Abstract
Background
Prodromes of Parkinson's disease (PD) include both motor and non-motor symptoms. Although questionnaires have been established for non-motor symptoms, no quantitative self-assessment tool has been developed to assess subtle motor symptoms during the prodromal stage.
Objective
To develop a self-administered questionnaire to assess subtle motor symptoms during the prodromal stage.
Methods
We created the Screening Questionnaire for Subtle Parkinsonism (SQSP). The SQSP and questionnaires on non-motor symptoms were collected from health checkup examinees. Individuals with ≥ 2 non-motor symptoms, including autonomic dysfunction, hyposmia, and REM sleep behavior disorder, were classified as high-risk, while those without these symptoms were low-risk. We also conducted comprehensive evaluations, including neurological examinations and imaging tests, on 30 patients with PD, 71 high-risk, and 24 low-risk subjects.
Results
Among 1183 health checkup examinees, high-risk subjects had higher SQSP scores than low-risk (9 [4–15] vs. 3 [1–6]). Patients with PD had the highest SQSP scores, followed by high-risk subjects and then low-risk. SQSP scores correlated with MDS-UPDRS II and III scores and specific binding ratios of DaT-SPECT. High-risk subjects with abnormal DaT-SPECT had higher SQSP scores than those with normal imaging (9 [7–19] vs. 5.5 [2–10]). Although 26 of the 71 high-risk and 23 of the 24 low-risk subjects scored zero on the MDS-UPDRS II, most high-risk and half low-risk subjects had SQSP scores above zero.
Conclusions
The SQSP was deemed effective for assessing subtle motor symptoms during the prodromal stage of PD and identifying prodromal PD cases within the general population.
Plain language summary
Parkinson's disease (PD) presents several non-motor symptoms and subtle motor symptoms before developing the disease. Early diagnosis during this prodromal stage is essential for developing novel therapies to slow disease progression. While questionnaires to assess non-motor symptoms are established, no self-assessment tool has been developed to evaluate subtle motor symptoms at this stage. Therefore, we created a new questionnaire named the Screening Questionnaire for Subtle Parkinsonism (SQSP) for self-evaluation of mild motor symptoms. We collected the SQSP and questionnaires on non-motor symptoms from 1183 health checkup examinees. People with ≥2 non-motor symptoms, including autonomic dysfunction, olfactory dysfunction, and REM sleep behavior disorder (high-risk people), had higher SQSP scores, indicating they were aware of subtle motor symptoms. We also conducted comprehensive evaluations, including neurological examinations and imaging tests, on 30 patients with PD, 71 high-risk, and 24 low-risk people (those without non-motor symptoms). SQSP scores were highest in patients with PD, followed by the high-risk, and lowest in the low-risk people. SQSP scores correlated with MDS-UPDRS III scores, which measure parkinsonism, MDS-UPDRS II scores, which assess motor experiences of daily living of patients with PD, and the specific binding ratios of DaT-SPECT, which indicate dopamine neuron loss. Among the high-risk people, 23 cases with abnormal DaT-SPECT had higher MDS-UPDRS II, III, and SQSP scores. These findings suggest that the SQSP is a useful tool for evaluating subtle motor symptoms during the prodromal stage of PD.
Introduction
Parkinson's disease (PD) has a prodromal stage characterized by non-motor symptoms, such as autonomic dysfunction, hyposmia, REM sleep behavior disorder (RBD), depressive symptoms, and daytime sleepiness. 1 Additionally, mild motor symptoms that are not severe enough to be diagnosed as parkinsonism may also appear during this period. 2
Among the non-motor symptoms at the prodromal stage, RBD is considered the most indicative because of its high likelihood of future development of neurodegenerative disorders.3,4 Consequently, many studies on the prodromal stage of PD have focused on patients with idiopathic RBD (iRBD). 5 However, the clinical progression of PD from the prodromal stage exhibits substantial heterogeneity from case to case, and recent studies indicate certain subtypes of the prodromal progression of PD. 6 For example, in the “body-first” subtype, alpha-synuclein pathology and degeneration start in the peripheral autonomic nervous system, most likely the enteric nervous system, and spread upward to the sympathetic trunk and brainstem, whereas the “brain-first” subtype is associated with pathology starting in the olfactory bulb or amygdala, reaching the substantia nigra, and then spreading downward to the middle and lower brainstem, and eventually to the peripheral nervous system. Therefore, to identify subjects at risk of developing PD, it is necessary to evaluate not only RBD but also various non-motor and motor symptoms.
Our research group has been conducting a cohort study on at-risk subjects with PD who were identified through a questionnaire survey in a general population (the NaT-PROBE study).7–9 We reported that approximately 5.7% of healthy participants had ≥2 prodromal symptoms, and they were defined as high-risk subjects for PD. 7 At the baseline evaluation, approximately one-third of the high-risk subjects presented with abnormalities on either MIBG myocardial scintigraphy or DaT-SPECT. Those with abnormal MIBG myocardial scintigraphy results presented non-motor symptoms such as lower OSIT-J scores and higher RBDSQ scores, and those with abnormal DaT-SPECT results presented higher MDS-UPDRS III scores, 8 indicating that this cohort included various subtypes of prodromal PD. This cohort also focused primarily on non-motor symptoms because self-report questionnaires to evaluate subtle motor symptoms had not been established.
