Abstract
Importance
Unlike other head and neck cancers, head and neck soft tissue sarcoma (HN-STS) is staged similarly to sarcomas in the trunk and extremities. The current American Joint Committee on Cancer (AJCC) staging system has limitations that hinder accurate prognosis prediction for HN-STS.
Objective
We aimed to develop a novel location-grading-node-metastasis (LGNM) staging system based on the primary tumor location to more accurately stratify prognosis for HN-STS.
Design
A retrospective case series from 1990 to 2021.
Setting/Participants
This study included 471 patients diagnosed with HN-STS at Sun Yat-sen University Cancer Center between 1990 and 2021.
Main outcome measures
In the primary analysis, we obtained the overall survival (OS) rate. Secondary measures included area under the receiver operating characteristic curve, Harrell’s C, Somers’ D, Gönen and Heller’s K, O’Quigley’s ρ2k, Royston’s R2, the Bayesian information criterion for concordance, and variation in patient outcomes.
Results
The eighth edition of AJCC T classification for tumor size inadequately conveys prognosis information. In contrast, the primary tumor location and local invasion are prognostic factors for HN-STS and categorized into 4 stages: L1 (low risk: scalp, face, supraclavicular, ear), L2 (intermediate risk: neck, paravertebral, pharynx, tonsil, eye, orbit), L3 (high risk: cavity, lip, palate, buccal mucosa, salivary gland, maxilla, mandible), and L4 (any location with local invasion). The new LGNM staging system effectively distributed patients into stages I to IV, with statistically-significant survival differences among these stages. Five-year OS rates were 96.9% for stage I, 78.4% for stage II, 37.1% for stage III, and 7.1% for stage IV (P < .001). Additionally, the LGNM staging system demonstrated superior predictive ability and concordance compared with the seventh and eighth editions of AJCC staging systems.
Conclusions/Relevance
The LGNM staging system shows better homogeneity and discriminatory power than the AJCC system, improving risk stratification and prognosis prediction in HN-STS.

Introduction
Soft tissue sarcomas (STSs) are rare, heterogeneous, and mesenchymal originated with low incidence and high mortality.1,2 Head and neck-STS (HN-STS), which accounts for ~5% to 10% of STS and about 1% of head and neck cancers,3-5 has worse oncologic outcomes than STS that occurs in other primary sites. 6 With a poor 5 year overall survival (OS) of 31% to 80%,7-9 HN-STS shows a local recurrence of 20% and a distant metastasis of 10% to 40%. 9 Among HN-STS, fibrosarcoma and undifferentiated polymorphic sarcoma are characterized as a high tendency of local aggressiveness and distant metastasis. 10 Considering effective treatments are limited for HN-STS, surgical resection still plays a crucial role. However, even in the early stages, complex anatomical structures and adjacent critical sites in head and neck make obtaining negative surgical margins challenging. Therefore, there is an urgent need for identifying validated biomarkers predicting prognosis in HN-STS to stratify risks.
Tumor staging systems guide severity evaluations and treatment decisions. Most head and neck cancers follow the specialized American Joint Committee on Cancer (AJCC) staging systems. The characteristics and prognosis of HN-STS is different from other head and neck cancers, and it is not distinguished as separate entities and staged consistently to the STS located in trunk and extremities.11,12 Apparently, the lack of a dedicated HN-STS staging system has negative impacts on patient risk stratification and prognostic prediction. Although, in 2017, the AJCC staging system was revised to the eighth edition, proposing the development of an HN-STS staging system with the updated definition of T classification. However, except for the renewed T classification based on both previous issues and expert consensus and the elimination of the primary tumor depth, the definition of nodal involvement (N), histological grade (G), and distant metastasis (M) went unchanged. 12 It is also noteworthy that due to its rarity and heterogeneity, the eighth edition failed to develop prognostic stage grouping. As a transition staging system for data collection, further improvement in and optimization of the existing classification are expected. Moreover, published literature regarding HN-STS staging system contains small numbers of cases or derived from the public database, whose G definition was usually inconsistent with the AJCC staging system.
