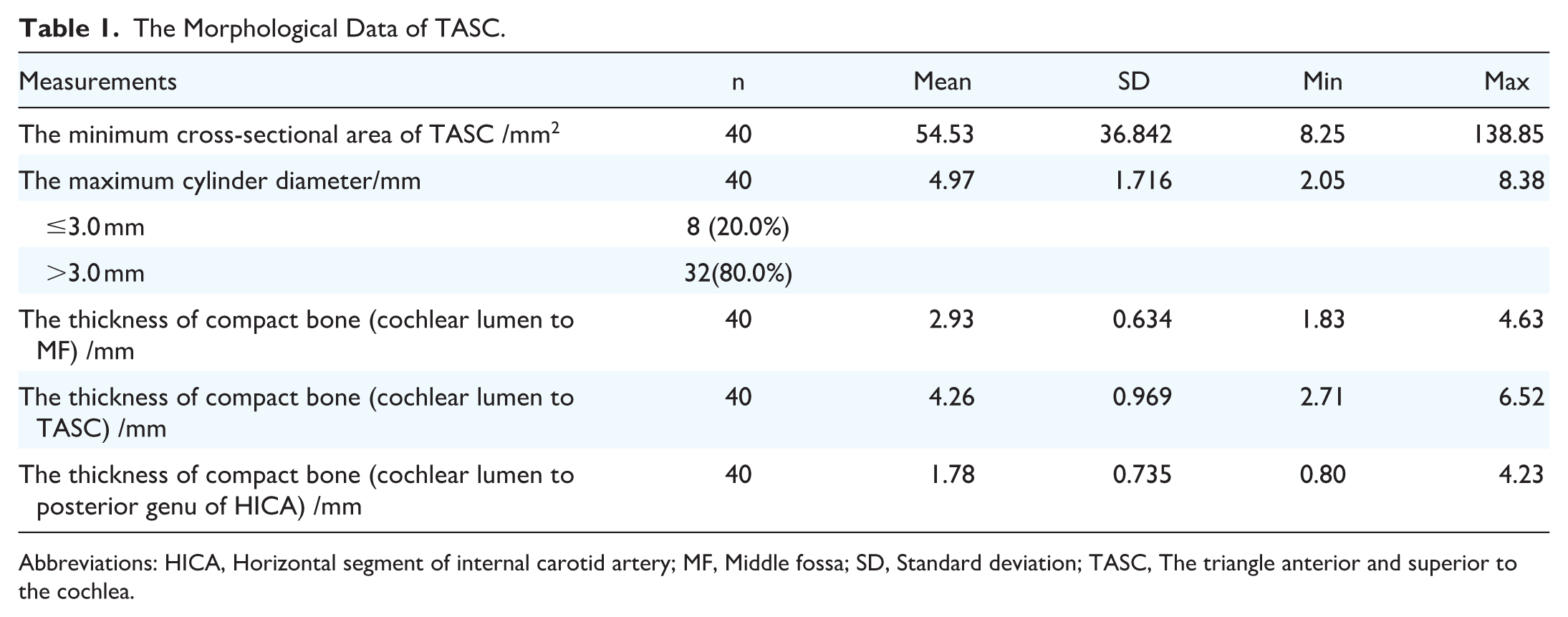
Abbreviations: HICA, Horizontal segment of internal carotid artery; MF, Middle fossa; SD, Standard deviation; TASC, The triangle anterior and superior to the cochlea.
During the measurement, we found that the trigeminal nerve impression (TI) greatly affected the size of the TASC. The TIs clung to the bone wall of the horizontal segment of the ICA in 20 of 40 samples, and the average minimum cross-sectional area of the TASC in these samples was 35.6 ± 20.39 mm2. For samples in which the TI did not cling to the bone wall of the horizontal segment of the ICA, the average minimum cross-sectional area of the TASC was 74.39 ± 42.33 mm2. The difference between these 2 groups was significant (P = .0018) (Figure 3B and C).
