Abstract
Background
Type 2 diabetes mellitus (DM) can influence the phenotype and progression of Parkinson's disease (PD), as both conditions share inflammation as a common pathogenic mechanism.
Objective
To explore peripheral inflammatory indices that reflect the impact of DM on PD.
Methods
We analyzed 52 drug-naïve PD patients with DM and 182 without DM, along with age- and sex-matched healthy control (HC) with and without DM in a 1:1 ratio. Clinical features were evaluated, including the Hoehn and Yahr (H&Y) scale and the Unified Parkinson's Disease Rating Scale (UPDRS). Peripheral inflammatory markers included the count of leukocyte subpopulations, high-density lipoprotein-cholesterol (HDL-C), and markers derived from these including neutrophil-to-HDL-C ratio (NHR), monocyte-to-HDL-C ratio (MHR), and lymphocyte-to-HDL-C ratio (LHR).
Results
There were no significant differences in age, sex, or disease duration between PD with DM and PD without DM group. The PD with DM group showed more symmetric motor features (p = 0.007) compared to the PD without DM group. NHR, MHR, and LHR were elevated in the PD with DM group compared to the other groups. Notably, MHR was highest in the PD with DM group, followed by the HC with DM group and the PD without DM group, and HC without DM group (9.73 vs. 8.30 vs. 7.63 vs. 6.46, p < 0.001). MHR positively correlated with clinical scales, including H&Y and UPDRS, across all PD patients (p < 0.05 for all).
Conclusions
Our study suggests that MHR effectively reflects the peripheral inflammatory status related to both PD and diabetes.
Plain language summary
Type 2 diabetes mellitus (DM) can affect the phenotype and progression of Parkinson's disease (PD) due to shared inflammation as a pathogenic mechanism. This study examined peripheral inflammatory indices in 52 PD patients with DM, 182 PD patients without DM, and matched healthy controls with and without DM. The peripheral inflammatory markers analyzed were leukocyte subpopulations, high-density lipoprotein-cholesterol (HDL-C), and ratios derived from these. Results showed no significant differences in age, sex, or disease duration between PD groups. However, PD with DM exhibited more symmetric motor features compared to PD without DM. Inflammatory markers, particularly monocyte-to-HDL-C ratio (MHR), were significantly elevated in the PD with DM group compared to other groups (PD without DM, HC with DM, HC without DM). MHR was highest in PD with DM (9.73), followed by HC with DM (8.30), PD without DM (7.63), and HC without DM (6.46). Furthermore, MHR positively correlated with disease severity, as reflected by H&Y and UPDRS scores, across all PD patients. These findings suggest that MHR effectively represents peripheral inflammation associated with both PD and DM, highlighting its potential as a biomarker for inflammation-driven disease progression in PD patients with DM. This study underscores the importance of monitoring inflammatory indices to better understand and manage the interaction between PD and DM.
Keywords
Introduction
Since its first report in 1993, numerous studies have examined the relationship between type 2 diabetes mellitus (DM) and Parkinson's disease (PD). 1 DM may serve as a risk factor for developing PD and is associated with a more severe PD phenotype, faster clinical progression, or worse prognosis.2–5 While not all studies have reported consistent findings, several large-scale cohort studies have shown that individuals with DM have a slightly higher risk of developing PD.5,6 Although these cohort studies do not indicate a strong association (odds ratios : 1.29, 1.41), the Mendelian randomization studies provide some evidence suggesting a potential causal effect. 5 Additionally, case-control studies found that PD patients with DM exhibited higher scores on PD severity scales and lower cognitive abilities.7–11 Longitudinal studies have identified diabetes as a contributing factor to disease progression in PD.12–22
There are various theories about the mechanisms linking PD and DM. Atherosclerosis in DM can exacerbate neurodegeneration in PD,23,24 but the connection between PD and DM remains significant even in the absence of established vascular disease. 25 Recent research indicates that PD and DM share commonalities in their pathophysiology.2–4 Both conditions are associated with aging and involve the selective depletion of cell populations with high metabolic demands, such as β-cells and substantia nigra dopaminergic neurons. Molecular pathways involved in DM, such as insulin signaling, mitochondrial dysfunction, impaired autophagy, and inflammation may overlap with and exacerbate PD pathophysiology. Additionally, DM can contribute to neurodegeneration via indirect mechanisms like chronic systemic inflammation.2–4
Inflammation is a well-established mechanism in PD pathogenesis. Both the peripheral and central immune systems undergo changes from the early stage of PD, which are associated with the development, severity, and prognosis of PD.2–4,26,27 Although both DM and PD are closely associated with inflammatory mechanisms, markers reflecting peripheral inflammation based on the presence of DM in PD have not been thoroughly evaluated.