Therefore, our study aimed to assess the predictive value of the T classification of the eighth edition of AJCC staging system, in terms of both tumor size and invasion. We sought to propose a novel prognostic location-grading-node-metastasis (LGNM) staging system and compare the ability of prognosis stratification between the LGNM staging system and the existing AJCC staging system.
Materials and Methods
Patient Characteristics
From January 1990 to January 2021, 536 patients with primary HN-STS were diagnosed and reviewed at Sun Yat-sen University Cancer Center. Prior to analysis, 31 presented a histology (mainly represented by angiosarcoma, desmoid-type fibromatoses, and embryonal rhabdomyosarcoma) that was not recommended for staging by AJCC criteria, therefore excluded from the study. Similarly, patients with incomplete medical records (n = 8), ages <16 years old (n = 12), nonmetastatic without R0 resection (n = 3), and those lost to follow-up (n = 11) were also excluded. Finally, 471 patients were included Figure S1), including 443 patients without distant metastasis who underwent R0 resection, defined as complete surgical resection with a microscopically-negative margin.
Demographic data (sex and age), past medical history, and medical examinations of patients were collected retrospectively from electronic medical records. Tumor features (size, location, lymph invasion, distant metastasis, surgical margin, histopathological subtype) are defined primarily by surgical findings and ultimate pathologic analysis by an experienced pathologist. Patients with HN-STS were staged according to the AJCC seventh and eighth editions. 5 The grading system proposed by Fédération Française des Centres de Lutte Contre le Cancer (FNCLCC) was used for histological grading. 13 This retrospective study was approved by the Ethical Committee and Institutional Review Board of the Sun Yat-sen University Cancer Center (no. B2021-314-01) with informed consent exemption.
Follow-Up
All patients were routinely examined at least every 3 to 6 months in the first 3 years; after that, a follow-up examination was performed every 6 to 12 months until September 1, 2021, or death. Disease-free survival (DFS) was defined as the interval between R0 resection and the first detection of local recurrence, lymph node or distant metastasis, death, or recent follow-up. Tumor progression was confirmed by imaging and/or pathology. OS ranged from the date of initial histological diagnosis to the date of death for any cause or final follow-up.
Statistical Analysis
The Kaplan-Meier method was used to map the survival curve, and log-rank tests were used to examine the intergroup differences in survival time. Multivariate analyses with the Cox regression proportional hazards model were used to calculate the hazard ratio (HR) with 95% confidence intervals and evaluate the independent significance of related variables.
Several different statistical methods were used in evaluating different staging schemes. The prediction accuracy of the 5 year OS of each staging system was evaluated by comparing the area under the receiver operating characteristic curves (AUCs) generated from logistic regression analysis. The concordance between 2 staging systems was examined using Harrell’s C, 14 Somers’ D, 15 and Gönen and Heller’s K. 16 The extent of variation in patient outcomes interpreted by the regression models were assessed using O’Quigley’s ρ2k 17 and Royston’s modification thereof (R 2 ).18,19 Bootstrap techniques with 750 replications were used to calculated the estimates’ standard errors. The Bayesian information criterion (BIC) was calculated systematically for each staging system as a measure of model fit. All results were obtained using a 2-way hypothetical test of α = .05. P < .05 were referred as significant. SPSS version 26.0 (IBM Corporation, Chicago, IL, USA), Stata version 15.1 (StataCorp LLC, College Station, TX, USA), and R version 3.4.0 (Bell Laboratories, Murray Hill, NJ, USA) were used for the statistical analysis.
