Abstract
Parkinson's disease (PD) is a rapidly developing neurodegenerative disorder characterized by degeneration of dopaminergic neurons in the basal ganglia of the brain. Its prevalence is estimated to exceed 1.2 million cases in the United States by 2030. Emerging evidence suggests that PD may originate in the gut and hence is linked to gastrointestinal (GI) dysfunctions such as inflammatory bowel disease and GI cancers. Colorectal cancer (CRC) is one of the most common cancers seen worldwide. It shares several risk factors with PD, including advancing age, male gender, genetic predispositions, and environmental exposures. Although these shared factors indicate a possible correlation, the studies evaluating this relationship have suggested inconsistent results. Some research proposes an increased risk of CRC in PD patients, potentially due to overlapping genetic and inflammatory pathways. Alternatively, others argue an inverse relationship due to opposing underlying mechanisms of neuro degeneration in PD and cellular proliferation in CRC. This narrative review explores the intricate relationship between PD and CRC and seeks to understand how neurodegenerative and malignant diseases may overlap.
Plain language summary
Parkinson's disease (PD) is a brain disorder that mainly affects movement by damaging the brain cells that produce dopamine. It is one of the fastest-growing neurological conditions, and experts expect more than 1.2 million people in the United States to have PD by 2030. Colorectal cancer (CRC), often called colon cancer, is one of the most common cancers worldwide. At first glance, these two conditions may not seem related. However, new research suggests there may be important connections between them. Both PD and CRC share common risk factors such as older age, being male, certain inherited genetic changes, and environmental exposures like pesticides. Some studies suggest people with PD may have a higher risk of developing CRC because of overlapping genetic changes, gut inflammation, or imbalances in gut bacteria. Other studies suggest the opposite, that PD may actually lower the risk of CRC. This may be due to the protective effects of medications used in PD or because the processes that cause brain cells to die in PD may counteract the uncontrolled cell growth that drives cancer. Because the findings are mixed, researchers still do not know for certain whether PD increases or decreases the risk of colon cancer. What is clear is that there are shared biological pathways—such as genetic mutations in PARK2 and LRRK2 genes, that may link the two conditions. Understanding this possible connection could have important benefits for patients. If a link is confirmed, doctors might be able to screen patients with PD more effectively for colon cancer, or even use new therapies that target the gut-brain axis with approaches such as diet changes, probiotics, or other treatments. Larger, carefully designed studies are needed to clarify this relationship and to guide prevention and treatment strategies for both PD and CRC.
Keywords
Introduction
Parkinson's disease (PD) is the second most common neurodegenerative illness worldwide with a steadily rising incidence that is expected to double every decade, making it one of the world's most rapidly developing neurological disorders. It is predicted that the number of patients affected by PD worldwide rose from 2.5 million in 1990 to 6.2 million in 2015 and further to 8.5 million in 2019, according to WHO estimates.1,2 The Parkinson Foundation estimates that currently one million people in the U.S. are living with PD and this is expected to rise to 1.2 million by 2030, likely due to the aging population. PD predominantly affects older adults, with more than 1% of people over the age of 60 and more than 5% of those above 85 diagnosed with the disease.3–5 Men are affected 1.5 times more than women, which is likely the result of the neuroprotective effects of estrogen.6,7 The disease is also more prevalent in the Western population than in Asians and Africans, owing to increased amounts of neuromelanin in dark-skinned individuals which is speculated to have a neuroprotective effect.8,9 Apart from age and gender, approximately 15% of PD cases have a familial inheritance pattern, pointing towards genetic influences on disease onset and progression. 