Abstract
Background
While ultrasound-guided fine-needle aspiration cell block (FNACB) is a cost-effective, expeditious, and reliable procedure used routinely in the initial evaluation of head and neck masses, it has limited efficacy in diagnosing lymphoproliferative disorders such as non-Hodgkin lymphoma (NHL). Flow cytometry performed on an fine-needle aspiration (FNA) sample [ultrasound-guided fine-needle aspirate flow cytometry or flow cytometry performed on an FNA sample (FNAFC)], has been shown to be a valuable adjunct to FNACB in the diagnosis of lymphoproliferative disorders of the spleen, kidney, and thyroid. The objective of this study was to appraise FNAFC’s utility as an ancillary tool to detect NHL arising in the head and neck region in adult patients.
Methods
This is a retrospective study involving 52 adult patients with head and neck lymphadenopathies and masses suspicious for lymphoproliferative disorders, who underwent ultrasound-guided FNACB and ultrasound-guided FNAFC between January 2017 and November 2022. Patient demographics, FNACB histopathological and immunophenotypic results, postoperative histopathology results (when available), and follow-up information until May 2023 were reviewed.
Results
Of the 52 FNACB samples, 23 samples (44.2%) yielded a diagnosis negative for carcinoma, 20 samples (38.5%) were nondiagnostic on account of scant cellularity, 8 samples (15.4%) were suspicious for malignancy, and a single sample (1.9%) was compatible with malignancy. Regarding FNAFC samples, 37 samples (71.2%) were diagnosed as showing no evidence for a lymphoproliferative disorder, 4 samples (7.7%) as nondiagnostic because of insufficient cell count, 4 samples (7.7%) as suspicious for a lymphoproliferative neoplasm, and 7 samples (13.5%) as compatible with a lymphoproliferative neoplasm, most frequently a B-cell lymphoma. 7 of the 11 patients (63.6%) with a suspicious/positive FNAFC result underwent excisional biopsy for additional work up. Postoperative histopathology reports corroborated FNAFC’s findings in 6 patients (85.7%), while the remaining patient’s (14.3%) suspicious FNAFC result was discordant with postoperative histopathology results. The other 4 patients (36.4%) did not require excisional biopsy as the hemato-oncologist deemed the information provided by the FNAFC as sufficient for the diagnosis and treatment of an NHL in the specific clinical contexts of those patients. All patients with nondiagnostic (due to insufficient cell count), inconclusive, or negative FNAFC (ie, nondiagnostic of a lymphoproliferative disorder) were followed up for a mean follow-up period of 11.9 months (range: 61.2 months; SD: 10.2 months), during which no new lymphadenopathies/masses nor progression was observed.
Conclusions
FNAFC is a useful and practical supplementary tool in the diagnosis of lymphoproliferative disorders in the head and neck region, principally B-cell lymphoma. While conventional FNACB offers a valuable insight into the initial work up of head and neck masses, FNAFC can routinely detect small abnormal cell populations. Furthermore, in specific clinical contexts, it can reliably diagnose NHL, thereby averting the need for an excisional biopsy in a subset of patients.

