Abstract
Background
Isolated REM sleep behavior disorder (iRBD) is a prodromal stage of α-synucleinopathies, including Parkinson's disease (PD), dementia with Lewy bodies (DLB), and multiple system atrophy (MSA). Evaluating trajectories of phenoconversion is crucial in this population. Plasma neurofilament light chain (NfL), a marker of axonal injury, has emerged as a promising candidate for tracking disease progression.
Objectives
To assess plasma NfL levels in iRBD patients, examine their associations with clinical features, particularly neurogenic orthostatic hypotension (nOH), and explore their potential role in predicting phenoconversion.
Methods
Plasma NfL was measured in 54 iRBD subjects, 54 PD, 54 healthy controls and 16 MSA. Participants underwent motor, cognitive, and non-motor symptoms assessments. Longitudinal follow-up data have been collected for iRBD subjects. NfL was quantified using the Ella platform; analyses were performed with Prism 9.
Results
NfL levels were significantly higher in iRBD compared to controls and similar to PD. iRBD-nOH+ patients had significantly higher NfL than nOH− counterparts, with values overlapping PD and MSA. nOH was inversely associated with hyposmia, supporting phenotypic divergence. NfL correlated positively with SCOPA-AUT and BDI scores. At follow-up, all four MSA converters had nOH at baseline.
Conclusions
Plasma NfL elevation in iRBD supports its role as a marker of early neurodegeneration. Its association with nOH suggests that this autonomic feature may identify a biologically more severe iRBD phenotype, possibly on a trajectory toward MSA. These findings warrant extended longitudinal validation and support the integration of clinical and biological markers, including NfL, for stratifying conversion risk.
Plain language summary
Some people act out their dreams while sleeping, often moving or talking during vivid dreams. This condition is called REM sleep behavior disorder (RBD), and it can be an early warning sign of diseases such as Parkinson's disease (PD), dementia with Lewy bodies (DLB), or multiple system atrophy (MSA). These brain conditions are known as alpha-synucleinopathies. However, not all people with RBD develop these diseases, and researchers are trying to find ways to identify those who are most at risk — especially before they show symptoms like tremor or memory loss. In this study, we looked at a blood protein called neurofilament light chain (NfL). This marker can indicate damage to nerve cells and may reflect how fast a disease is progressing. We measured NfL levels in the blood of people with RBD, PD, MSA, and healthy volunteers. We found that: People with RBD had higher NfL levels than healthy controls, but similar to those with PD. Those with RBD and orthostatic hypotension (a drop in blood pressure when standing up) had even higher NfL levels, similar to patients with MSA — a more severe condition. RBD patients with orthostatic hypotension were more likely to later develop MSA. These results suggest that combining a simple blood test with clinical signs like blood pressure changes may help identify which RBD patients are at higher risk of developing aggressive forms of neurodegenerative disease. This could be important for early diagnosis and for choosing participants in future clinical trials.
Keywords
Introduction
Isolated REM sleep behavior disorder (iRBD) is a well-established prodromal manifestation of α-synucleinopathies such as Parkinson's disease (PD), dementia with Lewy bodies (DLB), and multiple system atrophy (MSA). Over 80% of iRBD patients eventually convert to a defined synucleinopathy, with an estimated annual conversion rate of 6–12%.1,2 Identifying early biomarkers that can capture neurodegeneration before the onset of motor symptoms, and potentially differentiate among disease trajectories, is critical for prognosis and therapeutic stratification.
Among candidate biomarkers, plasma neurofilament light chain (NfL), a structural axonal protein released upon neuronal damage, has shown promise in tracking neurodegeneration across parkinsonian syndromes. Notably, NfL levels are substantially higher in atypical parkinsonisms like MSA compared to PD, reflecting the more aggressive axonal injury observed in these conditions.3,4
Recent studies suggest that autonomic dysfunction, particularly neurogenic orthostatic hypotension (nOH), may delineate a more severe phenotype of prodromal α-synucleinopathy.5–7 nOH is prevalent in MSA, often preceding motor symptoms, and has been associated with worse prognosis and faster disease progression in PD and iRBD.5,8 There is growing interest in whether nOH in iRBD reflects a more aggressive disease trajectory, potentially associated with increased NfL levels and risk of MSA, though this remains under investigation.9–12
Building on this evidence, the present study focused on three main objectives: (1) to characterize plasma NfL levels in iRBD patients; (2) to examine their association with nOH; and (3) to investigate whether nOH identifies a biologically more severe iRBD phenotype, as reflected by elevated NfL levels in the absence of motor signs.
Methods
Study design and participants
This study included both cross-sectional and longitudinal components. Between 2018 and 2021, fifty-four patients with iRBD, confirmed by video-polysomnography, were prospectively recruited at our centers. For comparison, we enrolled fifty-four patients with PD, of whom thirty-seven had a documented comorbid history of RBD (PDRBD), as well as fifty-four age-matched healthy controls with no history of neurological or sleep disorders. In addition, sixteen patients with a diagnosis of MSA were included.