In the present study, we created a questionnaire on subjective subtle motor symptoms related to parkinsonism. We evaluated the utility of the questionnaire for assessing subtle motor symptoms at the prodromal stage of PD by conducting a questionnaire survey on health checkup examinees. We further conducted comprehensive evaluations, including DaT-SPECT and MIBG myocardial scintigraphy, in high-risk and low-risk subjects with prodromal PD as well as in patients with PD.
Methods
Screening Questionnaire for Subtle Parkinsonism (SQSP)
We created a self-administered questionnaire, the “Screening Questionnaire for Subtle Parkinsonism (SQSP)”, to investigate subtle motor symptoms associated with parkinsonism according to several prior studies10–13 (Table 1).
English version of the Screening Questionnaire for Subtle Parkinsonism (SQSP).

Study design and participants
The present study was done as a part of the Nagoya-Takayama preclinical/prodromal Lewy body disease (NaT-PROBE) study, which is a prospective, longitudinal, multicenter, and community-based cohort study coordinated by the Nagoya University School of Medicine. This study consists of two parts. In Study 1, we conducted a cross-sectional questionnaire survey on health checkup examinees to examine the distribution and validity of the SQSP in the general population and to investigate the relationship between the SQSP and the questionnaires on non-motor symptoms. On the other hand, in Study 2, we performed comprehensive cross-sectional evaluations on neuroimaging and motor, cognitive, and autonomic functions in the high-risk and low-risk subjects with prodromal PD who met the criteria defined below, as well as patients with PD to investigate the relationship between the SQSP and imaging findings or motor symptom.
Criteria of low-risk and high-risk subjects
In the NaT-PROBE study, since March 2017, healthy individuals aged ≥ 50 years who have undergone annual health checkups at Kumiai Kosei Hospital, Daido Clinic, or Chutoen General Medical Center in Japan have been surveyed via the following questionnaires on non-motor symptoms: the Japanese version of the Scale for Outcomes in Parkinson's disease for Autonomic Symptoms (SCOPA-AUT) for autonomic dysfunction; 14 the Self-administered Odor Question (SAOQ) for hyposmia; 15 the RBD screening scale (RBDSQ) for RBD; 16 the Beck Depression Inventory-Second Edition (BDI-II) for depression; 17 and the Epworth Sleepiness Scale (ESS) for excessive daytime sleepiness. 18 As previously reported, 7 individuals were categorized based on their scores on the SCOPA-AUT, the SAOQ, and the RBDSQ: those with ≥2 abnormal scores on the three questionnaires were classified into a high-risk group with prodromal PD, those without abnormal scores were classified into a low-risk group. The cutoff values were 10 points for the SCOPA-AUT, 90.0% for the SAOQ, and 5 points for the RBDSQ.
Study 1. Questionnaire survey for health checkup examinees
We obtained answers to the SQSP and the questionnaires on non-motor symptoms from 1497 individuals aged
Study 2. Comprehensive evaluations of low-risk and high-risk subjects in the NaT-PROBE study and patients with PD
We conducted comprehensive evaluations of low-risk and high-risk subjects undergoing annual evaluations, including motor and cognitive function examinations, physiological function tests, and isotope imaging tests, in the NaT-PROBE study and patients with PD. For low-risk and high-risk subjects, those who underwent evaluations from October 2022 to January 2024 were analyzed in the present study. There was no overlap between Study 1 participants and Study 2 participants. For patients with PD, those who visited Nagoya University Hospital from October 2022 to October 2023 and met the United Kingdom Parkinson's Disease Society Brain Bank Diagnostic Criteria 19 were evaluated. We excluded those with psychiatric disorders other than depression or neurological disorders, those with MRI abnormalities, and those who had experienced diseases that could affect motor symptoms within the last five years.
Questionnaires on non-motor symptoms
We utilized the following established questionnaires: the SCOPA-AUT to evaluate autonomic dysfunction, the SAOQ to evaluate olfactory dysfunction, the RBDSQ to evaluate RBD, the BDI-II to evaluate depressive symptoms, the ESS to evaluate excessive daytime sleepiness, and the Parkinson's Disease Questionnaire-39 (PDQ-39) to evaluate the PD-specific health-related quality of life.
Calculation of the total likelihood ratios for prodromal PD
We calculated the total likelihood ratios (LR) for prodromal PD using the updated version of the MDS Research Criteria for Prodromal Parkinson's Disease issued by the Movement Disorder Society in 2019. 4 In Study 1, among the defined risk and prodromal markers, only those obtainable from health checkups and self-administered questionnaires were used in the calculation. Detailed information is provided in the previous report. 7 In Study 2, following prodromal markers were also included in the calculation: “Dopaminergic PET/SPECT clearly abnormal” defined as DaT-SPECT SBR below −2; “Subthreshold parkinsonism” defined as MDS-UPDRS III score (excluding rest and action tremor) greater than 6; “Olfactory loss” defined as OSIT-J score below 8; “Orthostatic hypotension (OH)” defined as Schellong test showing a systolic blood pressure drop of 20 mmHg or more, or a diastolic blood pressure drop of 10 mmHg or more; and “Global cognitive deficit” defined as Japanese version of the Montreal Cognitive Assessment (MoCA-J) score below 26. According to the MDS criteria, cases with a probability of 80% or higher were defined as probable prodromal PD.
