Abstract
Background
Both Mediterranean and ketogenic diets have been proposed as nutritional interventions in Parkinson's disease (PD). Combined approaches may offer maximal benefits.
Objective
Assess the feasibility, safety and exploratory efficacy of two ketogenic interventions, using a Mediterranean diet base, in individuals with PD (PwP).
Methods
In this Phase II, random-order crossover study, PwP followed two 8-week dietary interventions, separated by an 8-week washout: 1) a high-fat, low-carbohydrate Mediterranean diet (MeDi-KD) and 2) a standard Mediterranean diet supplemented with medium chain triglycerides (MeDi-MCT).
Results
Of 52 participants randomized, 48 started the trial. Forty-one (79%) participants completed at least one, whereas only 33 (63%) completed both intervention phases. There were no intervention-related serious adverse events, nor any significant changes in plasma lipid profiles. Seventy-three percent and 92% of participants reported deviating from the MeDi-KD and MeDi-MCT no more than a few times per month, respectively. Moderate Mediterranean Diet Adherence Screener scores of 6.7 (SD: 1.6) and 7.2 (SD: 2.3) were achieved during the MeDi-KD and MeDi-MCT, respectively, out of a maximum of 14. Fifty percent of participants were in nutritional ketosis ([beta-hydroxybutyrate] ([BHB]) > 0.5 mM) at follow-up for the MeDi-KD, as compared with only 1 (3%) participant following the MeDi-MCT. MDS-UPDRS Part II and IV scores decreased by a mean of −1.4 (SD: 4.2; p = 0.039) and −1.0 (SD: 3.0; p = 0.044) points, respectively, following the MeDi-MCT.
Conclusions
While both Mediterranean-ketogenic interventions appear safe in the short-term in PwP, their feasibility is called into question by a high study dropout rate (37%) and modest adherence. Preliminary benefits observed in patient-reported motor experiences were paradoxically limited to the MCT-supplemented MeDi, in which ketosis was not reliably achieved. Together, our findings indicate the need to refine behavioral strategies to optimize dietary awareness and adherence in future trials.
Trial Registration
The trial was registered on ClinicalTrials.gov: NCT05469997.
Plain language summary
Many patients want to know what the best diet is to follow for their Parkinson's disease (PD). Previous studies suggest broad advantages of Mediterranean-style diets (MeDis), which emphasize vegetables, fruits, whole grains, legumes, fish, and olive oil, with smaller amounts of red meat and sweets. New research also hints at potential benefits of “ketogenic” nutritional approaches, which aim to increase availability of fat breakdown products that the brain uses for energy. This can be achieved either through high-fat, low-carbohydrate diets, or by supplementation with medium-chain triglyceride (MCT) oil. Adding either of these elements to a MeDi base may represent an optimal approach.
The aim of this study was to test whether these combined “Mediterranean-ketogenic” diets would be safe and practical for patients with PD to follow. Early indicators of potential clinical benefit were also assessed. Fifty-two participants were randomly assigned to follow two 1) A high-fat, low carbohydrate MeDi (MeDi-KD) and 2) A MeDi administered with MCT oil (MeDi-MCT) for eight weeks each in random order, with an eight-week break in between.
Forty-eight people started the study, 41 completed at least one and 33 completed both diets. No serious side effects were reported. Although most participants felt they followed the diets closely, objective adherence measures suggested room for improvement.
The MeDi-MCT diet led to small but significant improvements in patient-reported motor activities of daily living and motor complications. Longer-term studies are needed to validate these findings. Future trials should incorporate more dietitian-led behavioral coaching techniques to better help participants follow the diets.
Keywords
Introduction
Parkinson's disease (PD) is the second most common neurodegenerative disease,1,2 and fastest growing neurological disorder worldwide.3,4 Current PD treatments primarily target cardinal motor symptoms, with no therapies yet found to slow or halt disease progression, other than potentially exercise.5,6 In this context, nutritional interventions such as the ketogenic diet (KD) have gained increased attention as a potential complementary approach to target multiple disease pathways in tandem, with putative disease-modifying and symptomatic benefits.7,8
Interest in ketogenic interventions for PD originated from preclinical models, which demonstrated that ketone bodies can attenuate neurodegeneration by supplying an alternative fuel source to glucose for bioenergetically compromised nigral neurons. 9 Simultaneously, ketone bodies have been shown to reduce disease-related mitochondrial dysfunction, oxidative stress and neuroinflammation. 9 Supply of ketone bodies to the brain may be increased through adherence to a high-fat, low-carbohydrate KD (70–90% calories from fat, 2–10% from carbohydrates, and 8–20% from protein), 10 or through consumption of ketogenic supplements such as medium-chain triglycerides (MCTs).11,12 Previous short-term pilot studies of ketogenic interventions in PD have reported variable levels of improvement across multiple symptom domains, including voice quality, 13 exercise performance, 14 anxiety, 15 and self-reported non-motor,15–18 motor,17,18 and cognitive 19 symptoms.
While this preliminary evidence is encouraging, several important safety concerns have been raised regarding the long-term implementation of standard ketogenic regimens in persons with PD (PwP). Standard KDs may be associated with poor diet quality and micronutrient deficiencies,20–22 for which PwP are at elevated baseline risk. 23 Standard KDs are often high in saturated fats, which may increase levels of cardiovascular risk biomarkers,24–27 and have been shown to increase the risk of major adverse cardiovascular events over long-term follow-up. 28 KDs may induce fluid loss through increased urination, leading to hypotension and increased risk of falls, which is of particular concern in the elderly population to which many PD patients belong and postural hypotension due to PD. 29 Weight loss, which has been associated with reduced health-related quality of life, 30 and increased mortality in PD, is a common effect of KDs,24,25 and has been consistently observed in five of the six PD ketogenic trials wherein this was reported.15–19 Additionally, KDs have been associated with shifts in gut microbiota composition which may be specifically or especially unfavorable in the setting of PD, 31 wherein gut dysbiosis may play a causative role in disease through interactions with the immune and enteric nervous systems.32–35
Combining KD principles with those of the Mediterranean diet (MeDi) represents one strategy to improve the safety profile of ketogenic regimens,36,37 particularly in the setting of PD wherein MeDis have been repeatedly associated with favorable outcomes.38–46 MeDis encompass a range of plant-based eating patterns, encouraging vegetables, fruits, whole grains, legumes, nuts/seeds and olive oil, while allowing for moderate intake of poultry, fish, wine and dairy, and limiting red and processed meats, sweets, sugar-sweetened beverages, refined grains and saturated fats.47–49 Relative to standard KDs, combined “Mediterranean-ketogenic” diets (MeDi-KDs) permit some increased carbohydrate consumption, primarily in the form of non-starchy vegetables, thereby protecting against KD-related micronutrient and/or fiber insufficiency, and promoting the growth of healthy gut flora. 50 In keeping with the MeDi framework, MeDi-KDs emphasize meeting the KD's high fat requirements using unsaturated sources, such as olive oil and fatty fish, which may minimize cardiovascular risks associated with standard KDs high in saturated fats. 37
Various neurologic, metabolic and gastrointestinal health benefits of MeDi-KDs have been reported in mild cognitive impairment,37,50–53 and Alzheimer's disease. 54 However, only one study to date has focused on adherence to MeDi-KDs in community-dwelling older adults, 37 which is of particular relevance given the substantial adherence challenges associated with intense carbohydrate restriction in trials of standard KDs.21,55–57 As an alternative Mediterranean-ketogenic approach, 58 the comparative safety and acceptability of an MCT-supplemented MeDi (MeDi-MCT) remains to be evaluated. A MeDi-MCT offers the potential to achieve mild nutritional ketosis with a focus on a MeDi-compatible diet composition, without the need for intensive macronutrient tracking nor carbohydrate restriction. 12
In this “Mediterranean-Ketogenic Interventions in Parkinson's Disease” (KIM) Trial, we describe the safety and feasibility outcomes from a phase II, random-order crossover trial of an 8-week MeDi-KD and 8-week MeDi-MCT in PwP, separated by an 8-week washout.
We assess feasibility in terms of recruitment capability, retention and dietary adherence, and report on safety indicators including adverse events, changes in fasting lipid profiles, and weight loss. Adherence was assessed using a multimodal combination of self-report, validated questionnaire-based assessment of MeDi adherence, 48 and gold standard objective biomarker assessment of KD adherence in the form of blood beta-hydroxybutyrate concentration ([BHB]). 59 Changes in scores on the Movement Disorder Society-Unified Parkinson's Disease Rating Scale (MDS-UPDRS) Parts I-IV, 60 Montreal Cognitive Assessment (MoCA), 61 and Parkinson's Disease Cognitive Rating Scale (PD-CRS), 62 were examined on an exploratory basis.
Methods
Participants and recruitment
The study protocol was approved by the University of British Columbia Clinical Research Ethics Board (Protocol #: H21-03747) and was registered on clinicaltrials.gov (
Functionally independent individuals aged 40–85 years, with mild-to-moderate PD without dementia (MoCA ≥21), who were on stable or no dopaminergic medication for ≥1 month, met the study's principal inclusion criteria (Supplemental File 1; Supplemental Table 1). Participants on warfarin were excluded due to potential adverse effects of diet-induced changes in vitamin K on warfarin efficacy, 63 and those on insulin were excluded due to the risk of hypoglycemia. 64 Individuals with inflammatory bowel disease and/or a recent history of probiotic, antibiotic or immunomodulatory medication usage were excluded due to potential interference with gut microbiome-related readouts, 65 which will be reported in a separate publication.
Prospective participants were contacted via phone. Inclusion and exclusion criteria were reviewed during the initial phone call, after which interested participants were scheduled for consenting and first study visits. Final eligibility checks were conducted in person at the first study visit.