10 Several monogenic variants have been identified which account for around 30% of familial and 3%–5% of sporadic cases. 11 The remaining cases of sporadic PD are thought to be the result of environmental pollutants that act as neurotoxins and cause oxidative stress and disruption of neurotransmission with detrimental effects in the basal ganglia. Various chemicals in agricultural pesticides such as 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP), dieldrin, and rotenone are implicated in increased risk of PD. Oxidative damage is also caused by heavy metals like Iron, copper, and manganese which in turn increase the risk of PD. 12 The disease is characterized by alpha-synuclein aggregation, a presynaptic neuronal protein that disturbs cellular homeostasis and triggers neuronal death. 13 It results in degeneration of dopaminergic neurons in substantia nigra pars compacta of basal ganglia leading to hallmark motor symptoms- tremors, bradykinesia, rigidity- causing gait and postural disturbances. 14 Recent research points towards the gut as the possible site of origin of PD and hypothesizes a gut-brain axis as the pathway for disease initiation and progression. Evidence suggests alpha-synuclein may first accumulate in the intestines, leading to non-motor symptoms such as constipation, difficulty swallowing, and psychosis. The retrograde transmission of this alpha-synuclein to the central nervous system (CNS) via the vagus nerve is expected to drive the disease in the brain. Due to genetics and environmental factors, gut dysbiosis also causes local and systemic inflammation, affecting various organs, including the brain. As a result, PD has been associated with several gastrointestinal (GI) illnesses, including irritable bowel syndrome, inflammatory bowel disease, GI infections, and even GI neoplasms. 15 The strong evidence linking PD to GI dysfunction has raised speculations about how PD can affect the risk of colorectal cancer (CRC). CRC is one of the most common cancers, with an anticipated global incidence of 1.9 million and 0.9 million deaths in 2020. It is ranked as the third most frequent cancer in males and the second most common in women. 16 Most of the cases are seen in developed countries but a sharp rising trend is visible in developing countries of Asia and Africa, making it a global health issue. 17 The Centers for Disease Control and Prevention estimates that the United States reported 141,902 new cases in 2021 with 52,967 deaths in 2022, making CRC one of the most common malignancies and the second most common cause of cancer-associated deaths. 18 The global estimate of incidence in 2020 was 547,619 and 288,852 cases of colon and rectum cancer in females, respectively, and 600, 896, and 443,358 cases of colon and rectum cancer in males, which indicates a greater incidence in males as compared to females. 19 The median age of CRC is 64 years, and approximately 90% of patients are older than 50 years. 20 While about 75% of CRC cases lack a genetic predisposition, the remaining cases are associated with genetic pathways such as chromosomal instability, microsatellite instability, and the CpG island methylator phenotype. 21 Specific gene mutations in PD have been linked to an increased risk of CRC. Shared risk variables, such as age, gender, and environmental effects, may imply a link between PD and CRC. However, certain research also suggests an inverse relationship between PD and CRC, arguing that the processes of neurodegeneration in PD vary from those of cellular proliferation in carcinogenesis. This review attempts to evaluate the potential relationship between PD and CRC.
Pathophysiology
PD develops due to a multitude of factors including genetics, environment, chemical alterations within the neurons, and the resulting immune system activation causing cellular dysregulation and eventual degeneration. Each of these factors plays a key role in ultimately destroying the cellular architecture within a part of the brain called substantia nigra where nerve cells that release dopamine neurotransmitters are permanently affected. Dopamine is a neurotransmitter released in deep brain structures like the basal ganglia which either increases or decreases movements. The inhibition and subsequent destruction of cells that produce dopamine is the primary reason why patients develop PD.
