Abstract
Deep brain stimulation (DBS) can improve Parkinson's disease symptoms; however, its effectiveness depends on selecting optimal settings. DBS parameter selection can be challenging, as objective metrics to guide the process are lacking. This n-of-1 study explored using functional near-infrared spectroscopy (fNIRS) to guide DBS programming. Led by the participant, the study embedded a patient perspective at the center of the project. Multiple DBS settings were tested, and their effects on gait and cortical functional connectivity were measured. The DBS program that best supported gait had the lowest functional connectivity with the left dorsolateral prefrontal cortex as seed. This suggests fNIRS may be used to guide individual optimization of DBS treatment.
Plain language summary
This study involved one person with Parkinson's who led the research to explore how brain imaging might help neurologists to choose deep brain stimulation (DBS) settings. People with Parkinson's often need to think actively to move. This thinking for movement can be seen in surface brain activity. Our study found that the DBS program that best supported walking showed the least functional connectivity between brain areas involved in thinking about movement. This is important, because there are currently no clinical tools to adequately guide DBS programming for gait, and such imaging could help neurologists select more effective DBS settings.
Dear Editor,
Parkinson's disease (PD) is a neurodegenerative disorder with heterogeneous, progressive symptomatology. 1 While deep brain stimulation (DBS) can be an effective PD therapy, individual tailoring of DBS settings relies on real-time clinical assessments, which can be complicated by delays until symptoms are impacted by changed DBS settings. 2
This patient-led n-of-1 PD-DBS study, undertaken at Queen's University, Ontario, benefited from the patient's insights being embedded within the project team. The patient was diagnosed with Parkinson's in 2010 at age 40 years, with bradykinesia and predominantly right-sided rigidity, and had DBS implanted in the subthalamic nucleus (STN) in 2020. 3 The first report from the present study described the impact of DBS frequency on symptoms and found that DBS frequency led to objectively measured alterations in gait, speech and sensorimotor performance. 4 The patient had also noted that DBS settings less supportive of his gait led to an inability to engage in conversation while walking. We hypothesized that changing DBS settings may result in differences in functional connectivity between regions of the brain responsible for concentration and motor control.
To investigate this hypothesis, we used functional near-infrared spectroscopy (fNIRS) to measure resting-state functional connectivity. After the initial experiment in May 2024, a second five-day experiment was conducted in October 2024. This second experiment allowed us to repeat measurements and vary the order and timing of DBS program tests.
A markerless motion capture system (Theia3D, Theia Markerless Inc., Kingston, ON, Canada) was employed to record movement. 5 Experiment 1 gait protocol involved 5 min walking briskly (1.7 m/s) on a treadmill. Measurements were successfully taken for five DBS programs during one day. Experiment 2 employed the Timed Up and Go (TUG) test to assess gait under three DBS programs. Each program included 12 TUG trials, half of which incorporated a verbal fluency task for the duration of each trial to impose a moderate cognitive load. Arm-swing analysis was chosen to determine the effectiveness of DBS settings to support gait. 6 While stride length, speed and cadence provide a set of typical measurements for gait, the participant reported that inter- and intra-arm variation in arm swing provided a more meaningful assessment of the effort he needed to make to co-ordinate walking.
An fNIRS device (NIRSport 2, NIRx Medizintechnik GmbH; 16 sources, 16 detectors; 54 channels including 8 short channels) was used to assess resting-state functional connectivity. The montage covered the frontal lobe, and some parietal and temporal lobe areas. Measurements lasted 10 and 12 min for Experiments 1 and 2, respectively, with the participant seated in a quiet, dimly-lit room, with eyes closed. The cap was placed using the standardized 10–20 landmarks method. Data processing followed established protocols.7,8 Short-channel spectra were produced using Welch's method, selecting those for further analysis that had clear heart-rate–frequency peaks (∼1 Hz). Sampling frequency was 5.086 Hz. An age-adjusted differential pathlength factor was applied. 9 One minute was trimmed from each end to reduce edge effects. Optical density was computed, followed by spline and wavelet corrections to remove baseline shifts and motion artifacts, then converted to concentration values. The spline parameter was optimized per session by varying the value (2–4, in 0.5 increments) and visually inspecting baseline shift removal. After detrending and band-pass filtering (0.009–0.08 Hz), 100 samples were discarded to reduce border effects. Physiological noise was removed via general linear model regression with short-channel regression, cross-validation, and pre-whitening. 10 Correlation matrices were computed and total hemoglobin correlations averaged. Connectivity patterns were visualized with 3D projections.