Results
Clinicopathologic characteristics of the cohort (N = 471) are described in Table 1. Males represented 63.3% of the total study population, and median age was 44.0 (range: 16.0-87.0). Histologic subtypes consisted of fibrosarcoma (n = 220, 46.7%), rhabdomyosarcoma (n = 67, 14.2%), and undifferentiated pleomorphic sarcoma/malignant fibrous histiocytoma (n = 45, 9.6%). The common primary sites were neck and parotid (n = 94, 20.0%), followed by scalp (n = 91, 19.3%), sinuses (n = 57, 12.1%), and face (n = 46, 9.8%). The median tumor size was 4.0 cm (interquartile range [IQR]: 2.5-5.5 cm), and 113 patients (24.0%) had invasive tumors. Considering the T category distribution according to the AJCC seventh edition, 131 (27.8%) patients were classified by T1a, T1b (n = 206, 43.7%), T2a (n = 29, 6.2%), and T2b (n = 105, 22.3%). By the AJCC eighth edition, T-stages were distributed as T1 (n = 82, 17.4%), T2 (n = 132, 28%), T3 (n = 144, 30.6%), T4a (n = 97, 20.6%), and T4b (n = 16, 3.4%). G1 tumors were identified in 185 (39.3%), G2 in 221 (46.9%), and G3 in 65 (13.8%) patients. Over half (56.7%) of the patients underwent R0 resection alone. After an operation (including R0 and palliative surgery), 55 patients (11.7%) underwent chemotherapy, 78 patients (16.6%) had radiotherapy, and 43 patients (9.1%) used chemoradiotherapy. Chemotherapy and radiotherapy were given to 89 of 471 (18.9%) patients before operation. The 5 year survival rate was 64.2% and the 10 year rate was 57.1% in the entire patients. Two hundred three (43.1%) patients had died with a median follow-up time of 134.6 months (IQR: 81.0-181.6). Furthermore, for the nondisseminated cohort, the 3 and 5 year DFS rates were 49.8% and 43.4%. Two hundred twenty-five (50.8%) patients suffered from local recurrence, and 41 (9.8%) patients had experienced distant metastasis.
Baseline Characteristics of All Patients (N = 471).

Values are presented as n (%) or median (range).
Abbreviations: AJCC, American Joint Committee on Cancer; G, grade; IQR, interquartile range; N, lymph nodes; NOS, not otherwise specified; T, primary tumor.
For the nonmetastatic cohort.
For the entire cohort.
