Abstract
Introduction
There are conflicting findings regarding the influence of sex on idiopathic REM sleep behavior disorder (iRBD) in terms of prevalence and associated clinical characteristics. This study, conducted as part of the Italian multicenter longitudinal FARPRESTO project, aims to explore sex-related differences in the age of onset, iRBD diagnosis, and phenoconversion, as well as in cognitive and non-motor features and the occurrence of RBD-related injuries among male and female patients with iRBD.
Methods
The FARPRESTO study included 536 iRBD patients recruited from 13 Italian centers. This analysis assessed the age at iRBD diagnosis, diagnostic delay, motor and non-motor symptoms, global cognitive performance, conversion rates to neurodegenerative disorders, and the prevalence of RBD-related injuries at the time of iRBD diagnosis, stratified by sex.
Results
Female patients were older at iRBD diagnosis compared to males (males: 67.8 years, IQR 62.5–72.6; females: 69.8 years, IQR 65.1–74.8; p = 0.003). Compared to male patients, female patients exhibited a higher prevalence of orthostatic hypotension (27.9% vs. 16.2%; p = 0.019), depression (43.9% vs. 26.6%; p = 0.010), and hallucinations (43.9% vs. 26.6%; p = 0.010) at iRBD diagnosis. Additionally, self-directed injuries were significantly more frequent in females compared to males at the first visit (71% vs. 53.2%; p = 0.034). No significant differences were observed in the phenoconversion rate between sexes.
Conclusion
Although research on sex-related differences in iRBD remains limited, this study highlights the importance of understanding sex-specific characteristics. As diagnostic and therapeutic approaches evolve, incorporating these differences will be essential for tailoring clinical strategies and improving patient outcomes.
Plain language summary
Keywords
Introduction
Rapid eye movement (REM) sleep behavior disorder (RBD) is a REM sleep parasomnia characterized by dream enactment behaviors due to the loss of physiological REM sleep muscle atonia. It typically occurs in individuals over 50 years old and is frequently associated with neurological diseases, primarily synucleinopathies such as Parkinson's disease (PD), dementia with Lewy bodies (DLB), and multiple system atrophy (MSA). RBD can also present in an isolated form as idiopathic RBD (iRBD). However, with sufficient follow-up, the majority of iRBD patients eventually develop a neurodegenerative disorder in a process referred to as “phenoconversion”. 1
From early observations, a male predominance in RBD presentation has been reported. 2 However, findings in the literature remain inconsistent, likely due to methodological variations, including differences in study samples (clinical sample vs. general population) and diagnostic methods, e.g., confirmation through video-polysomnography (vPSG) versus probable RBD diagnosis. 3 According to the diagnostic criteria outlined in the International Classification of Sleep Disorders, Third Edition (ICSD-3) 4 an RBD diagnosis requires both a history of recurrent sleep-related vocalizations and/or complex motor behaviors and the presence of REM sleep without atonia confirmed by vPSG. In contrast, many population-based studies rely on clinical history alone, classifying individuals as having possible or probable RBD (pRBD) without vPSG confirmation.
Takeuchi et al. 5 recently investigated 43 female and 141 male patients with vPSG-confirmed iRBD, finding that female patients had an older estimated age of onset compared to males, while no differences were observed in age at diagnosis. This study also reported no sex-related differences in olfactory function or cognitive performance in the same cohort. 5 Similarly, Fernàndez-Arcos 6 analyzed a larger cohort of 41 female and 162 male iRBD patients and found no sex-related differences in age at diagnosis or phenoconversion. While male predominance in RBD remains incompletely understood, other sex-related differences, including those in cognitive or non-motor symptoms, age at onset and phenoconversion, or the presence of self-inflicted or bed partner injuries, are even less clearly defined. Larger longitudinal studies are needed to elucidate the clinical and prognostic trajectories of iRBD based on sex. 7
The Risk Factors Predictive of Phenoconversion in Idiopathic REM Sleep Behavior Disorder Italian Study (FARPRESTO) is a multicenter, longitudinal, retrospective, and prospective Italian project promoted by the Italian Association of Sleep Medicine (AIMS; www.sonnomed.it) and endorsed by the Italian Association of RBD Patients (www.associazionerbd.it). The project currently involves 14 Italian sleep centers. 8
The aim of this study was to investigate sex-related differences within the FARPRESTO cohort of patients with vPSG-proven iRBD in terms of: a) age at disease onset, age at first clinical visit, and age at phenoconversion; b) cognitive and non-motor characteristics, and c) the occurrence of dream enactment-related injuries. 8
Patients and methods
All participants in this study were patients with iRBD enrolled in the FARPRESTO study. The data presented in this article were collected as of February 2023, with 536 iRBD patients consecutively recruited from 13 accredited Sleep Medicine Centers across Italy (Bologna – two centers, Cagliari, Genoa, Milan, Pavia, Pisa, Rome Gemelli, Rome Tor Vergata, Verona, Troina, Udine, and Pescara), all members of the Italian Association of Sleep Medicine. The cohort included both previously diagnosed and newly diagnosed iRBD patients, all meeting the ICSD-2 or ICSD-3 diagnostic criteria and aged ≥18 years. Because the FARPRESTO cohort includes patients diagnosed both before and after 2014, iRBD diagnoses were made according to ICSD-2 criteria for earlier cases and ICSD-3 criteria for later cases, with the latter providing more detailed specifications for REM sleep without atonia. Because the ICSD-3-Text Revision (2023) postdated most enrollments, diagnoses were adjudicated under ICSD-2 or ICSD-3 as appropriate. The ICSD-3-TR 9 does not substantively alter the core case definition of vPSG-confirmed iRBD (recurrent REM sleep–related behaviors with REM sleep without atonia), and our case ascertainment is therefore consistent with ICSD-3-TR. Moreover, as this is a clinical rather than a general population sample, potential selection bias, particularly regarding sex distribution, has been considered in the interpretation of the results. All participants provided signed informed consent.