Across the two experiments, the patient was examined on fifteen periods involving nine different DBS programs (Supplementary Table S1). Detailed arm-swing assessments from Experiment 1 demonstrated Program #1 provided better gait support than the four alternative DBS programs assessed during that stage. 4 For Experiment 2, the TUG tests with a verbal task for Program #1 had mean left-arm/right-arm swing ratio of. 1.0 (i.e., symmetrical), whereas Program #4 yielded a ratio of 1.7 (Supplementary Table S2). Consistent with Experiment 1, in Experiment 2, the participant clearly had difficulty walking on Program #2, and Program #1 was the most supportive for gait of the DBS programs assessed.
fNIRS recordings were attempted during a total of fifteen resting-state periods. Three periods were excluded due to failure to complete the session and two due to sub-standard quality of short channels; ten were considered valid for analysis, involving six of the nine DBS programs. For each of these ten sessions, three sets of resting-state functional connectivity total hemoglobin Pearson correlation coefficients (r) were calculated: (1) using the left dorsolateral prefrontal cortex (DLPFC) as seed, (2) right DLPFC as seed, and (3) no seed (Supplementary Table S3). The left DLPFC seed provided a clearly delineated separation of correlation coefficients relative to gait quality and was highly replicable, with Program #1 consistently having the lowest functional connectivity (Figure 1(b)). Visual inspection of the image maps highlighted that the increased correlation relative to Program #1 was primarily due to increased activity with the motor cortex (MC) (Figure 1(a)). The exception was Program #4, Experiment 2. That result (r = 0.37) was unusually high and did not appear to be driven solely by left DLPFC-MC functional connectivity; observational notes indicated some participant distraction during that session, which may have contributed to the observed discrepancy. This value was excluded from the analysis. From the three other DBS programs assessed, Program #7 had stimulation to the distal contacts turned off, and had the highest functional connectivity (r = 0.26).
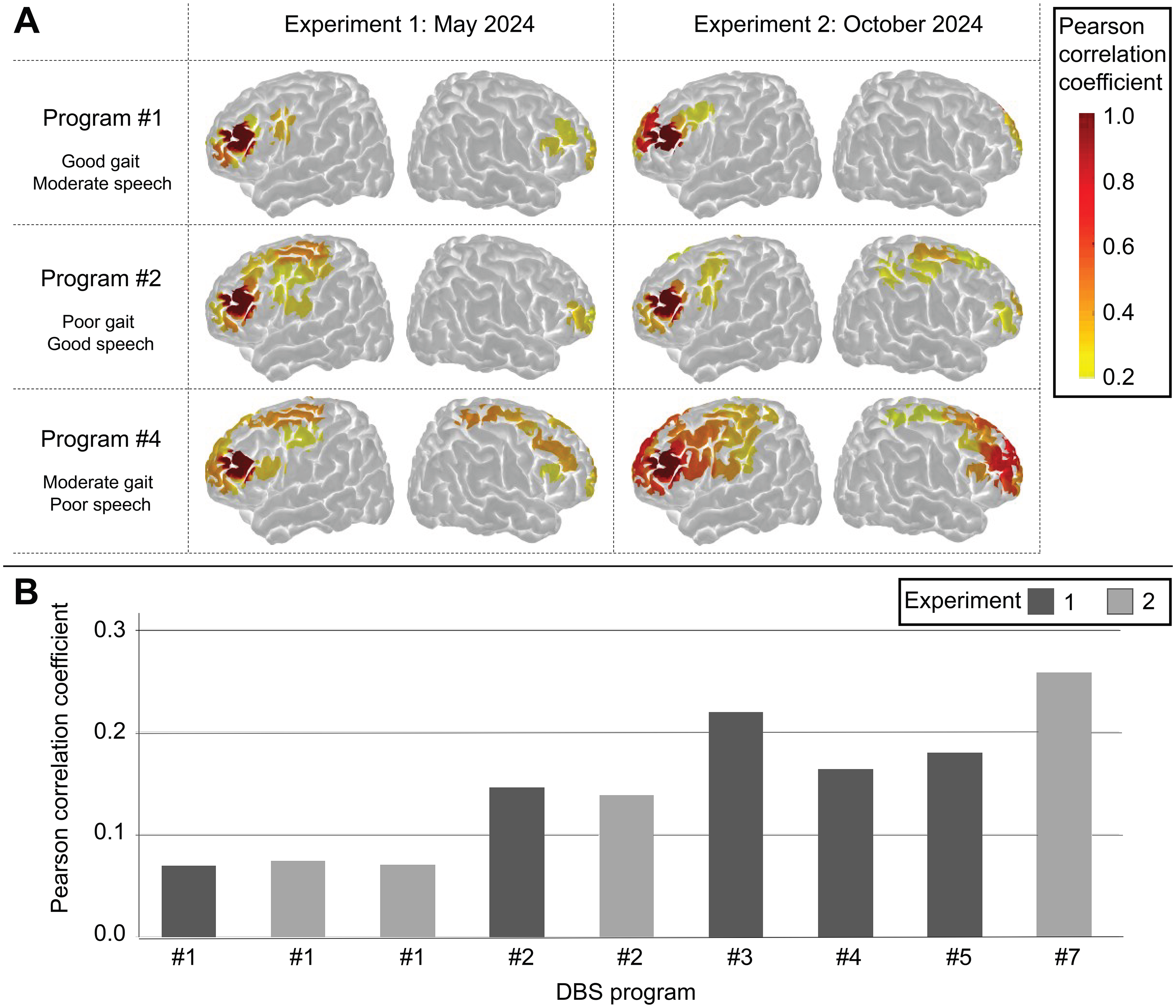