Genetics
A small percentage of PD cases (10–15%) indicate a family history, with around 5% showing Mendelian inheritance, although PD is typically idiopathic. Additionally, various polygenic risk factors, though not yet well-defined, also play a role in an individual's risk of developing PD. PD is associated with 23 “PARK” genes, which are listed in the order that they were discovered. Mutations in these PARK genes follow either autosomal recessive inheritance patterns (e.g., PRKN, PINK1, and DJ-1) or autosomal dominant inheritance patterns (e.g., SCNA, LRRK2, and VPS32). 22
Environmental agents
Compelling evidence suggests that exposure to environmental hazards like pesticides, herbicides, and nearby industrial plantations can directly alter dopamine levels. 23 Insecticides such as chlorpyrifos and organochlorines, and herbicides (like paraquat) are commonly implicated in the mechanism that causes neuronal proteins to fold unnaturally. 24 Misfolded proteins known more infamously as Lewy bodies, are aggregates of fibrillar proteins that comprise a major constituent called a-synuclein, a protein that clumps up within the neurons and subsequently gets passed from one neuron to the next resulting in the pathological spread of the disease. 25 Recent literature has pointed towards another substance that is found to play a key role in the development of PD, called neurotoxin MPTP following self-injection of intravenous heroin in drug abusers plays a significant role in the development of PD. MPTP naturally crosses the blood-brain barrier unchecked and once inside the brain, it gets converted to a molecule called 1-Methyl-4-phenylpyridinium (MPP+) which is a potent neurotoxin. 26 MPP + accumulates within the mitochondria of the Substantia nigral neurons increasing the oxidation reactions and generating free radicals which can significantly destroy thalamic nuclei and the permanent nerve cells that release dopamine. 23 Immune system activation further accelerates cytotoxicity, triggering microglial cell death by releasing destructive mediators such as hydroxyl radicals, superoxide radicals, nitric oxide the cerebrospinal fluid, signaling immune-mediated destruction of the substantia nigra and hence contributing to the pathogenesis of PD. 27 Inflammation of the neurons can cause cellular death in the CNS and the peripheral nervous system (PNS) which is explained by the gut-brain axis.
Gut-brain axis
Due to decreased dopaminergic synapse at the level of the GI tract regions, the resulting constipation causes alterations in the makeup of the gut microbiota. An imbalance occurs between beneficial and detrimental microbial metabolites, ensuing an inflammatory state that subsequently triggers increased gut permeability. This GI inflammation stimulates neurons inside the enteric nervous system via the Vagus nerve, transporting chemical signals from the GI tract to the brain. Therefore, any altered proteins within the neuronal components in the enteric nervous system such as alpha-synuclein could indirectly get shuttled into the brain disrupting signals within the substantia nigra. This retrograde transport of the pathological Lewy bodies from the gut to the brain is referred to as the microbiota-gut-brain axis. 15
