Abstract
Background:
Blood uric acid level represents an emerging biomarker in Parkinson’s disease (PD). Whether uric acid levels change in genetic forms of PD is just beginning to be explored.
Objective:
The aim of the present study was to assess differences in serum uric acid level among PD patients harboring the p.A53T mutation in the alpha-synuclein gene, idiopathic PD, and healthy controls.
Methods:
Longitudinal 5-year serum uric acid measurement data of 369 de novo idiopathic PD patients and 174 age- and gender-matched healthy controls have been downloaded from the Parkinson’s Progression Markers Initiative (PPMI) database. Furthermore, we assessed baseline serum uric acid measurements of 24 p.A53T alpha-synuclein PD patients enrolled in PPMI and followed in our site as compared to 24 age-, gender- and disease duration-matched sporadic PD patients and 24 healthy controls.
Results:
Longitudinal serum uric acid measurements did not differ statistically between idiopathic PD patients and healthy controls (despite a trend for lower uric acid in the PD group) (p = 0.879). This was also true when male and female subgroups were assessed separately. The p.A53T SNCA mutation carrier PD group exhibited lower baseline serum uric acid level as compared to their matched healthy controls (p = 0.025).
Conclusion:
In the present study we did not replicate the established lower serum uric acid measurements in PD patients as compared to controls using PPMI data, possibly due to the fact that PD patients in baseline visit were de novo and the average disease duration was shorter than that observed in most epidemiological PD studies. The faster progression rate and increased disease severity in p.A53T PD possibly correlate with the lower serum uric acid observed in this subgroup.
INTRODUCTION
Oxidative stress is thought to play a crucial role in the pathogenesis of Parkinson’s disease (PD). The findings of a recent meta-analysis demonstrated high blood level of oxidation markers and lower blood concentrations of catalase, uric acid and glutathione in PD patients, supporting the clinical evidence that PD is accompanied by increased oxidative stress [1]. On the other hand, uric acid represents an important endogenous antioxidant agent. Previous studies have highlighted its role as a putative PD biomarker. Uric acid exerts its antioxidant function mainly by means of ferrum chelation, although other mechanisms may also contribute to this effect. In certain PD population studies, patients exhibited lower serum uric level in comparison to healthy controls [2, 3].
The Parkinson’s progression markers initiative (PPMI) study evaluated the 5-year change of clinical, imaging and biochemical parameters in de novo PD patients, in patients with genetic forms of PD (including carriers of the p.A53T mutation in the alpha-synuclein gene) and in matched healthy controls.
The aim of the present study was to determine whether there are baseline and longitudinal differences in serum uric acid level between sporadic PD patients and healthy controls enrolled in the PPMI study. Moreover, we assessed differences among PD patients harboring the p.A53T mutation in the alpha-synuclein gene, idiopathic PD, and healthy controls. LRRK2 and GBA carriers are additional genetic cohorts assessed in PPMI study, however in the present analysis we focused only on the alpha-synuclein genetic cohort.
MATERIALS AND METHODS
Data used in the preparation of this article were obtained from the Parkinson’s Progression Markers Initiative (PPMI) database (http://www.ppmi-info.org/data). For up-to-date information on the study, visit http://www.ppmi-info.org. The present study was conducted in agreement with the principles of the Declaration of Helsinki. Signed informed consent was obtained from all participants recruited. The study was approved by the Scientific Board of all PPMI sites involved (including the Scientific Board of Eginition hospital). Longitudinal 5-year serum uric acid measurement data of 421 de novo PD patients and 174 age- and sex- matched healthy controls have been downloaded from the PPMI database (Table 1). We have reviewed data regarding GBA and LRRK2 variants in the PPMI de novo PD cohort and have removed 44 PD carriers of GBA variants and 8 PD carriers of LRRK2 variants so as to eliminate the impact of genetic forms on uric acid levels in this cohort (remaining sporadic PD patients N = 369). Biochemical analyses (including measurements of uric acid level in serum) have been carried out in Covance laboratories in a uniform fashion, as per the study protocol.