Although recent studies have reported altered lipid metabolism in PD, including a significant reduction in high-density lipoprotein cholesterol (HDL-C) levels, peripheral inflammatory markers involving HDL-C have not yet been widely studied in the context of PD.28,29 Among these markers, the neutrophil-to-HDL-C ratio (NHR) has been reported to be elevated in PD patients compared to healthy controls in several studies, with some demonstrating a positive correlation between NHR levels and disease severity.28,30,31 Similarly, although absolute values of the monocyte-to-HDL-C ratio (MHR) vary across studies, multiple investigations have shown that higher MHR levels are associated with greater disease severity in PD.28,29,32 By contrast, the lymphocyte-to-HDL-C ratio (LHR) remains a relatively unexplored peripheral inflammatory marker in PD and requires further investigation.
Therefore, this study aims to identify peripheral inflammation markers that most accurately reflect systemic inflammation in PD patients, categorized by the presence of DM, in comparison to healthy controls (HC) using cost-effective routine lab tests. Additionally, we assess the correlation between these markers and the clinical phenotype of PD, encompassing symptom characteristics and disease severity.
Methods
Study design and subject
This case-control study recruited individuals diagnosed with PD and age- and sex-matched HC to investigate differences in peripheral inflammation levels, with a focus on the presence of DM. PD patients were recruited from the Hallym PD registry, a prospective database that has been enrolling all individuals presenting with parkinsonism at the neurology clinic of Hallym University Sacred Heart Hospital since May 2011.32,33 The registry includes computerized demographic, clinical, imaging, and laboratory records of all patients admitted for disease assessment.
In this study, drug-naïve patients in the early stages of PD were selected from this registry spanning from May 2011 to Aug 2023. The diagnosis of PD was established according to the Movement Disorder Society Clinical Diagnostic Criteria by two movement disorders specialists. 34 Confirmation of PD diagnosis was supported by decreased nigrostriatal tracer uptake observed in 18F-N-(3-fluoropropyl)-2β-carboxymethoxy-3β-(4-iodophenyl) nortropane (18F-FP-CIT) PET scans. We excluded cases diagnosed with Parkinson-plus syndrome, PD with other etiologies affecting parkinsonism, such as drug-induced parkinsonism, stroke, or normal pressure hydrocephalus.
During the same period, HC participants without PD were recruited from affiliated health screening centers linked to our institution. PD and HC were age- and sex-matched in a 1:1 ratio, with age matched within ± 1 year. To ensure the validity of the cumulative effect of PD and DM on inflammation levels, we categorized HC group into subgroups of individuals with and without DM, similar to the stratification applied to the PD group. We defined individuals who were taking diabetes medications or had a hemoglobin A1c (HbA1c) level of 6.5% or higher as the PD with DM group, and those with an HbA1c level below 6.5% and not taking diabetes medications as the PD without DM group. This study included only individuals with type 2 DM, excluding those with type 1 DM.
Furthermore, all individuals presenting with conditions that could induce chronic or active inflammatory states were excluded. This exclusion criterion encompassed individuals with active cancer, hematologic diseases, autoimmune diseases, recent infectious disease within the last 3 months, and unstable metabolic conditions (including acute renal failure, hepatic failure, and thyrotoxicosis). The patient selection process is detailed in Supplemental Figure 1.
Ethical statement
This study received approval from the institutional review board of Hallym University Medical Center (IRB 2023-12-025). Informed consent was waived due to the retrospective nature of the study, and all assessments were conducted in accordance with the ethical standards of the institutional committee, as well as the 1975 Helsinki Declaration and its later amendments or comparable ethical standards.
Clinical assessment
At the time of diagnosis, demographic information was collected from each participant, including age, sex, underlying diseases (hypertension status, DM status, and other conditions that can affect inflammation levels, including cancer and autoimmune diseases). Additionally, medication history was documented, covering the use of antihypertensive medication, antidiabetic medication, antihyperlipidemics, nonsteroidal anti-inflammatory drugs (NSAIDs), and immunosuppressants. The duration of parkinsonism was also recorded.
Motor and nonmotor symptoms were evaluated using the Hoehn and Yahr (H&Y) stage and Unified Parkinson's Disease Rating Scale (UPDRS). Cognitive functions were assessed through the Korean version of Montreal cognitive assessment (K-MoCA). A motor Asymmetry Index (AI) was calculated as the relative value of the difference between the right and left scores from the UPDRS-III score to assess the degree of lateralization. 35 In other words, a higher AI indicates greater asymmetry, while a lower AI indicates a more symmetric motor symptom.