Participants with a Charlson Comorbidity Index (CCI) greater than 3 or with relevant systemic comorbidities were excluded to minimize potential confounding effects on plasma NfL levels.
All iRBD participants were free of parkinsonism at baseline and fulfilled diagnostic criteria for idiopathic RBD. 13 All participants provided written informed consent in accordance with the Declaration of Helsinki, and the study protocol was approved by the local institutional review boards.
Healthy controls were included as methodological comparators for biochemical analyses only. They were free from any acute or chronic medical conditions and were not taking medications at the time of blood sampling; however, they did not undergo the same clinical and neuropsychological evaluations as the patient groups. MSA participants were similarly selected to ensure the absence of relevant comorbidities, and clinical information was collected during neurological evaluation at the time of sampling, but they were not administered the same battery of standardized assessments as the iRBD and PD cohorts.
The iRBD cohort underwent comprehensive clinical assessment and blood sampling for biomarkers analyses at baseline and was longitudinally followed with regular clinical reassessments every 6 to 9 months to monitor for possible phenoconversion to an overt α-synucleinopathy; thirteen patients were lost to follow-up.
Clinical Assessments: At baseline, all participants underwent a comprehensive battery of evaluations to assess motor, cognitive, and non-motor features. Motor function was evaluated using the motor subsection of the Movement Disorder Society-Unified Parkinson's Disease Rating Scale (MDS-UPDRS Part III), while global cognitive performance was assessed with the Montreal Cognitive Assessment (MoCA). In the iRBD and PD groups, additional assessments included the Beck Depression Inventory (BDI), the Non-Motor Symptoms Scale (NMSS), and the Scale for Outcomes in Parkinson's Disease–Autonomic Dysfunction (SCOPA-AUT). Olfactory function was assessed using the single item from the Non-Motor Symptoms Scale (NMSS, Domain 9) addressing hyposmia, and through a brief self-administered anamnestic questionnaire (see SM-1) specifically designed to identify secondary causes of smell loss. Participants reporting more than two risk factors for non-neurodegenerative hyposmia were classified as non-hyposmic to minimize potential confounding effects. Orthostatic hypotension was defined according to the current consensus criteria as a sustained drop in systolic blood pressure of ≥ 20 mmHg or in diastolic pressure of ≥ 10 mmHg within 3 min of standing. 14 Blood pressure and heart rate were measured manually after at least 5 min in the supine position and again at 1 and 3 min after standing. Neurogenic orthostatic hypotension was confirmed by assessing the heart rate response, with a ΔHR/ΔSBP ratio < 0.5 bpm/mmHg. 15
Plasma NfL measurement
Blood samples were collected in EDTA tubes at baseline, centrifuged (1000 g, 20 min, RT), and plasma was aliquoted and stored at −80°C until analysis. Plasma NfL levels were measured using Human NF-L Simple Plex cartridge (Ella®, ProteinSimple, CA, USA), in accordance with the manufacturer's instructions. Plasma samples were diluted 1:2 in Sample Diluent (ProteinSimple, CA, USA) before measurement. The lower limit of quantification was 2.70 pg/ml, and the upper limit of quantification 10.290.00 pg/ml.
Statistical analysis:
Statistical analyses were performed using Prism 9 (GraphPad Software). Group comparisons of plasma NfL levels were conducted using Kruskal–Wallis tests followed by Dunn's correction for multiple comparisons, due to non-normal data distribution. Within the iRBD group, Mann–Whitney U tests were used to assess differences in NfL based on the presence or absence of nOH, hyposmia, constipation, depressive symptoms, and mild cognitive impairment. Spearman's rank correlation was used to explore associations between NfL levels and clinical scales including MDS-UPDRS Part III, MoCA, BDI, and SCOPA-AUT.
To explore the relationship between nOH and other prodromal features (hyposmia, depression, constipation, and cognitive impairment), contingency tables were analyzed using Fisher's exact test. For descriptive purposes, baseline NfL levels were compared among iRBD phenoconverters (PD, DLB, and MSA). A p-value < 0.05 was considered statistically significant.
Results
A total of 178 participants were included: 54 with iRBD, 54 with PD, 54 age-matched HC, and 16 with MSA. Although the MSA group was smaller, it was analyzed alongside the other cohorts to reflect the full clinical continuum of α-synucleinopathies. Despite a significant overall difference in age across groups, post hoc analyses showed no significant pairwise differences, indicating that age distribution was largely comparable. Demographic and clinical characteristics are reported in Table 1.
Demographic, clinical, and biochemical characteristics of HC, iRBD, PD, and MSA groups.