Genetic mutations leading to CRC in PD
PD may raise the risk of CRC in several ways, including genetic predispositions in leucine-rich repeat kinase 2 (LRRK2) and procollagen type 1 intact N terminal propeptide (P1N1) neuronal components, and changes to the proteasome, an organelle that is involved in protein breakdown. These pathways have led to notable declines in the preventive mechanisms that the gut microbiota offers, particularly in individuals with PD, which has caused the etiology of CRC in a large number of patients. It has also been demonstrated that any recent or repeated antibiotic exposure also increases the risk of CRC by 20%, as does the bacterial dysbiosis that follows. 3 Park 2 gene mutation is the most common cause of early-onset PD. Park 2 gene encodes for an enzyme called E3 ubiquitin ligase and any alteration of its tumor-suppressing function will thereby result in unchecked cellular proliferation. E3 ligase ubiquitinates an enzyme called Cyclin E which is needed to keep the cellular cycle in check. E3 ligase may be indirectly impacted by any inactivating somatic mutation on the Park 2 gene, leading to mitotic instability and eventually, oncogenesis in multiple different parts of the body such as the colon causing CRC. 28 CRC risk is closely associated with genetic variants such as the a-synuclein gene (SNCA), PARKIN gene, peptidyl-prolyl isomerase pin 1 (PIN) gene, and LRRK family of genetic predispositions. CRC is also significantly associated with patients who experience chronic constipation and bacterial overgrowth from a species called Streptococcus Bovis. 3 On the contrary, genetic variants can otherwise also act as a protective barrier in the prevention of cancer. The pathways such as phosphoinositide 3 kinase (PI3K)/protein kinase B (Akt)/mechanistic target of rapamycin (mTOR) PI3K/AKT/mTOR are overactive in patients with CRC as illustrated in Figure 1. By preventing apoptosis, (mTOR) PI3K/AKT/mTOR pathways increase the survival of dopaminergic neurons and lower the risk of PD. 29 Dopamine agonists act through the D1-like receptors to alter the angiogenic properties of vascular endothelial growth factor (VEGF) thereby fully halting the blood vessel formation in tumor cells. The G-alpha subunit route is used by a complex network of G-protein-coupled receptors to activate the adenylyl cyclase enzyme, which inhibits cell proliferation and disease progression in malignancies such as those of the colon. The dopamine agonists’ anti-cancerous properties through activation of the G-alpha subunit pathway play a key role in the downstream effects that take place in the CNS and the PNS. By acting on growth factors like VEGF, these medications can both carry out their peripheral antiangiogenic properties as well as increase dopamine levels centrally, which significantly helps to lessen PD symptoms. 30
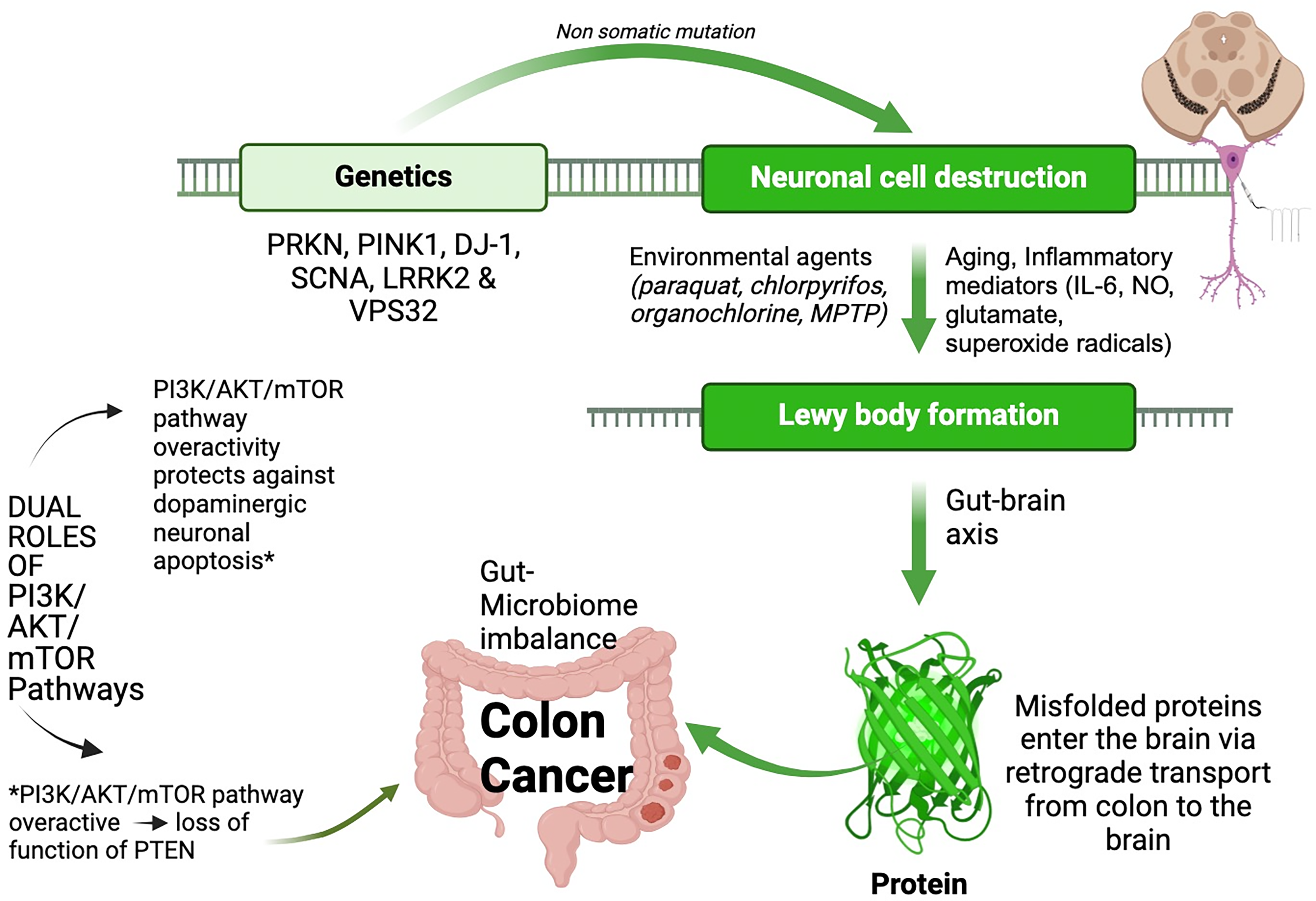
Pathophysiological connection between colorectal cancer and Parkinson's disease. Role of genes, inflammatory mediators, and environmental agents in causing CRC.
Molecular pathogenesis