For each participant, we recorded sex, marital status, age at iRBD diagnosis, and diagnostic delay. Age at iRBD diagnosis referred to the age at the first clinical visit in which the diagnosis of iRBD was established according to ICSD-2 or ICSD-3 criteria. Diagnostic delay was calculated as the time interval between the self-reported onset of RBD symptoms and the date of iRBD diagnosis. Motor symptoms were assessed using the Unified Parkinson's Disease Rating Scale part III (UPDRS III) or the Movement Disorders Society-UPDRS part III (MDS-UPDRS III). Non-motor symptoms (including hyposmia, constipation, urinary dysfunction, erectile dysfunction, orthostatic hypotension, depressive symptoms, and hallucinations) were evaluated in agreement with the FARPRESTO protocol 8 through semi-structured interviews and/or validated questionnaires, including the Scales for Outcomes in Parkinson's Disease-Autonomic Dysfunction (SCOPA-AUT), the Hospital Anxiety and Depression Scale (HADS), and the Beck Depression Inventory (BDI). 8 Hyposmia was assessed by self-report unless otherwise documented by an objective smell test in the medical record. 8
To standardize the evaluation of cognitive impairment, the ‘MoCA-to-MMSE’ conversion tables by Aiello 10 were applied. Similarly, to harmonize UPDRS III and MDS-UPDRS III scores, the formula developed by Goetz et al. was used. 11
This study adhered to the principles outlined in the Declaration of Helsinki. Ethical approval was obtained from the local ethics committees of all participating FARPRESTO centers, and informed consent was obtained from all participants. The FARPRESTO study is registered at clinicaltrials.gov (NCT05262543).
Statistical analysis
Due to the non-parametric distribution of data, the Mann–Whitney U test was used to evaluate differences between males and females at the first medical examination (for the overall sample and phenoconverted subjects) and at the second medical examination (for phenoconverted subjects) in the following variables: age at iRBD diagnosis, age at MMSE or MoCA testing, age at RBD onset, age at conversion, age at post-conversion MMSE or MoCA testing, years of education, RBD-related injuries, UPDRS III or MDS-UPDRS III scores, MMSE scores, and BDI scores. In the FARPRESTO protocol, follow-up visits are scheduled at least every 12 months; for the present analysis, we considered the first visit (diagnosis) and the visit at phenoconversion, or the most recent follow-up in non-converted patients, with a median follow-up duration of 2.57 years across participants, reflecting variability in observation periods. Differences in age at iRBD diagnosis and age at RBD onset were also analyzed between patients with and without a history of phenoconversion.
The Pearson's chi-squared test was used to analyze differences in categorical variables between males and females at the first medical examination (for the overall sample and phenoconverted subjects) and at the second medical examination (for phenoconverted subjects). These variables included marital status, presence/absence of lesions, oneiric status (defined as the presence of a recollection of dream content), hyposmia (yes/no), constipation (yes/no), urinary dysfunction (yes/no), orthostatic hypotension (yes/no), depression (yes/no), presence/absence of hallucinations, and injuries (to the patient, the partner, or both). For the purpose of statistical analysis, injuries were stratified into self-directed (affecting only the patient) and other-directed (affecting the bed partner) to examine potential differences in their occurrence and associated factors.
Information on injuries was collected for all participants at the first visit using a standardized semi-structured interview, following the FARPRESTO protocol. Injuries were classified as affecting the patient, the bed partner, or both, and self-directed injuries were defined as those sustained by the patient during dream-enactment behaviors. This classification was applied consistently in all analyses. At the second medical examination, lesion data were collected in the same manner, through self-report during a structured medical interview; no additional vPSG was performed.
For the survival analysis, we included all participants with complete follow-up data (n = 320), comprising individuals who experienced phenoconversion (n = 121) and those who had not converted by their most recent follow-up (n = 199), who were treated as censored. In accordance with the FARPRESTO protocol, follow-up visits are scheduled at least every 12 months. The time-to-event variable was defined as the interval between the date of iRBD diagnosis (first visit) and either the phenoconversion date (for events) or the date of the second clinical follow-up visit (for censored cases). Participants excluded from the Cox regression (n = 217) were removed due to missing values in key variables (n = 216) or automatic software exclusion before the first observed event in their stratum (n = 1). The sex distribution did not differ significantly between included and excluded participants (χ² = 0.86, p = 0.353), indicating no evidence of sex-related exclusion bias. This approach allowed us to account for variable follow-up durations across participants, rather than relying on two fixed time points. To reduce the risk of model overfitting, covariates were preselected a priori based on clinical relevance and baseline sex-related differences, and only two predictors (sex and age at iRBD diagnosis) were included, ensuring an event-per-variable ratio greater than 10, consistent with recommended standards for Cox regression.
A two-tailed α error threshold of p < 0.05 was used to reject the null hypothesis. Statistical analyses were conducted using SPSS® version 20.0 (IBM Corporation, Armonk, NY, USA).
Results
Demographic and clinical data by sex for the overall study sample and the subgroup of subjects with conversion are presented in Tables 1a, 1b, 2a, and 2b.
Demographic and clinical data by sex for the overall study sample (continuous variables).

Note: The phenoconverted subgroup (n = 121) is included within the overall cohort in this table. Subgroup-specific values are presented in Tables 1b and 2b. IQR = interquartile range; MMSE = mini-mental state examination; MoCA = Montreal cognitive assessment; RBD = REM sleep behavior disorder; UPDRS = Unified Parkinson's Disease Rating Scale; MDS-UPDRS = Movement Disorder Society UPDRS.