Motor and cognitive function examinations
The Movement Disorder Society-Unified Parkinson's Disease Rating Scale (MDS-UPDRS) was scored by neurologists who were certified by the International Parkinson and Movement Disorder Society. The MoCA-J was used to assess general cognitive function.
Physiological function tests
CVRR was measured as previously described 20 to assess cardiac parasympathetic dysfunction. The Odor Stick Identification Test for Japanese (OSIT-J), a stick-type olfaction test consisting of 12 odorants typically familiar to Japanese people,21–23 was conducted to assess objective olfactory dysfunction.
Isotope imaging tests
DaT-SPECT imaging with (123I) FP-CIT and (123I) MIBG myocardial scintigraphy were performed to detect presynaptic dopamine neuronal dysfunction and assess postganglionic cardiac sympathetic denervation, respectively. DaT-SPECT and MIBG myocardial scintigraphy were performed as previously described. 8 DaT-SPECT was considered abnormal when a decreased specific binding ratio (SBR) or abnormal visual findings were observed. The reference values of Japanese volunteers were used to evaluate the decrease in SBRs. 24 MIBG myocardial scintigraphy was considered abnormal when early or delayed H/M ratios were <2.2. 25 For high-risk and low-risk subjects, the results of isotope imaging tests performed at the time of evaluation were used; for patients with PD, abnormalities in isotope imaging tests were judged according to previous examinations, and only those patients who had undergone DaT-SPECT at Nagoya University Hospital within two years of study enrollment were included in the analyses of SBRs and z-scores.
Statistical analysis
Numerical variables that demonstrated a normal distribution are presented as the means ± SDs, whereas those not normally distributed are presented as medians [IQRs]. For two-group comparisons of normally distributed numerical variables, the t test was used, and for multiple comparisons, analysis of variance (ANOVA) with the Tukey–Kramer post hoc test was used. For two-group comparisons of nonnormally distributed numerical variables, the Wilcoxon test was used, and for multiple comparisons, the Kruskal–Wallis test with the Steel–Dwass post hoc test was employed. Fisher's exact test was used for comparisons of categorical variables. Spearman's correlation test was used for correlation analysis of nonnormally distributed numerical variables. The Quade test was used for multivariate analysis of nonnormally distributed numerical variables. Logistic regression analysis was used for multivariate analysis of categorical variables. P values < 0.05 were considered statistically significant. The correlation coefficients (r) were interpreted as follows: > 0.8, ‘very strong’; 0.5–0.8, ‘moderately strong’; and 0.3–0.5, ‘weak’. All the statistical analyses were conducted using R version 4.2.0. Figures were generated using the R package ggplot2.
Results
Study 1. Questionnaire survey for health checkup examinees
Among the 1497 individuals, 1340 completed the SQSP and answered that they had not experienced diseases that could affect motor symptoms within the last five years. Of those, 1183 individuals completed all the questionnaires on non-motor symptoms (Supplemental Figure 1).
Table 2 shows the validity of the SQSP. The overall Cronbach's α coefficient for the SQSP demonstrated high reliability. Additionally, each item exhibited moderately strong or weak correlations with the total score, except for Q8 (resting tremor), which did not show an apparent correlation. The variance inflation factors indicated no issues with multicollinearity.
Item statistics and reliability of each SQSP question.