The PINK1 gene plays a crucial neuro-protective role by safeguarding mitochondria from damage and preventing cellular apoptosis in response to stress. 31 When PINK1 gets mutated, it can lead to cellular proliferation and accumulation of toxic proteins such as a-synuclein and synphilin-1, consequently leading to cancer progression. Normally, this gene helps to maintain mitochondrial membrane potential and prevents the release of factors that trigger apoptosis. 32 A deficiency in PINK1 has been linked to reduced plasticity in the striatum and hippocampus, potentially leading to neuro-degeneration and cognitive decline in PD patients. 33 Additionally, more than 100 loss-of-function mutations in the PINK1 gene have been associated with early-onset, autosomal recessive PD. 34
On the other hand, a gain-of-function mutation in the oncogene called DJ-1 promotes cancer progression by activating Akt/mTOR, MEK/ERK, NF-κB, and HIFα pathways and inhibiting tumor suppressors like PTEN and p53. Overexpression of these oncogenes has been observed in PD patients who were later diagnosed with Colon cancer. 35 Similarly, point mutations in an autosomal dominant inherited gene called LRRK2 have emerged as the most prevalent variant in PD patients, causing colon cancer. This gene increases the number and size of tumors by promoting intestinal epithelial cell proliferation and inflammation, which in turn causes colon cancer. 36
Discussion
PD patients who have cancer, such as colon cancer, benefit from the neurodegeneration and cell cycle disruption brought on by upregulated a-synuclein, which also inhibits carcinogenesis. 37 We delve more into the causal relationship between how PD patients, in some cases, can be either very highly susceptible or genetically protected from mechanisms that ultimately decrease the risk of developing CRC.
A study by Fang et al. demonstrated low overall risk of developing CRC in PD patients (0.78, with a significant p value of <0.001) Additionally, this reduced risk remains consistent regardless of the tumor location, whether rectal or colonic origin (with a relative risk of 0.89)—reinforcing the overall protective association between PD and CRC. 29 The reduction in the risk of PD patients developing CRC was tested in different regions of the world, yielding interesting results for those living in the West. The American population diagnosed with PD showed significantly lower odds of CRC (OR = 0.58) in comparison to the European population (OR = 0.82). 29
According to study results concluded by Xie et al., consisting of 343,226 PD patients, analysis performed in the western population revealed a significant inverse relationship between PD and CRC (with an RR of 0.79 and a p value of 0.006). 38
Further analysis was performed in a South Korean study, which thoroughly examined this inverse relationship using an age-matched and sex-matched male population. A total of 52,009 PD patients between 2010 and 2016 were matched, revealing a significantly lower rate of cancers in different regions of the body. Organs such as the larynx (HR = 0.45), gastric region (HR = 0.72), pancreas (HR = 0.75), liver (HR = 0.80), lung (HR = 0.73), prostate (HR = 0.78), and CRC (HR = 0.65) all indicated a very low likelihood of developing cancer in a known PD patient. 39 Therefore, the findings of the Korean study are in line with those of the Western studies previously cited, indicating a globally decreased risk.