Baseline demographic, motor and non-motor characteristics in idiopathic PD vs. healthy controls (N/A, not available)
MOCA, Montreal Cognitive Assessment test; MDS-UPDRS, Movement Disorder Society-Unified Parkinson’s Disease Rating Scale; SBR, Specific Binding Ratio.
Furthermore, we assessed baseline serum uric acid measurements of 24 p.A53T alpha-synuclein PD patients enrolled in PPMI and followed in our site as compared to 24 age-, sex – and disease duration-matched idiopathic PD patients and 24 healthy controls, again matched for age and sex (Table 2).
Demographic characteristics in p.A53T SNCA PD, idiopathic PD and healthy controls (N/A: Not available)
MDS-UPDRS, Movement Disorder Society-Unified Parkinson’s Disease Rating Scale.
Statistical analysis for the idiopathic PD cohort was performed using repeated measures ANOVA (tests of within- and between-subjects effects) to examine the longitudinal changes of uric acid level for the 2 subgroups (idiopathic PD and healthy controls). Independent samples Mann- Whitney U test was used for the comparison among matched p.A53T SNCA mutation genetic PD, idiopathic PD, and healthy controls baseline data. Statistical significance was set at p < 0.05.
Spearman correlations were calculated between uric acid level and various baseline parameters (MDS-UPDRS score parts I, Ia, II and III, Montreal Cognitive Assessment MoCa score and DATSCAN binding ratios (SBR) of the contralateral and ipsilateral caudate and putamen respectively) in the idiopathic PD cohort.
Finally, in order to assess the putative impact of uric acid levels on clinical progression of patients in the idiopathic PD group and determine if high and low uric acid groups follow different trajectories, we compared high serum uric acid level- and low serum uric acid level- subgroups (at baseline) in terms of motor deterioration (UPDRS III score at baseline, UPDRS III score on year 3 and the 3-year score change) using one way ANOVA. A significance level of 0.05 was used. The statistical analyses were performed using commercially available software (SPSS, Version 20.0).