Based on the Kaplan-Meier analysis, there were no statistical differences in OS among T1, T2, and T3 classifications and between T4a and T4b, indicating that T classification by the AJCC eighth edition is less important for prognosis (Figure 1A). However, pairing comparisons across the categories revealed a significant separation between T3 and T4a (P < .001), demonstrating that structural invasiveness portends a uniquely-poor prognosis. As expected, the examination of the invading structures category demonstrated that there was fair discrimination between OS with a statistical significance (P < .001) (Figure 1B). The primary tumor location was categorized as low, intermediate, and high risk according to Kaplan‑Meier OS curves (Figure 1C) and previous studies.9,20 The high-risk anatomical sites included the sinuses, nasal cavity, oral cavity, lip, palate, buccal mucosa, salivary gland, maxilla, and mandible. The intermediate-risk locations consisted of the neck, paravertebral, pharynx, tonsil, eye, and orbit, and the low-risk location was composed of the scalp, face, supraclavicular, and ear.
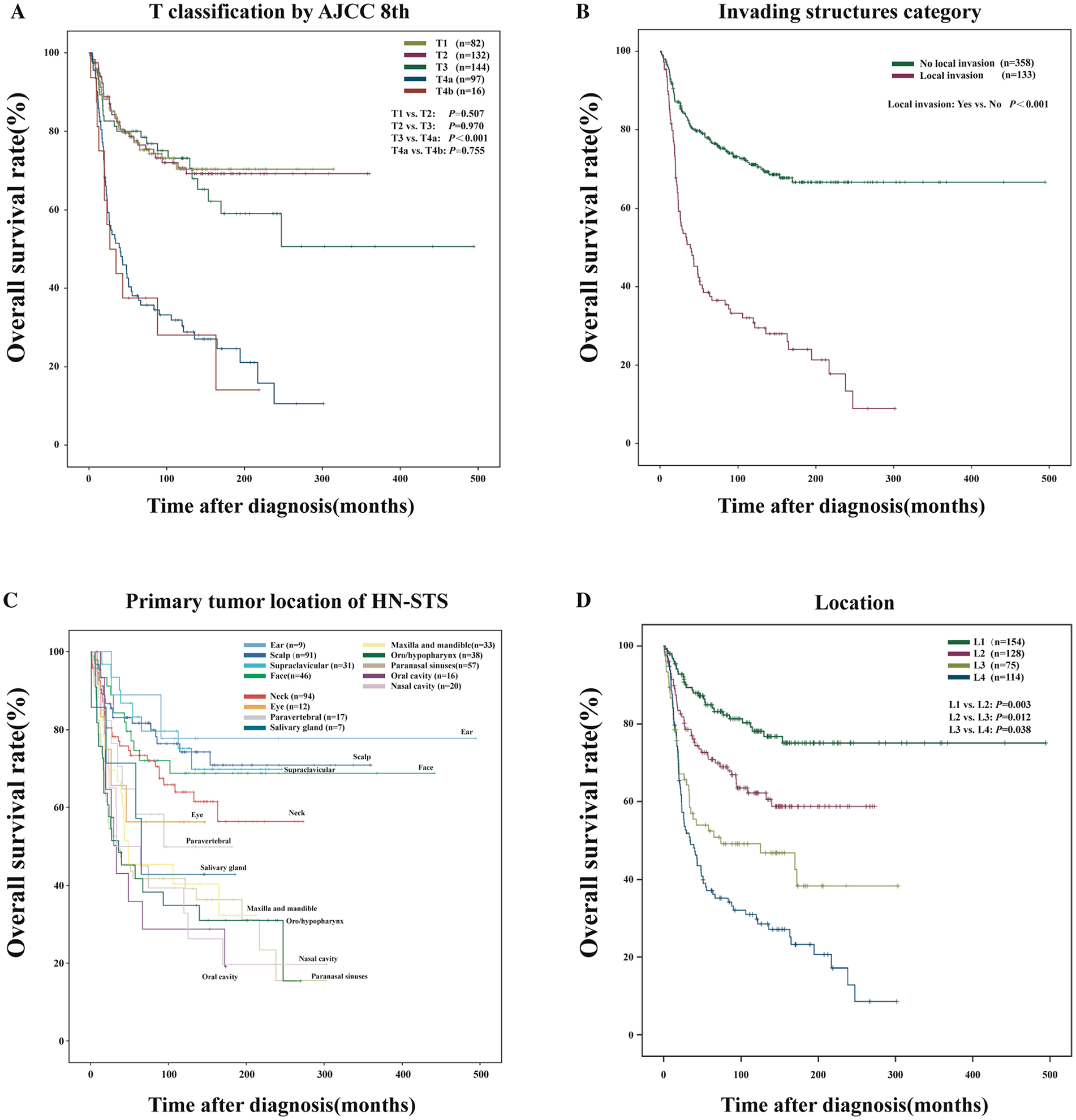
Kaplan-Meier curves showing the overall survival of patients with HN-STS according to (A) T classification by the AJCC eighth edition (T1-4), (B) invading structures category, (C) different primary tumor location, and (D) L1-4 stages in the entire cohort. AJCC, American Joint Committee on Cancer; HN-STS, head and neck soft tissue sarcoma; L, location; T, primary tumor.
Then, we combined the invasion status with primary tumor location subgroups to create L classifications, consisting of 4 stages: L1 (low-risk location: scalp, face, supraclavicular, and ear), L2 (intermediate-risk location: neck, paravertebral, pharynx, tonsil, eye, and orbit), L3 (high-risk location: cavity, lip, palate, buccal mucosa, salivary gland, maxilla, and mandible), and L4 (any location with local invasion) (Table S1). As shown in Figure 1D, the long-term OS shows segregation between L classifications, and the prognostic outcome of HN-STS patients exhibited a significant downward trend with the higher L stage (all: P < .05), with a 5 year OS rate of: 84.0% for L1, 70.9% for L2, 52.4% for L3, and 36.2% for L4, respectively. Additionally, pairing comparison reveals stark difference (P < .005) between categories (Table S2).
