Abstract
Importance
Effective anesthesia with minimal side effects is essential for pediatric patients undergoing adenoidectomy.
Methods
A single-center, prospective, randomized controlled trial was conducted at Gaoming District People’s Hospital (China, January 2022-October 2023). Two-hundred fifty-six children (ASA I-II, age 3-12 years) scheduled for elective plasma radiofrequency adenoidectomy were randomized to 4 groups (n = 64 each). Group A received 0.9% normal saline; groups B, C, and D received continuous infusions of benzenesulfonate remimazolam at 0.4, 0.6, and 0.8 mg/kg/h, respectively, combined with 6% sevoflurane for induction and 2% to 3% for maintenance. Primary outcomes were emergence time and postanesthesia care unit (PACU) stay. Secondary outcomes included incidence of adverse events, peak Pediatric Anesthesia Emergence Delirium (PAED) scores, and parental satisfaction.
Results
Median awakening time was longer in groups B, C, and D than in group A (P < .001). PACU stay was shorter in groups C and D versus group A (P < .05). Postoperative pain scores (Face, Legs, Activity, Cry, and Consolability) were lower in groups C and D than in groups A and B (P < .05). Peak PAED scores were lower in groups B, C, and D than in group A (P < .05); negative behavioral changes on postoperative day 3 were also reduced (P < .05). Parental satisfaction was higher in groups B, C, and D than in group A (P < .001). No serious adverse events occurred.
Conclusion and Relevance
Benzenesulfonate remimazolam (0.6 mg/kg/h) combined with sevoflurane provides effective, safe anesthesia for pediatric plasma radiofrequency adenoidectomy, balancing induction efficacy, sedation depth, and safety. This regimen reduces emergence delirium, postoperative pain, and negative behavioral changes while enhancing parental satisfaction, supporting its use in pediatric adenoidectomy and warranting further investigation in other pediatric procedures.

Key Messages
Remimazolam combined with sevoflurane provides effective and safe anesthesia for pediatric plasma radiofrequency adenoidectomy.
A medium dose of remimazolam (0.6 mg/kg) offers the optimal balance between induction efficiency, sedation depth, and safety.
Introduction
Adenoid hypertrophy is a condition caused by pathological enlargement of the adenoids and is common in children. Multiple techniques have been described for adenoid removal, with plasma adenoidectomy demonstrating potential advantages in terms of reduced intraoperative bleeding and decreased residual tissue.1 -3 Anesthesia management plays a crucial role in pediatric surgeries. Given the younger age, incomplete physical development, and weaker immune and cardiopulmonary reserve of children, the incidence of anesthesia-related complications and mortality remains relatively high. Choosing an appropriate anesthesia plan is not only essential for the smooth progress of the surgery but also directly affects postoperative recovery and long-term prognosis.
Currently, sevoflurane-based inhalation anesthesia is widely used in pediatric adenoidectomy. Sevoflurane is a novel inhalation anesthetic, that is, colorless, transparent, and has a mild fragrance. It induces anesthesia quickly and has minimal impact on the respiratory and circulatory systems, making it well-tolerated by children and one of the most-commonly-used anesthetics in pediatric surgeries in recent years. 4 However, emergence delirium (ED) is a common neurological complication and behavioral disorder after sevoflurane general anesthesia in children, often occurring in the early-postanesthesia period and manifesting as crying, convulsions, restlessness, and disorientation. 5 The incidence of ED in children after sevoflurane anesthesia can be as high as 80%, much higher than in adults. 6 Additionally, postoperative pain from tonsillectomy and adenoidectomy can increase the incidence of ED. Furthermore, it can lead to an increased risk of self-harm, wound dehiscence, accidental removal of intravenous catheters, and other adverse postoperative behaviors, posing a threat to patients and their parents, leading to a poor treatment experience, and potentially increasing the workload of postanesthesia care unit (PACU) nurses. 