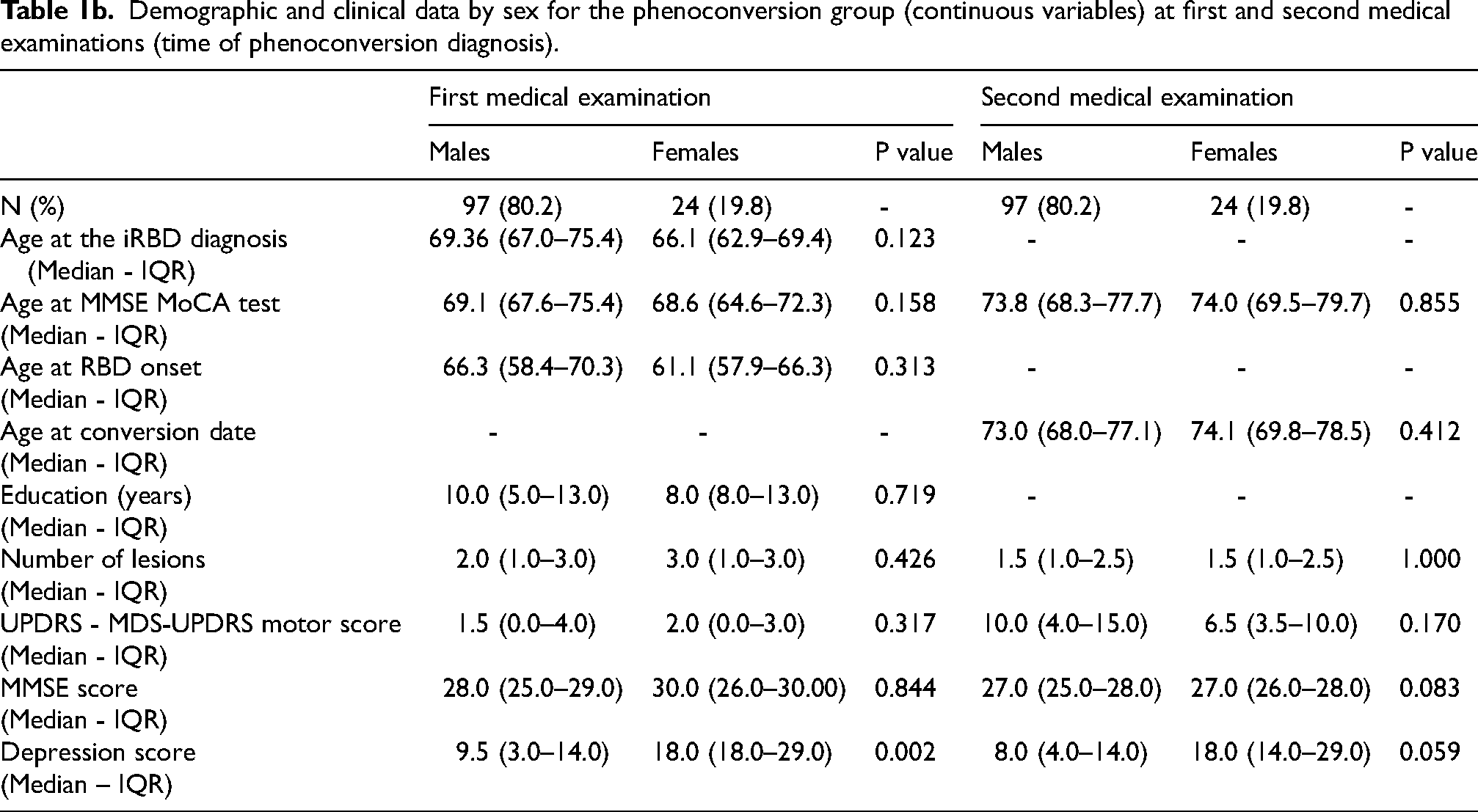
Demographic and clinical data by sex for the phenoconversion group (continuous variables) at first and second medical examinations (time of phenoconversion diagnosis).
Clinical data by sex for the overall study sample (categorical variables).
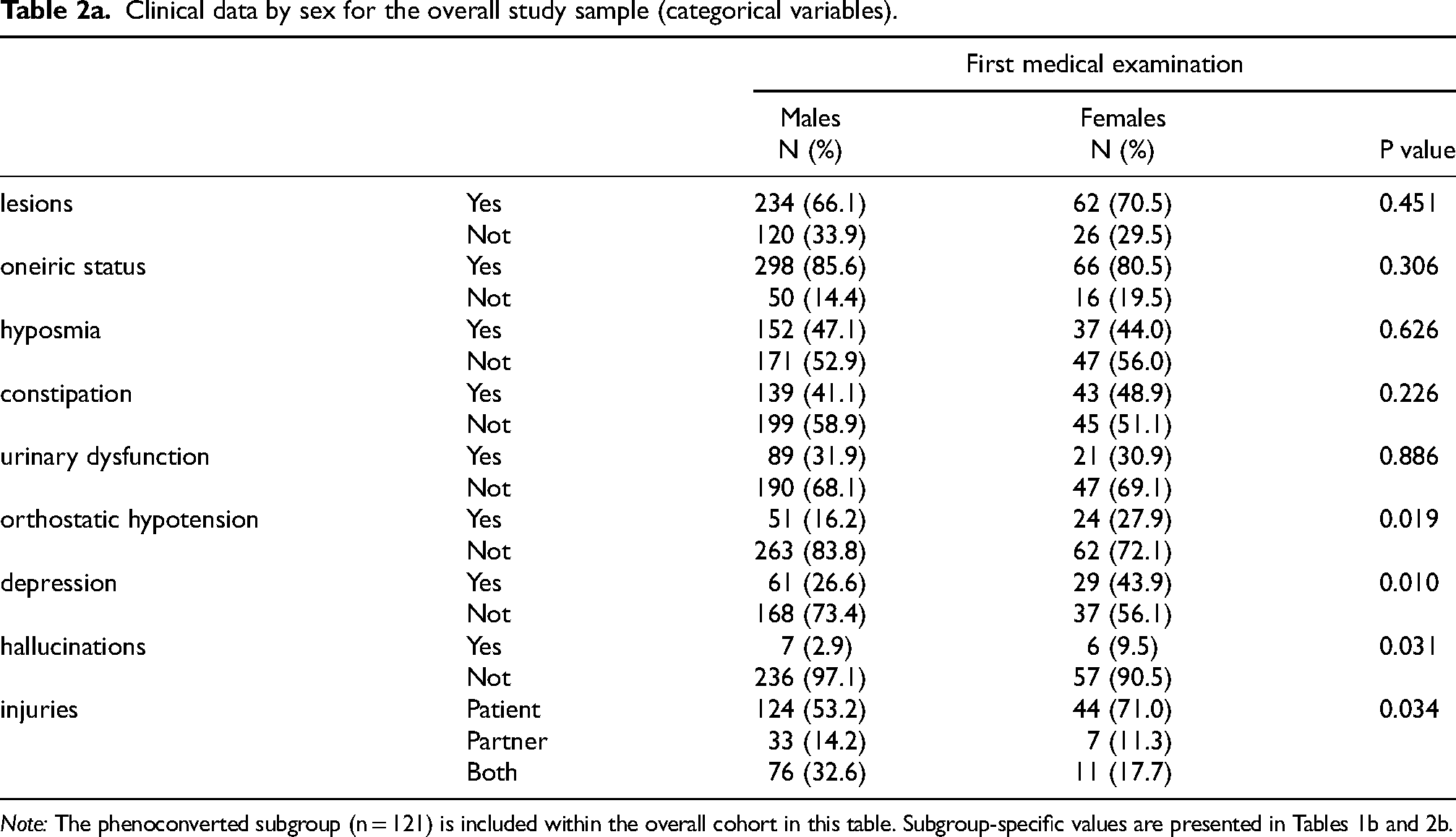
Note: The phenoconverted subgroup (n = 121) is included within the overall cohort in this table. Subgroup-specific values are presented in Tables 1b and 2b.