Introduction
Non-Hodgkin lymphoma (NHL) encompasses clinically and histopathologically heterogenous malignancies arising in diverse nodal and extranodal sites in the head and neck and elsewhere. The head and neck region has copious lymphatic tissue, making it the second most conducive anatomical site for the development of lymphoproliferative disorders, chiefly in the cervical lymph nodes, Waldeyer’s ring, ocular adnexa, sinonasal tract, salivary glands, oral cavity, larynx, and less frequently, the thyroid gland and eye.1,2 Lymphoma is the most frequent nonepithelial malignancy arising in the head and neck region, accounting for 5% of all malignancies, with NHL constituting 90% of them.3-5 NHL subtypes are classified according to the World Health Organization (WHO) Classification of Hematopoietic and Lymphoid Neoplasms as precursor B-cell neoplasms, mature B-cell neoplasms, precursor T-cell neoplasms, and mature T-cell and NK-cell neoplasms.2,6,7
Current literature asserts that ultrasound-guided fine-needle aspiration cell block (FNACB) is the customary first-line diagnostic modality for head and neck masses owing to its reliability, swiftness, cost-effectiveness, and minimally-invasive nature, while providing easy sample acquisition for numerous ancillary techniques. 8 Regarding lymphoproliferative neoplasms however, over the past 2 decades, there has been widespread dissent on the efficacy of FNACBs in diagnosing lymphoma; some praise their utility for the diagnosis of common types of lymphoma, while others disparage the procedure on account of potentially-misguiding treatment.9-12 Regardless, the consensus remains that cellblock alone is inadequate in diagnosing NHL due to many limitations including aspiration of scant material, sampling error, and loss of tissue architecture, which hinders accurate sub-classification of lymphoma.9,13
Excisional biopsy of enlarged lymph nodes or extra lymphatic tissue is widely accepted as the gold standard for diagnosing lymphoma, as also recommended by the WHO.1,2,6,14,15 Excisional biopsy may sometimes be carried out under local anesthesia in a clinical setting for patients with superficial lymphadenopathy. However, more frequently, in-patient surgery under general anesthesia is required for deep-seated lesions. 16 Excisional biopsy therefore often harbors operative risks, with a reported surgical complication rate of 5.9%, further compounded by comorbidities, which are increasingly present in an aging patient population. 17 Furthermore, the cost associated with surgery imposes a financial burden on the healthcare system. To offer solutions to such issues, flow cytometry performed on a fine-needle aspiration (FNA) sample (FNAFC) is emerging as an easier, safer, more affordable, and efficient ancillary diagnostic technique that permits the assessment of cellular antigen expression to rapidly provide immunophenotypic information that is critical for the diagnosis and classification of NHL.18-20 Studies of FNAFC of the spleen, kidney, and thyroid lesions have shown promising results and have delineated its diagnostic efficacy, reproducibility, and safety as an adjunct to FNACB in diagnosing certain subtypes of lymphoma.21-23 However, studies evaluating the function of FNAFC as a diagnostic procedure for NHL in the head and neck are scarce. The objective of this study was hence to assess FNAFC’s utility as an ancillary tool to detect NHL arising in the head and neck region.
Materials and Methods
Study Design
This retrospective study is based at the Jewish General Hospital, a quaternary level hospital in Montreal, Canada, between January 2017 and November 2022, with follow-up information until May 2023. It encompasses 52 adult patients on whom FNACB and FNAFC were performed in the evaluation of a head and neck mass. Data on patient demographics, FNACB histopathological and immunophenotypic results, postoperative histopathology results (when available), and follow-up information were reviewed. Ethics approval was granted by the Medical-Bioethics Research Ethics Committee of the Integrated Health and Social Services Network for West-Central Montreal (MP-05-2023-3630).
Patient Population
We reviewed medical charts of adult patients who initially presented with head and neck lymphadenopathies or masses suspicious for lymphoproliferative disorders, and upon consultation underwent ultrasound-guided FNACB and ultrasound-guided FNAFC on these suspicious lesions. The decision to perform a FNAFC was based on the clinical history and imaging findings, when the suspicion for a lymphoproliferative disorder was deemed high enough. Both FNACB and FNAFC procedures were often performed concurrently. In a few instances, a FNAFC was additionally performed after receiving a negative FNACB histopathology result to augment diagnostic specificity by combining these diagnostic modalities sequentially.
All patients with a suspicious/positive FNAFC result were referred to the hemato-oncology service, which either accepted the diagnosis of NHL based on FNAFC results and clinico-radiological findings, or based on a combination of FNAFC, peripheral blood flow cytometry, and cytogenic testing results. Alternatively, hemato-oncology staff requested a subsequent excisional biopsy to supplement FNAFC findings. All patients with an inconclusive or negative (ie, nondiagnostic of a lymphoproliferative disorder) FNAFC result were followed up every 3 to 4 months with physical examinations and serial neck ultrasound assessments, and were observed for any evidence of new head and neck masses or progression of the known lymphadenopathy/mass.
Consent for FNACB and FNAFC was obtained through a general written consent form used at our institution for all surgery, anesthesia, diagnostic, and therapeutic procedures.