Resting-state functional connectivity. Measurements were taken from a participant with Parkinson's disease treated with deep brain stimulation (DBS) of the subthalamic nucleus on a selection of DBS programs (Supplementary Table S1). Panel A: Images of brain outer cortex generated using a functional near-infrared spectrograph, with the left dorsolateral prefrontal cortex as the seed. The three DBS Programs shown were those successfully imaged in both experiments. Panel B: The functional connectivity correlation coefficients for the six DBS programs successfully measured. Each bar represents the mean functional connectivity for an individual session.
Our primary finding was that the DBS program that most effectively supported gait consistently exhibited the lowest mean functional connectivity with the left DLPFC as the seed. This finding was consistent with our hypothesis.
The DLPFC, particularly in the left hemisphere, has been associated with an important compensatory role in gait dysfunction in PD, especially under dual-task conditions. Elevated DLPFC activity and functional connectivity with motor regions reflect increased executive demands and reduced gait automaticity in PD.11–13 STN-DBS indirectly modulates cortical regions, including the DLPFC, by altering effective connectivity within basal ganglia–thalamocortical loops.14,15 Our observed association between reduced left DLPFC–motor cortex connectivity and improved gait with optimal DBS settings supports the notion that effective DBS may restore motor automaticity by reducing maladaptive prefrontal overactivation.
We employed an n-of-1 approach to explore whether a potentially useful fNIRS signal could be detected. While the findings are promising, they may not be generalizable. A next step could involve a small study to confirm the signal in multiple patients, followed by a clinical trial to test whether this technique can be used to improve gait outcomes by guiding DBS programming. Such a trial, possibly including a healthy control group, would be better facilitated by an easy-to-use, real-time and ideally open-source fNIRS data pipeline. We recommend a technology development project to create such a pipeline. Even with technological advances, fNIRS would likely remain a specialized tool offered in dedicated DBS clinics for patients with refractory gait issues post-DBS.