Clinical data (categorical variables) for the phenoconversion group by sex at first medical examination and second medical examination (time of diagnosis of phenoconversion).
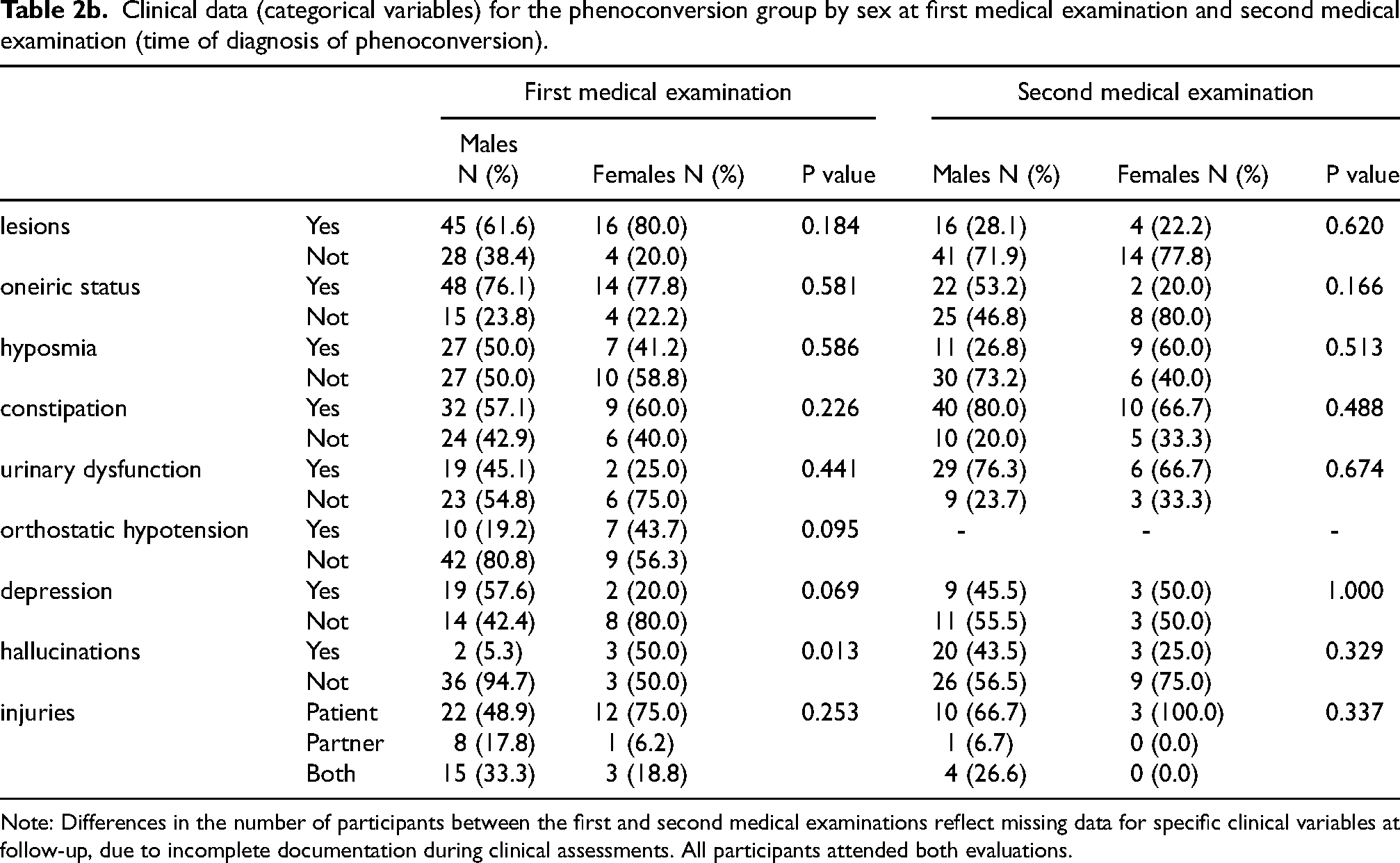
Note: Differences in the number of participants between the first and second medical examinations reflect missing data for specific clinical variables at follow-up, due to incomplete documentation during clinical assessments. All participants attended both evaluations.