7 Several drug interventions have been studied to prevent ED after sevoflurane anesthesia to achieve a smooth awakening, such as midazolam and propofol.8,9 However, midazolam has a slow onset of action, a half-life of over 1 hour, and active metabolites, leading to a longer awakening time and possible residual effects after sedation. 10 Propofol can cause injection pain, 11 hypotension, and hypoxia, 12 and lacks a specific antidote. In cases of drug accumulation and delayed awakening, it takes a long time to metabolize. Long-term, high-dose infusion may lead to propofol infusion syndrome, causing serious complications such as metabolic acidosis, hyperlipidemia, hepatic steatosis, and muscle injury, and even death. 13
Benzenesulfonate remimazolam is a novel ultra-short-acting benzodiazepine that acts on the gamma-aminobutyric acid type A receptor. It changes the concentration of chloride ions on both sides of the cell membrane, increasing the membrane potential and causing hyperpolarization, thereby inhibiting neuronal electrical activity and producing a sedative effect. The metabolism of benzenesulfonate remimazolam does not depend on liver or kidney function. It is hydrolyzed by nonspecific esterases in the body into zaleplon propionic acid, which is pharmacologically inactive. It has the advantages of rapid onset and offset, short sedation recovery time, no injection pain, and minimal inhibitory effects on the cardiovascular and respiratory systems. 14 Its awakening time and safety profile are superior to midazolam or propofol, making it particularly suitable for special populations such as children, the elderly, obese patients, and those with impaired liver or kidney function. Additionally, the sedative effect of benzenesulfonate remimazolam can be reversed by the specific antagonist flumazenil.15,16 Numerous national and international studies have shown that benzenesulfonate remimazolam can be used for sedation in outpatient gastrointestinal endoscopy,17,18 bronchoscopy, 19 hysteroscopy, 20 and as an adjunct for spinal anesthesia, achieving appropriate depths of anesthesia sedation with high success rates. 21 Liu et al found that remimazolam can reduce the incidence of hypoxia in elderly patients undergoing gastroscopy. 22 It has also been reported that remimazolam can be safely used in patients with heart disease and cardiovascular patients. 23 Foreign studies on the use of remimazolam for general anesthesia induction and maintenance have shown that it is safe and feasible for tracheal intubation under general anesthesia, with a sedation success rate similar to propofol, and seemingly-better safety and hemodynamic stability. 24
Previous studies have shown that benzenesulfonate remimazolam has been safely used in pediatric general anesthesia.25 -27 However, most current research focuses on adult dosages, with fewer studies on pediatric dosages and unclear dose-effect relationships. Therefore, this study chose to combine benzenesulfonate remimazolam with sevoflurane as an anesthesia plan for pediatric surgeries, aiming to align with the medical concept of safe medication use, rapid awakening, and quick recovery for children. By evaluating the effects of different doses of benzenesulfonate remimazolam combined with sevoflurane in pediatric plasma radiofrequency adenoidectomy, this study aimed to provide scientific evidence for individualized medication for anesthesiologists, reduce surgical anesthesia risks, and accelerate the recovery process for children. Plasma radiofrequency adenoidectomy was chosen as the surgical technique due to its institutional prevalence (78% of pediatric adenoidectomies at our center) and documented benefits over cold steel and coblation, including reduced thermal injury (40 °C-70 °C vs 80 °C-120 °C), lower blood loss (<5 mL), and faster recovery (mean hospital stay 1.2 days). While these factors may influence postoperative pain and ED, the remimazolam-sevoflurane regimen’s rapid offset and hemodynamic stability are expected to remain effective across techniques. Additionally, this study fills the gap in research on the effective dosages of continuous infusion of benzenesulfonate remimazolam and sevoflurane inhalation in pediatric plasma radiofrequency adenoidectomy, providing important reference value for clinical rational drug use.