Randomization
Eligible participants were randomized to start on either the MeDi-KD or MeDi-MCT intervention using variable block sizes with a 1:1 ratio.
Dietary intervention
At the first study visit, personalized nutritional guidance was provided to participants during a 1-h consultation with the study's registered dietitian (YNW). Baseline dietary intakes were assessed using the Canadian Diet History Questionnaire II. 66 All participants were provided with online access to recipes via the KetoDietCalculator website (https://www.ketodietcalculator.org/about/). Participants were initially assigned to one of four meal templates (1500 kcal/day, 1750kcal/day, 2000kcal/day, or 2250 kcal/day) based on anthropometry-based estimations of maintenance caloric requirements using the Harris-Benedict equation 67 ; however, these caloric levels were used only to guide meal plan design. Participants were not restricted to a fixed energy intake and were free to consume meals and snacks from the recipe guides ad libitum. If participants struggled to maintain their desired body weight for successive weeks, they were either reassigned to a more appropriate plan, or advised by the dietitian to add or remove one or more 200 kcal snacks from the original meal plan to which they were assigned.
Participants were provided gram scales to measure out ingredients. Coconut-derived MCT oil (Nutiva MCT oil (Nutiva Inc.); NPN: 80086912) was supplied to participants starting the MeDi-MCT intervention. All participants were shown how to use the Abbott Freestyle Precision Neo device for at-home weekly blood ketone self-monitoring. Participants were instructed to test their blood ketone levels 2 h after their evening MCT dose (MeDi-MCT) or evening meal (MeDi-KD). Participants were supplied with study journals in which they recorded bowel movements, food intake on pseudo-random days once weekly, daily MCT oil intake (MeDi-MCT arm only), and any adverse side effects. Food records were used exclusively for dietary coaching purposes and not for data collection, as participants were ultimately free to “randomly” select which day of each week to record as representative. Participants completed weekly 30-min check-in phone calls with the dietitian throughout each intervention phase, wherein study journal entries were reviewed and individualized modifications made as needed. Care partners were invited to join all dietitian intake consultations and subsequent follow-ups.
In both intervention phases, the MeDi component emphasized minimally processed plant-based foods, nuts, seeds, fish, poultry, non-starchy vegetables, and olive oil, while limiting red meat, animal-based culinary fats, sweets, and carbonated beverages. In the MeDi-KD, carbohydrates were restricted to 4–10% of daily calories, with the remaining calories derived from lean proteins (10–20%) and (primarily unsaturated) fats (69–82%). The target ketogenic ratio (the ratio of fats to carbohydrates and protein by weight) was increased from 1:1 to 1.5:1 or 2:1 over the first two intervention weeks, depending on participants’ tolerance to the diet and ketone reading. As the Mediterranean diet does not explicitly provide specific guidance on macronutrient intake, participants in MeDi-MCT phase were asked to have food groups in proportions based on Canada's Food Guide (1/2 of their plate fruit and vegetables, 1/4 protein, and 1/4 whole grain), while within each food group, the food selection is guided by the Mediterranean diet. During the MeDi-MCT phase, all participants started the intervention by taking 1 teaspoon (5 mL) of MCT oil twice daily for the first day to acclimate to the supplement. The dose was then increased to 2 teaspoons twice daily on day 2 and 1 tablespoon (15 mL) twice daily on the subsequent days of the first week. If the supplement was deemed tolerable by the end of the first week, MCT doses were then further increased during the second week until one of the following thresholds was reached: 1) The dose was not tolerable, 2) the limit of the recommended dose on the supplements’ label was reached (i.e., ≤40 mL of MCT oil supplement/day), or 3) previously set target dose was reached (30 ml/day) and ketosis (blood [BHB] > 0.5 mM) was achieved. Participant handouts containing education and instructions for each diet are provided in Supplemental File 2. An overview of the study timeline is provided in Figure 1.
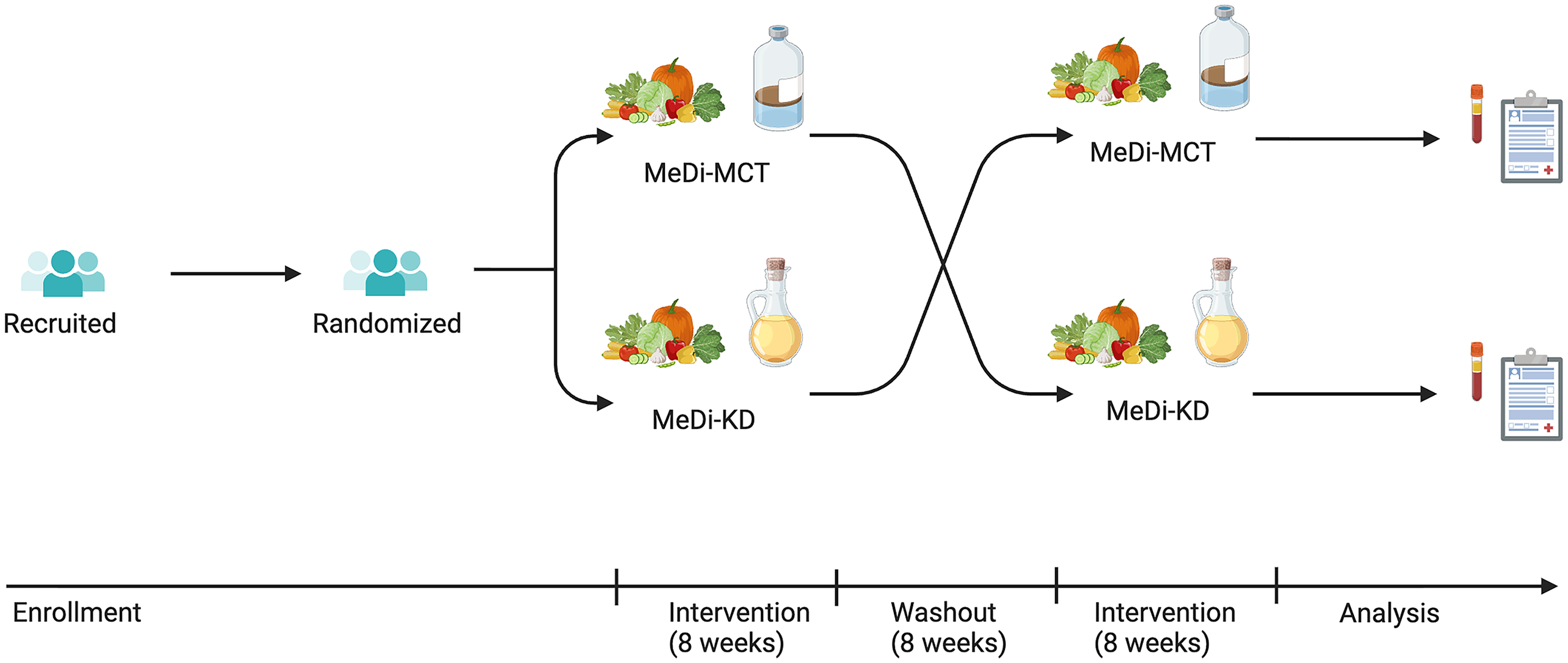
Study timeline. Created in BioRender. Tosefsky, K. (2026) https://BioRender.com/jvpeum4.
Feasibility assessment
We focused on three key domains of feasibility: recruitment capability, retention and dietary adherence.
Recruitment capability was assessed based on whether or not our target enrollment of 50 participants was met within 24 months of the study start date. Retention rates were tracked through each study stage, with a prespecified conservative 75% target completion rate based on previously reported retention rates for 6–12 week KD trials in adults with neurological disease, ranging from 73–86%.17,50,68,69 Reasons for withdrawal were documented in all cases where participants remained contactable. Dietary adherence was tracked using a combination of once-weekly at-home measurements of blood [BHB], pre- and post-intervention fasting plasma [BHB], pre- and post-intervention scores on the 14-item Mediterranean Diet Adherence Screener (MEDAS),
70
and self-assessed adherence according to a 4-point scale administered at post-intervention visits. The MEDAS score awards points for olive oil, fruit, vegetable, legume, fish, nut, and red wine consumption as well as preference for white over red meat, while deducting points for consumption of animal fats, pastries and sugar-sweetened beverages.
70
The 4-point scale asked participants how often they followed the diet, with the following answer options: 1) Followed the diet every single day; 2) Deviated from the diet several times a month; 3) Deviated from the diet more than one time a week; 4) Could barely execute the diet. Elevated blood [BHB] was defined as 0.30 mM in the non-fasting, and 0.50 mM in the fasting state.36,57,71,72
Intervention acceptability was also assessed through semi-structured interviews, which will be analyzed qualitatively and reported in a separate publication.
Safety assessment
Participants were instructed to report adverse events to study team members at the time of occurrence and were asked about adverse effects at each check-in and study visit. All adverse events were reported to the Principal Investigator (SAC) and graded according to severity, persistence and probability of relatedness to diet. Fasting plasma lipid panels (total cholesterol, triglycerides, high-density lipoprotein cholesterol (HDL-C), and low-density lipoprotein cholesterol (LDL-C)) were obtained at pre- and post-intervention visits to monitor cardiovascular risk. Plasma lipid analysis was performed by the UBC Hospital Outpatient Laboratory.
Anthropometric measurements
Weight and height were measured by research staff at the first study visit to calculate BMI, and weight was measured at each follow-up visit using the same calibrated digital scale. We did not rely on participant-measured at-home weights for any of our data collection; however, participants were asked about any noticed or voluntarily measured changes in body weight as a part of routine nutrition care and safety monitoring in weekly dietitian check-in sessions. Participants were discouraged from changing anti-Parkinsonian medications during the trial unless medically necessary and were instructed to inform the study team of any such changes during weekly check-in phone calls and follow-up study visits.