Acquisition of laboratory findings
Blood samples were collected in the early morning following a minimum of 8 h of fasting during baseline visits prior to the initiation of medication. All patients underwent testing for two parameters related to diabetes status: fasting blood sugar (FBS) and HbA1c. In addition, Peripheral blood was analyzed for various parameters, including total leukocyte count (WBC), count and proportion of leukocyte subpopulations (neutrophils, lymphocytes, monocytes), and HDL-C. Additionally, the following inflammatory indices were calculated: neutrophil-to-lymphocyte ratio (NLR) (neutrophils/lymphocytes), monocyte-to-lymphocyte ratio (MLR) (monocytes/lymphocytes), and systemic inflammation response index (SIRI) (neutrophils × monocytes/lymphocytes). The NHR, MHR, and LHR values were expressed by dividing leukocyte count (10 3 /uL) by HDL-C (mg/dL). We selected these peripheral inflammatory markers that have been investigated in previous studies.26,28–32
A Beckman AU5821 automatic biochemical analyzer was used to measure HDL-C by selective inhibition method (Beckman Coulter, Brea, CA, USA). Other laboratory parameters were assessed with the ADVIA 2120 (Siemens Healthcare Diagnostics, Tarrytown, NY, USA) automated hematology counter.
Statistical analysis
Categorical variables were presented as absolute frequency (proportions) and compared between groups using Pearson's chi-square test. Continuous variables were expressed as median [interquartile range (IQR)].
We performed analyses based on four-group stratification for inflammatory markers: (1) PD with DM, (2) PD without DM, (3) HC with DM, and (4) HC without DM. Meanwhile, comparisons of clinical parameters such as disease duration, UPDRS scores, and Hoehn and Yahr stage were conducted only between the PD with DM and PD without DM groups. Kruskal-Wallis test followed by the Bonferroni correction were employed to compare differences between groups. Subsequently, nonparametric analysis of covariance (ANCOVA) was conducted to adjust for potential confounders such as age, sex, use of antihyperlipidemic agents and use of NSAIDs. Relationships between variables were assessed by calculating Spearman's rank correlation coefficient (rho). All significance levels were adjusted using the Bonferroni correction for multiple testing. Additionally, receiver operating characteristic (ROC) curve analysis was performed to assess the diagnostic performance of MHR between groups. Statistical analyses were carried out using SPSS version 28.0 (IBM Corp., Armonk, NY, USA), GraphPad Prism 10 software (GraphPad software, www.graphpad.com) and R version 4.3.3. A p-value below 0.05 was considered statistically significant.