Specimen Analysis
An experienced head and neck surgeon performed ultrasound-guided FNAs on the suspicious lymphadenopathy or mass. Samples were sent to the pathology department at the Jewish General Hospital separately for FNACB histopathology and FNA flow cytometry analyses.
The FNACB sample was sent to the laboratory in 10% buffered formalin, spun down in a centrifuge in a conical tube, and integrated into a gel to create a cell block, which was then paraffin-embedded. The cellblock was sectioned by microtome and slides were stained with conventional hematoxylin and eosin (H&E). Additional immunohistochemical studies were applied by the surgical pathologist to selected cases based on the initial cytomorphological evaluation of the cell material.
The FNA sample destined for flow cytometry analysis was preserved in RPMI Medium 1640 and sent to the pathology laboratory within 2 hours of collection. The flow cytometry sample was processed for analysis in a dedicated flow cytology laboratory at the Jewish General Hospital, and data was acquired on a 10-color BD FACS Canto II analyzer equipped with 3 lasers. Cells were manually disaggregated into a cell suspension and washed with phosphate buffered saline. They were processed using the same procedures, cytometer settings, and panels (modified lymphocyte screening tubes 1 and 2). 24 The BD FACS Diva™ software (v6.1.3) was used for data acquisition and analysis (BD Biosciences, San Jose, CA, USA). CD45/SSC gating strategy was used to gate on the lymphocyte populations.
If a patient underwent excisional biopsy, the sample was sent fresh for a lymphoma protocol to be triaged for flow cytometry, conventional H&E preparation ± molecular studies, and a board-certified hematopathologist subsequently analyzed the surgically-resected specimens histologically and immunohistochemically for features of a lymphoproliferative disorder.
Statistical Analysis
Descriptive statistics, including mean, standard deviation, 95% confidence intervals (continuous variables), and percentages (categorical variables), were performed using nonparametric testing of the Mann-Whitney U test. For frequency analysis in contingency tables, statistical analyses of associations between variables were performed using the chi-squared test or Fisher’s exact test (with significance set at p < .05). Statistical analyses were performed using STATA® (STATA Corp, College Station, TX, USA).
Results
Baseline Characteristics
In our study of 52 patients with head and neck lymphadenopathies and masses suspicious for lymphoproliferative disorders, the mean age was 54 years (±18.06); 32 (61.5%) patients were female, and 20 (38.5%) patients were male. Of the 52 masses, 4 (7.7%) were thyroid masses, 4 (7.7%) were parotid masses, and 44 (83.8%) were cervical lymphadenopathies.
FNACB Histopathological Analysis
Of the total 52 FNA samples, FNACB diagnosed 23 (44.2%) as negative for carcinoma, 20 (38.5%) as nondiagnostic on account of scant cellularity, 8 (15.4%) as suspicious for malignancy, and 1 (1.9%) as compatible with malignancy, specifically anaplastic thyroid carcinoma. Additional immunohistochemical studies on the cellblock further classified 2 of the 8 (25%) samples suspicious for malignancy as suspicious for B-cell lymphoproliferative neoplasms, as illustrated in Figure 1.

Histopathological diagnoses for head and neck lymphadenopathies and masses suspicious for NHL by ultrasound-guided FNA cellblock.
FNAFC Immunophenotypic Analysis
As for FNAFC, 37 (71.2%) samples were diagnosed as nondiagnostic for a lymphoproliferative disorder, 4 (7.7%) as nondiagnostic because of insufficient cell count, 4 (7.7%) as suspicious for lymphoproliferative neoplasm, and 7 (13.5%) as compatible with a lymphoproliferative neoplasm (in the appropriate clinic-opathological context). Of the samples suspicious for a lymphoproliferative neoplasm, 3 (75%) were suspicious for the presence of an abnormal B-cell population and 1 (25%) was suspicious for the presence of an abnormal T-cell population. All 7 samples with a positive FNAFC result revealed a B-cell lymphoproliferative neoplasm (Figure 2).

Immunophenotypic diagnoses for head and neck lymphadenopathies and masses suspicious for NHL by ultrasound-guided FNAFC.