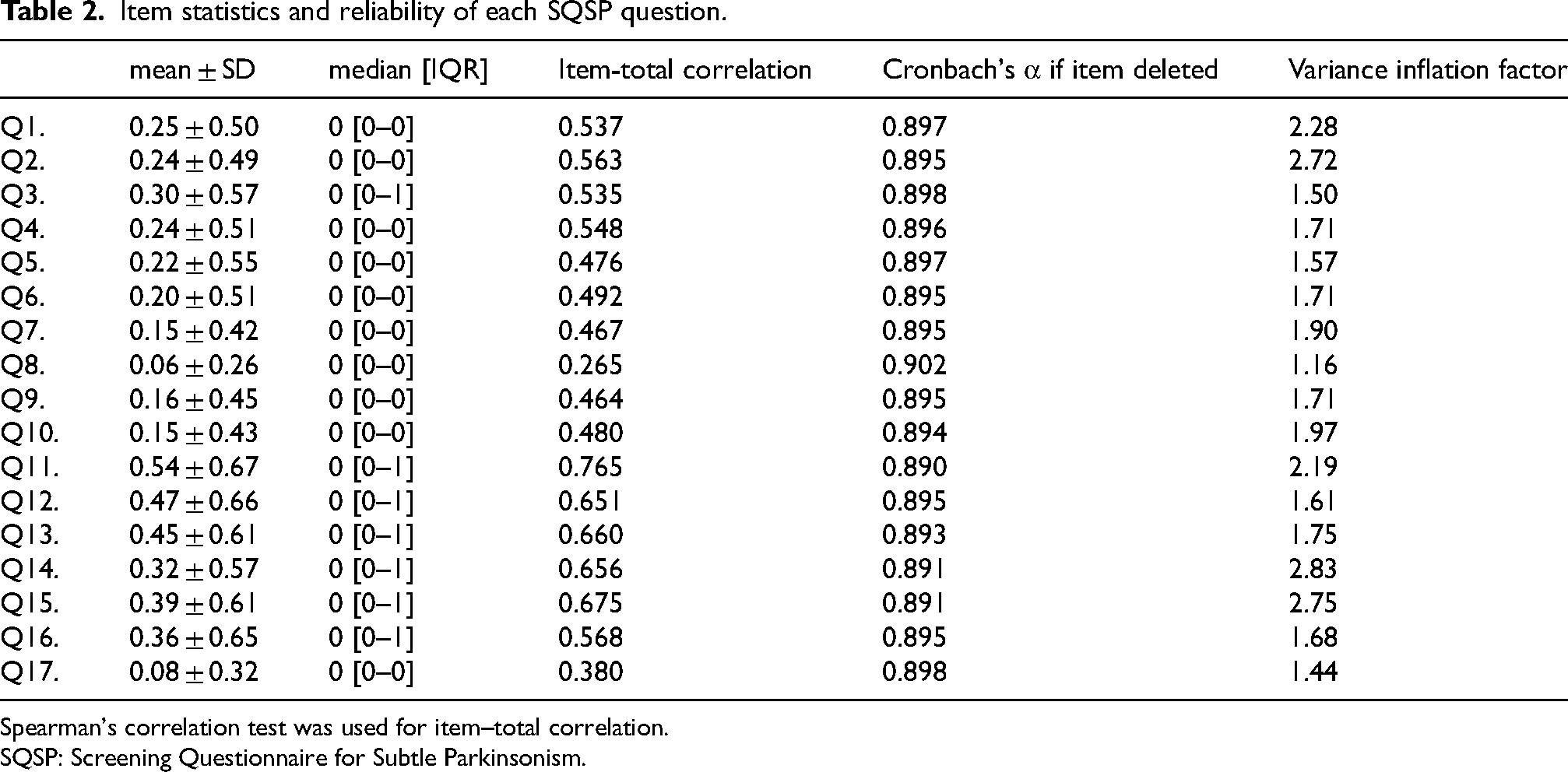
Spearman's correlation test was used for item–total correlation.
SQSP: Screening Questionnaire for Subtle Parkinsonism.
Among the study participants, SQSP scores were 0 in approximately one-fourth of the cases, showing a rightward decline, with a median of 4 [1–8], a mean of 5.5 ± 5.9, and a 90th percentile value of 13 (Figure 1(a)). No apparent correlation was found between SQSP scores and ages (Figure 1(b)). SQSP scores were significantly highest in the high-risk group, followed