Multivariable analysis of the entire cohort demonstrated that the redefined L-stage remained an independent factor for OS along with N and G. In contrast, the T classification by the AJCC eighth edition is not. Specifically, the risk of mortality in L3 or L4 patients was 2.21 times and 3.69 times higher than in L1 patients (Table S3). Therefore, we devised the LGNM staging system using L classifications, FNCLCC grade, nodal involvement, and metastasis. Cases were grouped according to the LGNM, and stage groupings were assigned based on the evaluation of Kaplan-Meier survival curves and 5 year OS rates as follows (Figure 2A and Table S4): stage I (L1-3N0M0G1), stage II (L1-2N0M0G2-3 and L4N0M0G1), stage III (L3-4N0M0G2-3 and any LN1M0anyG), and stage IV (any LanyNM1anyG). Table S1 shows the AJCC and LGNM classifications. Finally, of the 471 patients, 140, 129, 174, and 28 were classified as stages I to IV based on the LGNM staging system, respectively. Schema of the changes from the AJCC seventh edition and LGNM staging systems for HN-STS are shown in Figure 2B and Table S5. Migration from the AJCC seventh edition to the proposed the LGNM stage system resulted in a decrease in the stage I and stage II populations and an increase in the stage III population. Stage I of the AJCC seventh edition showed the most diversified stage migration by the LGNM. Specifically, in 158 cases classified as stage I by the AJCC seventh edition, 140 cases remained as stage I, 17 cases were upstaged as stage II, and 1 case was upstaged as stage III by the LGNM. Two hundred eighteen cases classified as stage II based on the AJCC seventh edition were redistributed into 107 cases as stage II and 111 cases as stage III by the LGNM. Stages IV remained the same in those 2 staging systems.

(A) Staging algorithm for the proposed staging system and (B) stage migration from the AJCC seventh edition to the proposed LGNM staging system in the entire cohort. AJCC, American Joint Committee on Cancer; LGNM, location-grading-node-metastasis.
The 3 year OS by the novel LGNM system from stages I to IV were 98.5%, 86.5%, 46.3%, and 17.9% (P < .001), respectively, and the 5 year OS were respectively 96.9%, 78.4%, 37.1%, and 7.1%, (P < .001; Figure 3A). The HRs of the death risk based on the LGNM staging system were 7.45 for stage II, 26.34 for III, and 71.82 for IV, with stage I as the reference (Table S6). In accordance with the AJCC seventh staging scheme, the 5 year OS for each stage is as follows: 94.5% for stage I, 56.5% for stage II, 38.8% for stage III, and 7.1% for stage IV. Although the difference in the survival of stage I versus stage II (P < .001) and stage III versus IV (P < .001) was similarly significant, there was no significant difference between stage II and stage III comparisons (P = .072; Figure 3B). The AUC calculation showed better predictive ability for the LGNM staging system than the AJCC seventh staging system. The prognostic accuracy of the LGNM was notably higher than the AJCC seventh edition (0.853 vs 0.789, P = .018; Figure 3C).