RESULTS
Longitudinal serum uric acid measurements did not differ statistically between idiopathic PD patients and healthy controls (despite a trend for lower uric acid in the PD group) (Between subjects effects, p = 0.879). Moreover, there was no significant effect of time and time*group interaction on uric acid level (Within subject effects, Greenhouse-Geisser correction, p = 0.522 and p = 0.115 respectively). This was also true when male and female subgroups were assessed separately (Between subjects effects, p = 0.774 and p = 0.367 respectively for men and women) (Fig. 1A–C).
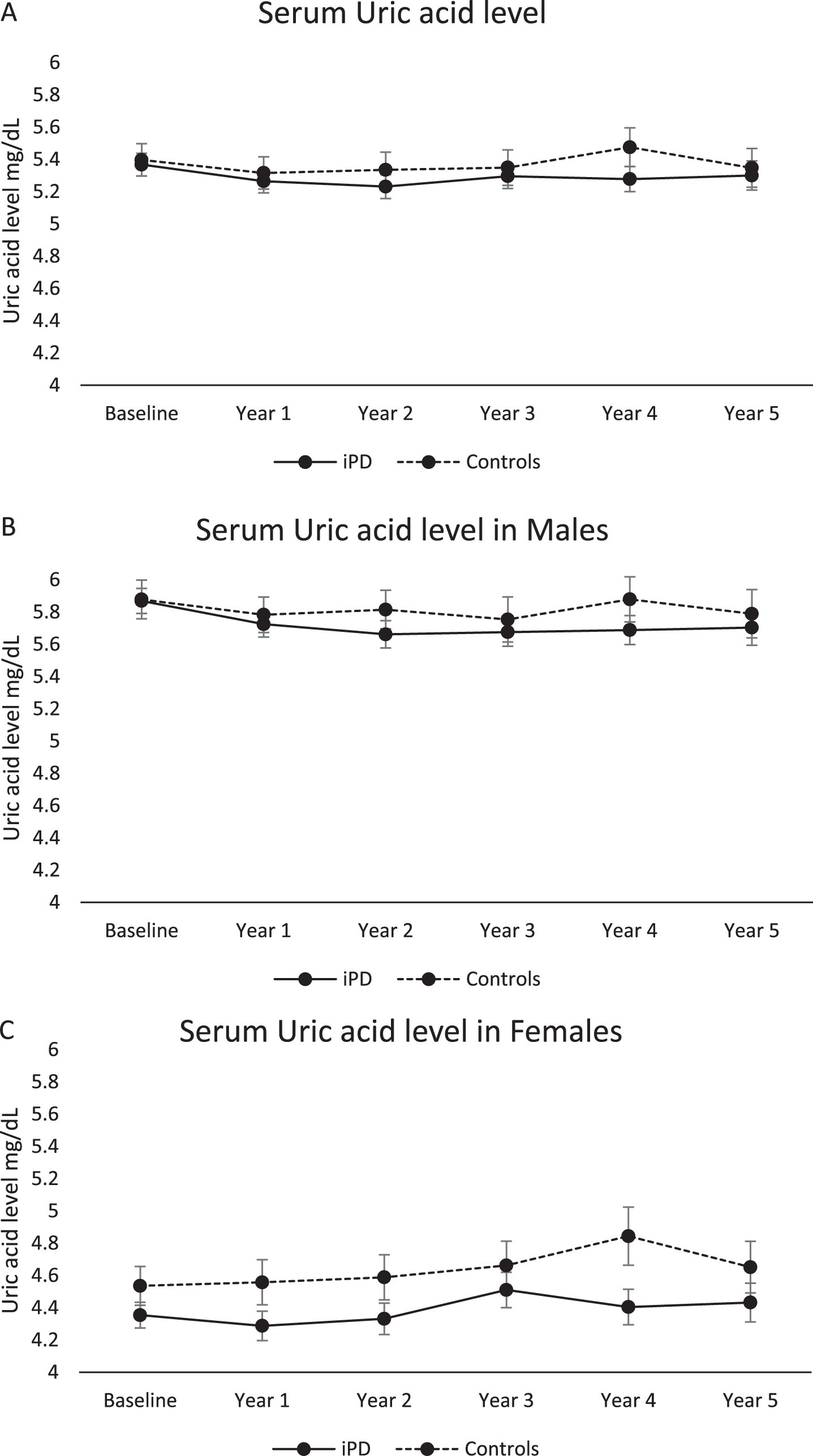
Longitudinal measurements of serum uric acid measurements in sporadic PD vs healthy controls (1A total study population, 1B males, 1C females). Statistical analysis has been performed using repeated measures linear ANOVA. PPMI timepoints of visits BL, V04, V06, V08, V10 and V12 correspond to Years 0, 1,2,3, 4, and 5, respectively.
In the PD group, we observed a modest negative correlation between baseline uric acid level and MDS-UPDRS score part I [Spearman correlation: r = –0.147 (p = 0.005)]. No other parameter correlated to baseline uric acid level in a statistically significant way. Moreover, we failed to reveal any significant impact of initial uric acid levels on the severity of motor deterioration when comparing high and low uric acid subgroups [baseline UPDRS III score 19.9 vs 20.4 (p = 0.654), Year 3 UPDRS III score 27.7 vs 28.8 (p = 0.432) and 3-year change + 8.9 vs + 8.8 (p = 0.939)].
The p.A53T SNCA mutation carrier PD group (N = 24) exhibited lower baseline serum uric acid levels compared to their matched healthy controls (4.39±1.44 vs 5.13±1.46 mg/dL, p = 0.025). Sporadic PD serum uric acid level was intermediate (4.82±1.56 mg/dL) and did not differ from either p.A53T PD (p = 0.392) or healthy controls (p = 0.332) (Fig. 2).