by the intermediate-risk group, which had one abnormal score on the three questionnaires such as the SCOPA-AUT, the SAOQ, and the RBDSQ, and then the low-risk group (Figure 1(c)). SQSP scores were moderately strongly correlated with SCOPA-AUT and BDI-II scores (Table 3).
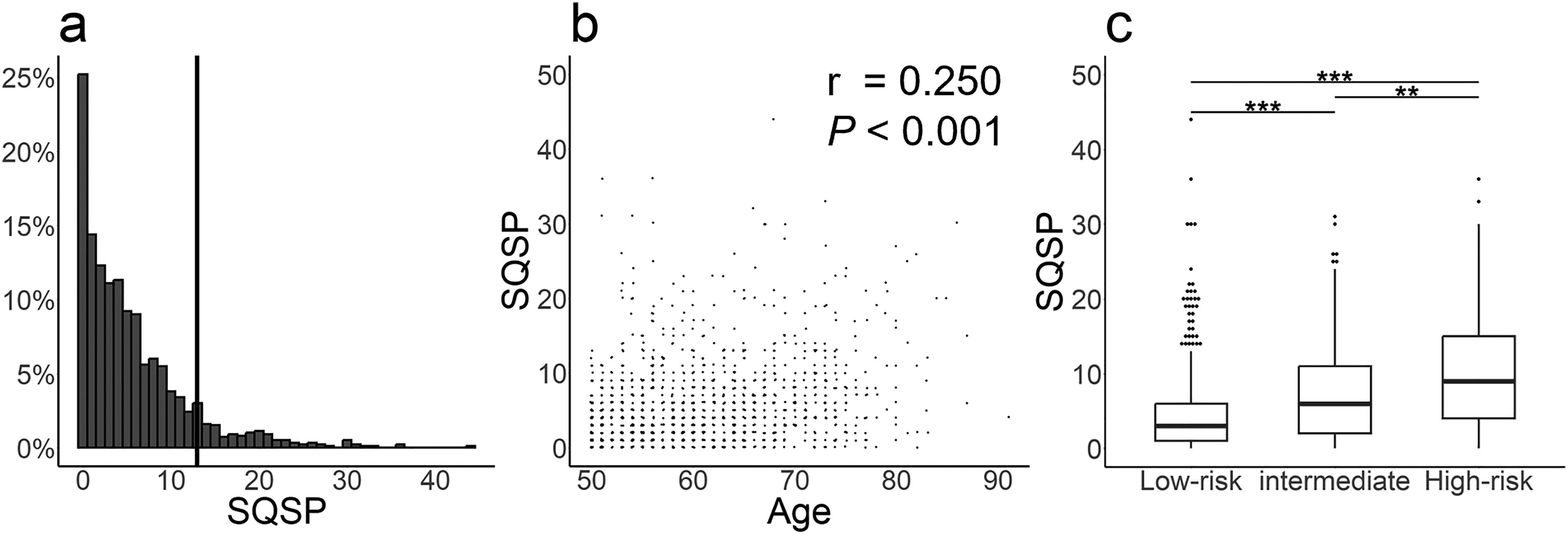
Distribution of SQSP scores in health checkup examinees. (a) Histogram of SQSP scores in individuals aged ≥ 50 years. The vertical line indicates the 90th percentile value. (b) Scatter plot of SQSP scores and ages.