Epidemiological and Genetic research conducted by Tian et al. further supports the inverse relationship between PD and CRC. Their study overlapped 16 genes contributing to PD and CRC, with only 1 positive association between PD and CRC. However, 15 other genetic sequences also collected indicated a negative association between PD and CRC patients, highlighting an overall genetically decreased risk of CRC in patients with PD. 40 A study conducted by Holtz in 2024 indicates that there is no iatrogenic transmission of the pathogenic protein known as a-synuclein during a colonoscopy operation, highlighting a net neutral correlation between a significant GI procedure and its associated risk of developing PD. 41
Genetic forms of PD patients have often been associated with the development of several malignancies, for instance, the familial, autosomal recessive form of PD caused by germline mutations in the PARK2 gene. In a study by Veeriah et al., they performed an array comparative genomic hybridization on 98 colonic tumor samples, 24 of which (24.4%) had PARK2 copy number loss. In 100% (24 out of 24) of colon cancer samples with loss on chromosome 6q, loss of the PARK2 gene was observed within the area of copy number alterations. Unlike in neuronal dysfunction caused by germline mutations, when altered in non-neuronal somatic cells, PARK2 may contribute to oncogenesis. Tumorigenic mutations neutralize the growth-suppressive effects of the PARK2 protein. These mutations decrease PARK2's E3 ligase activity, creating mitotic instability, concluding that the PD-associated gene PARK2 is a legitimate tumor suppressor gene that is inactivated and mutated in glioblastoma multiforme, colon cancer, and lung cancer. 28
Agalliu et al. studied the cancer outcome in PD patients carrying Leucine-rich repeat kinase 2 (LRRK2) mutations, idiopathic Parkinson's disease (IPD) patients, and non-affected controls. In their primary analysis of 257 LRRK2-PD patients, 712 IPD patients, and 218 controls, the self-reported prevalence of Colon cancer in the LRRK2-PD group was 2.3%, while 0.8% in IPD and 0.9% in controls. 42 Evidence suggested an increase in colon cancer risk, although not statistically significant. Their pooled data analysis revealed a statistically significant 2.34-fold increased risk of colon cancer in LRRK2-PD patients when compared with IPD patients (p = 0.018). 43 These results corroborate previous evidence of the association of R1441G/C mutation (Type of LRRK2 mutation) with colon cancer. 44
Chen et al. also investigated the role of the PD-associated protein ATP13A2 in colon cancer progression. They found significantly higher mRNA levels of ATP13A2 in colon cancer tissue and demonstrated its role in colon cancer stemness and invasion. ATP13A2 functionally serves as a novel enhancer of stem-like characteristics (stemness) in colon cancer cells in vitro. Knockdown of ATP13A2 decreased the number of colonies formed by colon cancer cells and markers for cancer stem cells, such as SOX2 and OCT4, while overexpression increased them. Increased ATP13A2 levels also increased the cell mobility, thus increasing the invasiveness of colon cancer. ATP13A2 expression is associated with poor patient outcomes, shorter overall survival, advanced stages (Stage III and IV) of colon cancer, and lymph node metastasis. The findings suggest the potential of ATP13A2 as not only a novel prognostic biomarker for colon cancer but also a target for colon cancer therapy. 45
PD has been known to cause multiple different GI symptoms with varied frequency, like hypersalivation, dysphagia, nausea, constipation, and defecatory dysfunction. GI manifestations of PD lead to altered colonic transit time and gut microbiota. These alterations give rise to inflammatory metabolites and altered permeability of the colonic barrier, leading to an increased risk for developing Colonic neoplasms. The altered microbiota also affects intra- and extraneuronal alpha-synuclein clearance mechanisms, further increasing the risk of PD.46,47 The accumulation of alpha-synuclein in the intestinal mucosa indicates the early onset of PD in colon cancer patients 48 and implies a risk of colon cancer in individuals with PD. 49