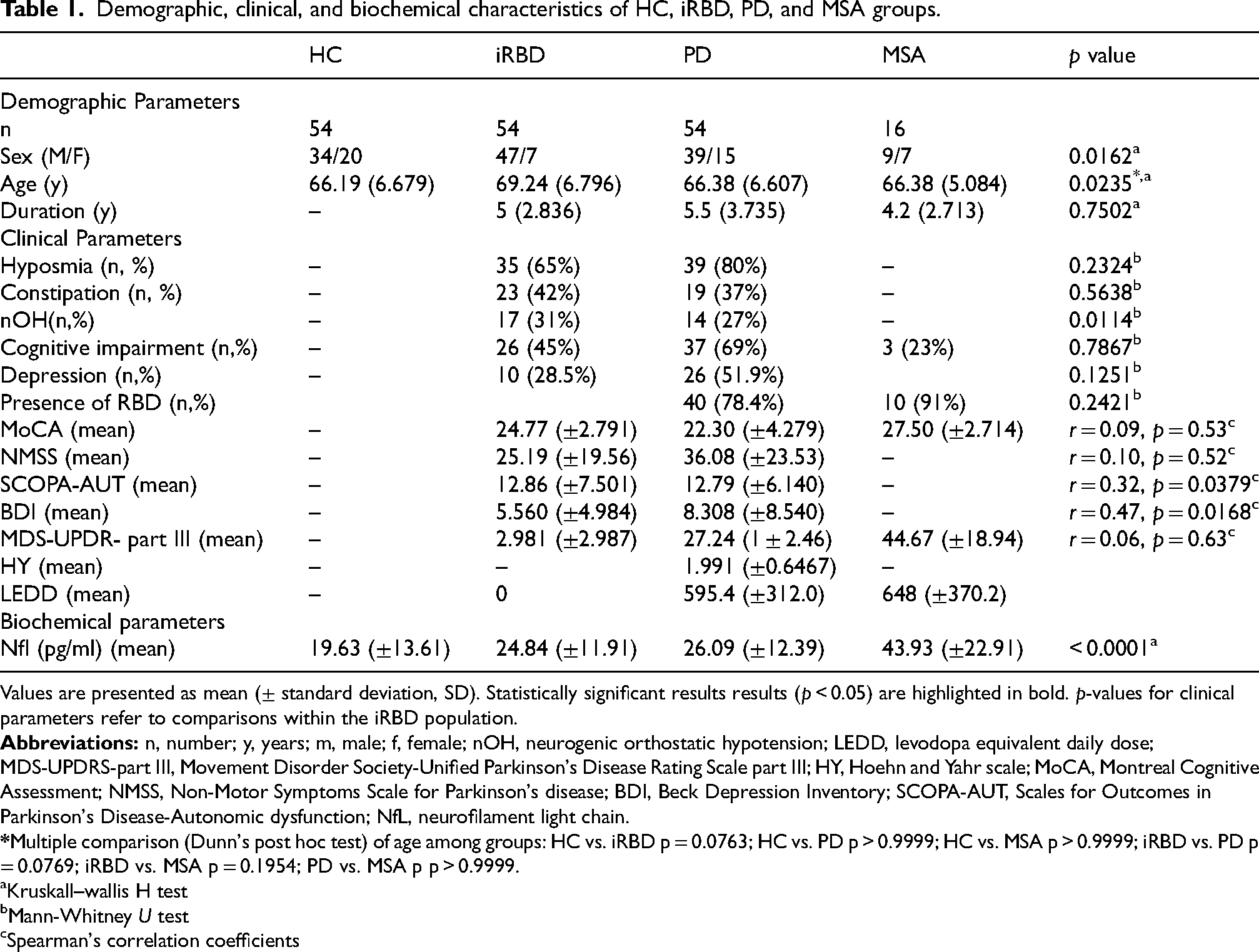
Values are presented as mean (± standard deviation, SD). Statistically significant results results (p < 0.05) are highlighted in bold. p-values for clinical parameters refer to comparisons within the iRBD population.