Clinical assessment
The MDS-UPDRS, MoCA and PD-CRS were assessed in the “on” state at each study visit by certified research personnel.60–62
Statistical analysis
All analyses were conducted in RStudio (Version 2024.12.1 + 563). Statistical significance was set a priori at p < 0.05. Differences in baseline characteristics between randomization groups were compared using Wilcoxon rank sum tests, Pearson's Chi-squared tests, Fisher's exact tests and Wilcoxon rank sum exact tests dependent on variable type. Between- and within-intervention comparisons were performed using linear mixed effects models and Wilcoxon signed rank tests, respectively. Mixed effects models were adjusted for period-specific baselines and intervention sequence, with participant IDs included as a random effect. Reported results derive from available-case analyses. Linear associations between continuous variables were calculated using Pearson correlation. Relationships between ordinal variables were described using Spearman rank correlation. Statistical approaches were reviewed by a staff statistician.
Results
Recruitment
Sixty-eight participants were recruited to the study between April 2023 and April 2024, with the final participant completing the study in October 2024. Figure 2 presents the flow of study participants according to the CONSORT guidelines. Fifty-two participants were randomized to start either the MeDi-KD or the MeDi-MCT intervention. Four of these participants withdrew before starting the first dietary intervention to which they were randomized, leaving a total of 48 participants having started one of the two diets.
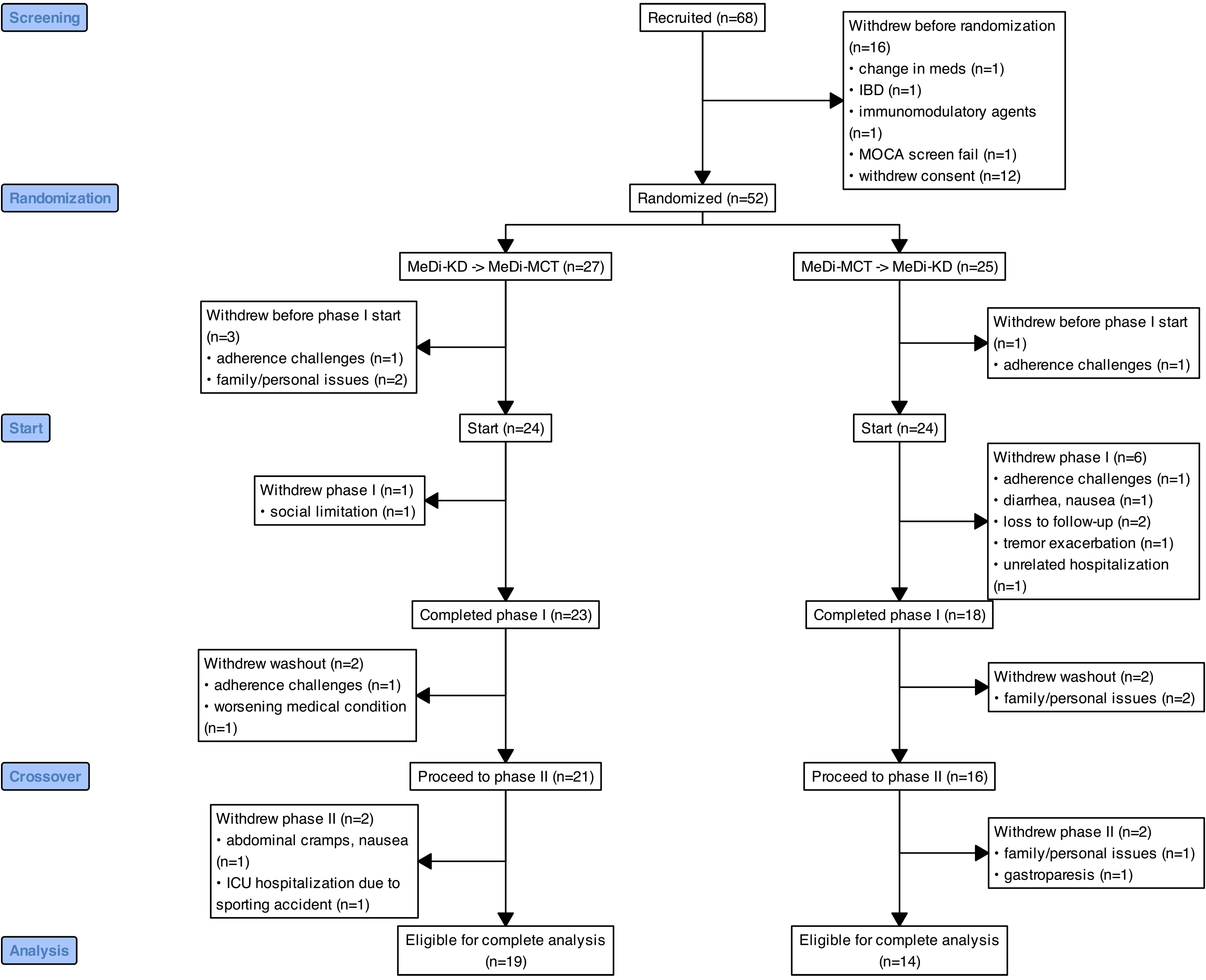
Consort diagram.
Baseline characteristics
The median age of randomized participants was 68 (interquartile range [IQR]: 63–74), with a median disease duration of 7 (IQR: 4–10) years. Sixteen (31%) participants were female and 84% were Caucasian (Table 1). Most participants had completed post-secondary education, with a median total of 18 (IQR: 16–20) years of education across the cohort. Eighty percent of participants were retired or not working. Most (76%) participants were married or in a long-term relationship, of whom 67% were living with a partner and 18% were living with children.
Baseline characteristics.
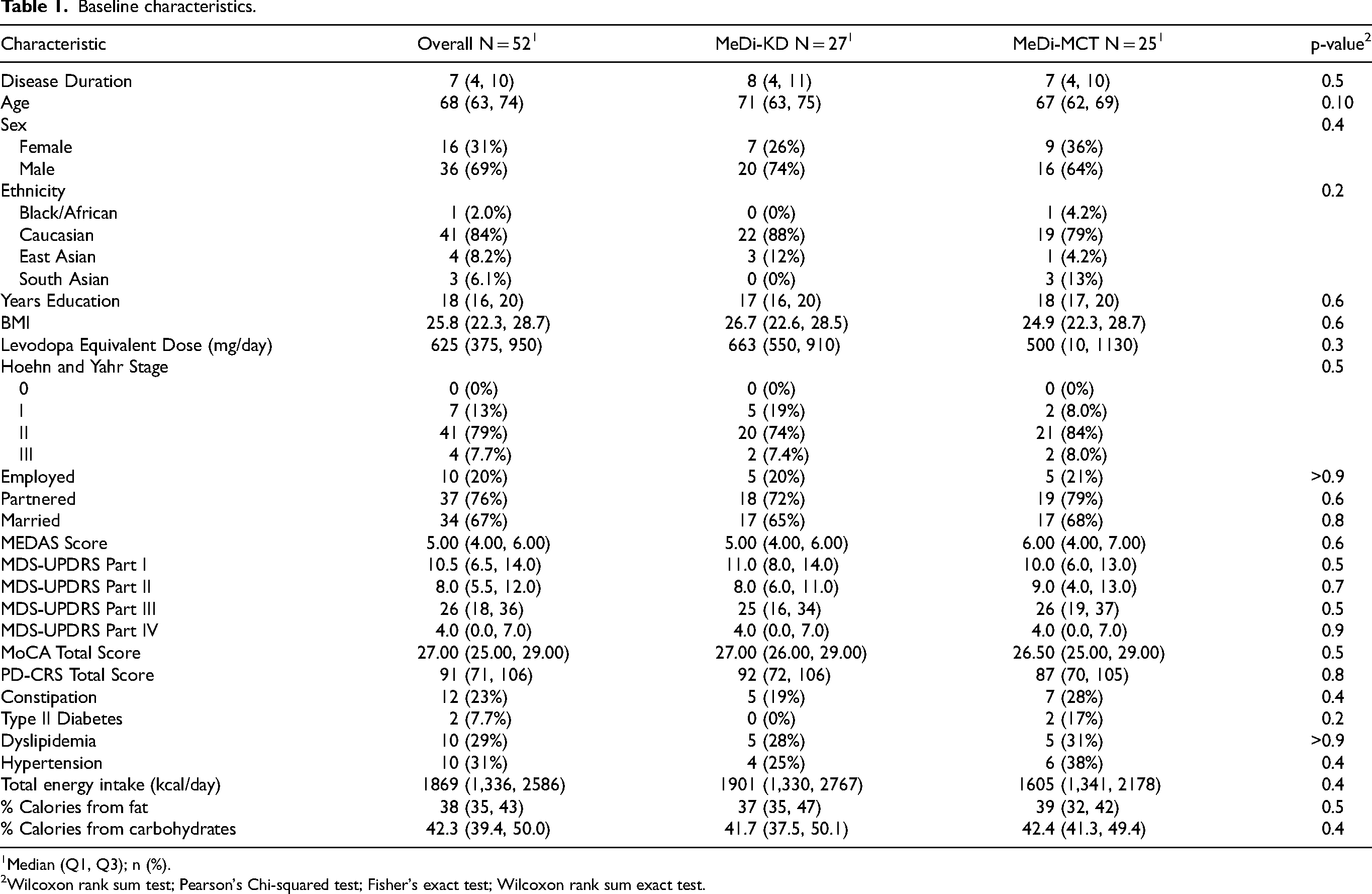
Median (Q1, Q3); n (%).