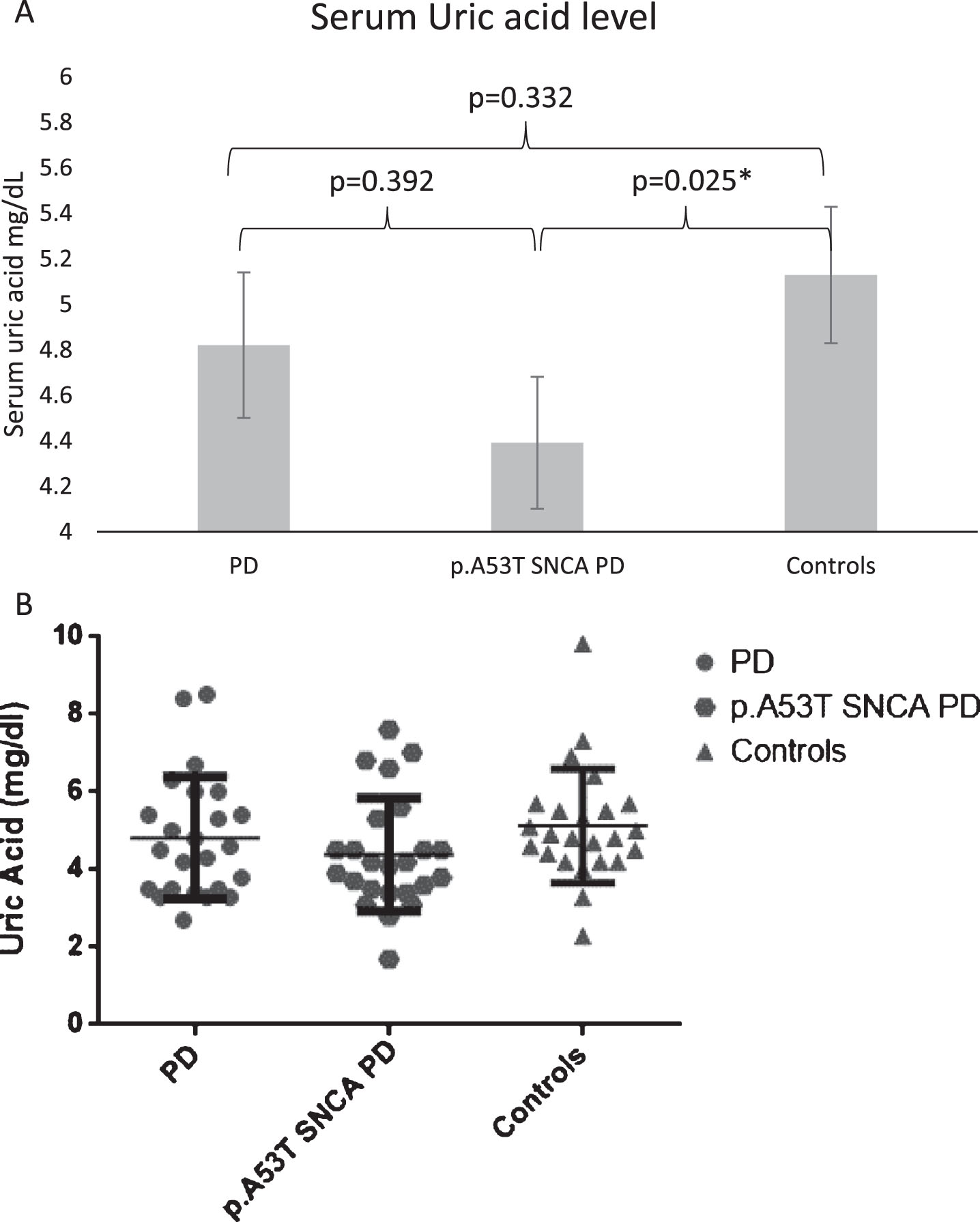
Baseline measurements of serum uric acid in healthy controls, idiopathic PD and p.A53T SNCA PD (statistical significance is marked by *).
DISCUSSION
The role of uric acid as a biomarker in PD has been established in recent literature. Its physiological role in PD still remains elusive. In vitro studies have shown that uric acid could induce autophagy activation and ameliorate alpha- synuclein (SNCA) accumulation [4]. In an in vivo rat PD model, uric acid demonstrated neuroprotective properties for dopaminergic neurons through modulation of neuroinflammation and oxidative stress [by means of increased mRNA and protein expression of Nrf2 (NF-E2-related factor 2) and three Nrf2-responsive genes] [5]. Large retrospective and prospective studies have demonstrated decreased serum uric acid in PD patients or in individuals that eventually developed PD during follow up [6, 7].