In a large-scale nationwide cohort study by Lin et al. conducted in Taiwan (East Asian population), a population of 62,023 patients with newly diagnosed PD and without any cancer at baseline was investigated for the risk of various cancers in the PD patients. Their analysis showed a statistically significant (p < 0.001) risk of CRC among PD patients with a hazard ratio of 1.47 (95% CI,1.31–1.65). Contrary to the studies done on Western populations, Lin et al. inferred that PD is a risk factor for the majority of malignancies in Taiwan. 50
However, several studies found no discernible difference in the risk of colon cancer among those with PD. Wirdefeldt et al. in a nationwide survey of the Swedish patient population of PD found that the risk of colon cancer in PD patients is not significantly different from that in PD-free individuals. The hazard ratio (HR) for colon cancer up to one year before the PD index date is 0.75, and from one year after the PD index date, it is 0.74. Colon cancer was rather neutral, while other cancers still carried a higher risk among PD patients. Although an increased risk was noted around the time of the PD index date at most cancer sites, it was likely the effect of ascertainment bias due to increased medical attention when a person is being diagnosed with a chronic disease like PD. The study also investigated the siblings of PD patients compared to siblings of PD-free individuals, there was no significant difference in the risk of colon cancer (HR = 0.98, 95% CI: 0.81, 1.19). This finding suggests that shared familial factors (like genetics or early-life environment) may not be the primary explanation for the observed association between PD and colon cancer incidence. 51
The striking differences between studies in Asian countries and Western countries suggest the importance of ethnicity and environmental exposures in disease pathogenesis and colon cancer risk among PD patients. The data on Asian studies is still insufficient to derive any conclusive evidence. More research on the long-term risks of colon cancer in PD patients is needed in Asian countries. The existence of robust evidence in Western countries enables the development of methods for diagnosing, preventing, and enhancing the prognosis of malignancy in PD (Table 1).
Key Studies evaluating relationship of colorectal cancer and Parkinson disease.

Conclusion
The association between PD and CRC presents a complex and multifaceted landscape with conflicting evidence and research gaps. While a definitive causal relationship remains unclear, several lines of evidence suggest an intricate interplay between PD and CRC. Shared genetic predispositions, including mutations in genes such as PARK2 and LRRK2, can increase the susceptibility to both conditions. Environmental exposures, such as pesticide exposure, may also contribute to the development of both PD and CRC. The increasing incidence of PD patients in the United States and worldwide offers a larger data set for the examination of risk factors, the progression of malignancies, and the therapeutic techniques utilized to mitigate the risk of CRC. Understanding this complex interplay may have significant implications for clinical practice, disease management strategies, and the development of targeted therapies. Early identification and interventions like targeting the gut-brain axis through dietary interventions, probiotics, or other therapeutic strategies may improve patient outcomes. Addressing the common risk factors and targeting the putative underlying pathophysiological mechanisms will be essential for establishing effective preventative and therapeutic treatments, ultimately enhancing the management and prognosis of both PD and CRC. Some studies have indicated a potential protective effect of PD against CRC, possibly due to the anti-cancer properties of dopamine agonists or the opposing mechanisms of neurodegeneration and cellular proliferation. Other studies indicate a positive association of PD with CRC providing possible causal mechanisms like common genetic origin, gut dysbiosis causing inflammation, and tumorigenesis by shared gene mutation. These conflicting findings underscore the need for further research, particularly large-scale prospective studies, to fully elucidate the intricate relationship between PD and CRC.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