Kruskall–wallis H test
Mann-Whitney U test
Spearman's correlation coefficients
NfL across diagnostic groups
Plasma NfL concentrations differed significantly across the four diagnostic groups (Kruskal–Wallis p < 0.0001). Post hoc comparisons revealed that NfL levels were significantly higher in iRBD subjects compared to HC (p = 0.0181), and significantly lower than in MSA patients (p = 0.0089). PD and MSA patients also exhibited significantly elevated NfL levels compared to HC (p = 0.0021 and p < 0.0001, respectively). No significant difference was observed between iRBD and PD (p > 0.9999), while MSA patients showed significantly higher levels than those with PD (p = 0.0340). Mean plasma NfL levels were: HC 19.63 ± 13.61 pg/mL, consistent with previously reported values in the literature 16 ; iRBD 24.84 ± 11.91 pg/mL; PD 26.09 ± 12.39 pg/mL; MSA 43.93 ± 22.91 pg/mL. (Table 1, Figure 1, ST-1)
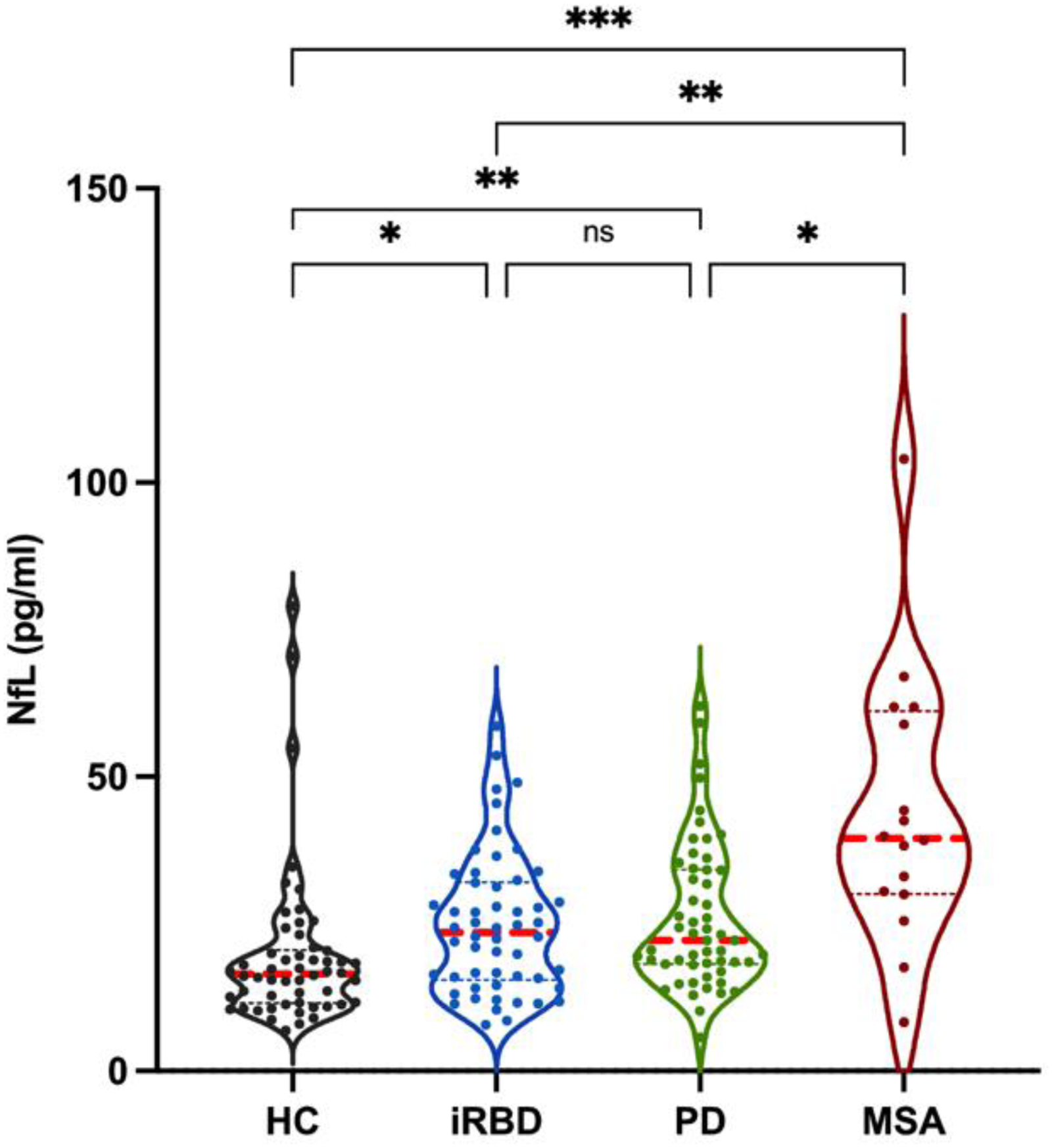
Violin plot showing plasma NfL levels across diagnostic groups.
NfL in iRBD: association with autonomic dysfunction
Among the iRBD cohort, 17 (31%) subjects were classified as having nOH (iRBD-nOH+), consistent with previous prevalence studies, 17 while 37 (69%) did not (iRBD-nOH−). Plasma NfL levels were significantly higher in iRBD-nOH+ compared to iRBD-nOH− patients (p = 0.0114).
Stratified comparisons using Dunn's test showed that: iRBD-nOH+ patients had significantly higher NfL levels than healthy controls (p = 0.0015), and did not differ significantly from PD or MSA patients (p > 0.9999 for both). iRBD-nOH− patients had significantly lower NfL levels than MSA patients (p = 0.0014), with no significant difference from either PD (p = 0.0990) or healthy controls (p = 0.2247) (Figure 2, Table 2).