In our present report, we failed to replicate the reported lower serum uric acid measurements in PD patients as compared to controls using longitudinal PPMI data although there was a trend for lower uric acid in the PD group. This negative result should be interpreted with caution as it might stem from lack of statistical power to detect differences in early stages of the disease. The lack of a statistically significant difference in uric acid levels could be partially explained by the fact that as per the study protocol, PD patients in baseline visit were de novo and the average disease duration was much shorter than that observed in most epidemiological PD studies. Our finding is supported by a recent study, in which uric acid levels and the ratio uric acid/creatinine in advanced stage PD patients were significantly lower than in early and medium stage PD patients [8]. Also, remarkably, serum uric acid levels dropped in an experimental PD model of 6-OH-DA lesioning of the median forebrain bundle, in a manner dependent on the magnitude of the behavioral impairment, as assessed by the apomorhine-induced rotation test, and the degree of nigrostriatal dopaminergic neurodegeneration [9]. Thus, an early PD stage, as was the case in de novo PD PPMI participants, could account for the relatively high uric acid level in this group. We did not however observe a decrease of uric acid levels with disease evolution over 5 years, except for a trend towards this direction in the first two years only in males; possibly, follow-up for longer periods of time may be needed to observe a decline.
High serum uric acid levels have been correlated with lower risk of PD and a milder disease course [10]. Uric acid level has also been implicated in the disease subtype and clinical characteristics. A study examining the effect of uric acid on motor and non motor symptoms in PD demonstrated an association of higher serum uric acid levels with the more benign tremor dominant (TD) motor subtype rather than with the postural instability gait disorder (PIGD) and mixed phenotypes. Regarding non motor symptoms there was less fatigue in patients with higher uric acid level, which could be related to its anti-oxidative properties [11]. Regarding cognitive outcomes, lower levels of serum uric acid in the early disease stages were reported to be associated with the later occurrence of mild cognitive impairment (MCI) in an early PD cohort [12].
In our current study we failed to observe a relationship between indices of disease severity and uric acid levels, except for an inverse relationship with MDS-UPDRS part I. However, this association was quite weak and did not survive multiple comparisons. As far as clinical progression of patients in the idiopathic PD group, when comparing PD subgroups with baseline high and low uric acid level, we failed to reveal any significant impact of uric acid level on the severity of motor deterioration. A lack of statistical power to detect clinical differences in early stages of the disease (in PPMI de novo patients) might again account for this finding. It is possible that a prospective follow-up could reveal different trajectories between these subgroups. A recent study assessing the putative prognostic value of 4 blood biomarkers in PD (uric acid, apolipoprotein A1, C-reactive protein and vitamin D) supports our results since none predicted motor or nonmotor prognosis. A weak association which was observed between higher uric acid and lower MDS-UPDRS III intercept/higher MDS-UPDRS III slope did not survive multiple comparisons [13].
Considering the fact of previously reported sex differences in the impact of uric acid level on PD, we assessed males and females separately but again failed to demonstrate any statistically significant effect in either sex (there was an apparent trend of lower uric acid level though not significant in female PD patients as compared to female controls, Fig. 1C). A Japanese study showed decreased serum uric acid levels in males with PD-related disorders (PD, MSA, and PSP) compared with male controls (but not in female patients) [14]. Moreover, a prospective study of plasma urate measurements demonstrated that men, but not women, with higher urate concentrations had a lower future risk of developing PD, suggesting that urate in high levels present in males could be protective against PD risk or could slow disease progression during the preclinical stage of the disease [15].
An important finding of our study are the decreased serum uric acid levels at baseline visits in patients carrying the p.A53T mutation of the alpha-synuclein gene, as compared to matched healthy controls. Idiopathic PD showed intermediate uric acid levels and did not differ significantly from either subgroup. This is the first account of uric acid assessment in a genetic cohort carrying a mutation in alpha-synuclein gene. The faster progression rate and increased disease severity in p.A53T SNCA PD [16, 17] possibly correlate to the lower serum uric acid observed in this subgroup. The higher Levodopa Equivalent Daily Dose (LEDD) administered to such patients [17] may also play a role. Another possibility is that somehow alpha-synuclein metabolism is linked to the urate pathway, and that the p.A53T mutation plays a direct role in modulating urate levels. A recent study implicates a putative association between these two pathways. In a cell line overexpressing wildtype or A53T mutant SNCA, uric acid has been shown to decrease SNCA/α-synuclein accumulation and this was blocked by Bafilomycin A1 co-treatment. Furthermore, uric acid administration was able to reduce SNCA accumulation in the midbrain of SNCA A53T transgenic mice in vivo. These results provide evidence that uric acid could induce autophagy activation via an mTOR-dependent signaling and thus ameliorate alpha-synuclein accumulation [4].