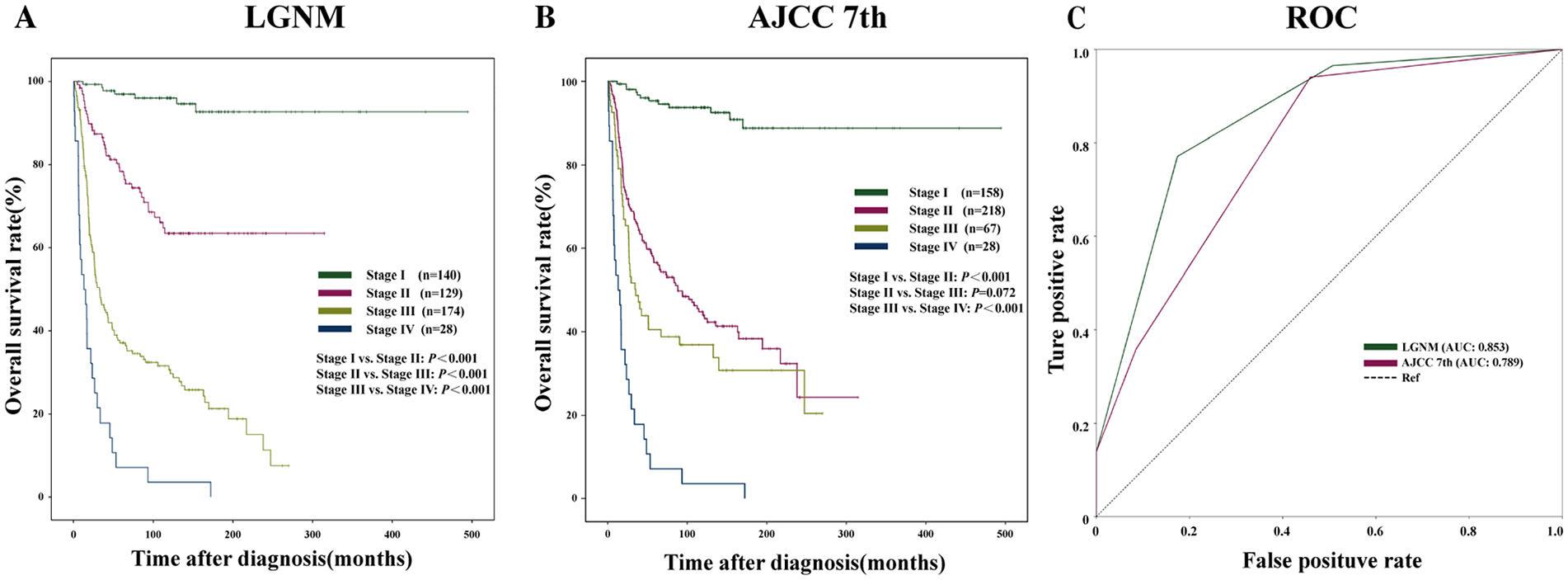
Kaplan-Meier plots of OS by (A) the proposed LGNM staging system and (B) the AJCC seventh edition staging system. For the LGNM, differences among the stages I to IV were significant (P < .001). (C) ROC curve analysis of the proposed LGNM staging system and the AJCC seventh edition staging system for the prediction of OS. AJCC, American Joint Committee on Cancer; AUC, area under the ROC curve; LGNM, location-grading-node-metastasis; OS, overall survival; Ref., reference line; ROC, receiver operating characteristic.
The LGNM staging system showed better concordance (larger Harrell’s C, larger Somers’ D, and larger Gönen and Heller’s K) and higher measures of explained variation (larger O’Quigley’s ρ 2 k and Royston’s R 2 ) than the AJCC seventh staging system. Moreover, the LGNM staging system BIC value was lower than that of the AJCC seventh staging system, suggesting that the proposed staging system provides a better fitting and more parsimonious regression model (Table 2).
Concordance Indices, Measures of Degree of Variation Accounted for, and BIC for Each Staging System for HN-STS (N = 471).

Abbreviations: AJCC, American Joint Committee on Cancer; BIC, Bayesian information criterion; HN-STS, head and neck soft tissue sarcoma; LGNM, location-grading-node-metastasis; NA, not applicable.
Discussion
In this study, we constructed a novel L-stage in the present study by incorporating the primary tumor location and local invasion. Moreover, we replaced the T classification of the eighth staging system with L-stage to set up the LGNM staging system for patients with HN-STS. To the best of our knowledge, this represents the only attempt to integrate the anatomical site with the stage strategy of HN-STS to date and represents the largest, single-center reported series of HN-STS in such a heterogeneous and unusual condition.
Based on the reliable cancer staging system, clinicians could uniformly describe the severity of illness, effectively communicate therapeutic plans, and informs patients on expected survival outcomes. An inferior staging system complied by physicians would waste enormous health care resources and lead to inappropriate treatment regimens. In addition, it is generally known that HN-STS have unique biological behaviors tending to present with smaller tumor sizes, disproportionately-greater risk of local recurrence, and a lower likelihood of distant metastasis at presentation than the other anatomical sites. However, HN-STS was staged identically to the trunk and extremity STS criteria for a long time until the AJCC eighth edition staging system was developed. This new dedicated regime proposed separate disease staging for HN-STS for the first time but, unfortunately, did not provide the prognostic stage grouping strategy due to a lack of data. Therefore, a location-specific staging algorithm for HN-STS based on population evidence was urgently needed.