Wilcoxon rank sum test; Pearson's Chi-squared test; Fisher's exact test; Wilcoxon rank sum exact test.
Forty-one (79%) participants had Hoehn and Yahr Stage 2 disease with a median MDS-UPDRS total score of 46 (IQR: 41–63) at the first study visit. The median levodopa equivalent dose was 625 (IQR: 375–950) mg/day. The median BMI was 25.8 (IQR: 22.3–28.7). Twelve (23%) participants experienced clinically significant constipation prior to starting the trial according to the ROME III module criteria.73,74
No participants were following a ketogenic diet or taking an MCT supplement at enrolment, as per the study's exclusion criteria. At baseline, participant's diets were generally not aligned with a Mediterranean pattern, with a median MEDAS score of 5 (IQR: 4–6) out of a maximum of 14. 48
The two groups (those randomized to start on the MeDi-KD vs. MeDi-MCT intervention in phase I) were well balanced at baseline with respect to pertinent clinical and demographic variables, indicating that randomization was successfully achieved.
Retention
Forty-one (79%) participants completed at least one intervention, and 33 (63%) completed both intervention phases, falling short of our initial retention target of 75%. Seven of 16 (44%) female and 12 of 36 (33%) male participants withdrew (p = 0.5). Study completion status did not differ based on randomization group (p = 0.6). Study completion rate was higher (76%) among those with long-term partners or spouses as compared to participants who were single (33%) (p = 0.027). Reasons for study withdrawal at each study stage are detailed in Figure 2.
Adherence
MEDAS scores
Modest increases in MEDAS scores were seen following both the MeDi-KD (p = 0.005) and MeDi-MCT (p = 0.001) interventions (Figure 3), from mean pre-intervention scores of 5.1 (standard deviation [SD]: 1.4) and 5.5 (SD: 2.0) to mean post-intervention score of 6.7 (SD: 1.6) and 7.2 (SD: 2.3), respectively. No significant carryover effects in MEDAS scores were observed, as might have been expected if participants maintained a Mediterranean-style diet during the washout period. Examining MEDAS scores at each study visit according to previously established categories for weak, moderate and good adherence, 48 we see that the majority of participants (76% on MeDi-KD; 54% on MeDi-MCT) were only able to achieve moderate adherence during either intervention phase, although 8 (23%) achieved good adherence during the MeDi-MCT intervention (Supplemental file 1; Supplemental Table 2).
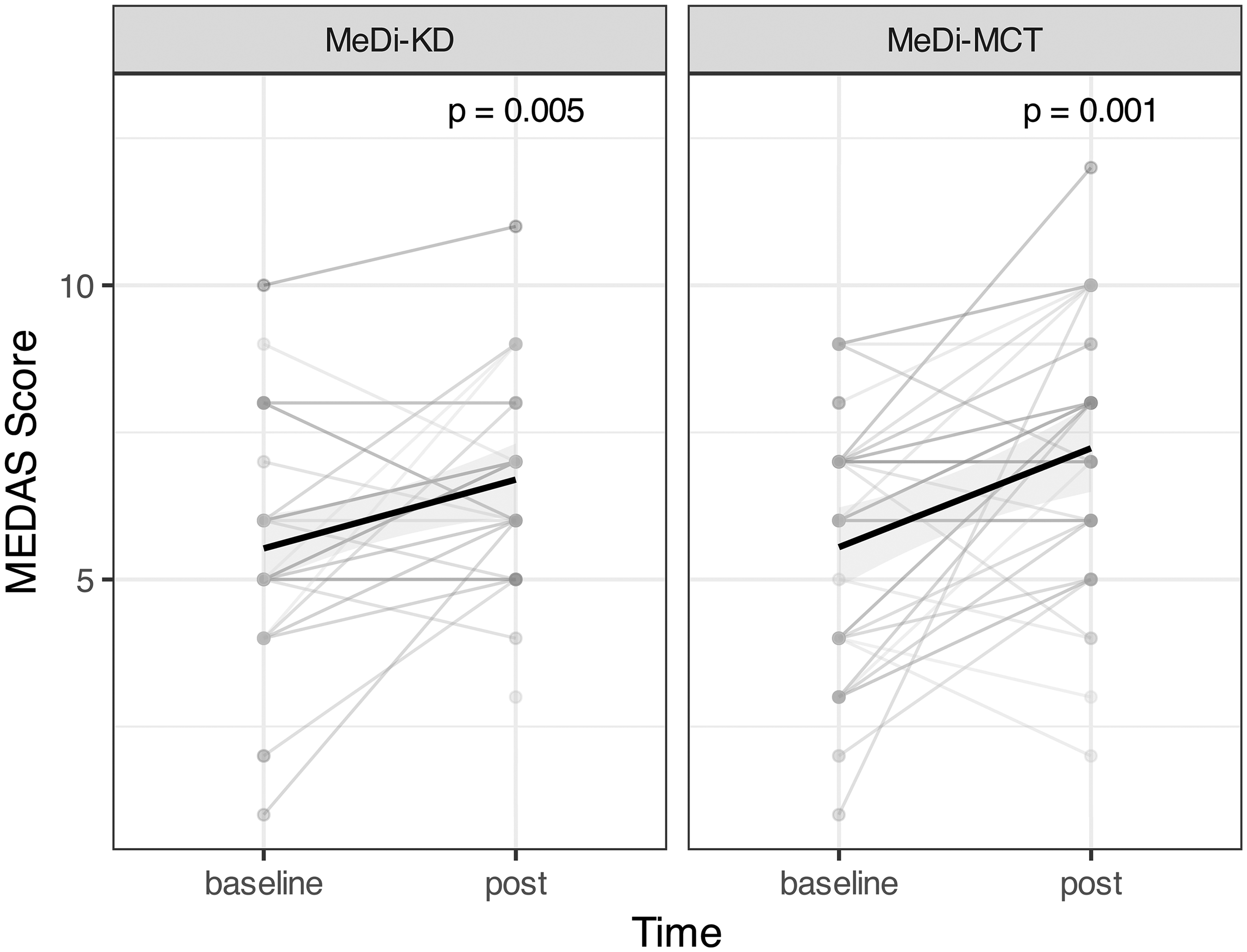
MEDAS scores increase from baseline during each intervention phase. Black lines represent means, gray shading represents the 95% confidence interval, and gray lines represent individual trajectories.
Blood BHB
Participant-monitored non-fasting blood BHB concentrations were consistently higher during the MeDi-KD phase compared with the MeDi-MCT phase throughout all 8 weeks of the intervention (p < 0.001) (Figure 4A). This significant difference held following removal of outliers (p < 0.001), and adjustment for period effects and ketone levels at baseline (p < 0.001) in sensitivity analysis. Average non-fasting blood [BHB] in the MeDi-KD group consistently exceeded the threshold of 0.3 mM each week, with a mean of 0.51 (SD: 0.36) mM across study weeks. In contrast, only week 5 and week 7 average [BHB] readings were above 0.3 mM in the MeDi-MCT group, with an overall mean of 0.19 (SD: 0.11) mM. The proportion of participants with mean at-home ketone levels >0.3 mM was 71.4% in the MeDi-KD group, as compared with 22.9% in the MeDi-MCT group (p < 0.001).
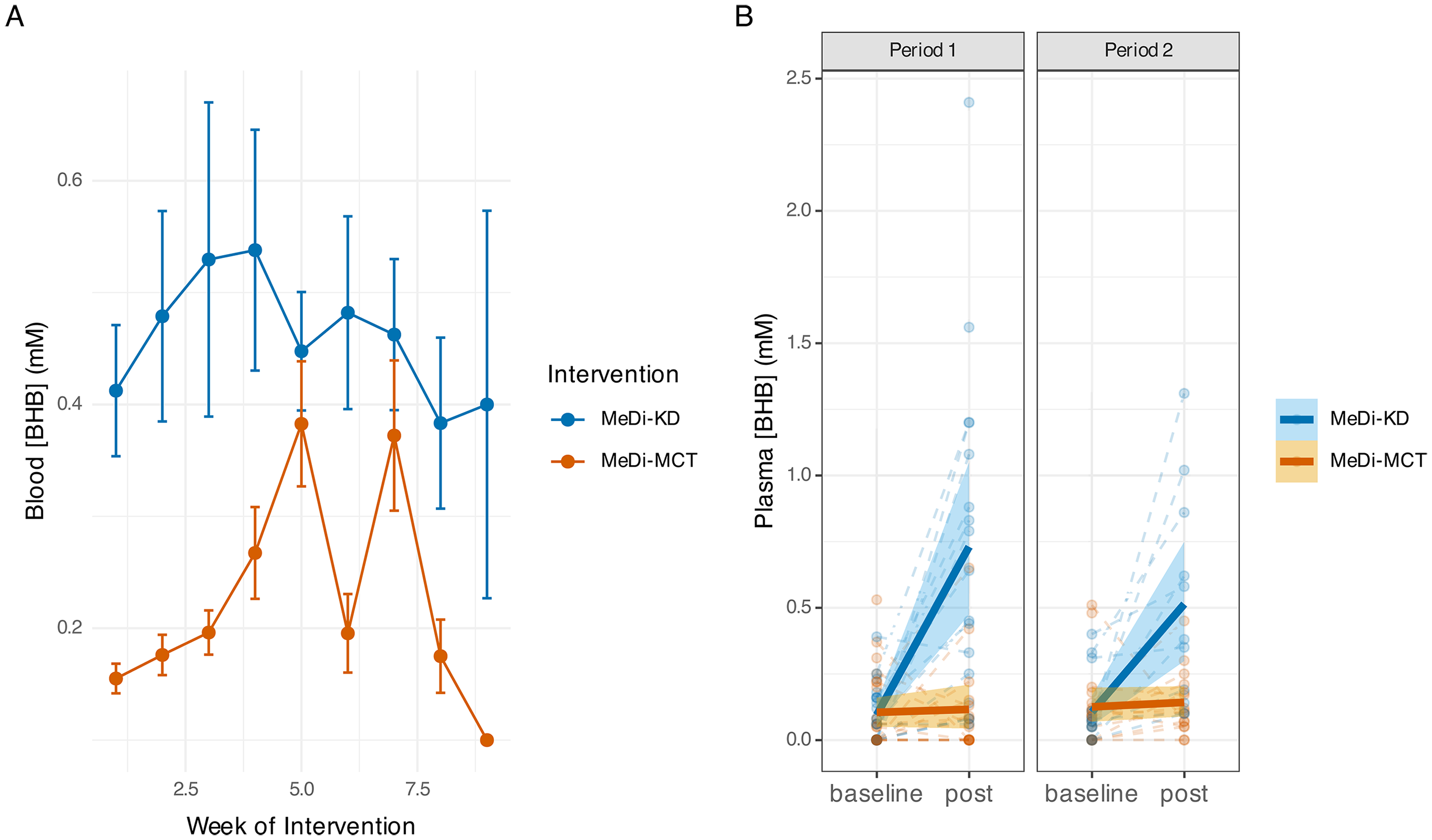
Ketone levels increase relative to baseline following MeDi-KD, but not MeDi-MCT, regimens. A) At-home non-fasting blood [BHB] (mM) over the course of each 8-week intervention. Error bars represent standard error of the mean (SEM). B) Laboratory measurements of fasting plasma [BHB] (mM) at baseline and post-intervention follow-up visits. Solid lines represent means surrounded by 95% confidence intervals.