Materials and Methods
Study Design and Subjects
A single-center, prospective, randomized controlled study was conducted, during the period from January 2022 to October 2023, and 256 consecutive pediatric patients were enrolled from the otolaryngology surgical schedule at Gaoming District People’s Hospital. All patients underwent isolated adenoidectomy without tonsillectomy. Each day, research staff reviewed the following day’s operating list to identify all children aged 3 to 12 years scheduled for elective plasma radiofrequency adenoidectomy; electronic health records were then used to exclude candidates with a BMI ≥25 kg/m2, recent upper respiratory infection, or a history of difficult airway. Eligible families were approached in the preoperative holding area by a dedicated research nurse, who explained the study protocol and obtained written informed consent from legal guardians before any study procedures were initiated. Randomization was performed using a computer-generated block randomization scheme with block size of 8, allocating participants in a 1:1:1:1 ratio to group A, B, C, or D (n = 64); allocation concealment was ensured through sequentially-numbered, opaque, sealed envelopes. This study has been approved by the Ethics Committee of Gaoming District People’s Hospital in Foshan City, with the ethics number: 2020117. Informed consent has been obtained from the parents of all participants. Sample size was calculated using the HHU G*Power 3.1 (F-test, ANOVA) based on awakening time (primary outcome). Pilot data indicated a mean difference of 2.5 minutes among groups with a common standard deviation (SD) of 3.5 minutes. With α = .05 (Bonferroni-corrected to 0.0083 for 6 pairwise comparisons) and power = 0.80, 56 participants per group were required. To allow for a 10% dropout rate, 64 participants per group were enrolled (total n = 256). The clinical trial is registered with the number: ISRCTN67586415.
Inclusion criteria: ASA physical status I to II; age 3 to 12 years; no difficult airway, Mallampati grades I to II; no upper respiratory tract infection in the past month, no neurological, cardiopulmonary, hepatic, or renal systemic diseases; routine blood tests, coagulation function, electrolytes, hepatic and renal function tests, chest X-rays, and electrocardiograms show no significant abnormalities; children scheduled for pediatric plasma radiofrequency adenoidectomy; informed consent from the patient’s family. All children underwent preoperative airway assessment (Mallampati I-II, adequate thyromental distance, and no history of difficult intubation or obstructive sleep apnea) to ensure suitability for oral intubation and to minimize airway-related complications.
Exclusion criteria: BMI ≥25 kg/m2; upper respiratory tract infection, cardiopulmonary insufficiency within 1 month; perioperative allergies; unpredicted difficult airway; failed intubation on the first attempt; severe complications in children, such as hemorrhagic shock, anaphylactic shock, and cardiac arrest; changes in surgical or anesthesia plans due to medical conditions; withdrawal from the study at the request of the patient’s family; loss to follow-up of the subjects were considered.
Treatment
Preoperative preparation: All children were routinely fasted for 6 hours and restricted from drinking for 2 hours. The modified Yale Preoperative Anxiety Scale (m-YPAS) 28 was used to assess preoperative anxiety in the waiting area before surgery. On the day of surgery, intravenous access was established and fluids were administered as needed. Routine monitoring of vital signs was conducted after entering the operating room.
Grouping: Children in groups A, B, C, and D were all induced with sufentanil 0.3 µg/kg, benzenesulfonate remimazolam 0.3 mg/kg, benzenesulfonic acid rocuronium injection 0.15 mg/kg, and atropine 0.1 mg/kg for intravenous anesthesia induction. After successful intubation, groups B, C, and D immediately started continuous infusion of benzenesulfonate remimazolam at rates of 0.4, 0.6, and 0.8 mg/kg/h, respectively, and maintained with sevoflurane inhalation with oxygen in medical air. Continuous infusion of benzenesulfonate remimazolam was maintained intraoperatively and discontinued at the conclusion of skin closure. The control group A received an equal volume of 0.9% normal saline. The Bispectral Index value was maintained between 40 and 60 throughout the procedure. Nitrous oxide was not administered in any group; anesthesia was maintained exclusively with sevoflurane and remimazolam infusion.
The surgical incision was made after the minimum alveolar concentration of sevoflurane was stabilized at 0.65. All vital signs were monitored and reported to the attending anesthesiologist. During the trial, in cases of hypotension, ephedrine 0.1 mg/kg was administered; for bradycardia, atropine 0.01 mg/kg was given; and for any psychiatric symptoms, comfort and symptomatic treatment were provided.