A total of 121 out of 536 subjects (22.6%) underwent phenoconversion, including 24 out of 105 females (22.9%) and 97 out of 431 males (22.5%). Among males, the diagnoses were: 55 PD (12.8% of males and 10.3% of the total), 31 DLB (7.2% of males and 5.8% of the total), and 9 MSA (2.1% of males and 1.7% of the total). In the female group, 13 were diagnosed with PD (12.4% of females and 2.4% of the total), 8 with DLB (7.6% of females and 1.5% of the total), and 3 with MSA (2.9% of females and 0.6% of the total), according to current diagnostic criteria. 8 Additionally, two male subjects had an undefined diagnosis because, although they did not meet specific criteria, an extrapyramidal condition was evident. There was no significant difference in the distribution of phenoconversion phenotypes between female and male patients (p = 0.894). Among the 415 subjects who did not undergo phenoconversion at the first follow-up visit, there were 81 females and 334 males.
No significant differences were observed in age at iRBD diagnosis or age at RBD onset between patients with and without a history of conversion. In this study, age at iRBD diagnosis refers to the age at the first clinical visit when the iRBD diagnosis was established, while diagnostic delay represents the interval between the self-reported onset of RBD symptoms and the date of iRBD diagnosis.
We also calculated the prodromal duration, defined as the interval between reported RBD symptom onset and phenoconversion. The median duration was 6.92 years in females and 7.11 years in males, with no significant sex-related difference (Mann–Whitney U = 475.0, p = 0.427).
Female participants were older at diagnosis (mean ± SD: 69.23 ± 6.57 years vs. 67.00 ± 7.52 years; p = 0.019) and showed higher MMSE scores (28.17 ± 2.04 vs. 27.60 ± 2.39; p = 0.049) and MoCA scores (25.74 ± 3.38 vs. 24.43 ± 4.21; p = 0.012) compared with males. Notably, there was no significant difference in the age of RBD onset.
Education level was significantly higher in males at the time of iRBD diagnosis (median 13.0 years, IQR 8.0–13.0) compared to females (median 8.0 years, IQR 8.0–13.0; p = 0.015). However, no significant differences in education level were found among female and male patients who underwent phenoconversion. Aside from females being older at iRBD diagnosis, no significant sex differences were observed in age at RBD symptom onset, age at phenoconversion, or