Quantification of Morphometric Data Around the TAIC
For the MPR slices, the minimum cross-sectional area of TAIC (Figure 4A) was approximately 22.35 ± 18.939 mm2. The average maximum cylinder diameter that could be accommodated within the TAIC was 3.85 ± 1.52 mm. Among these 40 cases, the maximum cylinder diameter of 15 cases (37.5%) was smaller than the diameter of the conventional ear endoscope (3 mm). The bony shell of the cochlea was usually in contact with the bony wall of the ICA, accounting for 95% (n = 38) of all the cases in this study. In contrast, the distance between the cochlear bone shell and the JB bone wall was variable and greatly affected the shape and size of the TAIC. In this study, the JB bone wall clung to the cochlear bone shell in 37.5% (n = 15) of all the samples; in this instance, the cross-section of the TAIC was a small and typical triangle. In the other 25 samples, there was a distance between the cochlear bone shell and the JB bone wall. In such an instance, the cross-section of TAIC had a large area and an irregular shape. Moreover, the space of the TAIC usually communicated with the mastoid cavity posteriorly in these samples (Figure 4B and C).

(A) Morphological data from TAIC. (B) An example in which the JB bone wall clings to the cochlear bone shell. (C) An example in which there is a distance between the JB and the cochlear bone shell. TAIC, Triangle anterior and inferior to the cochlea; JB, Jugular bulb.
Furthermore, we investigated other anatomical relationships that are important for safe exposure of TAIC on reconstructed slices. The average angle between the JB and the ICA was 55.76° ± 17.712°. The thickness of the cochlear bone shell, which separates the cochlear lumen and TAIC, was 2.78 ± 0.700 mm (Table 2).
The Morphological Data of TAIC.

Abbreviations: ICA, Internal carotid artery; JB, Jugular bulb; SD, Standard deviation; TAIC, The triangle anterior and inferior to the cochlea.
Clinical Case
We retrospectively reviewed 1 patient with massive temporal bone cholesteatoma who underwent preoperative measurement of cochlear‒carotid recesses. 3D reconstructed CT images of the cochlear‒carotid recesses were acquired from this patient preoperatively. We found that the TASC and TAIC were interconnected due to bony destruction of the cholesteatoma. The bone wall of the middle fossa was partly deficient, while the bone covering the ICA was completely gone. The compact bone, which acts as a divider between the TASC and the cochlear lumen, was also absorbed, and its thickness was only 1.59 mm. Moreover, we observed that the TI did not cling to the bone wall of the horizontal segment of the ICA. Thus, the maximum cylinder diameter that could be accommodated within TASC reached 10.76 mm. For the TAIC, the JB also did not cling to the cochlear bone shell. Thus, the maximum cylinder diameter that could be accommodated within the TAIC was 4.22 mm. According to the measured data, we assumed that the TASC and TAIC could function as spaces to place the endoscope and surgical instruments during surgery. The intraoperative findings were consistent with the preoperative reconstructed image data (Figure 5). Through these 2 anatomical spaces, the cholesteatoma located on the inner side of the cochlea and around the ICA was successfully resected via an endoscope, with the cochlea in situ. The patient was followed for 6 years without recurrence.

(A) Overall image of the interconnected TASC and TAIC on a reconstructed CT image (T top, L left, P posterior, A anterior). (B) Image and morphological data of TASC. (C) Image and morphological data of TAIC. (D) Intraoperative image of the cochlear‒carotid recesses. (E) Intraoperative image of TASC; the white arrow indicates the cholesteatoma. (F) Intraoperative image of TAIC. TASC, Triangle anterior and superior to the cochlea; TAIC, Triangle anterior and inferior to the cochlea; CT, Computed tomography.