Results
Demographic and clinical characteristics of study groups
The study included a total of 234 PD patients, consisting of 52 with DM and 182 without DM. Additionally, 234 age- and sex-matched controls without PD were enrolled, including 52 individuals with DM and 182 without DM.
Table 1 presents the clinical features and detailed demographic profiles of each group. There were no statistical differences in age and sex between the DM and non-DM group in both PD and HC. The disease duration of PD was also similar between the PD patients with DM and those without DM.
Demographic properties and clinical features of the groups.
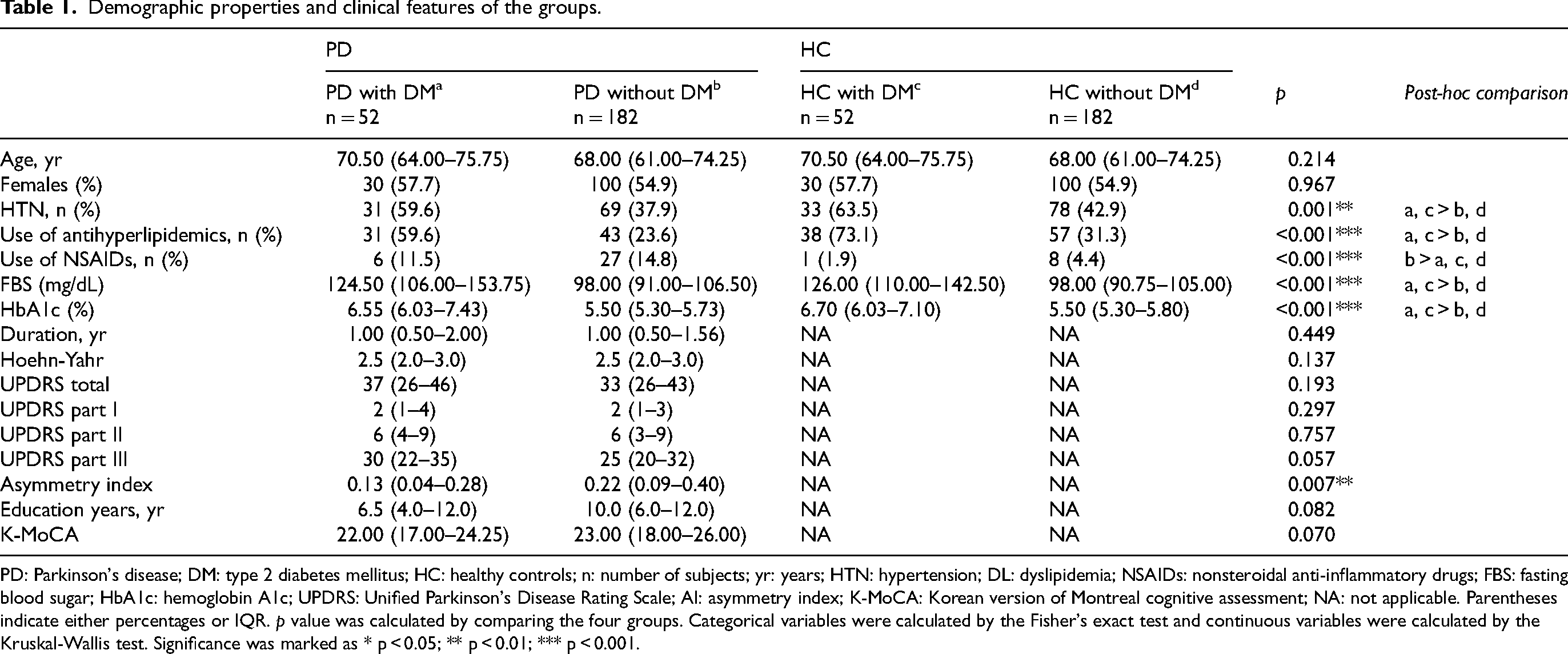
PD: Parkinson's disease; DM: type 2 diabetes mellitus; HC: healthy controls; n: number of subjects; yr: years; HTN: hypertension; DL: dyslipidemia; NSAIDs: nonsteroidal anti-inflammatory drugs; FBS: fasting blood sugar; HbA1c: hemoglobin A1c; UPDRS: Unified Parkinson's Disease Rating Scale; AI: asymmetry index; K-MoCA: Korean version of Montreal cognitive assessment; NA: not applicable. Parentheses indicate either percentages or IQR. p value was calculated by comparing the four groups. Categorical variables were calculated by the Fisher's exact test and continuous variables were calculated by the Kruskal-Wallis test. Significance was marked as * p < 0.05; ** p < 0.01; *** p < 0.001.
The prevalence of hypertension and the proportion of patients taking antihyperlipidemics were significantly higher in the DM group compared to the non-DM groups in both PD and HC (p ≤ 0.001 for all). The prevalence of NSAIDs use was higher in the PD without DM group compared to the other three groups (p < 0.001). Additionally, FBS and HbA1c levels were higher in the DM group compared to non-DM group (p < 0.001).
There were no differences in global cognition as measured by K-MoCA and education level between the PD with DM group and PD without DM group (p = 0.070, p = 0.082, respectively). Although there was no significant difference in motor symptoms between the PD with DM group and PD without DM group (p = 0.057), PD patients with DM exhibited lower AI values, indicating more symmetric motor symptoms compared to those without DM (p = 0.007).
Comparison of peripheral inflammatory markers in relation to PD and DM
The comparison of peripheral inflammatory markers across different groups is presented in Table 2 and Supplemental Figure 2. The monocyte count (×109/L) was significantly higher in both the PD with DM group and the PD without DM group compared to the HC without DM group (0.46, 0.40, 0.35, p < 0.001). The PD with DM group showed lower HDL-C levels (mg/dL) than both the PD without DM group and the HC without DM group (44, 53, 55, p < 0.001). Additionally, the NHR levels were elevated in the PD with DM group compared to the PD without DM group and the HC without DM group (73.83, 54.80, 54.19, p = 0.002).