Information Collection
The study compared the anesthesia effects, ED, postoperative pain intensity, parental satisfaction, adverse reactions, and behavioral changes 3 days after surgery among the 3 groups of children. (1) Anesthesia effects: Anesthesia time, awakening time (the interval from cessation of inhalation anesthesia to eye opening on verbal command), and PACU stay time were measured. (2) ED situation: ED was assessed using the Pediatric Anesthesia Emergence Delirium (PAED) scale, which includes 5 items: eye contact, purposeful movement, awareness of the surrounding environment, restlessness, and inconsolability. The scoring for items 1, 2, and 3 is as follows: 4 = not at all, 3 = a little, 2 = quite a bit, 1 = very much, 0 = extremely. Items 4 and 5 are scored in reverse. ED is defined as a total PAED score of ≥10. 29 (3) Postoperative pain intensity: The Face, Legs, Activity, Cry, and Consolability (FLACC) scale was used to assess pain intensity every 10 minutes during the first 30 minutes in the PACU. The FLACC scale consists of 5 behaviors (face, legs, activity, cry, and consolability), each rated by the observer on a scale of 0 to 2. 30 (4) Parental satisfaction was self-reported using a numerical rating scale (range 0-10) 24 hours after surgery, where 0 indicates dissatisfaction and 10 indicates complete satisfaction. (5) Adverse reactions included dizziness, hallucinations, restlessness, laryngospasm, nausea and vomiting, hypertension (an increase in mean arterial pressure of more than 30% above baseline), and tachycardia (tachycardia was defined as a heart rate increase of ≥30% above the preinduction baseline value, consistent with pediatric anesthesia standards). 31 (6) Behavioral changes 3 days after surgery: On the third day after surgery, negative behavioral changes were determined using the 27-item Post-Hospital Behavior Questionnaire (PHBQ) through telephone interviews. 32 The PHBQ includes the following subscales: general anxiety and regression, separation anxiety, sleep anxiety, feeding disorders, aggression toward authority, and withdrawal. For each item, parents were asked to compare the child’s current behavioral problems with those before hospitalization (1 = much less, 2 = less, 3 = no change, 4 = more, 5 = much more).
Statistical Analysis