age at post-conversion MMSE/MoCA testing. The presence of orthostatic hypotension (29.6% vs. 20.2%; p = 0.043), depression (29.6% vs. 17.3%; p = 0.006), and hallucinations (11.9% vs. 5.4%; p = 0.041) was more frequent in females than in males in the entire group. In the phenoconverted subgroup, hallucinations and depression were more frequently observed in females at the time of the first visit.
Additionally, injuries affecting patients were significantly more frequently reported in females (71.0%) compared to males (53.2%; p = 0.034). Injuries affecting the partner (11.3% females vs. 14.2% males) or both the patient and the partner (17.7% females vs. 33.6% males) were less common in females compared to males. To clarify the overall patient involvement, when injury categories were collapsed into “patient-involved” (self-directed or both) versus “partner-only,” the proportion of patient-involved injuries was 88.7% in women (55/62) and 85.8% in men (200/233), with no significant sex difference (Fisher's exact p = 0.68).
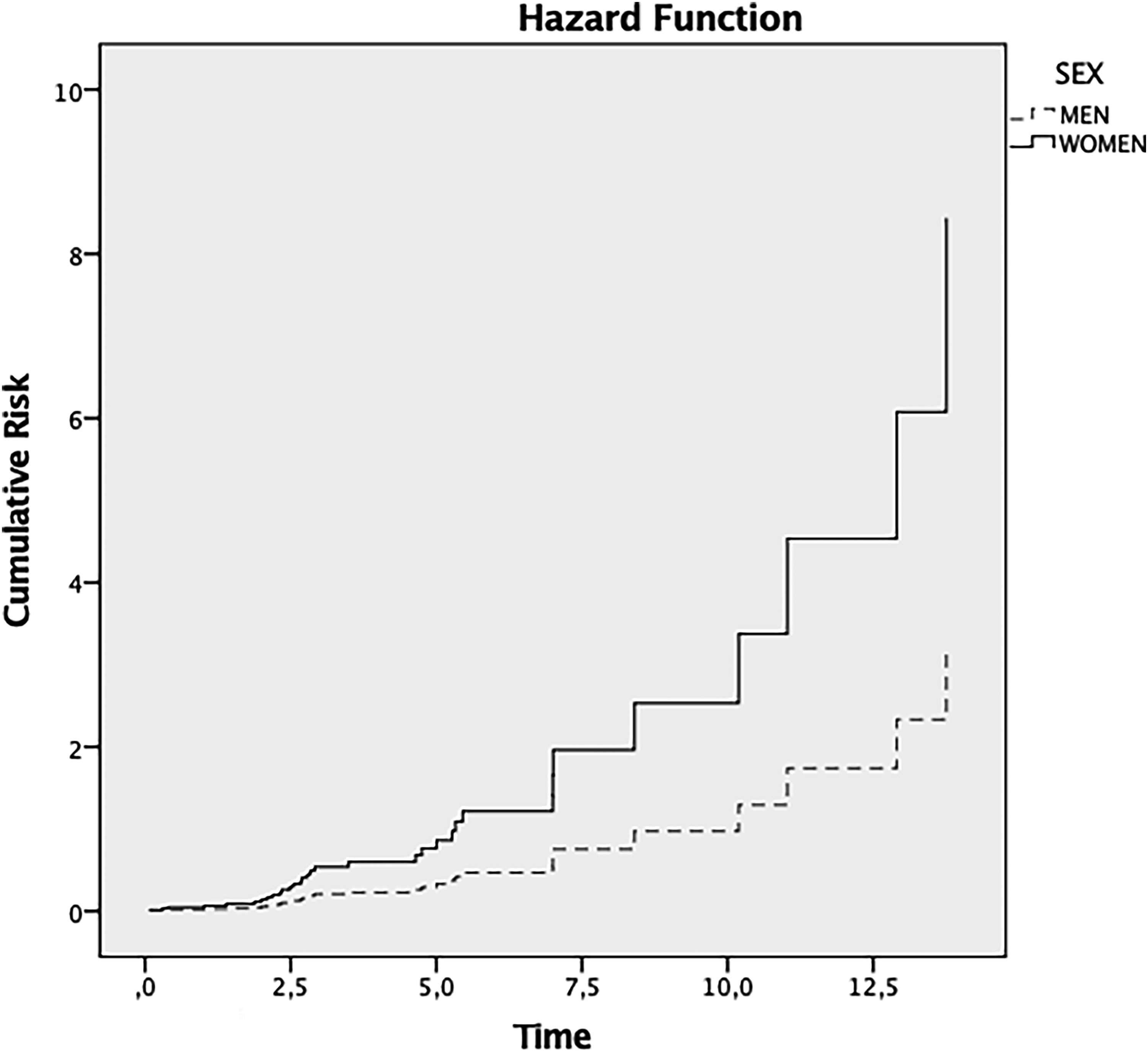
All 536 participants completed at least one follow-up visit; 217 were excluded from the Cox regression due to missing values in key variables (n = 216) or automatic software exclusion before the first observed event in their stratum (n = 1). The sex distribution did not differ significantly between included and excluded participants (χ² = 0.86, p = 0.353), indicating no evidence of sex-related exclusion bias. Overall, the median follow-up duration for the 319 participants included in the Cox regression analysis was 2.57 years. Follow-up intervals from iRBD diagnosis to the second clinical visit were also comparable between sexes: among converters, the median follow-up was 3.00 years in males and 2.73 years in females (p = 0.897), and among censored participants, 2.53 years in males and 1.89 years in females (p = 0.271). No differences in the risk of conversion between males and females were found (median time: females = 2.54 years; males = 2.64 years; β = 1.14, 95% CI 0.72–1.81, p = 0.571) (Figure 1).
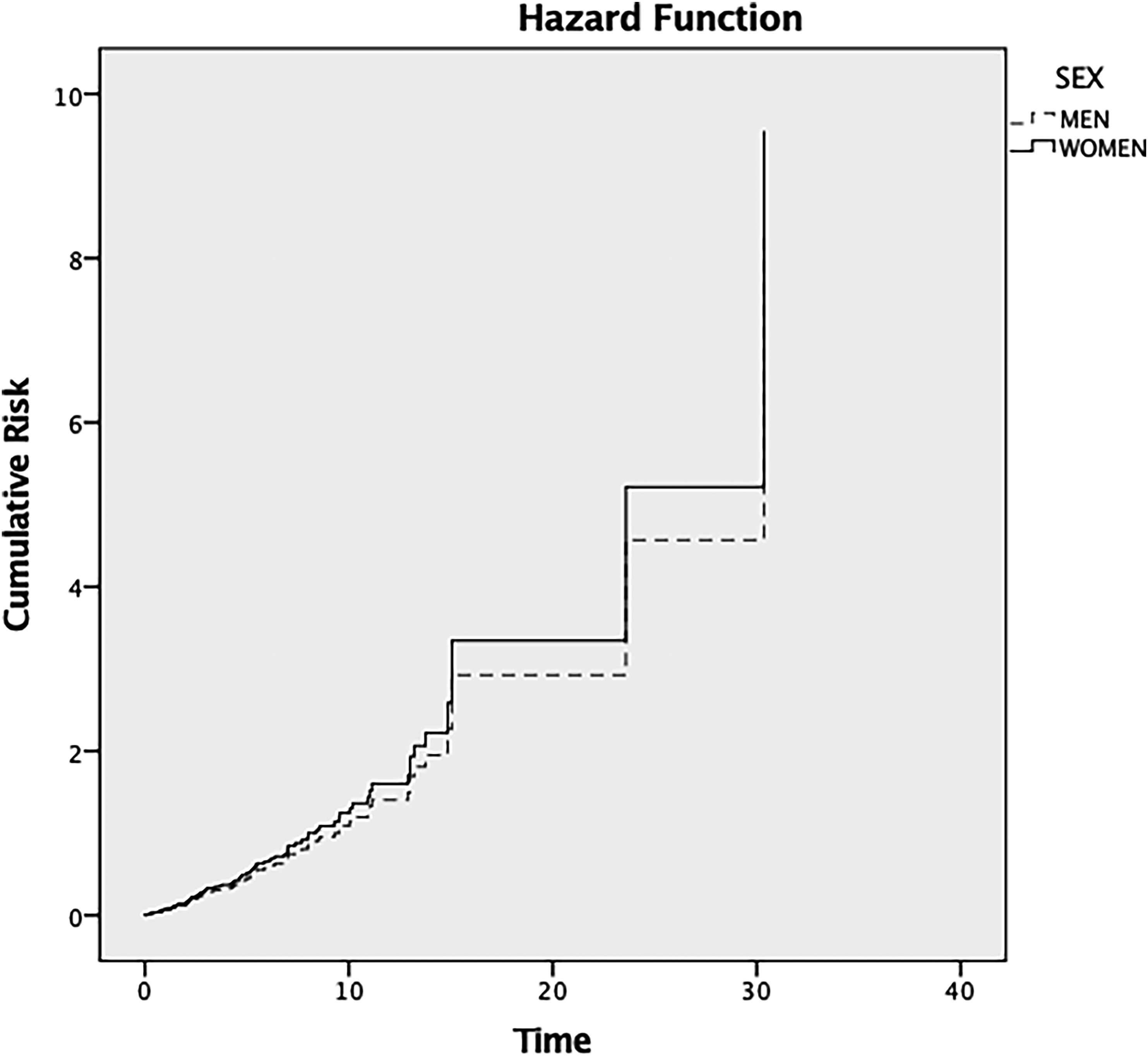
Hazard function of the cox regression analysis of risk of conversion in males and females (model corrected by age at iRBD diagnosis). The observation period (time in months) was defined as the time between iRBD diagnosis and the next medical examination). β = 1.14, 95% CI 0.72–1.81, p = 0.571.