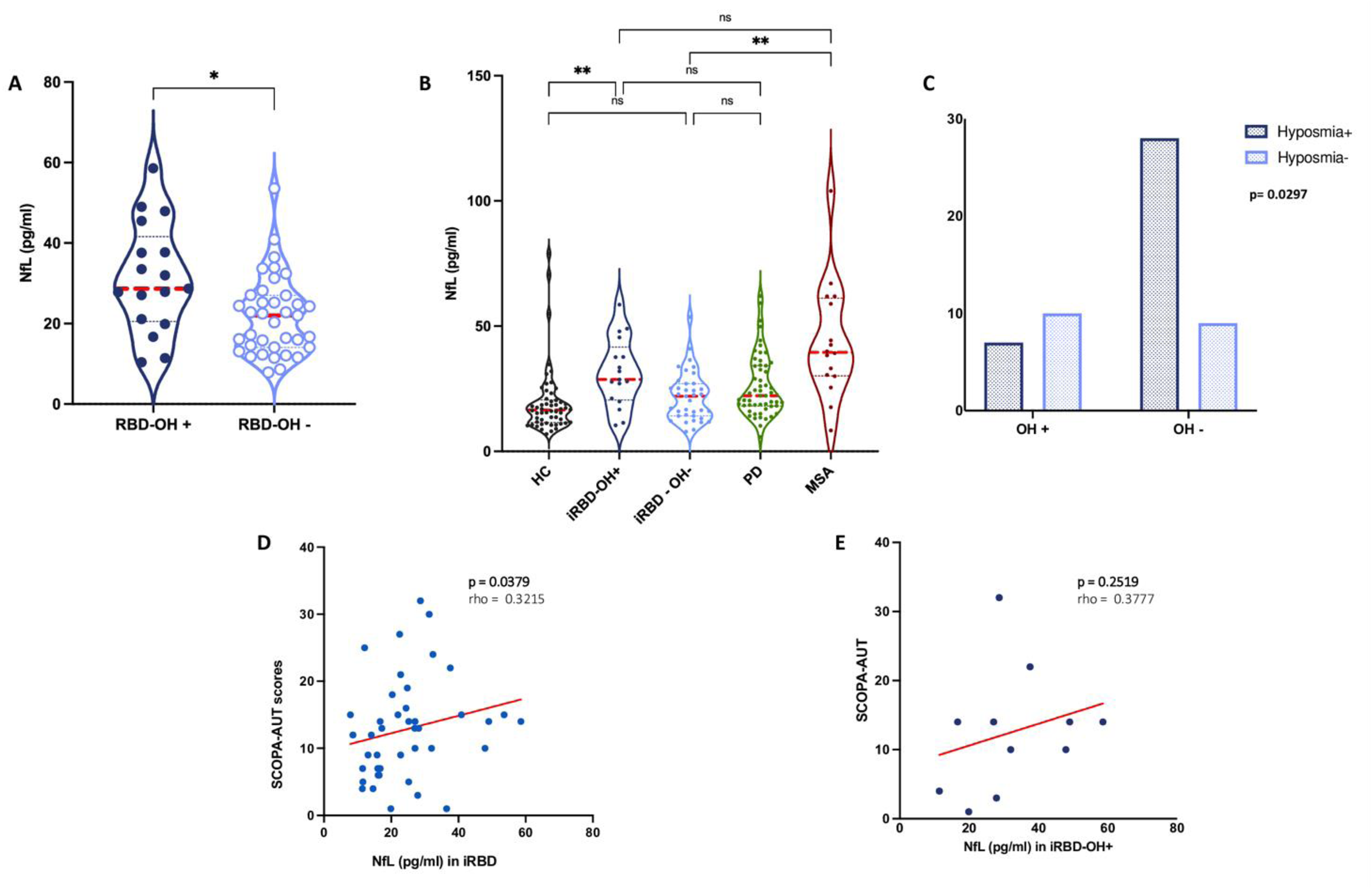
Association between plasma NfL levels and autonomic dysfunction in the iRBD cohort. (A) Violin plot showing plasma NfL levels in iRBD patients with and without orthostatic hypotension (nOH) (p = 0.0114). (B) Plasma NfL levels in iRBD-nOH+ and iRBD-nOH−compared to HC, PD, MSA. (C) Contingency analysis (Fisher's exact test) showing an inverse association between nOH and hyposmia in iRBD patients. (D) Scatter plot illustrating the correlation between plasma NfL and SCOPA-AUT scores in the overall iRBD cohort (r = 0.3215, p = 0.0379). (E) Correlation between plasma NfL and SCOPA-AUT scores in the iRBD-nOH+ subgroup (r = 0.3777, p = 0.2519).
Demographic, clinical, and biochemical characteristics of iRBD-nOH+ and iRBD-nOH− groups. Values are expressed as mean ± standard deviation (SD). Statistically significant results (p < 0.05) are shown in bold.

Mann-Whitney U test
Fisher's exact test
Contingency analysis using Fisher's exact test revealed a significant inverse association between nOH and hyposmia (p = 0.0297): only 41.2% of iRBD-nOH+ patients exhibited hyposmia, compared to 75.7% in the nOH− group. (Figure 2, supplementary Table 2). No significant associations were found between nOH and constipation (p = 0.7694), cognitive impairment (p = 0.2981), or depressive symptoms (p = 0.0752) (Table 2, SF-1).