Postoperative Histopathological Analysis and Final Diagnoses
Of 11 patients with a suspicious/positive FNAFC, excisional biopsy was performed in 7 (63.6%) patients. Among these 7 patients, 6 (85.7%) received results that were compatible with results obtained by FNAFC, yielding a diagnosis of 1 diffuse large B-cell lymphoma (DLBCL), 1 B-cell lymphoma (not otherwise specified), 2 follicular lymphomas, 1 marginal zone lymphoma, and 1 CD30+ T-cell lymphoma. The other mass that was suggestive of involvement by a monotypic B-cell population by FNAFC was in fact reactive lymphoid hyperplasia (RLH) on the final histopathology report of the subsequent excisional tissue biopsy.
2 (5.4%) patients with a negative FNAFC result (1 with a negative FNAFB result and the other with a nondiagnostic FNAFB result) underwent an excisional biopsy due to persistent lymphadenopathies and a high clinical suspicion for lymphoproliferative disorders. The histopathology reports corroborated the initial negative diagnosis suggested by FNAFC, confirming a diagnosis of RLH in both cases.
Diagnoses by each diagnostic procedure for the 9 patients with subsequent excisional biopsy are recapitulated in Table 1.
Preoperative Diagnoses According to FNACB and FNAFC for Patients That Underwent Excisional Biopsy.
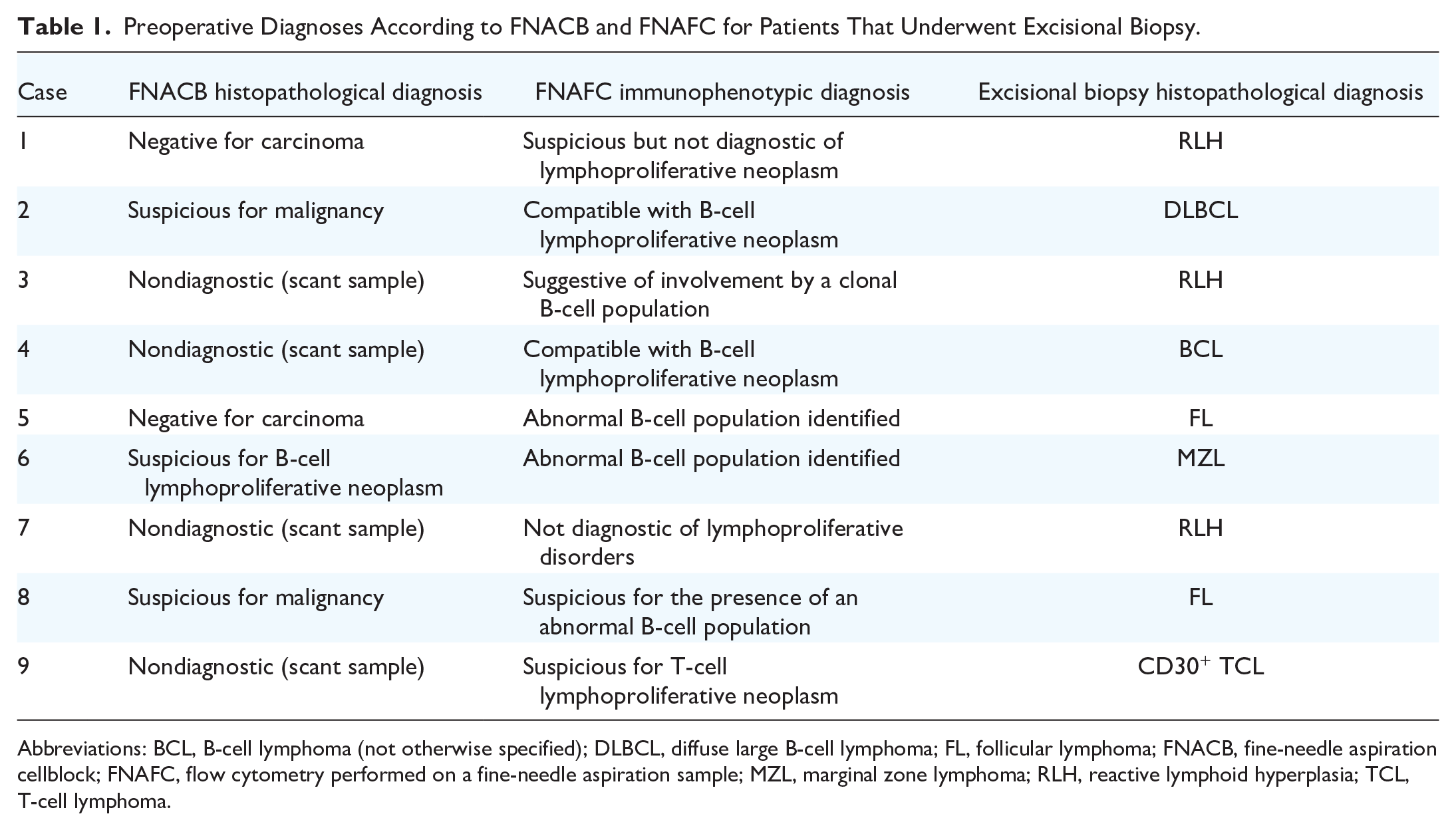
Abbreviations: BCL, B-cell lymphoma (not otherwise specified); DLBCL, diffuse large B-cell lymphoma; FL, follicular lymphoma; FNACB, fine-needle aspiration cellblock; FNAFC, flow cytometry performed on a fine-needle aspiration sample; MZL, marginal zone lymphoma; RLH, reactive lymphoid hyperplasia; TCL, T-cell lymphoma.
Of the 4 (36.4%) patients with a suspicious/positive FNAFC result who did not undergo excisional biopsy, the hemato-oncologist deemed the information provided by FNAFC as sufficient for the diagnosis of B-cell lymphoma and monoclonal B-cell lymphocytosis in 2 patients because of their history of B-cell lymphoma and mild lymphocytosis, respectively. Both patients had a positive FNAFC result: 1 compatible with CD5+ B-cell lymphoproliferative neoplasm and the other compatible with kappa clonal B-cell lymphoproliferative neoplasm. The former patient initially received a FNACB histopathological diagnosis of a salivary gland using Milan System for Reporting Salivary Gland