Discussion
In clinical practice, supralabyrinthine cholesteatoma is the most common type of cholesteatoma, and it is also considered favorable for preserving hearing. The middle cranial fossa approach, with its modified approach, the supralabyrinthine approach, and the subarcuate approach, are alternative surgical modalities for treating the supralabyrinthine subtype.4,7,9,12,13 For the selected apical, infralabyrinthine-apical, or infralabyrinthine subtype, hearing preservation can be achieved via retrolabyrinthine, infralabyrinthine, or infratemporal fossa approaches.9,13,14 However, for the management of PBC, preserving hearing may be associated with an increased recurrence rate. 4 In addition, according to Sanna’s newest report, 76% of patients with PBC still undergo resection via the TO or MTC approach,5,8 which are relatively more drastic approaches. Hence, we propose the concept of cochlear-carotid recesses. By carefully drilling out the cochlear‒carotid recesses, the lesion within it can be exposed, and the endoscope can be placed in the recesses for observation. Most importantly, the cochlea is left in situ for hearing preservation.
The cochlear‒carotid recesses are actually part of the petrous bone pneumatization. Allam and Schuknecht first divided pneumatization of the petrous bone (excluding the mastoid region) into 2 anatomical categories, namely, the perilabyrinthine region and the apical region. The perilabyrinthine region was further subdivided into the supralabyrinthine region and the infralabyrinthine region. 15 Subsequently, Wei et al 6 divided the perilabyrinthine recesses into 5 subregions, namely, the supralabyrinthine recess, the infralabyrinthine recess, the antralabyrinthine recess, the retrolabyrinthine recess, and the retroinfralabyrinthine recess. The antralabyrinthine and infralabyrinthine recesses are basically equal to the definitions of TASC and TAIC. Pneumatization has implications not only for the disease process but also for the selection of surgical access. Therefore, designing surgical approaches to the petrous bone requires a good understanding of pneumatization. 16
The development process of petrous bone pneumatization is divided into 3 stages: the infantile stage (from birth to 2 years), the transitional stage (from 2 to 5 years), and the adult stage.17 -19 The pneumatization of the petrous apex occurs as the last step, typically during late adolescence or early adulthood. 18 The degree of pneumatization is highly variable and is influenced mainly by the postnatal middle ear environment. 20 Some studies suggest that petrous bone pneumatization is closely linked to the degree of mastoid air cell development,17,19,21 while others propose that it may be an independent process. The mastoid is formed from the saccus superioris, whereas the petrous bone and the anterior epitympanic recess develop from the anterior saccule of the saccus medius. The type of anterior epitympanic recess significantly influences the pattern of petrous bone pneumatization.22,23 Besides, the lateral recess of the sphenoid sinus affects the size of the petrous bone pneumatization from the medial side. Generally, the bilateral petrous bone pneumatization is symmetric.24,25 Gender differences in pneumatization extent have been reported in several studies, while other findings do not support this observation. Similar controversy exists regarding the degree of pneumatization in the upper and lower parts of the petrous bone.26,27 Thus, the lack of consensus further emphasizes the importance of the preoperative CT scan for individualized assessment.
In our research, the minimum cross-sectional area of the cochlear‒carotid recess also varied widely among the samples. However, in the majority of samples, cochlear‒carotid recesses are feasible for operation and endoscopic observation. The proportions in TASC and TAIC are 80% and 62.5%, respectively. In the TAIC, our data are smaller than the results that have been published.28,29 The reason is that the round diameter they measured included the thickness of the cochlear bone shell. In our opinion, measurement without a cochlear bone shell better coincides with real-world clinical practice. Although previous studies reported only a 30% to 35% incidence of petrous apex pneumatization,19,21,25 this research investigates the space that can be safely drilled, and in pathological cases, the expansile process remodels the adjacent bone, increasing the utility of the recesses. 25 Therefore, in conclusion, recesses are qualified to be generalized to some degree.