The statistical analysis was conducted using the IBM SPSS 20.0 software. Normally-distributed continuous data are presented as mean ± SD (x ± s), and comparisons between groups were performed using the independent samples t-test. Non-normally-distributed continuous data are presented as median (M) and interquartile range (IQR), and comparisons between groups were performed using nonparametric rank-sum tests. Categorical data are presented as frequency (%), and comparisons were performed using the chi-squared (χ2) test. A P < .05 was considered statistically significant.
Results
General Patient Information
Figure 1 illustrates the eligibility, randomization, and follow-up of the participants. Between January 2022 and October 2023, a total of 260 participants were enrolled in the study. Twenty-three participants were excluded for the following reasons: 2 did not meet the inclusion criteria, and 2 declined to participate. In groups A to D, 4, 2, 6, and 7 participants, respectively, violated the study protocol. Consequently, the final analysis included 60 participants in group A, 62 in group B, 58 in group C, and 57 in group D. The demographic characteristics (age, height, weight, gender, and ASA status) and perioperative clinical data (m-YPAS scores, surgical duration, and anesthesia time) were similar across all groups (Table 1).
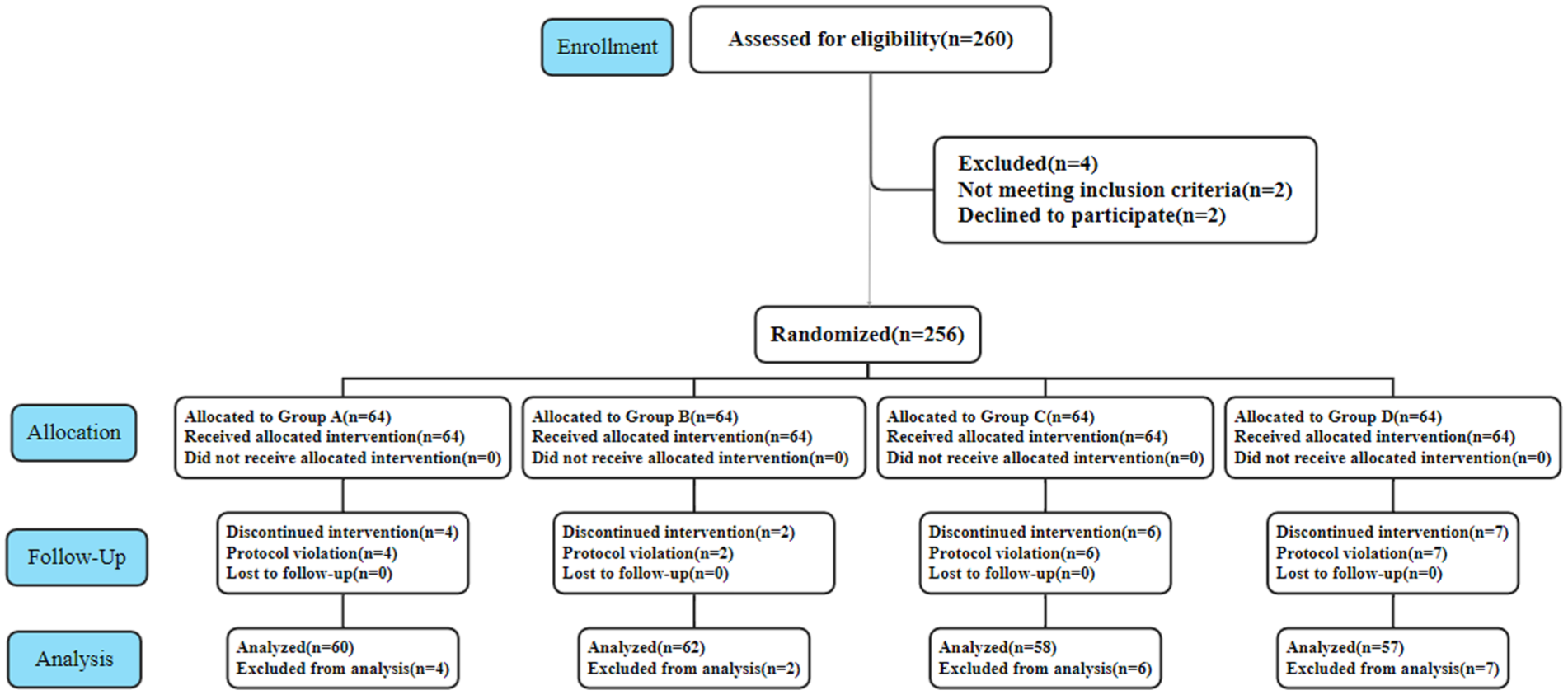
Screening process of subjects during the study.
Demographic and Perioperative Characteristics.

Data are presented as mean ± SD, P value; median (IQR, P value) or number (%, P value).
Abbreviations: ASA, American Society of Anesthesiologists; IQR, interquartile range; m-YPAS, modified Yale Preoperative Anxiety Scale; SD, standard deviation.
Postoperative Adverse Reactions
As shown in Table 2, compared with group A, the median awakening time was prolonged in groups B, C, and D (P < .001). However, the PACU stay time was similar between groups A and B (median difference −3 minutes, 95% CI 0-1 minutes; P = .217). In contrast, the PACU stay time was significantly shorter in groups C and D than in group A (P < .001), with no significant difference between groups C and D. Postoperative pain scores indicated no significant difference between groups A and B at 10, 20, and 30 minutes, while groups C and D had significantly-lower scores, indicating a significant alleviation of postoperative pain. The peak FLACC pain scores for groups A and B were 2.0 (IQR, 1.0-3.0), while for groups C and D, they were 2.0 (IQR, 0-2.0). The peak PAED scores [median (IQR)] for groups B, C, and D were 7 (6-7), 6 (4-8), and 5 (4-9), respectively, all significantly lower than group A (P < .001; Figure 2).
Postoperative Outcomes and Complications.