NfL in iRBD: relationship with clinical features
Within the iRBD group, plasma NfL levels did not differ significantly based on the presence of hyposmia (p = 0.23), cognitive impairment (MoCA < 26; p = 0.78), depressive symptoms (BDI > 7; p = 0.12) or constipation (NMSS item 6; p = 0.56). No significant correlations were found between NfL levels and MDS-UPDRS-III scores (r = 0.06, p = 0.63), MoCA scores (r = 0.09, p = 0.53) or NMSS total scores (r = 0.10, p = 0.52). Conversely, NfL levels positively correlated with BDI scores (r = 0.47, p = 0.01) and with SCOPA-AUT scores (r = 0.32, p = 0.0379). Notably, this latter correlation was not confirmed when the analysis was restricted to the iRBD-nOH+ subgroup (r = 0.37, p = 0.2519) (Table 1, SF-1).
There was no significant association between NfL levels and disease duration (p = 0.70)
NfL in pd: impact of comorbid RBD
In the PD group, the presence of comorbid RBD was not associated with differences in NfL levels (p = 0.24). (SF-2).
Phenoconversion in iRBD: clinical and biological insights
At the most recent follow-up (mean 3.5 years, range 1–6 years), 11 (27%) iRBD patients had phenoconverted: 3 to PD, 4 to DLB, and 4 to MSA. Phenoconversion to overt α-synucleinopathy was determined after at least one year of documented parkinsonism, based on clinical evaluation consistent with established diagnostic criteria: the MDS clinical diagnostic criteria for Parkinson's disease, the 2017 consensus criteria for dementia with Lewy bodies, and the 2022 MDS criteria for clinically established multiple system atrophy.18–20 All MSA converters had nOH at baseline. Additionally, nOH was present at baseline in one PD and one DLB converters. Although inferential statistics were limited due to small sample size, baseline NfL levels in MSA converters were descriptively higher (mean 34.2 ± 18.09 pg/mL) than in non-converters (mean 24.34 ± 10.02 pg/mL,) (Kruskal–Wallis overall p = 0.37; post hoc MSA-converters vs non-converters p = 0.94). A direct comparison between all converters and non-converters also showed no significant difference (Mann–Whitney p = 0.8110). (Figure 3). ROC analysis was not included due to the limited number of conversion events.
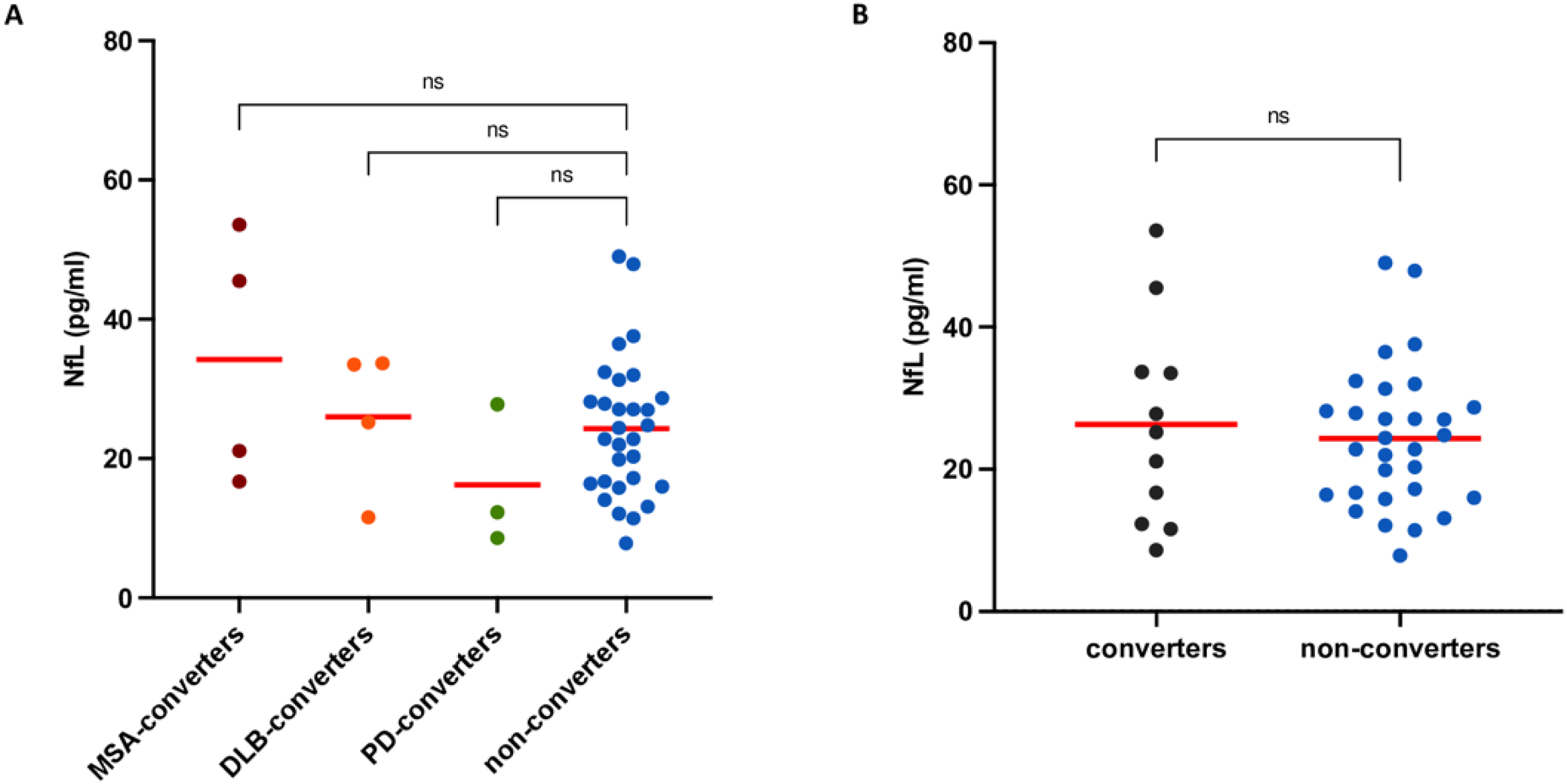
Baseline plasma NfL levels in iRBD converters and non-converters. (A) Scatter plot showing baseline plasma NfL concentrations in iRBD patients who later converted to MSA, DLB, or PD, compared to non-converters. MSA converters had descriptively higher NfL levels, though differences were not statistically significant (Kruskal–Wallis p = 0.3712; MSA vs. non-converters p = 0.9445; DLB vs. non-converters p > 0.9999; PD vs. non-converters p = 0.6363). (B) Scatter plot comparing baseline NfL levels between all converters and non-converters (Mann–Whitney p = 0.8110).
Discussion