Correlation matrix of the questionnaires on non-motor symptoms and the SQSP in health checkup examinees.

Spearman's correlation test with the Bonferroni post hoc correction was used. *p < 0.05, **p < 0.01, ***p < 0.001.
SCOPA-AUT: Japanese version of the Scale for Outcomes in Parkinson's disease for Autonomic Symptoms; SAOQ: Self-administered Odor Questionnaire; RBDSQ: RBD screening scale; BDI-II: Beck Depression Inventory-Second Edition; ESS: Epworth Sleepiness Scale; SQSP: Screening Questionnaire for Subtle Parkinsonism.
Table 4 shows comparisons between the low-risk and high-risk groups. Compared with the low-risk group, the high-risk group was older and had a greater proportion of males, individuals with a smoking history, and patients with type II diabetes mellitus. The high-risk group had significantly worse scores on all the questionnaires on non-motor symptoms than the low-risk group. SQSP scores and the LRs for prodromal PD were also significantly higher in the high-risk group. SQSP scores did not show an apparent correlation with the LRs for prodromal PD (r = 0.256, p < 0.001, Spearman's rank correlation). We next assessed whether the SQSP could predict whether each individual would be classified into the high-risk group. The AUC was 0.744 (0.6875–0.801), indicating moderate accuracy.
Demographics and scores of questionnaires on non-motor symptoms and the SQSP of the low-risk and the high-risk groups in health checkups.
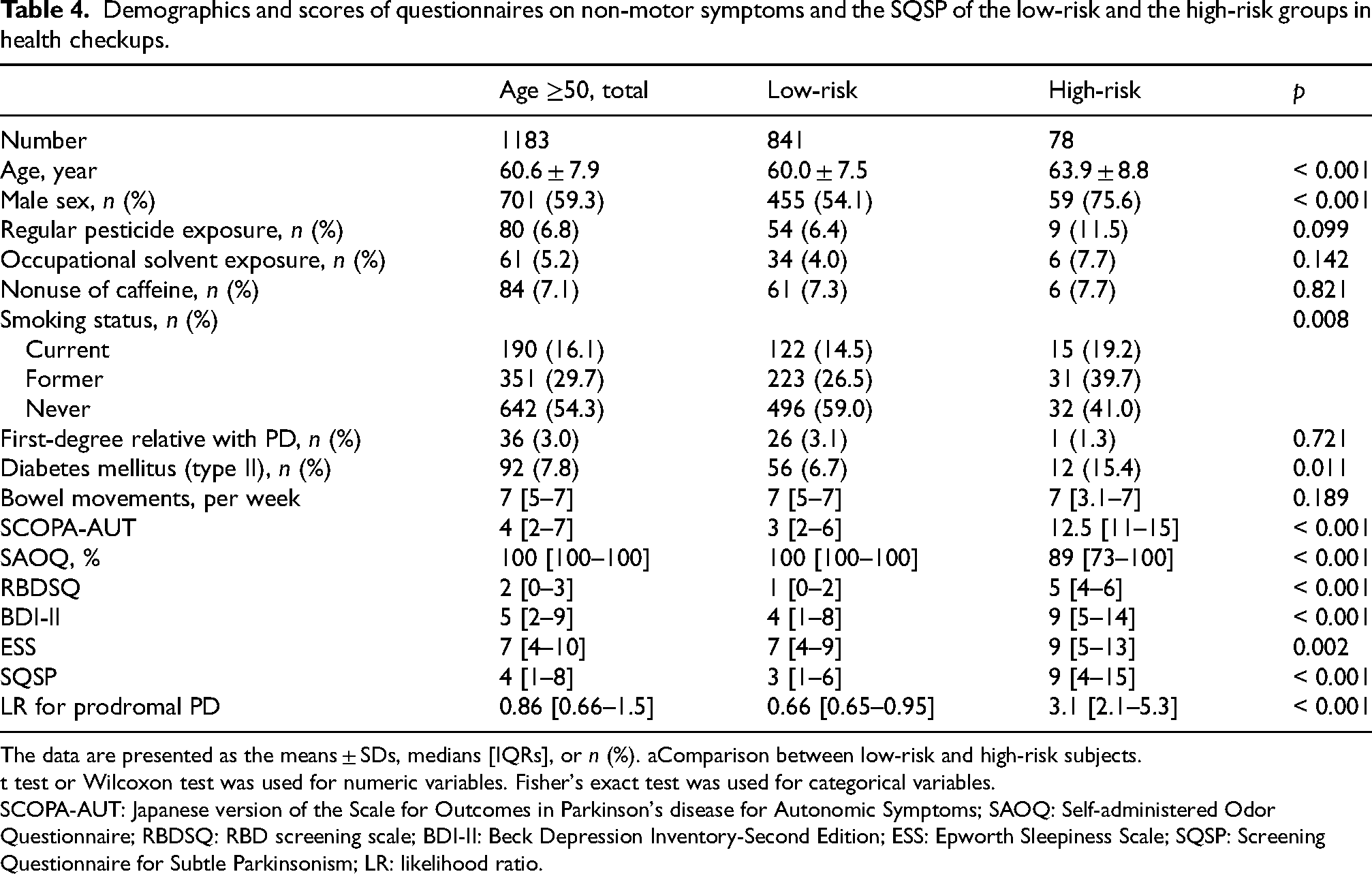
The data are presented as the means ± SDs, medians [IQRs], or n (%). aComparison between low-risk and high-risk subjects.
t test or Wilcoxon test was used for numeric variables. Fisher's exact test was used for categorical variables.
SCOPA-AUT: Japanese version of the Scale for Outcomes in Parkinson's disease for Autonomic Symptoms; SAOQ: Self-administered Odor Questionnaire; RBDSQ: RBD screening scale; BDI-II: Beck Depression Inventory-Second Edition; ESS: Epworth Sleepiness Scale; SQSP: Screening Questionnaire for Subtle Parkinsonism; LR: likelihood ratio.
Study 2. Comprehensive evaluations of low-risk and high-risk subjects in the NaT-PROBE study and patients with PD
Next, we conducted detailed evaluations, including motor and cognitive function examinations, physiological function tests, and isotope imaging tests, on low-risk and high-risk subjects in the NaT-PROBE study 8 as well as patients with PD. We enrolled 25 low-risk and 81 high-risk subjects and 35 patients with PD. After excluding those who had experienced diseases that could affect motor symptoms within the last five years, 24 low-risk and 71 high-risk subjects and 30 patients with PD were analyzed. Comparisons among the three groups are presented in Table 5. No significant differences in background information were observed among the three groups. Isotope imaging tests revealed that approximately one-third of the high-risk subjects had abnormalities on DaT-SPECT, and approximately one-fifth had abnormalities on MIBG myocardial scintigraphy, which was almost consistent with our previous reports.8,9 Approximately one-fifth of the high-risk subjects met the MDS research criteria for prodromal PD. Compared with the low-risk subjects, the high-risk subjects and the patients with PD had significantly worse scores on all the questionnaires on non-motor symptoms except for the ESS, where no significant differences were observed between the low-risk subjects and the patients with PD. Additionally, there were no significant differences between the high-risk subjects and the patients with PD. SQSP scores were significantly highest in the patients with PD, followed by the high-risk subjects and the low-risk subjects. The MDS-UPDRS II, the MDS-UPDRS III, and the PDQ-39 summary index followed the same pattern. MoCA-J scores were significantly lowest in the patients with PD, followed by the high-risk and then the low-risk subjects. OSIT-J scores were significantly lower in the patients with PD than in the high-risk and the low-risk subjects.
Demographic and clinical characteristics of low-risk and high-risk subjects and patients with PD.