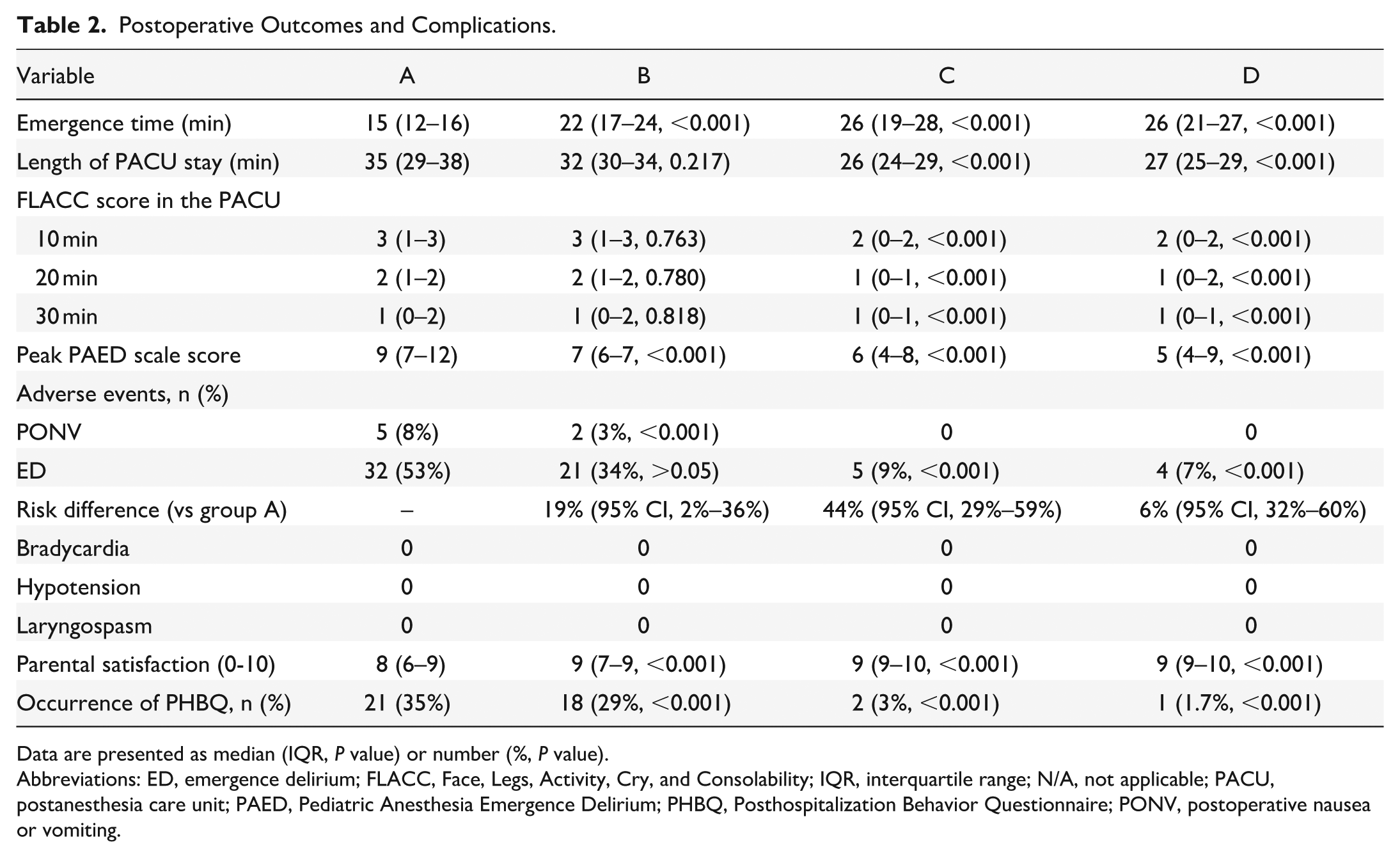
Data are presented as median (IQR, P value) or number (%, P value).
Abbreviations: ED, emergence delirium; FLACC, Face, Legs, Activity, Cry, and Consolability; IQR, interquartile range; N/A, not applicable; PACU, postanesthesia care unit; PAED, Pediatric Anesthesia Emergence Delirium; PHBQ, Posthospitalization Behavior Questionnaire; PONV, postoperative nausea or vomiting.

Box plot of peak PAED scores in each group. PAED, Pediatric Anesthesia Emergence Delirium.
As shown in Table 2 and Figure 3, the incidence of ED in group A was 32/60 (53%), while in groups B, C, and D, the incidences were 21/62 [34%, risk difference 19% (95% CI, 2%-36%), relative risk = 0.64 (95% CI, 0.42-0.97); P > .05], 5/58 [9%, risk difference 44% (95% CI, 29%-59%), relative risk = 0.17 (95% CI, 0.07-0.43); P < .001], and 4/57 [7%, risk difference 46% (95% CI, 32%-60%), relative risk = 0.13 (95% CI, 0.05-0.35); P < .001], respectively.

Bar graph of the incidence of delirium during recovery in each group.