Cytopathology Category III, which can encompass indefinite lymphoid lesions, especially in a suspected parotid gland lymph node. The other 2 patients had no history of lymphoproliferative disorders, and hence, the final diagnosis of chronic lymphocytic leukemia (CLL) for both patients was predicated on the FNAFC report in adjunct with a positive peripheral blood flow cytometry result and a positive cytogenic testing result.
All patients with an inconclusive or negative FNAFC result were followed up every 3 to 4 months with a mean follow-up period of 11.9 months (range: 61.2 months; SD: 10.2 months), during which no new lymphadenopathies, masses nor progression was observed.
Sensitivity and Specificity
FNACB and FNAFC were diagnostically concordant for 24 (46.2%) patients. In 2 patients with rapidly-progressive thyroid masses had suspicious/positive FNACB results (Bethesda Category III and Bethesda Category VI—anaplastic thyroid carcinoma), the FNAFC correctly did not detect any lymphoproliferative disorder. The combined diagnostic modalities ruled out the possibility of a lymphoproliferative disorder, indicating instead the possibility of an epithelial neoplasm. FNACB was not sufficient to establish a diagnosis due to scant cellularity in 4 (7.7%) cases, classified 2 (3.8%) patients as suspicious for malignancy, and 1 (1.9%) patient as negative for carcinoma, as compared to final diagnoses by histopathological analysis of an excisional biopsy or by clinico-laboratory correlation by hemato-oncologists. FNAFC accurately diagnosed and subclassified 8 of the 9 (89.9%) patients who underwent excisional biopsy: 2 as negative and 6 as suspicious/positive for a lymphoproliferative disorder. However, it also classified 1 (11.1%) patient as suspicious for the presence of an abnormal B-cell population, which was not corroborated on the final excisional biopsy histopathology. Our results reported a sensitivity and specificity of FNAFC in detecting NHL of 100% and 67%, respectively, in lymphadenopathies and masses with postoperative histopathological follow-up. It demonstrated a diagnostic accuracy of 90.9% for all patients with a suspicious/positive FNAFC result. Levels of diagnostic sensitivity and specificity of the combination of FNACB and FNAFC were 90% and 75%, respectively, as shown in Table 2.