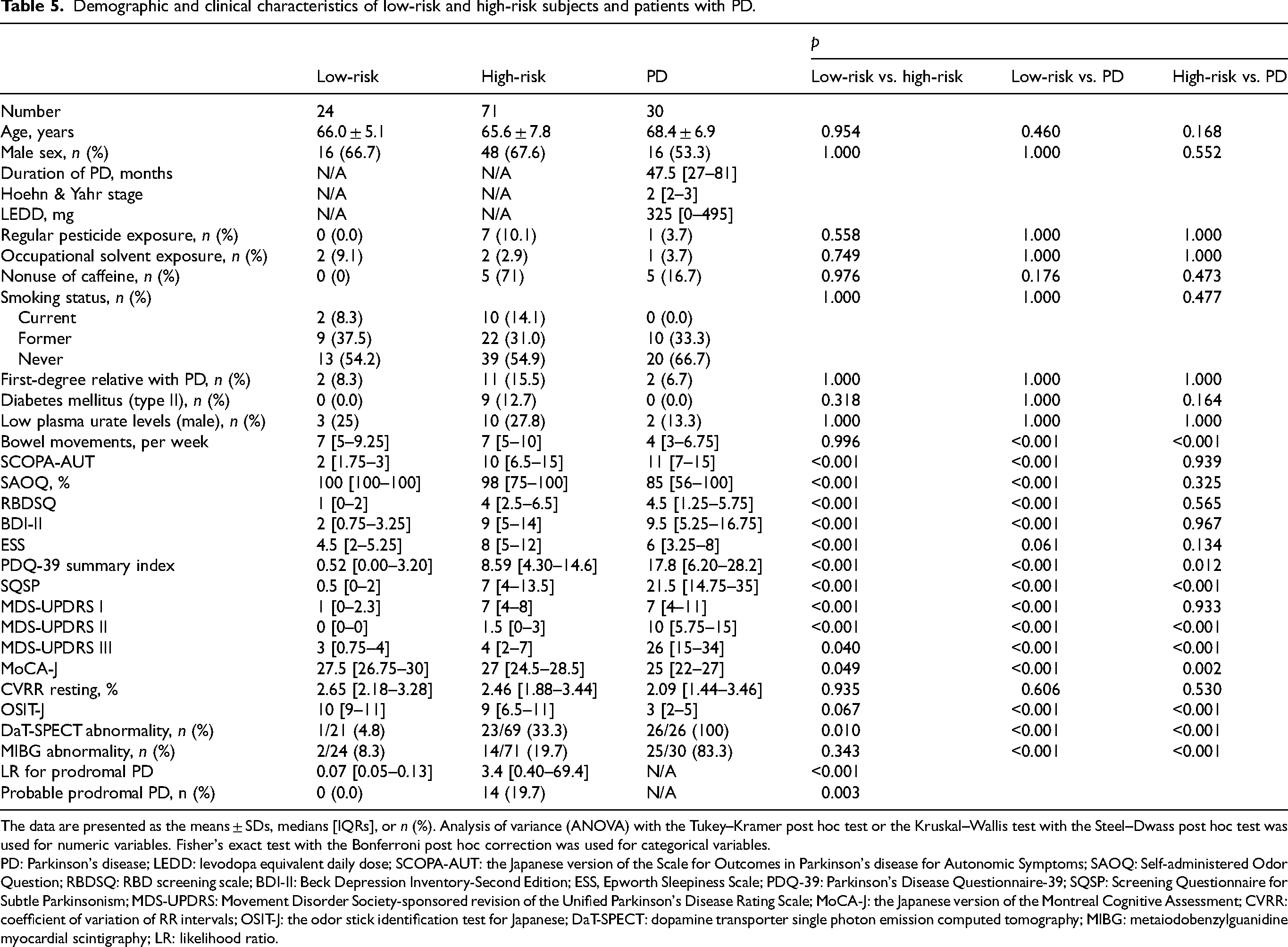
The data are presented as the means ± SDs, medians [IQRs], or n (%). Analysis of variance (ANOVA) with the Tukey–Kramer post hoc test or the Kruskal–Wallis test with the Steel–Dwass post hoc test was used for numeric variables. Fisher's exact test with the Bonferroni post hoc correction was used for categorical variables.
PD: Parkinson's disease; LEDD: levodopa equivalent daily dose; SCOPA-AUT: the Japanese version of the Scale for Outcomes in Parkinson's disease for Autonomic Symptoms; SAOQ: Self-administered Odor Question; RBDSQ: RBD screening scale; BDI-II: Beck Depression Inventory-Second Edition; ESS, Epworth Sleepiness Scale; PDQ-39: Parkinson's Disease Questionnaire-39; SQSP: Screening Questionnaire for Subtle Parkinsonism; MDS-UPDRS: Movement Disorder Society-sponsored revision of the Unified Parkinson's Disease Rating Scale; MoCA-J: the Japanese version of the Montreal Cognitive Assessment; CVRR: coefficient of variation of RR intervals; OSIT-J: the odor stick identification test for Japanese; DaT-SPECT: dopamine transporter single photon emission computed tomography; MIBG: metaiodobenzylguanidine myocardial scintigraphy; LR: likelihood ratio.
SQSP scores were moderately strongly correlated with MDS-UPDRS II and MDS-UPDRS III scores (Figure 2(a), (b)) and weakly correlated with SBRs and z-scores on the lower side of DaT-SPECT (Figure 2(c), (d)). Although most of the high-risk subjects and half of the low-risk subjects had SQSP scores above zero, there was a floor effect in the MDS-UPDRS II; 26 of the 71 high-risk and 23 of the 24 low-risk subjects had a score of zero on the MDS-UPDRS II (Supplemental Figure 2a). Among 46 individuals with a score of zero on the MDS-UPDRS II (26 high-risk and 20 low-risk subjects), DaT-SPECT abnormalities were found in 6 (5 high-risk and one low-risk subject), who had significantly higher SQSP scores compared with the subjects with normal DaT-SPECT (7.5 [6.25–8] vs. 2 [0–4.25], p = 0.018) (Supplemental Figure 2b).