No differences in the risk of conversion between males and females were found (median time: females = 2.54 years; males = 2.64 years; β = 1.14, 95% CI 0.72–1.81, p = 0.571) (Figure 1). After stratification for injuries (patient and partner), orthostatic hypotension, depression, and history of hallucinations, a significantly higher risk of conversion was observed in women with a history of injuries affecting patients at first iRBD diagnosis (β = 2.61, 95% CI 1.19–5.73, p = 0.017) (Figure 2).
Hazard function of the Cox regression analysis of risk of conversion in men and women in the subgroup with history of injuries affecting patients at first iRBD diagnosis (model corrected by age at the iRBD diagnosis) The observation period (time) was defined as the time between iRBD diagnosis and the next medical examination). β = 2.61 (CI 1.19–5.73 p value 0.017).
Discussion
In the present study, we observed a significant male predominance, with 4 out of 5 included patients being male, a finding consistent with previous research. 2 This disparity may be partially explained by the tendency for male patients to exhibit more aggressive and violent behaviors, potentially prompting them to seek medical attention more readily. 12 However, in women, injuries affecting patients at the first visit were significantly more frequent than injuries affecting the partner or both . Among men, the distribution was 53.2% for patient injuries, 14.2% for partner injuries, and 33.6% for injuries affecting both. This discrepancy may partly explain why women experience a longer latency between symptom onset and RBD diagnosis compared to men.
Biological factors such as the protective role of estrogen or a potential negative influence of androgens may also contribute to these differences. 13 However, previous studies have found no abnormalities in sex-linked serum hormones in male iRBD patients.14,15 Furthermore, our data revealed that while the age at iRBD diagnosis was significantly higher in the female group, no statistical difference was observed regarding the age of disease onset. This aligns with other studies and may reflect not only greater symptom intensity and severity in male patients but also socio-ecological factors influencing the detection of abnormal nighttime behaviors by bed partners.16,17
Gender disparity in education and cognitive performance
We identified a significant gender disparity in education, with males having higher educational attainment at the time of iRBD diagnosis. This finding reflects broader societal norms and historical educational policies, as cohorts born between 1920 and 1959 generally exhibited lower educational levels among women, a gap that has gradually narrowed. 18 Education level differences have been identified as a key factor contributing to the gender gap in late-life cognition, particularly in developing countries. 18
However, in our study, no significant gender differences were observed in cognitive test scores. Instead, we found a significant difference in the age at which these tests were administered, consistent with the older age at iRBD diagnosis in female patients.16,17
Educational attainment may influence recognition and help-seeking for nocturnal behaviors (via health literacy, symptom awareness, and facility in accessing specialist services) and could therefore contribute to sex-related differences in presentation and referral. This possibility should be considered when interpreting our findings.
Clinical symptoms and injuries
Orthostatic hypotension, depressive symptoms, and hallucinations were more frequently observed in females than in males at the time of iRBD diagnosis. The higher prevalence of both cortical and subcortical symptoms observed in female participants may suggest a broader distribution of alpha-synucleinopathy compared with males in our cohort. This could be related, at least in part, to their older age at the time of diagnosis, which might allow more time for pathological spread. Additionally, in the phenoconverted subgroup, hallucinations and depressive symptoms were more frequently reported in female patients at their first visit. However, the number of phenoconverted female patients with available data on hallucinations was relatively small, which may introduce a reporting bias. This limitation should be considered when interpreting this finding, and larger samples may help to confirm and better quantify this association.
Notably, PD is more prevalent in males than females and exhibits distinct sex-related pathophysiological and clinical differences. 19 Women with PD tend to experience more frequent and severe non-motor symptoms, including depression, autonomic dysfunction, pain, and hyposmia.19–22 Additionally, sex-related differences have been hypothesized to influence PD pathophysiology, particularly in mechanisms involving dopaminergic pathways, neuroinflammation, and oxidative stress. 19
In DLB, a recent longitudinal study revealed sex-specific differences in the timing and presentation of symptoms. Men were found to experience probable RBD earlier than women and prior to the onset of cognitive symptoms. 23 Conversely, women with DLB were observed to present RBD concurrently with cognitive dysfunction and were more likely to experience visual hallucinations, which occurred earlier and more frequently in the disease course compared to male patients with DLB. 23 Furthermore, in the general population, women exhibit a higher prevalence of depressive symptoms compared to men.24–26 This disparity may result from a complex interplay of social, behavioral, educational, and biological factors.24–26
At the time of iRBD diagnosis, women were more likely to report self-directed injuries, while men more frequently caused injuries to their bed partners. The longer diagnostic delay observed in females may be influenced both by the more frequent presence of self-directed injuries, which may be less apparent to others and thus less likely to prompt external concern, and by socio-ecological factors, such as living alone or being widowed, which may reduce the likelihood of early recognition by a partner. This pattern may partly reflect demographic differences, as women in our cohort were less likely to have a bed partner and more frequently divorced or widowed, which could influence the proportion of self-directed versus partner injuries reported. Examples of socio-ecological factors include sleeping separately from a partner; partner-related limitations to observation (e.g., poor health, sensory impairment, or deep sleep); lower awareness of RBD symptoms; and reduced access or delayed referral to accredited sleep centers, all of which may delay recognition and reporting of nocturnal behaviors. Interestingly, this is consistent with findings from a recent large cohort study involving vPSG-confirmed RBD patients without parkinsonism, dementia, or narcolepsy, which reported that men were more likely to injure their bed partners than women (45% vs. 17%). 27 Regarding dream-enactment behaviors and related injuries, findings in the literature remain inconsistent. Early reviews and cohort studies did not identify sex-related differences in dream-related behaviors in iRBD patients.6,28,29 A recent meta-analysis also found no significant differences in the risk of sleep-related injuries. However, a systematic review suggested that women with RBD exhibit less dream-enacting and disruptive behaviors compared to men, which may contribute to a lower likelihood of seeking medical consultation.6,28
In our cohort, these injury data were systematically collected through a standardized semi-structured interview, ensuring comparability across participants. This methodology allows a more reliable estimation of the prevalence of self-directed versus partner injuries at baseline and supports our finding of a higher frequency of self-directed injuries in women. Such differences may reflect sex-specific patterns of dream-enactment behaviors, differences in reporting, or sociocultural factors influencing symptom recognition and medical referral.