In the present study we observed significantly elevated NfL levels in iRBD subjects compared to HC, this finding is consistent with previous studies.11,12 Of note, NfL concentrations in iRBD were comparable to those observed in PD, reinforcing the notion that neurodegenerative processes are already underway in the prodromal phase, despite the absence of overt motor symptoms. As expected, PD and MSA patients exhibited higher NfL levels than controls, with MSA showing the highest concentrations, consistent with its rapid progression and widespread neuronal loss.3,9 Importantly, a key finding of this study was the association between nOH and plasma NfL within the iRBD group. Patients with nOH (iRBD-nOH+) showed significantly higher NfL levels than those without (iRBD-nOH−), suggesting that autonomic dysfunction may reflect earlier axonal damage even in the absence of manifest parkinsonism. This aligns with prior findings in PD, where OH has been associated with worse prognosis and faster disease progression.8,11 In our cohort, iRBD-nOH+ patients had NfL values comparable to PD and MSA, whereas iRBD-nOH− had significantly lower values than MSA. This finding indicates that iRBD-nOH+ patients already exhibit a degree of neuroaxonal injury comparable to overt α-synucleinopathies.
Evidence from pure autonomic failure (PAF) further supports a possible link between autonomic dysfunction and accelerated neurodegeneration: in a recent study, baseline NfL levels were higher in patients with PAF who subsequently converted to MSA, compared to those who remained stable. 21 While speculative, these parallels suggest that the coexistence of autonomic dysfunction and elevated NfL in iRBD may represent an early biological profile associated with a more aggressive disease trajectory, potentially aligning with an MSA-like pathway.
Additional support for phenotypic heterogeneity comes from the observed inverse association between nOH and hyposmia. Whereas hyposmia is commonly linked to a PD/DLB trajectory, nOH was more frequent among patients without olfactory dysfunction, suggesting the coexistence of distinct autonomic and sensory profiles within the iRBD spectrum. 22 Taken together, these findings are consistent with the emerging concept of early phenotypic divergence in iRBD, possibly reflecting different pathways of neurodegeneration.
Notably, although plasma NfL correlated with SCOPA-AUT scores across the entire iRBD group, this association was not retained when analyses were limited to the nOH+ subgroup, suggesting that NfL elevation in this group may indicate the presence - but not the severity - of autonomic dysfunction.
No significant differences in NfL were observed when stratifying the iRBD cohort by cognitive impairment, constipation, hyposmia, or depressive symptoms. However, a moderate correlation with BDI scores was identified, raising the possibility of an association between depressive symptoms and underlying neurodegeneration, though this remains speculative.
In the PD cohort, the presence of comorbid RBD was not associated with differences in plasma NfL, consistent with previous studies indicating that RBD reflects broader α-synuclein pathology but does not necessarily entail greater axonal injury.9,23
Lastly, at longitudinal follow-up, all MSA converters had nOH at baseline and showed higher baseline NfL levels than non-converters, although the difference was not statistically significant. While the limited number of conversion events precluded robust predictive modeling, this trend may suggest that combining NfL with autonomic markers could improve stratification of iRBD patients at risk for faster progression, a possibility that warrants confirmation in larger longitudinal cohorts.