The comparative analysis of peripheral inflammatory markers among the groups.
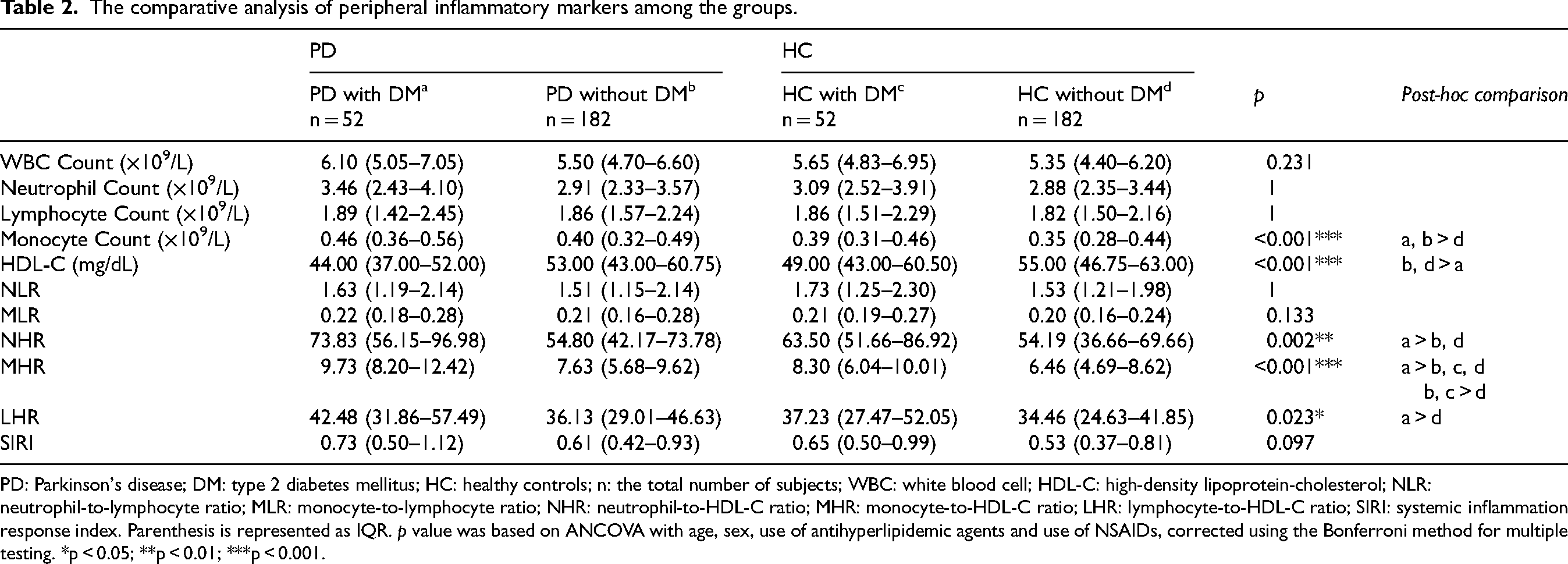
PD: Parkinson's disease; DM: type 2 diabetes mellitus; HC: healthy controls; n: the total number of subjects; WBC: white blood cell; HDL-C: high-density lipoprotein-cholesterol; NLR: neutrophil-to-lymphocyte ratio; MLR: monocyte-to-lymphocyte ratio; NHR: neutrophil-to-HDL-C ratio; MHR: monocyte-to-HDL-C ratio; LHR: lymphocyte-to-HDL-C ratio; SIRI: systemic inflammation response index. Parenthesis is represented as IQR. p value was based on ANCOVA with age, sex, use of antihyperlipidemic agents and use of NSAIDs, corrected using the Bonferroni method for multiple testing. *p < 0.05; **p < 0.01; ***p < 0.001.
Notably, the MHR levels were highest in the PD with DM group, followed by the HC with DM group and the PD without DM group, with the lowest levels observed in the HC group without DM (9.73 vs. 8.30 vs. 7.63 vs. 6.46, p < 0.001). However, MHR was not significantly different between the PD without DM group and the HC with DM group. The ROC curve of MHR between the groups was described in Supplemental Figure 3. The AUC was 0.675 for the comparison of PD with DM and PD without DM; 0.671 for PD with DM and HC with DM; 0.774 for PD with DM and HC without DM; 0.621 for PD without DM and HC without DM; and 0.654 for HC with DM and HC without DM.
The LHR value was higher in the PD with DM group compared to the HC without DM group (p = 0.023).
Association between PD phenotypes and laboratory results
The correlation analysis of clinical parameters and laboratory values for all PD participants, PD without DM, and PD with DM groups is summarized in Figure 1 and Supplemental Tables 1–3. Across all PD participants, MHR showed positive correlations with H&Y, UPDRS total and part III scores, and a negative correlation with K-MoCA score (p < 0.05 for all, Figure 1(a)). Several inflammatory markers, including neutrophil count, monocyte count, NLR, MLR, NHR, and SIRI, showed positive correlations with UPDRS total and part III scores, with and without H&Y scale, for all PD participants (p < 0.05 for all, Figure 1(a)). NHR was negatively associated with AI, while HDL-C showed a positive correlation with AI (p < 0.05 for all, Figure 1(a)). K-MoCA showed negative correlations with HbA1c, MLR, MHR, and SIRI (p < 0.05 for all, Figure 1(a)).