Given that participants were recruited from accredited Sleep Medicine Centers, our sample reflects a clinical sample rather than the general population. This recruitment context may introduce selection bias, including potential sex-related differences in referral patterns, symptom recognition, or healthcare-seeking behavior. These factors should be considered when interpreting the generalizability of our findings.
These discrepancies may stem from methodological differences, reporting biases, or variations in the rate of men and women with bed partners across studies. Regardless, a standardized approach to investigate this topic is essential, as RBD-associated behaviors can result in severe injuries to bed partners. Raising awareness about these risks and promoting education on preventive measures is critical.29–31
Conversion risk and long-term follow-up
Consistent with our Results, the overall time-to-phenoconversion did not differ between sexes (β = 1.14, 95% CI 0.72–1.81; p = 0.571). In our cohort, a history of injuries affecting patients at first iRBD diagnosis was associated with a significantly higher risk of conversion in women. This injury-stratified observation is exploratory and hypothesis-generating and will require confirmation in larger samples with longer follow-up. To our knowledge, this finding has not been previously reported. One possible explanation is that the diagnosis in women occurs at a later stage of neurodegeneration, when injuries are more prevalent, whereas in men, violent behaviours may manifest earlier. This is supported by evidence that men with iRBD exhibit more violent dream-enacting behaviours (e.g., punching or striking) and higher phasic muscle activity during REM sleep, suggesting earlier and more intense symptom onset. 6 In our cohort, the overall phenoconversion rate was consistent with figures reported in previous longitudinal studies. 32 As a detailed analysis of phenoconversion predictors in the FARPRESTO cohort is currently ongoing and not a primary aim of the present work, we provide here only a brief contextualization of these rates within the existing literature. A longer follow-up of our cohort is necessary to confirm this hypothesis.
Strengths and limitations
This study has several strengths and limitations. One limitation is the relatively small number of phenoconverted patients. In particular, the number of phenoconverted female participants was limited, which may reduce statistical power and increase variability, potentially underestimating sex-related differences. Other limitations include incomplete assessments for all subjects, the absence of objective biomarkers for disease progression, the lack of an injury assessment scale, and the absence of an evaluation of RBD symptom severity. However, these limitations may be addressed over time through ongoing recruitment and the prospective nature of the study.
The FARPRESTO study's standardized methodology and large, vPSG-confirmed iRBD cohort provide a valuable retrospective and prospective study opportunity. The inclusion of vPSG recordings, neuroimaging assessments, and socio-demographic criteria reduces biases such as diagnostic uncertainty and the presence of hypnological comorbidities or RBD mimics.
Conclusions
The present study highlights a male predominance in the iRBD sample, with female patients being older at iRBD diagnosis and more often reporting injuries affecting only themselves, while men more often reported injuries affecting both themselves and their bed partner; overall, the proportion of injuries involving the patient (self-directed or both) was similar between sexes. Females also showed higher rates of orthostatic hypotension, depressive symptoms, and hallucinations at iRBD diagnosis. These findings suggest that sex-related factors may influence the clinical expression of iRBD. Despite being older at iRBD diagnosis, women did not differ from men in time-to-phenoconversion in the overall analysis. In an exploratory stratified analysis, women with a history of injuries affecting the patient at diagnosis showed a higher conversion risk; this finding requires confirmation. This finding suggests a potential protective factor against alpha-synucleinopathies associated with RBD in women. Furthermore, despite having less formal education, being of older age at the time of cognitive evaluation, and exhibiting a higher prevalence of hallucinations, factors that are generally recognized as risk elements for the development of dementia, women were as likely as men to develop DLB.
Future research is essential to elucidate the underlying mechanisms and support the development of tailored diagnostic and therapeutic approaches as new treatments become available.
Footnotes
Ethical considerations
This study adhered to the principles outlined in the Declaration of Helsinki. Ethical approval was obtained from the local ethics committees of all participating FARPRESTO centers, and informed consent was obtained from all participants. The FARPRESTO study is registered at clinicaltrials.gov (NCT05262543).
Consent to participate
Informed consent to participate was written, for further information see clinicaltrials.gov (NCT05262543).
Author contributions
Michelangelo Maestri, Michela Figorilli, Francesca Ingravallo and Monica Puligheddu equally contribuited to the design and implementation of the research, to the analysis of the results and to the writing of the manuscript.
Federico Meloni processed the experimental data, performed the analysis, and designed the figures.
Elisa Casaglia, Marcello Mario Mascia, Martina Mulas aided in interpreting the results and worked on the manuscript.
All authors discussed the results and commented on the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Italian Ministry of health :PRIN: PROGETTI DI RICERCA DI RILEVANTE INTERESSE NAZIONALE – Bando 2022 Prot. 2022LSSAK7. This work in financed by Italian Association of Sleep Medicine who is the promoter of the FARPRESTO registry.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