The sizes of the TASC and TAIC are significantly affected by several anatomic factors. For TASC, the TI greatly affects the size of this area. TI is a bone depression formed by the trigeminal ganglia on the middle skull base. Previous studies have focused mainly on the width of the TI to estimate its obstruction of the surgical field. In Arslan’s report, the average width and diameter of the oval-shaped TI were 10.5 mm and 7.64 mm, respectively. 30 In our study, we emphasized that the depth of the TI was the main factor influencing the size of the TASC. If the TI is deep and tightly clings to the bone wall of the horizontal segment of the ICA, there will be little space for TASC. The studies from the Modena and Bologna groups introduce a novel transcanal endoscopic approach to TASC via the suprageniculate fossa (SGF). The applicability of this approach is contingent upon the degree of SGF pneumatization. 27 The TAIC is a corridor that has been used to access petrous apex lesions and the internal auditory canal in endoscopic surgery. Most of the previous studies on TAIC used width (from the JB to the carotid taken at a tangent to the basal turn of the cochlea) and height (from the jugulocarotid notch to the basal turn of the cochlea) to represent the size of the TAIC.28,31 In the only article that focused on the cross-sectional area of the TAIC, Giddings et al 32 reported that the TAIC was mostly shaped like a triangle or rectangle, and the mean area was 25.6 ± 16.3 mm2, which correlated well with our findings. In our study, we found that the shape and size of the TAIC were strongly affected by the distance between the cochlear bone shell and the JB bone wall. This observation aligns with the results of Ela’s research, which considered that the JB was the only factor that could determine the applicability of TAIC, and TAIC could be considered inapplicable if the jugular bulb was too close to the cochlea (distance < 3 mm). 33
The thickness of the compact bone shell covering the cochlea, which acts as a divider between the cochlear lumen and cochlear‒carotid recesses, directly indicates the safe range of bone drilling. Excessive removal of the compact bone might cause a cochlear fistula. Several studies have suggested that overdrilling of the cochlear lateral wall results in postoperative new bone formation and fibrosis of the cochlear lumen, which subsequently influences cochlear implantation.34,35 Inflammation and intracochlear bleeding are 2 possible pathogenic factors. Therefore, overdrilling around the cochlea should be avoided, and it makes sense to imperatively use antibiotics and glucocorticoids after surgery.
The common scenario in which the cochlear bone shell closely contacts the bone of the posterior genu of the ICA might induce some iatrogenic risks. In our study, the distance from the cochlear lumen to the posterior genu of the ICA was 1.78 ± 0.735 mm, which correlated well with previous studies.36,37 When TASC is opened, excessive bone removal in this region may cause the ICA to work as a third inner ear window, which could lead to some audiovestibular symptoms after surgery. Moreover, if a patient has any chance of an artificial cochlear implant, a defect in the bone may result in inadvertent penetration of the electrode into the carotid canal or cause severe tinnitus.37,38
One limitation of our research is the relatively small sample size, and we need to add more samples for analysis. What’s more, we should further evaluate the impact of the ear inflammatory diseases, the development stage, and the cholesteatoma on the cochlear‒carotid recesses. In future research, we can present a 3D model and parameters of cochlear‒carotid recesses in patients with massive PBC to illustrate their practical application.
Conclusion
Our study provided critical anatomic information on the cochlear‒carotid recess via 3D and slice reconstruction techniques. The low position of the TI and the small distance between the cochlea and the JB are 2 vital anatomical landmarks for predicting the narrow spaces of TASC and TAIC, respectively. Individualized evaluation of recesses on HRCT preoperatively enables surgeons to perform safe and accurate dissection with minimal trauma and optimal functional preservation in patients with massive PBC.
Footnotes
Author Contributions
M.M.Y. is a major contributor to writing the manuscript; G.Z. revises the manuscript; L.F.T. edits the manuscript and provides vital opinions; Z.W.X. provides help in gathering the data; J.X.H. analyzes the data; Y.Y.S. provides the idea and instructions. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Shanghai 2020 Science and Authors’ contributions Technology Innovation Action Plan One Belt One Road International Cooperation Project (20410740600 to Y.Y.), Clinical Research Plan of SHDC (SHDC2020CR1049B to Y.Y.), Youth fund of national natural science foundation of China (81400460 to Z.G.)