Although these findings support NfL as a reliable biomarker, we acknowledge several limitations of our study. First, the relatively small MSA group and the absence of a dedicated DLB cohort limit the representativeness of the full α-synucleinopathy spectrum, and the limited number of conversion events further reduced the statistical power for longitudinal inference. In addition, sex distribution differed across groups, with a higher proportion of males in the iRBD cohort, consistent with the known male predominance of the disorder. 24 Although the prevalence of neurogenic orthostatic hypotension was comparable between men and women - and the mean age of the cohort falls within the range where no sex-related differences in nOH prevalence are expected - the influence of sex on plasma NfL levels remains debated, although generally considered modest.9,25–27 Other physical covariates, such as BMI and hypertension, were not systematically available and should be considered in future studies. Furthermore, 13 iRBD subjects were lost to follow-up, preventing full ascertainment of phenoconversion. Olfactory function was assessed through self-reported measures, which are less accurate than standardized tools such as the UPSIT. Finally, the absence of α-synuclein seeding assays (e.g., RT-QuIC) limited our ability to biologically characterize potential PD/DLB-like versus MSA-like converters. Future studies integrating longitudinal biomarker trajectories with multimodal diagnostics, including SAA and imaging, will be essential to validate and extend these findings.
Conclusion
Our findings reinforce plasma NfL as a sensitive biomarker of early axonal damage in iRBD. Elevated NfL levels - particularly among patients with neurogenic orthostatic hypotension - suggest a more aggressive neurodegenerative profile and point toward underlying phenotypic heterogeneity within the α-synucleinopathy spectrum. These results underscore the potential of NfL to support individualized risk stratification and guide early disease-modifying interventions. Future longitudinal studies integrating NfL with clinical, imaging, and seeding-based biomarkers will be essential to validate these findings and to to clarify its role in the prodromal phase of synucleinopathies.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X261418988 - Supplemental material for Elevated plasma neurofilament light chain in isolated REM sleep behavior disorder: Associations with neurogenic orthostatic hypotension and implications for phenoconversion
Supplemental material, sj-docx-1-pkn-10.1177_1877718X261418988 for Elevated plasma neurofilament light chain in isolated REM sleep behavior disorder: Associations with neurogenic orthostatic hypotension and implications for phenoconversion by Alessandra Calculli, Deborah Di Martino, Piergiorgio Grillo, Gabriele Bellini, Stella Gagliardi, Elena Capriglia, Carlo Fazio, Davide Comolli, Gaetano Malomo, Simone Regalbuto, Michelangelo Maestri, Domeniko Hoxhaj, Enrica Bonanni, Roberto Ceravolo, Silvia Cerri, Antonio Pisani and Michele Terzaghi in Journal of Parkinson's Disease
Footnotes
Authors’ Roles
(1) Research project: A. Conception, B. Organization, C. Execution; (2) Statistical Analysis: A. Design, B. Execution, C. Review and Critique; (3) Manuscript: A. Writing of the First Draft, B. Review and Critique.
A.C: 1B, 1C, 2A, 2B, 2C, 3A, 3B D.DM: 1B, 1C, 3B P.G: 1B, 2C, 3B G.B: 2C, 3B S.G: 1C, 2C E.C: 1C, 3B C.F: 3B D.C: 3B G.M: 1C, 3B S.R: 3B M.M.: 2C, 3B D.H. 2C, 3B E.B: 3B R.C: 3B S.C: 1B, 3B A.P: 1A, 2C, 3B M.T: 1A, 1B, 2C, 3B
Acknowledgment
#NEXTGENERATIONEU (NGEU) and funded by the Ministry of University and Research (MUR), National Recovery and Resilience Plan (NRRP), project MNESYS (PE0000006) – A Multiscale integrated approach to the study of the nervous system in health and disease (DN. 1553 11.10.2022) “Fondazione Regionale per la Ricerca Biomedica (Regione Lombardia), project ID 3433068 – Acronym: LINKING PARK”.
Ethical considerations
The study was approved by the Comitato Etico Pavia at IRCCS Fondazione Istituto Neurologico C.Mondino (Protocol No.20210008499, Approval Date: 07/01/2021). All procedures were conducted in accordance with the Declaration of Helsinki and relevant institutional guidelines.
Consent to participate
All participants provided written informed consent to participate in the study, copies are available from the corresponding author upon reasonable request.
Consent for publication
Not applicable
Funding
Work was partially supported by:#NEXTGENERATIONEU (NGEU) and funded by the Ministry of University and Research (MUR), National Recovery and Resilience Plan (NRRP), project MNESYS (PE0000006) – A Multiscale integrated approach to the study of the nervous system in health and disease (DN. 1553 11.10.2022) “Fondazione Regionale per la Ricerca Biomedica (Regione Lombardia), project ID 3433068 – Acronym: LINKING PARK”.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