Moderate significant correlations between changes from baseline in non-fasting at-home and fasting laboratory ketone measurements were observed for the MeDi-KD (r = 0.65; p < 0.001) but not the MeDi-MCT (r = 0.17; p = 0.42) intervention. Fasting plasma [BHB] increased by a mean of 0.45 (SD: 0.41) mM from baseline following the MeDi-KD (p < 0.001) to a mean of 0.65 (SD: 0.55) mM at post-intervention visits, with 50% of participants having post-intervention fasting ketone levels >0.5 mM. In contrast, fasting plasma [BHB] rose by only 0.04 (SD: 0.19) mM (p = 0.18) following the MeDi-MCT (Figure 4B) to a mean of 0.13 (SD: 0.15) mM, with only 1 participant (3%) having a post-intervention fasting ketone level above 0.5 mM. As such, a significant difference was found in the effects of each intervention on fasting plasma [BHB] (p < 0.001).
Self-Rated adherence
Among participants completing both intervention phases, 73% reported following the MeDi-KD and 92% reported following the MeDi-MCT almost every day or skipping at most several times per month (Table 2). Self-rated adherence (p = 0.2) and likelihood of future adherence (p = 0.2) did not differ significantly between MeDi-MCT and MeDi-KD interventions. However, participants took significantly longer to adjust to the MeDi-KD regimen (p = 0.006), and rated their likelihood of adhering to a Mediterranean diet alone significantly higher than their likelihood of adhering to a ketogenic (p < 0.001) or MCT-supplemented (p < 0.001) diet.
Self-Rated adherence, adjustment period and likelihood of future dietary adherence.
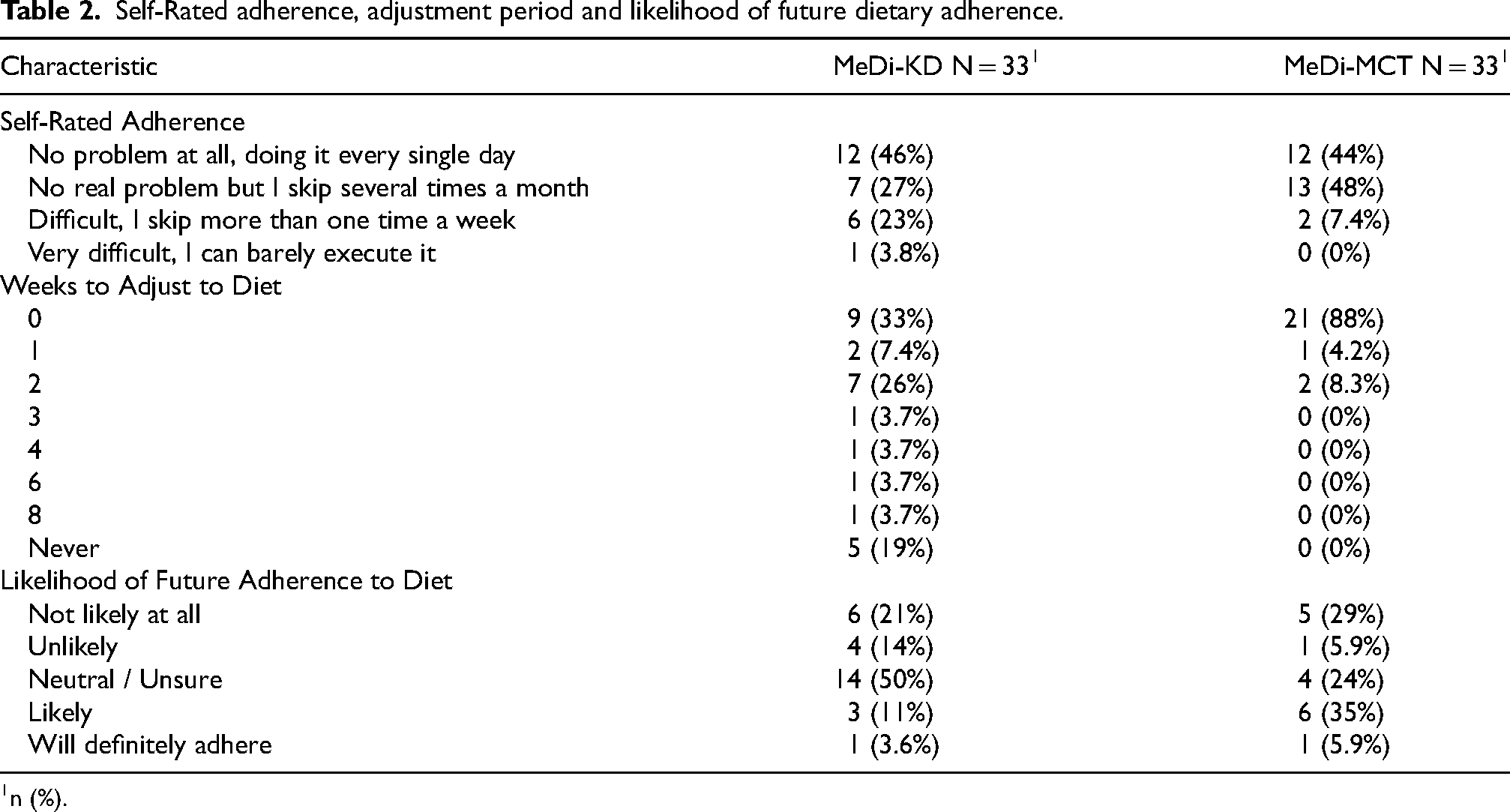
n (%).
When ranked into quartiles according to the proportion of non-trace (>0.3 mM) readings, participants’ at-home blood ketone levels were moderately correlated with self-rated adherence to the MeDi-KD (r = 0.50; p = 0.007), but not the MeDi-MCT (r = -0.09; p = 0.66). No significant correlations were observed between MEDAS scores and self-rated adherence to either diet.
Safety
Adverse events
Eight intervention-related adverse events were reported in the MeDi-KD arm and 10 were reported in the MeDi-MCT arm (Table 3). None were classified as serious adverse events. In the MeDi-KD arm, recurrent episodes of hypotension were reported by two participants, in association with vigorous exercise. Intermittent, mild tremor exacerbations were reported by two additional participants on the MeDi-KD, and one participant on the MeDi-MCT. Indigestion was reported by three participants on the MeDi-MCT and one on the MeDi-KD. Diarrhea was reported by two participants on the MeDi-MCT. Other symptoms reported with plausible causal links to the interventions included worsened fatigue and headaches on the MeDi-MCT, insomnia on the MeDi-KD, and a delayed response to levodopa medication on the MeDi-KD. One participant reported an allergic reaction deemed unrelated to the study diet on the MeDi-KD.
Adverse events.