Previous studies showed that tumor size strongly correlated with the prognosis of the solid tumor, including sarcomas.21-23 For the AJCC eighth edition staging system for HN-STS, the most predominant change was reflected in the T classification schema with 2.0 and 4.0 cm cutoff values. Cates analyzed the data of 2756 HN-STS patients from the Surveillance, Epidemiology, and End Results (SEER) database, which showed that HR synchronized with tumor size continuously and therefore establishing a more complex staging system to evaluate prognosis using the new T-stage. 20 In contrast, several studies reported that the current size classifications seem somewhat arbitrary and are not convincing. For example, a small retrospective series by Kim et al found no statistical significance among T1-2 and T3-4 tumors using the AJCC eighth edition staging system in terms of OS. 24 Similarly, Lee et al analyzed a 546 patients’ cohort from the SEER database, demonstrating that there were no differences in prognosis among stage T1-3, and a 5 cm tumor size cutoff point may be predictive of outcome. 25 In accordance with a previous study, 26 the present study underlined the negative prognostic factor of tumor size on HN-STS, showing that the dimensional threshold of T categories employed by the AJCC eighth edition contributes little to the prognostic model. A possible explanation is that the prognostic information of the tumor size is lost when it is trichotomized into a categorical variable instead of a continuous variable.
The tumor depth is prognostic in the staging system of STS of the trunk and extremity based on the presence or absence of investing fascia invasion.11,12 However, the definition of the superficial and deep tumor may be vague and less relevant in the head and neck, where many small and “less-aggressive” tumors will frequently invade the superficial investing fascia. Instead, in the eighth edition, the concept of adjacent anatomical structure invasion is included to define T4. Recently, Giannini et al found a significant decrease in 5 year OS for T4 (48.3%) than other T-stages (T1/T2: both 100%; T3:71.4%). 26 A similar conclusion was drawn from our study. Our results indicated fair discrimination between OS with a significant difference among tumors with or without invading structure and structural invasiveness as a prognostic factor of poor OS. Moreover, given the anatomical and functional characteristics of the head and neck region, the primary tumor location is considered a clinical prognostic impact, the multiple lesion location represents distinct biological behaviors. 27 Several authors have discussed and identified the potential impact of the tumor site.9,20 In this research, 13 anatomical sites were classified into 3 different risk groups, and higher risk locations tended to result in poorer OS.
Due to the intrinsic relation between the anatomical sites and the local invasion, we defined new L classifications incorporating these 2 factors into 4 categories. The L-stage could distinguish well between the adjacent stage, which had a stronger prognostic value. Therefore, based on an analysis of 471 patients with HN-STS, this research was aimed to use the L-stage to take the place of the T-stage, try to perfect the current AJCC system, and propose a novel LGNM staging system without changing the definition of the G, N, or M stage based on the AJCC cancer staging manual. It is worth noting that cases with N1 disease are specifically designated as stage III disease in the proposed LGNM staging system, not as stage IV according to the current staging system of the trunk and extremity.28,29 Consistent with a previous study, 20 our results suggest that HN-STS patients with lymphatic metastases have a better prognosis than those with distant metastases.
Then, the AJCC and LGNM staging systems were compared through prognostic performance and concordance analysis to determine which, if any, is more predictive of patient outcome. Multiple statistical analyses indicated the marked superiorities of the new proposed LGNM staging system. First, the new staging system was proposed based on a large cohort of HN-STS patients presenting with all disease stages. The extended follow-up duration allowed us to generate statistically-robust models predicting mortality. Also, the LGNM has outstanding stratification capacity to predict the survival of patients in each tumor stage and has better prognostic performance, demonstrated by the higher concordance score than the current AJCC staging system. Specifically, patients in LGNM stage II had a statistically-significant better survival than those in stage III. In contrast, the AJCC staging system was indistinguishable between stages II and III. It is also noted that patients are better distributed among the proposed stages and likely reflect the proportions of patients with HN-STS typically seen in practice. Third, tumor size, which could not precisely reflect the true disease burden in the trichotomy format as described above, was abandoned in the LGNM. Indeed, tumor size was accessed according to surgical and histopathological reports, while the preoperative tumor dimension was difficult to measure accurately as the shape of HN-STS is irregular. In contrast, the local invasion and tumor location clinical data were readily available and could even be specified from preoperative imaging, making the system more convenient and practical for clinical applications. This study powerfully supported adding the L-stage into the conventional staging system for a better prognostication approach, leading to more individualized risk-adaptive therapies in HN-STS.