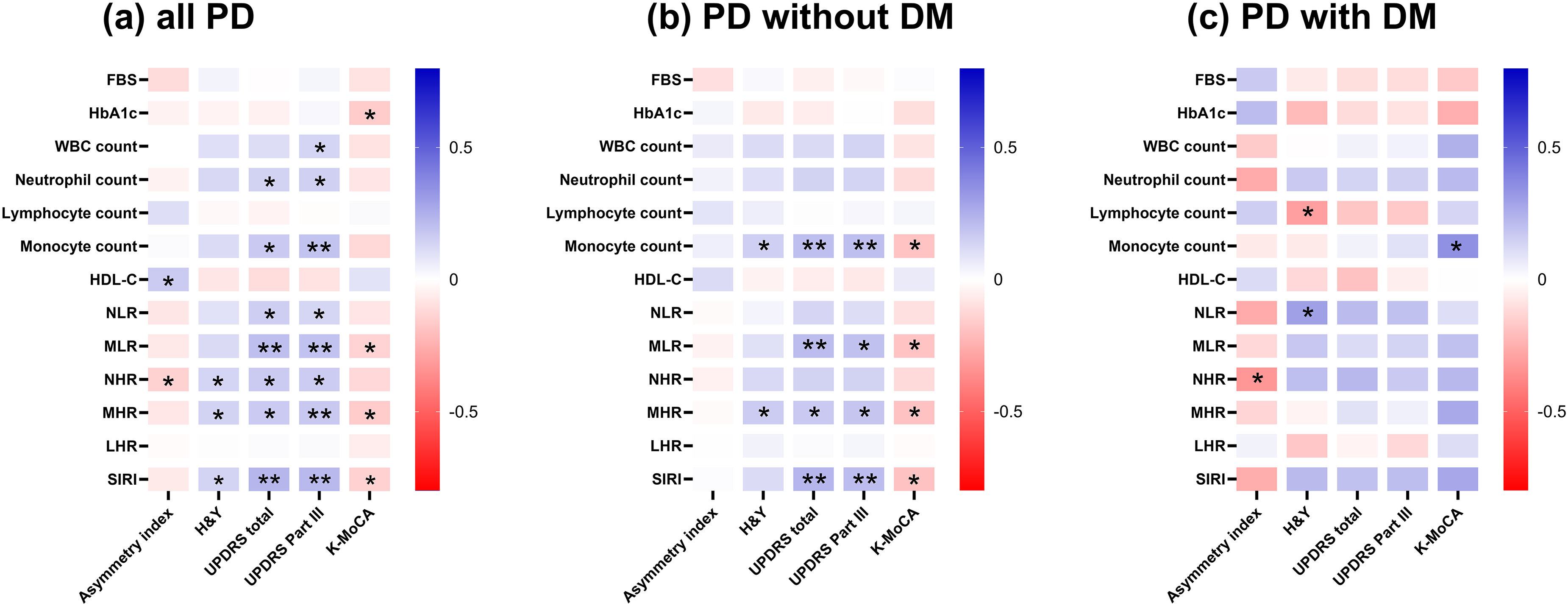
Correlation analysis of peripheral inflammatory markers and clinical parameters. The correlation analysis of peripheral inflammatory markers and the clinical parameters in (a) total PD population, (b) PD without DM patients, and (c) PD with DM patients. Color values indicate Spearman's rank correlation coefficient (rho). Asterisk (*) marks the cases where the value of rho is statistically significant. *p < 0.05; **p < 0.01.
When analyzed within each group, these associations were significant mainly in PD without DM group (Figure 1(b), (c)). In the PD without DM group analysis, monocyte count and MHR demonstrated significant correlations with disease severity, including with H&Y stage, UPDRS total and Part III scores, and K-MoCA (Figure 1(b) and Supplemental Table 2). Additionally, MLR and SIRI showed negative correlations with K-MoCA, and positive correlations with UPDRS total and Part III score (p < 0.05 for all). Meanwhile, in the PD with DM group, NHR showed a negative correlation with the AI, whereas no significant correlation was observed with MHR or other inflammatory indices in this subgroup analysis (Figure 1(c) and Supplemental Table 3).
Discussion
This study investigated various inflammatory parameters and clinical phenotypes in early-stage, drug-naïve PD patients categorized based on the presence of DM. The findings indicate that MHR level is a significant marker of systemic inflammation related to the existence of DM on PD, correlating with both motor and cognitive severity. MHR was developed to provide a more comprehensive indicator of inflammation which integrates the pro-inflammatory influence of monocytes and the anti-inflammatory role of HDL-C in various diseases. 36 There have been few studies on MHR as a potential marker of inflammation reflecting disease severity in PD and diabetes individually. In previous study of PD population, MHR levels positively correlate with disease severity, though the reported MHR levels and the difference in MHR value between PD and HC vary across studies.28,29,32 Among the three studies, one reported that MHR levels did not significantly differ between PD patients and healthy controls; however, correlation analysis demonstrated a positive association between MHR and H&Y stage. 28 In contrast, another study found that MHR values were lower in PD patients compared to healthy controls, although the relationship between MHR and disease severity within the PD group was not examined. 29 A third study showed that MHR levels were significantly higher in PD patients than in healthy controls, and that MHR was positively associated with H&Y stage and negatively associated with MMSE scores. 32 In diabetes, research has produced inconsistent results regarding whether MHR levels were higher compared to healthy individuals.37,38 Nevertheless, MHR is associated with an increased risk of type 2 diabetes and is linked to poor outcomes and complications in diabetic patients.39,40 In our study, we found that MHR levels were highest in the PD group with DM, intermediate in the PD group without DM and the HC group with DM, and lowest in the HC group without DM. Furthermore, MHR showed a clear positive correlation with disease severity both in the total PD group and in the PD group without DM. MHR may serve as a potential marker of systemic inflammation in the context of PD and DM, although further studies are needed to validate its clinical relevance and diagnostic utility.