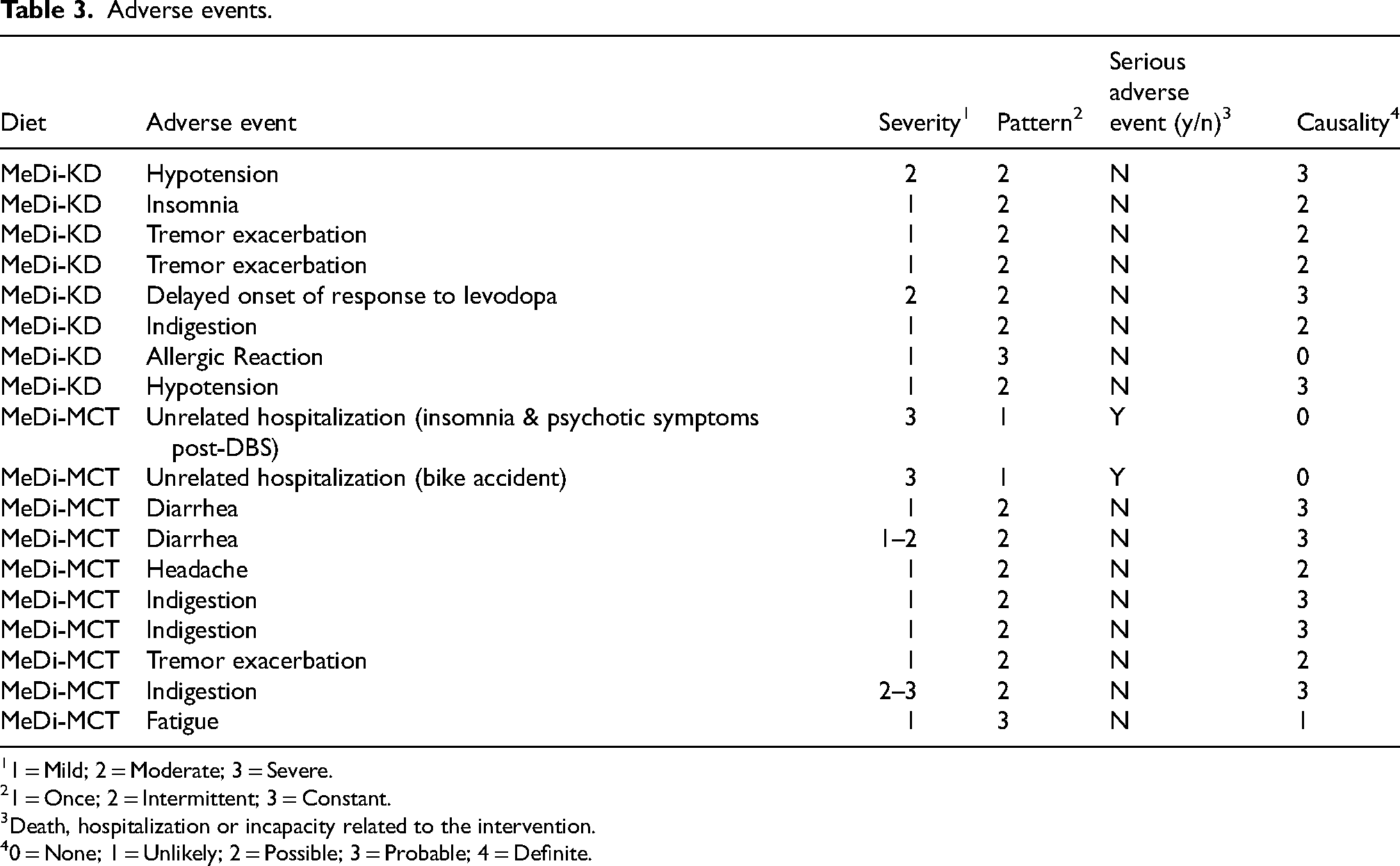
1 = Mild; 2 = Moderate; 3 = Severe.
1 = Once; 2 = Intermittent; 3 = Constant.
Death, hospitalization or incapacity related to the intervention.
0 = None; 1 = Unlikely; 2 = Possible; 3 = Probable; 4 = Definite.
All participants maintained blood ketone levels within or below the range for physiologic ketosis throughout the study, with the exception of one participant who obtained blood ketone readings of 5.1 mM and 3.1 mM on the same day when following the MeDi-KD, after skipping a midday meal.
Fasting lipids
No significant changes from baseline were observed in fasting triglycerides, LDL, HDL or total cholesterol following either intervention (Figure 5A-D).
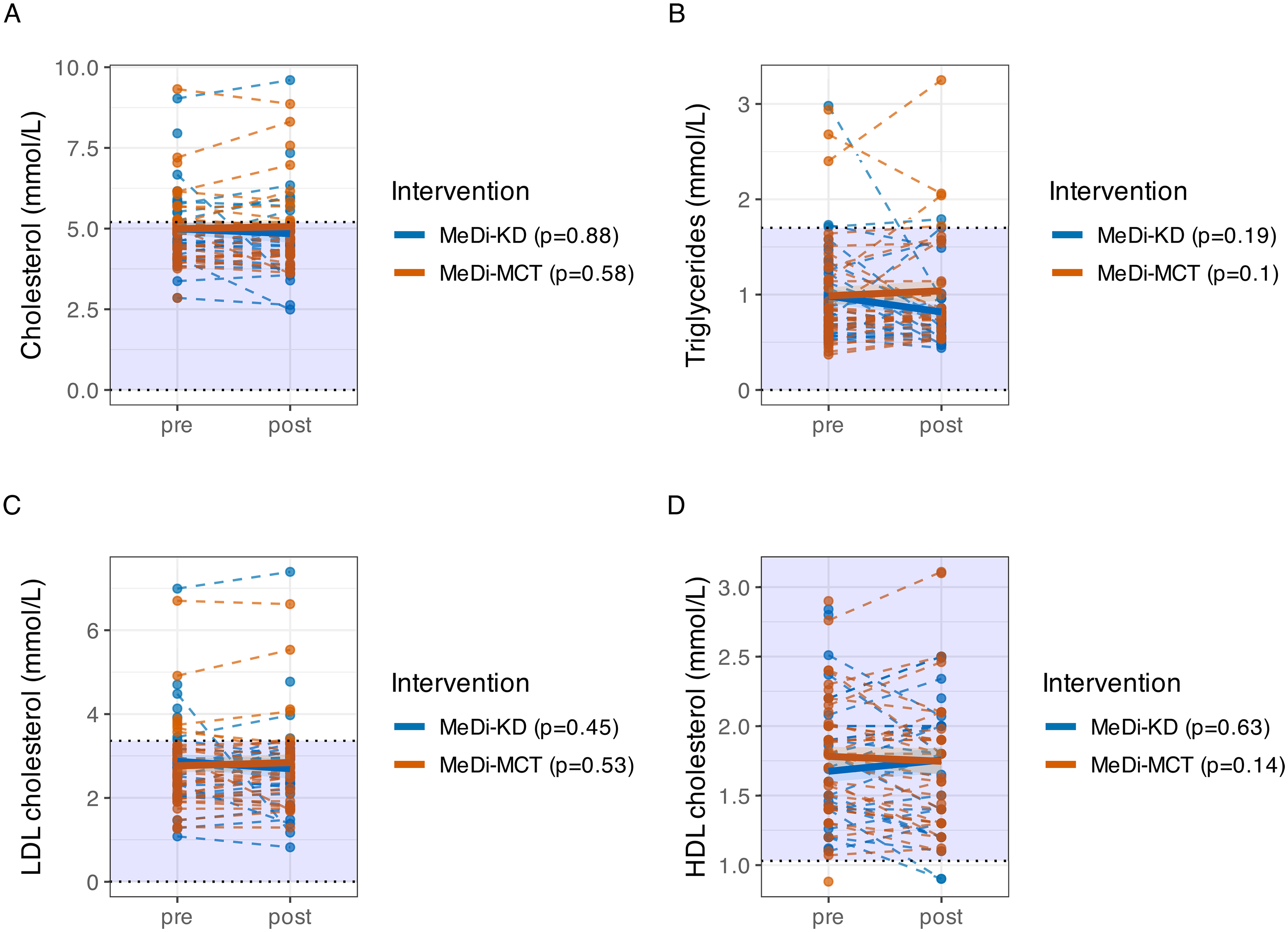
Intervention-associated changes in triglyceride, LDL, HDL and total cholesterol. Purple shading indicates the normal range for each laboratory value. 75 .
Body mass
A significant reduction in body mass of a mean −2.63 (SD: 1.86) kg from baseline was observed following the MeDi-KD phase (p < 0.001), whereas body mass remained stable during the MeDi-MCT phase (mean change from baseline: 0.03 (SD: 1.74) kg; p = 0.61) (Figure 6). In available-case analysis, there was a significant between-intervention difference in weight changes (p < 0.001) after adjusting for period effects. Weight loss did not result in an underweight status (BMI <18.5) for any participants in our study at any of the assessments.