On the third day after surgery, the incidence of negative behavioral changes was significantly lower in groups B to D than in group A (P < .001). Postoperative nausea or vomiting occurred in 5 out of 60 participants in group A (8%), 2 out of 62 in group B (3%), and none in groups C and D. No adverse events such as bradycardia, hypotension, laryngospasm, or hypoxia were observed during the study period. Additionally, parental satisfaction scores were significantly higher in groups B, C, and D than in group A (P < .001), and the scores in groups C and D were also significantly higher than in group B (P < .001), with no significant difference between groups C and D.
Discussion
This study aimed to investigate the application effects of different doses of benzenesulfonate remimazolam combined with sevoflurane in pediatric adenoidectomy. The results showed that compared with 0.9% normal saline, continuous infusion of remimazolam at 0.6 mg/kg/h during surgery could significantly reduce the possibility of ED after sevoflurane anesthesia, alleviate postoperative pain, and effectively improve parental satisfaction, without extending the PACU stay or increasing clinical adverse events. This is consistent with previous research findings. For example, Bai et al 33 compared the application effects of remimazolam and propofol in pediatric painless gastroscopy and found that the 2 were equivalent in sedative effects, but benzenesulfonate remimazolam had the advantages of low incidence of injection pain and fast awakening time. Yang et al demonstrated that remimazolam infusion (0.6 mg/kg/h) reduced ED after sevoflurane anesthesia in children aged 3 to 12 years, consistent with our results. 26 Fang et al reported equivalent induction success between remimazolam (0.3 mg/kg bolus) and propofol in 192 children aged 3 to 6 years, supporting the safety of remimazolam bolus dosing. 34 Dai et al showed remimazolam (0.2-0.3 mg/kg bolus) provided hemodynamic stability comparable to propofol during adult general anesthesia induction. 24 Zhang et al found remimazolam infusion (0.6-1.0 mg/kg/h) reduced postoperative pain and opioid requirements in adults undergoing hysteroscopy. 20
At present, the application of remimazolam in pediatric anesthesia is still in its infancy, and the dosage needs to be further studied in depth. Fang et al 34 conducted a clinical study in 192 children aged 3 to 6 years old and found that intravenous injection of remimazolam at 0.3 mg/kg for anesthesia induction was equivalent to intravenous injection of propofol at 2.5 mg/kg; while continuous infusion of remimazolam at a rate of 1 to 3 mg/kg/h for anesthesia maintenance was equivalent to continuous infusion of propofol at 4 to 12 mg/kg/h. The “Expert Opinions on the Clinical Application of Remimazolam” 35 pointed out that remimazolam can be used alone or in combination with propofol, sevoflurane, desflurane, etc, in conjunction with opioid drugs to maintain an appropriate depth of anesthesia. The dosage of remimazolam can be adjusted as needed during surgery, and the suggested infusion rate is 0.2 to 1.0 mg/kg/h. For weak patients, it is recommended to choose a smaller dose, and flumazenil can be given for antagonism if necessary, but the dosage in children is still unclear. The findings of this study further enrich the application data of remimazolam in pediatric anesthesia. Unlike other studies using intravenous bolus injection, this study chose the infusion method. With the increase in the infusion dosage of benzenesulfonate remimazolam, there was a better trend in PACU stay time, postoperative pain control, ED incidence, and parental satisfaction. This indicates that the infusion method can more accurately control the drug concentration, maintain a stable depth of anesthesia, and reduce drug adverse reactions. However, the median awakening time was extended, suggesting that the clinical need to balance the benefits brought by the increased dosage with the extended awakening time, and find the best dosage balance point to achieve optimal anesthesia management.
Although the application of plasma radiofrequency ablation in adenoidectomy has significant advantages, such as less intraoperative bleeding, shorter surgical time, and faster postoperative recovery, whether it is applicable to all types of adenoidectomy still needs further discussion.36,37 The findings of this study show that the anesthesia regimen of benzenesulfonate remimazolam combined with sevoflurane is not only suitable for plasma radiofrequency adenoidectomy but may also have similar clinical effects on other types of adenoidectomy. However, as there may be differences in intraoperative bleeding, surgical time, and postoperative recovery among different surgical techniques, it is necessary to take into account the characteristics of the surgical technique and the specific conditions of the patient when promoting this anesthesia regimen. For example, patients with coagulation disorders or severe cardiovascular diseases may need to carefully choose the surgical method. In addition, for some complex cases of adenoid hypertrophy, a combination of other surgical techniques may be needed to achieve the best therapeutic effect. In future research, we plan to further explore the optimized combination of different surgical techniques and anesthesia regimens to provide more personalized and precise clinical solutions for the treatment of adenoid hypertrophy.