Our data on serum uric acid level in asymptomatic SNCA carriers were very limited due to the small number of such subjects in the PPMI cohort (data not shown). Literature data on uric acid in other genetic forms of PD are very scarce as well. Johansen and co -authors assessed the metabolomic profiling in LRRK2-related PD and demonstrated reduced uric acid level in both mutation carrying PD and idiopathic PD patients as compared to controls along with aberrations in the purine pathway. This effect persisted even when unmedicated patients were examined separately. Notably, asymptomatic LRRK2 carriers also presented a tendency of reduced uric acid levels compared to controls which could not however be confirmed statistically [18]. Finally, a recent study comparing manifesting and non-manifesting LRRK2 mutation carriers concluded that unaffected carriers had significantly higher levels of urate than those who developed PD [19].
Whether genetic variants that modify circulating urate levels are also associated with altered PD risk or not, remains elusive. Age at PD onset has been shown to be influenced by polymorphisms in uric acid transporter SLC2A9 gene, which have an impact on serum uric acid levels [20]. In contrast, Hughes and co-authors failed to support an association between genetic variants associated with circulating urate levels and risk of PD [21].
Based on the aforementioned results, clinical studies focused on putative neuroprotective dietary manipulations (increase of dairy products resulting in uric acid increase) or pharmacological interventions, like inosine administration, are underway [22–24].
Future longitudinal studies targeted to PD patient cohorts carrying pathogenic or risk enhancing mutations (like mutations in the Glucocerebrosidase gene, GBA1) would shed light on the role of uric acid as a putative biomarker also for genetic PD with possible therapeutic implications.
CONFLICT OF INTEREST
Christos Koros received funding from the Michael J Fox Foundation for his participation in PPMI. Athina-Maria Simitsi received funding from the Michael J Fox Foundation for her participation in PPMI. Ion Beratis received funding from the Michael J Fox Foundation for his participation in PPMI. Maria Stamelou serves on the editorial boards of Movement Disorders Journal and Frontiers in Movement Disorders, received travel and speaker honoraria from Abbvie, UCB and Specifar Pharmaceuticals, and the International Parkinson’s disease and Movement Disorders Society, receives research support from the Michael J Fox Foundation (PPMI), royalties from Oxford University Press, Cambridge University Press, and Elsevier. Leonidas Stefanis is employed by the National and Kapodistrian University of Athens, Medical School and the Biomedical Research Foundation of the Academy of Athens. He has received the following grants: MULTISYN European Program (EU, FP7-HEALTH.2013.1.2-1, number 602646), PPMI (supported by the Michael J. Fox Foundation), IMPRIND-IMI2 Number 116060 (EU, H2020), SANTE Research Grant in Biomedical Sciences, NO-MND (EU-FP7-PEOPLE-2013-IRSES), NPF 2015 Investigator Award (Collaborator), “PBMC and urine collection in LRRK2 and idiopathic PD” Grant by the Michael J. Fox Foundation (Collaborator). No other authors have disclosures to report.
Footnotes
ACKNOWLEDGMENTS
This work was supported by funding from the PPMI study, Parkinson’s Progression Markers Initiative. PPMI, a public-private partnership, is funded by the Michael J. Fox Foundation for Parkinson’s Research and funding partners, including Abbvie, Allergan, Avid, Biogen, Biolegend, Bristol-Myers Squibb, Covance, GE Healthcare, Genentech, GlaxoSmithKline, Lilly, Lundbeek, Merck, Meso Scale Discovery, Pfizer, Piramal, Roche, Sanofi_genzyme, Servier, Takeda, Teva, UCB, Verily, Voyager-Therapeutics and Golub Capital.