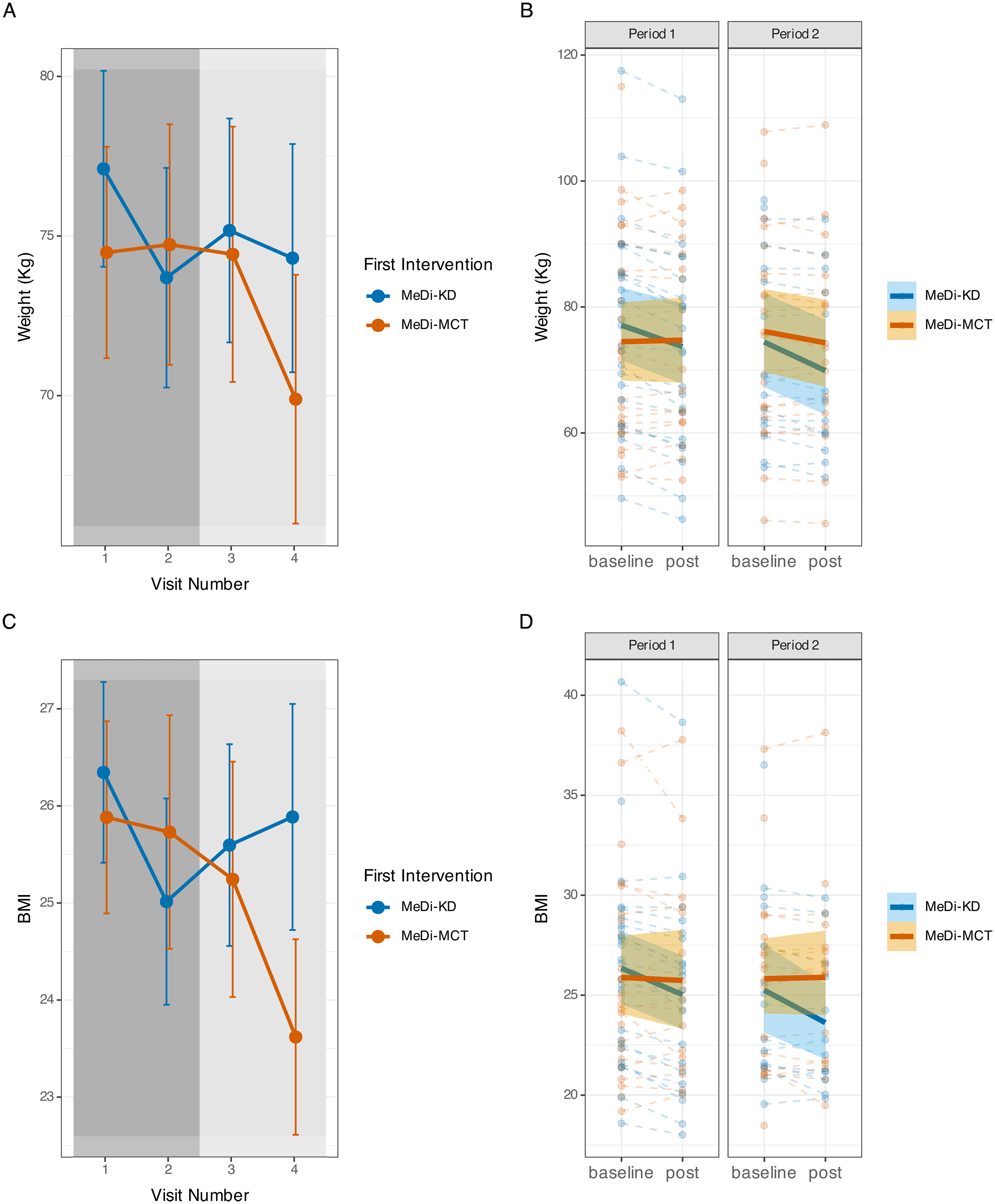
Modest weight loss occurred during the MeDi-KD, but not the MeDi-MCT phase. A and C) Mean body mass (A) and BMI (C) over the course of the four study visits. Error bars represent SEM. B and D) Changes from baseline in body mass (B) and BMI (D) for each intervention and period. Solid lines represent means surrounded by 95% confidence intervals.
Clinical outcomes
A significant mean within-participant decrease in MDS-UPDRS Part II scores of −1.4 (SD: 4.2; p = 0.039) points and Part IV scores of −1.0 (SD: 3.0; p = 0.044) points was observed following the MeDi-MCT intervention (Table 4). Non-significant improvements in MDS-UPDRS Parts II and IV scores were seen following the MeDi-KD intervention. No between-intervention differences were observed in any parts of the MDS-UPDRS nor in MDS-UPDRS total scores. No between- or within-intervention differences were observed in total MoCA scores. A statistically significant, but clinically negligible, 76 decrease of two out of a maximum of 134 points on the PD-CRS (p = 0.024) was observed following the MeDi-KD in available-case analysis, which was not reproduced in the intention-to-treat analysis. No changes to anti-Parkinsonian regimens were reported between assessments.
Changes in MDS-UPDRS, MoCA and PD-CRS scores.
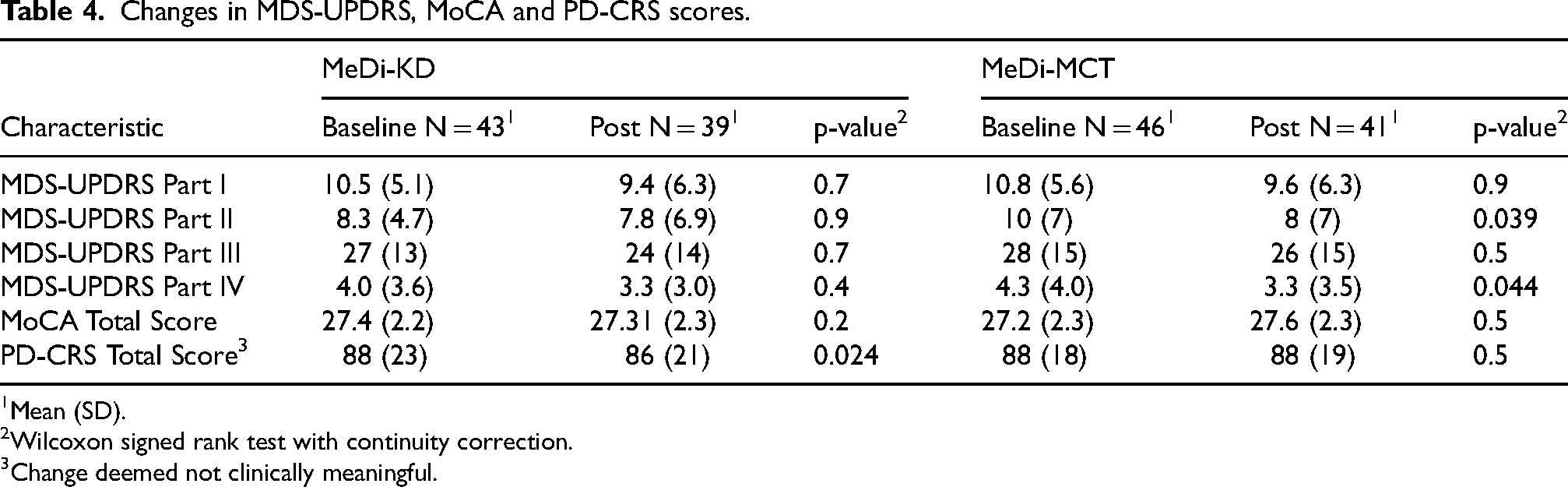
Mean (SD).
Wilcoxon signed rank test with continuity correction.
Change deemed not clinically meaningful.
Discussion
The KIM trial was designed as a response to patient inquiries regarding the utility of ketogenic diets as adjunct management strategies for their PD. This interest presumably stemmed from a growing body of pilot data suggesting variable beneficial effects of ketogenic diets on motor, non-motor, neuropsychiatric and cognitive symptoms of PD,13,15–19 and our research group's previous work on diet in PD. 39 Compelling as is the prospect of a complementary dietary self-management strategy for PD,8,77 none of these studies comprehensively assessed the potential long-term risks and barriers to implementation of ketogenic regimens in the PwP. The overarching goal of the KIM trial was to assess the feasibility and safety of both a dietary and MCT supplement-based ketogenic intervention in PwP, optimized from both an overall dietary quality and adherence perspective through their combination with Mediterranean diet principles.50,78 We demonstrate the feasibility of recruitment for this 6-month crossover study in an urban setting and the low short-term risk of major adverse health effects of these dietary approaches in PwP. However, our study's high dropout rate (37% vs. our target 25%), coupled with discordance between objective and self-rated intervention adherence, point towards key obstacles to the sustained implementation of these dietary changes in a real world PwP population.
The success of our recruitment effort attests to the high levels of interest in lifestyle self-management strategies among PwP seen at our tertiary Movement Disorders Clinic (Vancouver, British Columbia, Canada), as well as to the overall appropriateness of our eligibility criteria, screening and recruitment procedures.79–81 The identification of potentially interested and eligible participants by their neurologists during centre clinic-research hybrid meetings was particularly helpful in prioritizing initial contacts. A notable limitation to our recruitment strategy is reflected in the lack of demographic diversity of our cohort, constituted primarily by Caucasian males with post-secondary education living in proximity to our university campus. Established barriers associated with trial participation in PwP–including language barriers among ethnic minorities, travel distance, financial constraints, work conflicts, and reduced access to caregiver support among women–no doubt impacted our ability to recruit a demographically representative sample. 82 The apparent lack of cultural adaptability of both the Mediterranean, 83 and ketogenic diets, 84 employed in this study may have further contributed to the overrepresentation of those accustomed to a Western-style eating pattern in our cohort.
Similar participant and intervention-specific factors may have contributed to our sub-target retention rate. Compliance to standard ketogenic diet regimens is notoriously challenging. Even in the case of intractable epilepsy, which represents the strongest current indication for the ketogenic diet in adults, retention rates at 6 months in intervention studies are reported as low as 38–46%.68,85–87 In one study, over one-third of patients with no alternative treatment options for refractory epilepsy nevertheless refused to participate in a ketogenic diet intervention due to its perceived restrictiveness and complexity. 87 In PD, reported completion rates for 8–12 week ketogenic diet trials ranges from 70–88%, with ours being the first ketogenic intervention study longer than 3 months undertaken in this population.
While some evidence has indicated higher retention rates to Mediterranean adaptations of the ketogenic diet in adults with overweight, obesity and MCI,50,88,89 our retention data indicates unresolved challenges to its adoption by PwP. Particularly important to consider in PwP are issues related to the effects of high fat intakes on gastric motility and dopaminergic medication absorption, as was illustrated by one withdrawal from the MeDi-KD arm attributed to a delay in levodopa onset of action. Gastrointestinal disturbances may help to account for our lack of observed advantage of the MeDi-MCT over the MeDi-KD in minimizing dropouts, whereas retention rates as high as 95% have been reported for long-term MCT supplementation trials in older adults with probable Alzheimer's disease. 90 Specifically, the high rates of baseline gastrointestinal dysfunction in PD may make a subset of PwP particularly susceptible to more severe adverse gastrointestinal side effects of MCT oil.90,91 In our study, two out of a total of five such adverse events were sufficiently severe to prompt withdrawal from the MeDi-MCT arm.