Accuracy of the Studied Diagnostic Procedures.

Abbreviations: FNACB, fine-needle aspiration cellblock; FNAFC, flow cytometry performed on a fine-needle aspiration sample; NPV, negative predictive value; PPV, positive predictive value.
Discussion
NLH, especially B-cell lymphoma, is becoming increasingly diagnosed because of an aging population and may manifest as prominent painless cervical lymphadenopathies. Due to its rising prevalence, sampling of these suspicious masses with minimally-invasive modalities such as ultrasound-guided FNAs as opposed to diagnostic open surgical procedures is imperative. 25 Although excisional biopsy is the gold standard to obtain a large enough tissue sample for a conclusive diagnosis, there are invariably some risks associated with anesthesia and surgical intervention, including bleeding, hematoma, surgical site infection, and nerve damage. 26 Furthermore, surgical wait times delay the detection and treatment of disease. Excisional biopsy also entails an increased healthcare resource utilization and costs, while an FNA-based procedure can be swiftly performed in a clinical setting, making it more practical and cost-effective while not increasing the morbidity risk. 27
The proficiency of head and neck surgeons and their trainees in the management and timely delivery of FNA samples to the flow cytology laboratory is paramount. An integral component of this process is the education on the immediate use of RPMI Medium 1640 for specimen preservation and the crucial nature of communication with the technicians. While early submission of samples is advocated to ensure the viability of antigens and subsequent analysis, constraints related to laboratory workload and the metabolic demands of different lymphomas dictate the urgency and handling protocols. Particularly for high-grade lymphomas, immediate processing is vital due to the rapid metabolism of malignant cells, which can exhaust nutrients in RPMI and compromise cell viability. Conversely, low-grade or reactive processes may afford a wider timeframe for analysis without a significant loss of integrity. At our institution, we employ a technique of upfront formalin-fixed cell block preparation for FNA samples, which differs from the traditional cytopathology approach used in many institutions. Typically, conventional cytopathology involves the use of an alcohol-based fixative to prepare smears or thin-layer slides, which allows for immediate cytological evaluation. In contrast, our method focuses on the formalin fixation of cell blocks from the outset, which provides the advantage of preserving some of the cellular microarchitecture and enabling additional histological and immunohistochemical analyses. These additional analyses performed on the FNACB complement results obtained by the FNAFC to increase the diagnostic yield for this combined procedure.
The results of our study demonstrated FNAFC’s increased ability to routinely detect abnormal lymphoid populations in comparison with FNACB histopathology alone. FNACB did not conclusively diagnose 38.5% of samples because of scant cellularity, while FNAFC was unable to do so in only 7.7% of samples. In Hehn et al’s study evaluating FNAs utility in diagnoses of lymphoproliferative disorders from a clinician’s perspective, 88% of patients’ cytopathological diagnoses were clinically inadequate to determine patient treatment. 9 Such diagnoses included nondiagnostic, atypical/abnormal lymph node material, suggestive of lymphoma, or malignancy. Another 21% of patients received a broad, equivocal diagnosis of lymphoma, precluding adequate subclassification. 9 Of the 8 samples in our study suspicious for malignancy FNACB, only 2 were able to be further characterized as suspicious for a more specific subtype of B-cell lymphoproliferative neoplasm. The other 6 samples could not provide enough diagnostic information for a possible treatment plan for the patient. With FNAFC, samples suspicious or compatible with a lymphoproliferative neoplasm were further classified by their subtype, permitting clinicians to triage patients with indolent lymphomas from those with aggressive ones, especially to guide the timing of a subsequent excisional biopsy, if deemed necessary. FNAFC’s high sensitivity is due to its ability to detect small monotypic lymphoid cell population on a background of otherwise reactive lymphocytes. It allows a focused analysis of cell subpopulations as determined based on initial flow cytometry results. Flow cytometry has an additional advantage over immunohistochemistry (performed on FNACBs) as it allows an assessment of multiple lymphoid cell membrane antigens simultaneously, highlighting when ones are co-expressed. Receiving a conclusive diagnosis on the first sample eliminates the need for a repeat FNA biopsy and delays in treatment. While FNAFC provides a powerful adjunct to FNACB in the detection and characterization of lymphoproliferative disorders, it is essential to recognize its limitations. Specifically, its value is contingent upon the hematopoietic nature of the lesion being evaluated.