In PD, an increase in monocyte cell count and monocyte activation with an altered transcriptome is observed, 27 Recent investigations have also unveiled a noteworthy decrease in HDL-C levels among the PD population.28,29 In diabetes, the expression of monocyte activation markers such as CD11b and CD36 is elevated. 41 CD163 expression levels, which increase on monocyte in response to inflammatory stimuli, were significantly associated with insulin resistance in DM. 42 The upregulation of these markers in DM indicates augmented immune activity resulting from monocyte activation. In addition, changes in HDL-C are among the various abnormalities observed in DM. 43 Previous studies have exhibited accelerated catabolism of HDL-C particles, along with significant modifications in their size, lipidome, and proteome, likely contributing to functional defects. As a result, HDL-C exhibits diminished antioxidative function, including reduced ability to inhibit the nuclear factor kappa-B proinflammatory pathway. Hence, the hierarchy of MHR levels among PD with DM, DM without PD, PD without DM, and HC without PD and DM may reflect the cumulative effect of peripheral inflammation, which increases with the addition of PD or DM.
Moreover, the observed correlation between peripheral blood markers and clinical severity, including motor manifestations, activities of daily living, and cognitive function, suggests that increased peripheral inflammation may contribute to severe PD phenotype. Notably, this relationship varies depending on the subgroup. In the total PD group and the PD without DM group, there is a strong correlation between inflammation levels and clinical symptoms, but this correlation is not observed in the PD with DM group. In the PD with DM group, where inflammation levels were already high, it was challenging to observe a correlation between inflammation levels and clinical manifestations. In contrast, in the PD without DM group, a relationship between inflammation levels and clinical manifestations could be observed. This also implies that, in the absence of diabetes, inflammatory markers are more effective in predicting the disease phenotype compared to when diabetes is present. A hypothesis to explain this difference proposes the existence of factors in DM that influence inflammation, thereby modifying the relationship between peripheral inflammation and disease severity. These factors include the method of DM management (insulin or other glucose-lowering agents), the degree of DM control, and the duration of DM, all of which may alter this relationship.
The association between DM and a more severe PD phenotype, as well as a more rapid clinical progression and poorer prognosis, has been suggested by previous case-control and longitudinal study findings. 2 DM can contribute to higher UPDRS part III scores,7–9,12–14 and it also influences more pronounced and prevalent non-motor symptoms, comprising cognition, mood, and sleep disturbance. 13 In addition, PD patients with concomitant DM may demonstrate accelerated motor or cognitive decline, necessitating elevated doses of levodopa, and experiencing the earlier onset of motor complications during the disease course.10–22 Furthermore, the survival duration from diagnosis to death is markedly decreased in neuropathologically confirmed PD cases with concomitant DM. 15 There exists evidence indicating that DM can contribute to the deterioration of dopaminergic degeneration, the fundamental manner of PD pathology, as revealed by imaging studies. PD with DM patients exhibit lower striatal dopamine transporter availability compared to those without DM.14,44 Even in populations without PD, DM is associated with subclinical striatal dopaminergic dysfunction on dopamine transporter imaging. 14 However, despite these findings, studies comparing the clinical characteristics of PD patients with and without DM have reported inconsistent results.9,22,45 Such discrepancies may stem from differences in study design, sample size, and participant characteristics. Therefore, larger, well-designed prospective studies are needed to establish more definitive conclusions regarding the impact of DM on the clinical phenotype and progression of PD.
In our study, we analyzed motor asymmetry in addition to the other PD phenotypes. We found that PD patients with DM had a lower AI compared to those without DM, indicating that motor symptoms appear more symmetrically. Among the inflammatory markers, NHR value was correlated with symmetry, although MHR was not. Typically, the motor symptoms in PD start on one side of the body, which is a supporting diagnostic feature,34,46,47 though many patients may present with symmetric symptoms at diagnosis. This asymmetry is attributed to substantia nigra asymmetry, as evidenced by pathology and imaging studies.46–48 The asymmetric presentation of PD has been extensively studied. Some studies indicate that PD patients with asymmetric presentation show slower progression, superior quality of life, and good mental state.49–51 Recent research also suggests the association between asymmetry and non-motor symptoms, such as rapid eye movement sleep behavior disorder. 52 However, other studies have not found a clear link between asymmetry and the progression of PD.35,47,53 From this perspective, the presence of diabetes in PD could be considered a factor contributing to the more symmetrical presentation of parkinsonism.
Recently, a proposed model called the α-synuclein origin site and connectome model suggests categorizing PD into brain-first and body-first subtypes. 54 The location of the initial pathogenic α-synuclein deposition is crucial, along with the neural connectome, in determining the propagation of pathological α-synuclein through the nervous system. In the body-first subtype, α-synuclein pathology originates in the gut and spreads through the vagal nerves to the brainstem, resulting in predominantly symmetric nigrostriatal degeneration. In contrast, in the brain-first subtype, pathogenic α-synuclein forms unilaterally in one hemisphere, leading to asymmetric nigrostriatal degeneration. Body-first and brain-first phenotypes in PD differ significantly. Interestingly, as parkinsonism develops, it tends to become more symmetric on average. Although multiple mechanisms may influence the degree of asymmetry of PD pathology and symptoms, increased peripheral inflammation, as observed in DM, could contribute to a more symmetric motor presentation, similar to body-first PD phenotype. Additional longitudinal studies are required to investigate the long-term clinical impact of peripheral inflammatory changes.