However, this study also has certain limitations. The study was only conducted in a single center, and although the sample size reached a certain scale, there is still room for expansion. The open-label design (no blinding) may introduce observer and participant bias, potentially reducing internal and external validity—especially for subjective outcomes such as parental satisfaction. Additionally, there may be regional and sample selection biases, which limit the general applicability of the research results. In terms of research indicators, although many aspects are covered, the monitoring of some potential long-term neurocognitive effects and immune function changes is still insufficient. Moreover, this study lacks a direct comparison between plasma radiofrequency and traditional adenoidectomy techniques (such as cold curettage or electrocautery). Therefore, the observed perioperative benefits in children (such as less bleeding, shorter surgery time, or better anesthesia effects) may stem from the minimally-invasive approach. Future research directions can focus on multicenter and large-sample study designs to further verify the reliability and wide applicability of the research results. In-depth exploration of the pharmacokinetics and pharmacodynamics of benzenesulfonate remimazolam in children, combined with factors such as genetic polymorphism to analyze the impact of individual differences on drug reactions, will help more accurately determine individualized drug dosages. Additionally, the research can be expanded to different age groups and children with different comorbidities to improve its application system in the field of pediatric anesthesia and provide a more solid guarantee for the safety of pediatric anesthesia. Future studies should also directly compare plasma radiofrequency with traditional adenoidectomy techniques to confirm whether these perioperative advantages are technique-specific.
Conclusion
In conclusion, in pediatric adenoidectomy using plasma radiofrequency ablation, the combination of benzenesulfonate remimazolam and sevoflurane shows significant clinical effects. Continuous infusion of remimazolam at 0.6 mg/kg/h effectively reduces the incidence of ED, alleviates postoperative pain, and enhances parental satisfaction, without extending the PACU stay or increasing adverse events. Compared to 0.8 mg/kg/h, the 0.6 mg/kg/h dose offers greater safety and cost-effectiveness.
Footnotes
Acknowledgements
Here, we would like to express our sincere gratitude to all the people and institutions who have contributed to this study. We would like to thank the medical staff of the operating room, anesthesia department, pediatrics, and other related departments of Gaoming District People’s Hospital of Foshan, who provided professional assistance and support in patient recruitment, surgical operations and perioperative care, ensuring the smooth progress of the study. At the same time, we would like to thank the staff who participated in the data collection and collation of this study. Their rigorous and serious work attitude ensured the accuracy and completeness of the data. In addition, we would like to thank the patients and their families for their active cooperation and trust. Their participation was the key factor in the completion of this study. Finally, we extend our gratitude to Professor Tan Meiyun and her research team from the Department of Anesthesiology at Boai Hospital of Zhongshan for their invaluable assistance and guidance. Their professional opinions helped us continuously improve the research plan and analysis methods, and improved the quality and level of the research.
Authors’ Note
Whether it is economic interests, such as potential interest associations with pharmaceutical companies or medical device manufacturers, or noneconomic interests, such as academic competition, personal bias, no factors have had an improper impact on the design, implementation, data analysis, and result reporting of this study, ensuring the authenticity and reliability of the research results and maintaining the purity and scientific nature of academic research.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Research Project in Medical Science and Technology, Foshan Science and Technology Bureau (project number: 232000100734). The fund provided the necessary financial support for the study, covering the purchase of experimental equipment, reagent and consumables, data collection and analysis costs, and part of the labor costs of researchers. The fund sponsor did not intervene in the research direction, research design, and result analysis during the research process, ensuring the independence and autonomy of the research, allowing the research to proceed smoothly according to the established scientific goals, and providing a solid material guarantee for exploring the application effect of different doses of remimazolam combined with sevoflurane in pediatric adenoidectomy.
Ethical Considerations
This study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of Gaoming District People’s Hospital of Foshan.
Consent to Participate
Informed consent was obtained from all legal guardians.