Apart from gastrointestinal side effects, other adverse events deemed possibly or probably related to the study diets included hypotension (MeDi-KD) and tremor exacerbations (both MeDi-KD and MeDi-MCT). Hypotension is a known potential adverse effect related to KD-induced diuresis, 29 both of which may be of particular concern in PwP who often experience urinary dysfunction, 92 orthostatic hypotension, 93 and are at elevated risk of falls. 94 Clear instruction regarding fluid intake is therefore essential for any PwP interested in following a KD.17,95 Transient KD-related tremor exacerbations have been previously documented in PwP 17 ; however, the underlying mechanism remains unclear. From a cardiovascular risk perspective, the emphasis on unsaturated fats in the MeDi-KD appears to have mitigated against short-term KD-associated spikes in total and LDL cholesterol in our cohort on aggregate. 63 However, previously reported beneficial effects of Mediterranean-ketogenic diets on HDL-C were not observed in our study.53,88,96 Notably, there was considerable inter-individual heterogeneity in lipid profile changes induced by both diets, emphasizing the need for close monitoring of blood lipid levels in future ketogenic diet trials. 63 The modest weight loss observed in the MeDi-KD arm did not place any participants in a higher risk category according to BMI status over 8-week follow-up in our study; however, unintended weight loss may make KDs poor long-term options for PwP struggling to maintain adequate body mass.97,98 Together, while our findings suggest that combining ketogenic and Mediterranean principles minimizes some of the safety risks associated with standard KDs, PD care providers should be prepared to discuss the remaining possibility of various side effects with patients curious about such regimens.
The apparent improved safety profile of the MeDi-KD relative to standard KDs may come at the expense of “optimal” levels of nutritional ketosis (1.5–3.0 mM) achievable with more severe carbohydrate restriction.78,99 Nevertheless, at-home and laboratory based blood BHB measurements demonstrated that the majority of participants in our study were able to sustain “light” nutritional ketosis (0.3–1.0 mM post-prandial; 0.5–1.0 mM fasting) while following the MeDi-KD, comparable to levels observed in previous KD studies in PwP.16,100 Importantly, expected thresholds for nutritional ketosis with KDs of varying macronutrient composition have yet to be established in PwP. 16 Disease-specific thresholds may be relevant, as previous studies have demonstrated substantial inter-individual variability in responses to diets of identical ketogenic ratios, with older age representing a constraint on the levels of ketosis achievable. 101 Explanations for this phenomenon may relate to age-dependent elevations in postprandial insulin, decreases in plasma carnitine, decreases in plasma lipoprotein lipase activity and decreases in hepatic glucagon sensitivity. 102
High inter-individual variability in the ketogenic effect of MCT oil has likewise been demonstrated in human studies. 102 MCT supplementation is known to induce at most mild ketonemia in the absence of dietary carbohydrate and/or caloric restriction,12,103–105 with several studies reporting no MCT-induced increases in plasma, 106 or brain BHB.105,107 The subthreshold blood BHB elevations seen in our MeDi-MCT intervention, and their dissociation from self-rated adherence scores, was therefore not entirely unexpected, and were not interpreted as evidence that the MCT oil was not consumed as reported. Importantly, blood [BHB] may also be a poor predictor of the likelihood of neurologic benefit from MCT supplementation: emerging evidence suggests that MCTs may exert neuroprotective effects via the direct actions of plasma and brain medium-chain fatty acids,12,105 independent of changes in circulating ketones. Future studies of MCT supplementation should therefore consider alternative objective adherence measures to blood ketones, such as plasma medium chain fatty acids, whose levels may better correlate with primary bioenergetic and/or clinical outcomes. Together, these results suggest that the patient-reported clinical improvements seen in the MeDi-MCT phase were not related to nutritional ketosis itself, and require corroboration using primary objective outcome measures (such as the MDS-UPDRS Part III) in future efficacy studies.
Interestingly, self-rated adherence was also not associated with MEDAS scores during either intervention. Despite participants overwhelmingly claiming to have deviated from each diet program no more than a few times per week, MEDAS scores were predominantly in the “moderate adherence” category. Past studies have demonstrated similar disconnects between objective and subjective measures of adherence to Mediterranean diets. 108 Adherence may be remediable with more intensive dietary coaching, involving care partners where possible, 109 supplying ingredients or meal delivery, or by including a run-in period to test adherence and then only including those with satisfactory adherence in the main trial.72,78,89,110 The apparent ceiling effect for changes in MEDAS scores on the MeDi-KD may be additionally explained by the combined diet's inherent restriction of carbohydrates like fruits, vegetables, and legumes, which figure in to the MEDAS calculation.
Exploratory efficacy
Our secondary analyses revealed significant improvements in MDS-UPDRS Parts II and IV scores following the MeDi-MCT intervention, with similar trends observed in the MeDi-KD phase. Decreases in MDS-UPDRS Parts II and IV scores following a ketogenic diet was reported in a previous RCT in PwP. 17 While the mechanisms underlying these observations are not fully understood, improved gastrointestinal motility reported by several participants in our study may have optimized levodopa pharmacokinetics, alleviating motor complications and improving motor experience of daily living. 111 Diet-related changes in gut microbiota composition and metabolic function may have also contributed to this effect.17,111 Analysis of diet-induced shifts in the gut microbiota during the KIM trial is currently underway, and will be reported in a future publication. Finally, the unblinded nature of the trial makes it essential to consider the influence of participant expectations on clinical outcomes, particularly given that changes were observed only in self-reported parts of the MDS-UPDRS, and not in the objectively-rated MDS-UPDRS Part III. 112 The absence of any clinically meaningful changes in cognition scores was unsurprising given the short interval between baseline and follow-up visits, and the underrepresentation of participants with baseline cognitive impairment in our study. 113
Limitations
Several limitations need to be considered in evaluating our study findings. While we did not observe adverse effects of either diet on fasting lipid profiles nor major adverse events, our small sample size and short-duration interventions lower the likelihood that our study would have been able to detect such effects, as well as rarer or long-term complications of ketogenic diets and/or MCT supplementation, including vitamin and mineral deficiencies, osteopenia and kidney stones. 21 Future, longer-duration studies involving more precise assessment of dietary intake would be better poised to assess the true safety impacts of sustained nutritional ketosis in PwP. Likewise, such studies would help to clarify whether the expected levels of ketosis for given macronutrient compositions differ in PwP relative to the general population.
Conclusion
Mediterranean-ketogenic and MCT-supplemented Mediterranean diets appear to be generally safe in individuals with mild-to-moderate PD. Mild nutritional ketosis was achieved by approximately one-half of participants following the carbohydrate-restricted MeDi-KD, despite modest overall compliance. While ketosis was not consistently achieved with MCT supplementation alone in the absence of carbohydrate restriction (MeDi-MCT), this was interestingly the only phase in which modest clinical improvements were observed. Given our phase II study's suboptimal adherence and sub-target retention rates, thoughtful strategies to improve study diet feasibility are warranted before attempting to validate these findings in a phase III trial. Incongruence between participants’ self-reported versus objective measures of adherence indicate the likely utility of dietitian-led behavior change techniques under the domain of “Increase awareness of behavior” in any such future studies.89,114 Novel techniques could include photo-based food logs or brief daily digital checklists to prompt frequent reflection on dietary choices,115,116 coupled with tailored guidance for navigating social eating contexts.89,114 Finally, gastrointestinal side effects and cholesterol levels should be closely monitored in any forthcoming trials, as in clinical practice for patients opting to follow a ketogenic or MCT-supplemented diet.
Footnotes
Acknowledgements
The authors would like to thank all personnel who supported the trial's operations, and all the participants for the time and effort invested in our study. Dr Biljana Jonoska Stojkova and Frances Cheng are thanked for their statistical support. Dr Christopher Gardner and Dalia Perelman of Stanford Medicine are thanked for their guidance around the development of participant diet education materials. We would additionally like to acknowledge Ajinomoto Cambrooke Inc. (United States), a therapeutic nutrition company, which supplied the gram scales for participants and offered two Ketogenic Diet Professional Training sessions to YNW.
Ethical considerations
Ethical approval was granted by the University of British Columbia Clinical Research Ethics Board (Protocol #: H21-03747).
Consent to participate
All participants in the study provided written informed consent prior to enrolment.
Consent for publication
All participants consented to the de-identified dissemination of study findings as a part of the informed consent process.
Author contributions
SK, MSC, AMR, MAS, BBF, TRC and SAC were involved in study conceptualization, methodology and funding acquisition. KT, YNW and AJK were responsible for investigation, data curation and project administration. KT conducted the formal analysis. KT, JSTL and YNW prepared the original draft of the manuscript. All authors reviewed and approved of the final version of the manuscript. SAC supervised the project.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The KIM trial was funded by the Weston Family Foundation (Grant Number: F21-04689). SAC is supported by the Pacific Parkinson's Research Institute's Marg Meikle Professorship, and has received grant funding from the Pacific Parkinson’s Research Institute, the Weston Family Foundation, Parkinson Canada, Parkinson Society British Columbia, Rick’s Heart Foundation, Canadian Institutes of Health Research (CIHR), Jack and Darlene Poole Foundation, VGH and UBC Hospital Foundation and UBC Research Excellence Clusters program. KT is supported by a CIHR (Canadian Institutes of Health Research) Canada Graduate Scholarships - Doctoral Award (CGS-D) and the University of British Columbia MD/PhD Program.
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data supporting the findings of this study are available upon reasonable request to the corresponding author.
Supplemental material
The supplemental material is available in the electronic version of this article.