One of the major strengths of this study is its inclusion of only de novo patients who had not yet started dopaminergic medication, thereby avoiding the impact of levodopa or other dopaminergic drugs on glucose levels or inflammatory status. 2 This consideration is critical as levodopa has been shown to cause hyperglycemia, and even when combined with carbidopa, it can accumulate in the skeletal muscle, leading to β-adrenergic-dependent inhibition of insulin action. By focusing on newly diagnosed drug-naïve patients, the study aimed to eliminate the confounding effects of levodopa-induced glycemic change.
Additionally, we ensured more consistent results by using blood samples collected from all participants after an 8 h fast early in the morning, controlling for variations in blood glucose levels and diurnal changes in white blood cell count. This study also minimized the influence of asymmetry diminution during disease progression. As PD advances, bilateral cell losses become balanced, leading to an asymmetry that follows an “inverted U shape” peaking during prodromal or early PD and then declining as the disease progresses.47,52 Diabetes status was cross-verified using HbA1c and FBS measurements rather than relying solely on medical history. Lastly, Age- and sex-matched HC were incorporated into the analysis. This approach strengthens the study by ensuring that any observed differences in the outcomes can be more accurately attributed to the presence of PD and DM.
This study has several limitations. First, the laterality of striatal dopaminergic dysfunction was not assessed due to the absence of quantitative analysis of dopamine transporter imaging, limiting the understanding of regional brain changes. To confirm whether our findings correspond with pathological findings beyond clinical consistency, additional research including image analysis is necessary. Second, the study did not find which condition, DM or PD, has a greater impact on inflammation. Further investigation, including longitudinal studies, is necessary to identify the peripheral inflammatory markers that reflect the underlying mechanisms linking disease severity and the presence of DM in PD. Third, to better understand the specific effects of diabetes-related systemic inflammation in PD, it would have been beneficial to include long-term clinical outcome data for PD patients. Future research should evaluate the prognostic value of the MHR as a long-term predictor in PD patients with diabetes. In addition, the lack of detailed data on DM duration is another limitation of our study, as it might influence peripheral inflammation levels. Finally, the absence of lifestyle factors such as diet, exercise, and smoking further constrained our analysis.
In summary, the observed association between DM and PD extends beyond conventional understandings of individual disease pathology, highlighting a nuanced relationship between systemic conditions and neurodegenerative processes. By elucidating the intricate role of peripheral inflammation in PD and DM, MHR has notably been shown to reflect the increased levels of peripheral inflammation in both diseases and is well correlated with disease severity. These findings suggest that MHR may serve as a valuable integrative biomarker that reflects the burden of systemic inflammation in PD, especially when comorbid with DM. These insights pave the way for holistic approaches to PD management, emphasizing the importance of addressing systemic comorbidities and inflammatory pathways in mitigating disease burden and optimizing therapeutic outcomes.
Conclusion
Our study indicates that MHR effectively mirrors the heightened peripheral inflammation found in PD and DM. Both conditions are strongly associated with the clinical features of PD.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X251372392 - Supplemental material for Peripheral inflammatory markers and clinical phenotypes reflecting the impact of diabetes on Parkinson's disease
Supplemental material, sj-docx-1-pkn-10.1177_1877718X251372392 for Peripheral inflammatory markers and clinical phenotypes reflecting the impact of diabetes on Parkinson's disease by In Hee Kwak, Hyeo-il Ma, Yun Joong Kim, Hye-Mi Noh, Jeongjae Lee, Je Kook Yu and Young Eun Kim in Journal of Parkinson's Disease
Footnotes
Acknowledgements
We extend our heartfelt thanks to researcher Sion Lee from Medical Statistics Office at Hallym university medical center. Her invaluable contribution has been instrumental in advancing our research.
Ethical considerations
This study received approval from the institutional review board of Hallym University Medical Center (IRB 2023-12-025). Informed consent was waived due to the retrospective nature of the study, and all assessments were conducted in accordance with the ethical standards of the institutional committee, as well as the 1975 Helsinki Declaration and its later amendments or comparable ethical standards.
Consent to participate
This is an IRB-approved retrospective study, all patient information was de-identified and patient consent was not required. Patient data will not be shared with third parties.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: RS-2023-00265159), Basic Science Research Program through the National Research Foundation of Korea (NRF) grant funded by the Ministry of Education (grant number: RS-2023-00246655), the National Research Foundation of Korea (NRF) by the Korea government (MSIT) (grant number: 2022R1A2C2091254), and Bio & Medical Technology Development Program of the National Research Foundation (NRF) funded by the Korean government (MSIT) (grant number: RS-2023-00223501).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The datasets utilized in this study are not publicly accessible due to ethical or legal constraints. However, they can be obtained from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