Furthermore, FNAFC can accurately diagnose and subclassify lymphoproliferative disorders sampled in the head and neck region with a diagnostic accuracy of 90.9%, concordant with previously-published studies.28,29 A study by Savage et al reported an 88.4% independent diagnostic accuracy rate of flow cytometry obtained from 451 fine-needle aspirates. 28 FNAFC results corresponded to those by postoperative histopathology in 100% of lesions that were compatible with lymphoproliferative neoplasm, and in 66.7% of lesions that were suspicious for a lymphoproliferative neoplasm. Our results reported a sensitivity (when compared to the gold standard of excisional biopsy) of 100%. Barrena et al revealed a sensitivity of 96% for flow cytometry immunophenotyping diagnostic screening of B-cell NHL, adding to FNACB’s lower sensitivity of 94%. 30 In 1 case, FNAFC suspected a kappa monotypic B-cell lymphoproliferative neoplasm for a lymphadenopathy that was actually RLH on final excisional histopathology. This may be explained by the high detection sensitivity of the flow cytometry method as well as sampling, as the aspirate could have captured a collection of monotypic lymphoid cells of uncertain clinical significance (cell monotypia does not always correlate with cell clonality). Our study reported a specificity of 67%, which is lower than specificities reported in the literature which range from 92.7% to 100%.28,29,31,32 Interestingly, most articles evaluating FNAFC’s diagnostic capability demonstrated higher specificities than sensitivities; however, this trend was not reflected among our study population, and rather, the opposite phenomenon was observed. Senjug et al’s comparative study showed a higher sensitivity (97.7%) than specificity (85.7%) of FNAFC’s ability to diagnose lymphoma. 33 When they assessed FNAFC’s ability to accurately diagnose lymphoma within its subclassification, the sensitivity dropped to 74.4%. Hence, the incongruity between studies concerning sensitivities and specificities may be ascribed to the authors’ sternness regarding lymphoma diagnoses and subclassifications.
In 2 of our patients who underwent excisional biopsy despite a negative FNAFC because of a high clinical suspicion of lymphoma based on clinical history and imaging results, the postoperative histopathology reports reiterated the FNAFC’s findings (ie, only RLH was identified after a full histopathology work up with appropriate immunohistochemical panels). Cannon and Richardson’s (2001) study was the first to appraise the value of flow cytometry analysis on fine-needle lymphoid aspirates from the head and neck. 34 FNAFC performed on 3 of their 20 patients’ suspicious masses showed no evidence of lymphoproliferative disorder and were diagnosed with benign lymphoid proliferations. However, due to persistent clinical worry, they underwent excisional biopsy, and reactive or granulomatous conditions were corroborated in these patients, akin to our study. 34 Our study along with Cannon and Richardson’s suggests that FNAFC on a sufficient sample that does not show any evidence of a lymphoproliferative disorder may avert the need for an excisional biopsy to rule out lymphoma. If an excisional biopsy is performed in such instances, it should be carried out to rule out other malignancies, such as epithelial malignancies, or inflammatory, infectious, or granulomatous conditions.