The present study should highlight some limitations. First, given this study’s retrospective nature and long period, some confounding or unknown factors might influence all results, such as medical records with inconsistent or missing, especially in older cases. Second, the study did not consider some well-known prognostic factors (eg, histological subtype and perioperative adjuvant therapy) of the primary tumor that could influence the survival rates. There is a risk that such predictors may confound outcomes within our cohort. Third, the new LGNM staging system was evaluated in comparison with the AJCC seventh staging system and not to the latest AJCC eighth staging system. Prior to the publication of the eighth edition of AJCC guidelines, HN-STS was not recognized as distinct entities and consistently staged alongside STS located in trunk and extremities. 11 However, in 2017, the official guidelines for staging underwent revision to propose a separate category for HN-STS. Despite this update, the rarity and heterogeneity of HN-STS hindered the development of the eighth AJCC prognostic stage groups. 12 Consequently, we were unable to compare the prognostic efficacy and concordance between LNGM and the eighth AJCC staging system in our current study. Last, the new LGNM staging system was proposed using patients with HN-STS from South China. Validation in an external cohort is required to determine whether the results of this single-center study can be generalized to other populations with different ethnic backgrounds. However, due to the limited patients with HN-STS seen at even high-volume cancer centers and the missing fields (such as FNCLCC grade and anatomical tumor location) of the public databases, the LGNM staging system was not validated in an independent cohort. Although our results challenge conventional criteria, we believe they are built on a credible foundation and deem it necessary to confirm our results in a prospective cohort.
Conclusions
In summary, this study demonstrated that the T category in the AJCC eighth edition staging system is needed for further validation. In contrast, the primary tumor location and local invasion were prognostic for the HN-STS and were divided into 4 stages: L1 (low-risk location: scalp, face, supraclavicular, and ear), L2 (intermediate-risk location: neck, paravertebral, pharynx, tonsil, eye, and orbit), L3 (high-risk location: cavity, lip, palate, buccal mucosa, salivary gland, maxilla, and mandible), and L4 (any location with local invasion). The evidence-based LGNM staging system combining the L classification showed good discrimination of survival outcomes and provided a well-stratified prognosis for patients with HN-STS, which might be superior to the AJCC eighth edition staging system.
Supplemental Material
sj-docx-1-ohn-10.1177_19160216251333359 – Supplemental material for Novel Location-Grading-Node-Metastasis Staging System in Patients With Head and Neck Soft Tissue Sarcoma
Supplemental material, sj-docx-1-ohn-10.1177_19160216251333359 for Novel Location-Grading-Node-Metastasis Staging System in Patients With Head and Neck Soft Tissue Sarcoma by Dingfu Du, Shaojun Wu, Zilu Wang, Yuanxiang Guan, Ke Jiang, Bushu Xu and Yao Liang in Journal of Otolaryngology - Head & Neck Surgery
Footnotes
Author Contributions
D.D.: conduct of the study, patient recruitment, statistical analysis, and drafting of the paper. S.W.: conduct of the study, patient recruitment, and statistical analysis. Z.W.: conduct of the study, patient recruitment, and statistical analysis. Y.G.: conduct of the study and patient recruitment. K.J.: analysis and interpretation of patient data. B.X.: conduct of the study, statistical analysis, and drafting of the paper. Y.L.: conduct of the study, patient recruitment, statistical analysis, proofreading of the paper, and final approval. All authors have read and approved the final manuscript.
Consent for Publication
Written informed consent was waived.
Competing Interests
The authors declare that they have no competing interests.
Data Availability
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the Beijing Xisike Clinical Oncology Research Foundation (Y-Roche2019/2-0049); the National Natural Science Foundation of China (no. 82102776). The funding source had no role in the study design, data curation, or the analysis and interpretation of data.
Ethical Approval
The study protocol was approved by the Ethical Committee and Institutional Review Board of the Sun Yat-sen University Cancer Center (No. B2021-314-01).
Supplemental Material
Additional supporting information is available in the online version of the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