In conclusion, left DLPFC-MC resting-state functional connectivity may provide a useful biomarker when selecting DBS parameters to support gait. This is important because DBS sometimes leaves individuals with gait disability. For fNIRS to function in clinical practice, an automated pipeline for quantification of functional connectivity could be developed to help guide setting optimization. Replication beyond this n-of-1 study to other DBS patients is required to assess the generalizability of this approach. The participant's leadership of this study was key to drawing this conclusion.
Supplemental Material
sj-xlsx-1-pkn-10.1177_1877718X251381454 - Supplemental material for Parkinson's disease participant-led research: Towards neuroimaging-assisted deep brain stimulation programming
Supplemental material, sj-xlsx-1-pkn-10.1177_1877718X251381454 for Parkinson's disease participant-led research: Towards neuroimaging-assisted deep brain stimulation programming by Mark D McAuley, Alena A Ionova, Andreas Horn, Ethan Heming, Celestina A Onabajo, Madison M Solie, Sara A Stephenson and Susan E Boehnke in Journal of Parkinson's Disease
Footnotes
Acknowledgements
The authors would like to thank Doctors Richard Blazé (Australia) and Jennifer Sharma (Canada), neurologists who cared for the participant, and Matthew Jacobs, Sophie DeCoste, Abigail Ball, Khoi Tran, Roy Bleicher and Kerlas Samaan, for their contributions, and Androu Abdalmalak for his expert guidance of fNIRS.
ORCID iDs
Ethical considerations
This study was approved by the Queen's University's Health Sciences Research Ethics Board (approval no. 6041027) on May 10, 2024.
Consent to participate
Informed consent for participation was obtained on May 14, 2024.
Consent for publication
Informed consent for information published in this article was not separately obtained because the first author is the sole participant in the study.
Authors contributions
Mark D. McAuley: Conceptualization, Methodology, Formal analysis, Writing – original draft, Writing – review & editing. Alena A. Ionova: Formal analysis, Writing – original draft, Writing – review & editing. Andreas Horn: Methodology, Writing – review & editing. Ethan Heming: Project administration, Writing – review & editing. Celestina A. Onabajo: Formal analysis, Writing – review & editing. Madison M. Solie: Formal analysis, Writing – review & editing. Sara A. Stephenson: Formal analysis, Writing – review & editing. Susan E. Boehnke: Supervision, Methodology, Resources, Writing – review & editing.
Funding
The authors disclose receipt of the following financial support for the research, authorship, and/or publication of this article: AH was supported by the Schilling Foundation, the German Research Foundation (Deutsche Forschungsgemeinschaft, 424778381 – TRR 295), Deutsches Zentrum für Luft- und Raumfahrt (DynaSti grant within the EU Joint Programme Neurodegenerative Disease Research, JPND), the National Institutes of Health (R01MH130666, 1R01NS127892-01, 2R01 MH113929 & UM1NS132358) as well as the New Venture Fund (FFOR Seed Grant). This study was conducted as part of the Capstone Project Course of the Neurotech Microcredential Program, for which SB received funding from the Province of Ontario. SB also receives funding from the Connected Minds Program, supported by Canada First Research Excellence Fund, Grant #CFREF-2022-00010.
Declaration of Conflicting Interests
AH reports lecture fees for Boston Scientific, is a consultant for Modulight.bio, was a consultant for FxNeuromodulation and Abbott in recent years and serves as a co-inventor on a patent granted to Charité University Medicine Berlin that covers multisymptom DBS fiberfiltering and an automated DBS parameter suggestion algorithm unrelated to this work (patent #LU103178). All other authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting the findings of this study are available on request from the senior author (SB). The data are not publicly available due to privacy considerations.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