Cannon and Richardson proposed the use of a combined methodological approach in the assessment of head and neck lymphoid abnormalities as FNAFC reports provided adequate information for a diagnosis of certain lymphomas, obviating excisional biopsy in 46.7% of patients. Upon being referred to hemato-oncology, the management of 36.4% of patients in our study was based on the FNAFC results, avoiding excisional biopsy. 2 patients had a known history of a B-cell lymphoma and mild lymphocytosis, which were further supported by the newest immunophenotypic diagnoses. Congruously, in Cannon and Richardson’s study, 2 patients were given a diagnosis of recurrent lymphoma by FNA cytology and FNAFC analysis after being previously treated for NHL. 34 Research over the last decade suggests that cytomorphology in combination with the study of specific tumor cell antigens (by immunohistochemistry and/or flow cytometry) on a sufficient aspirated sample allow for reliable identification of certain selected B-cell lymphomas such as CLL, low-grade follicular lymphoma, mantle cell lymphoma, lymphoblastic lymphoma, and DLBCL.35,36 Other modalities used by hemato-oncologists at our institution to corroborate FNAFC’s results in provision for a final diagnosis include peripheral blood flow cytometry and cytogenic testing. We must acknowledge the nuanced reality that a subset of lymphomas may not present with sufficient cellularity or may display an atypical immunophenotype that eludes precise characterization through FNAFC alone. In such scenarios, FNAFC should be considered a complementary tool rather than a standalone diagnostic method.
Stacchini et al assessed the ancillary utility of FNAFC in diagnosing primary thyroid lymphomas and reported a sensitivity and specificity of 100% for the collective diagnostic capacity of FNA cytology and FNAFC. 37 Nonetheless, our study showed a combined sensitivity and specificity of 90% and 75% respectively. These discrepant values can be attributed to the high inconclusive rate of FNA cytology compared with FNAFC (20 inconclusive cases vs 4 inconclusive cases), which attenuated the patient pool for sensitivity and specificity analyses. Nevertheless, the combined diagnostic approach has the highest sensitivity, specificity, positive predictive value, and negative predictive value overall rather than each procedure separately.
Our study provides valuable insights into the ancillary utility of FNAFC in diagnosing NHL in the head and neck. However, there are some limitations to this study that should be considered. Primarily, only a subset of the study population underwent excisional biopsy; hence, the true diagnostic capability of FNAFC was only assessed in patients with a histopathology report for a comparison of diagnoses. By extension, as only a relatively-small pool of patients underwent an excisional biopsy, 1 inaccurate flow cytometry diagnosis disproportionately skewed FNAFC’s specificity. Additionally, the range of patients’ follow-up periods for those with a negative FNAFC result was wide. Owing to the variability of follow-up durations, the risk of missed diagnoses of indolent lymphomas, such as CLL and low-grade follicular lymphoma, is unlikely, but possible. From a technical point of view, flow cytometry results of individual patient samples may be hindered by the presence of necrosis (eg, in high-grade lymphomas), as well as a limited time window of reliable cell viability, even when the aspiration sample is transported in RPMI, especially if it is brought to the laboratory over 2 hours after sampling (eg, high metabolic rate of cells in high-grade lymphomas, RPMI medium freshness, etc).
Conclusion
The combination of FNACB and FNAFC is a useful, practical, and low-cost diagnostic modality for the work up of cervical lymphadenopathies and masses suspicious for NHL. FNAFC can identify immunophenotypic aberrancies even in a limited number of aspirated cells. In the appropriate clinical context, it can reliably diagnose and subclassify some types of non-Hodgkin lymphoproliferative disorders (mainly low-grade B-cell lymphomas) with an excellent sensitivity, thereby obviating the need for an excisional biopsy in some patients. Excisional biopsy may still be needed to investigate patients with a FNACB and/or FNAFC that is nondiagnostic of a lymphoproliferative disorder, to rule out other malignancies or inflammatory/infectious/granulomatous conditions. In our study, excisional biopsy was averted in over a third of patients, which further highlights the practicality and cost-effectiveness of the combination of FNACB and FNAFC to investigate patients for lymphoproliferative disease in anatomical sites easily accessible to FNA such as the head and neck.
Footnotes
Acknowledgements
None.
Author Contributions
S.B.: conceptualization, methodology, investigation, data curation, and writing—original draft; L.F., S.D., T.H.: resources, and writing—review and editing; M.P.H., A.M.M., M.A.M., R.J.P., and N.J.: writing—review and editing; S.D.S.: software, formal analysis, and writing—original draft; V.I.F.: conceptualization, supervision, and writing—review and editing. All authors read and approved the final manuscript.
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for Publication
Not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
Ethics approval was granted by the Medical-Bioethics Research Ethics Committee (REC) of the Integrated Health and Social Services Network for West-Central Montreal (MP-05-2023-3630).