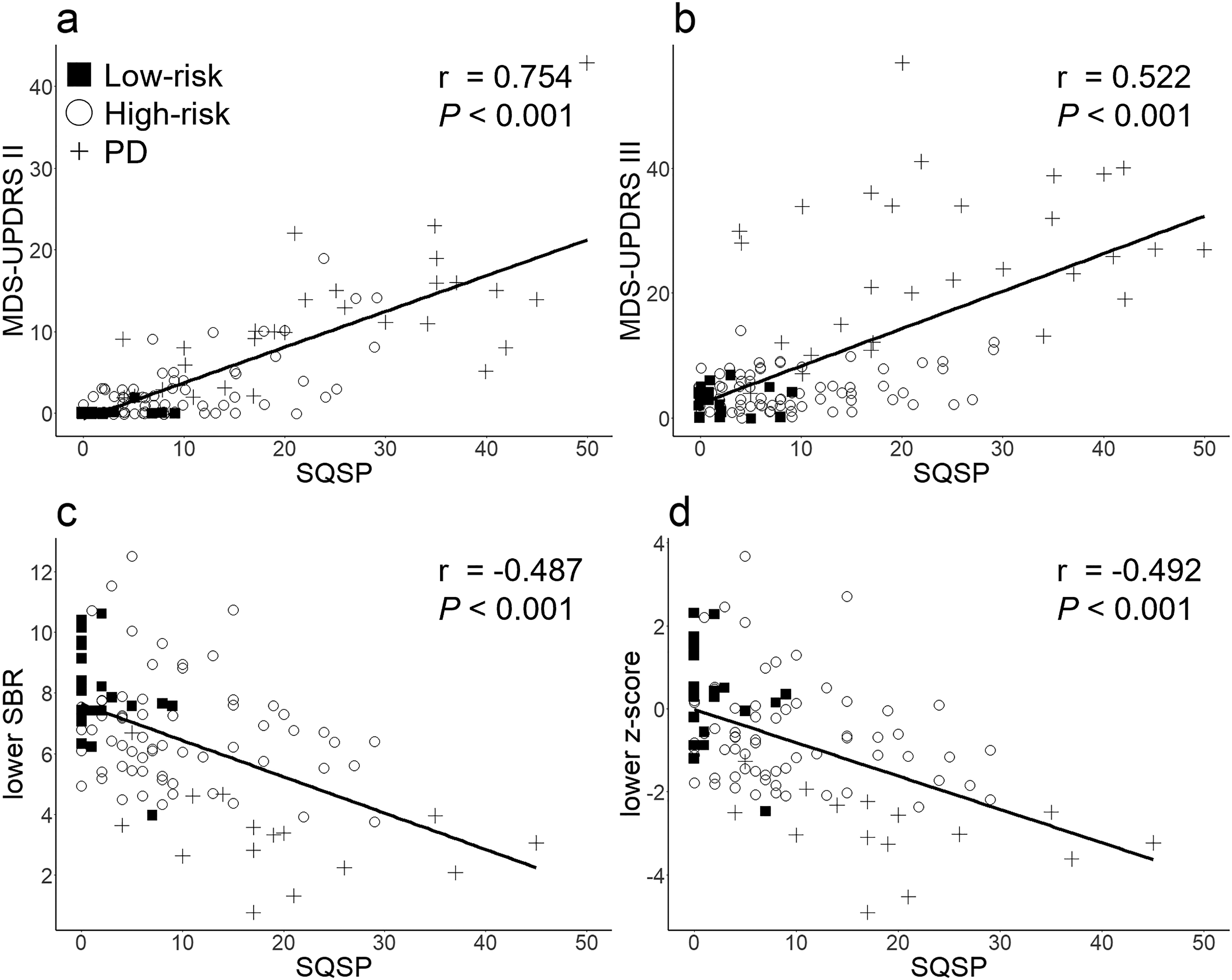
Correlation of the SQSP with the MDS-UPDRS and DaT-SPECT indices. Correlation between the SQSP and the MDS-UPDRS II (a), the MDS-UPDRS III (b), lower SBR of DaT-SPECT (c), and lower z-score of DaT-SPECT (d). SQSP: Screening Questionnaire for Subtle Parkinsonism; MDS-UPDRS: Movement Disorder Society-sponsored revision of the Unified Parkinson’s Disease Rating Scale; SBR: specific binding ratio; DaT: dopamine transporter.
In the high-risk subjects, SQSP scores showed moderately strong correlation with SCOPA-AUT scores and weak correlation with BDI-II and RBDSQ scores (Supplemental Table 1). High-risk subjects with abnormal DaT-SPECT were significantly older, had higher scores on the SCOPA-AUT, the BDI-II, the PDQ-39 summary index, the SQSP, the MDS-UPDRS II, the MDS-UPDRS III, and the LR for prodromal PD, and had significantly lower scores on the MoCA-J and the OSIT-J compared with those without DaT-SPECT deficits (Table 6). After adjusting for age and MIBG myocardial scintigraphy abnormalities, the high-risk subjects with abnormal DaT-SPECT had significantly higher scores on the SCOPA-AUT, the BDI-II, the PDQ-39 summary index, the SQSP, and the MDS-UPDRS II and significantly lower scores on the MoCA-J than those with normal DaT-SPECT.
Demographic and clinical characteristics of the high-risk subjects according to DaT-SPECT findings.
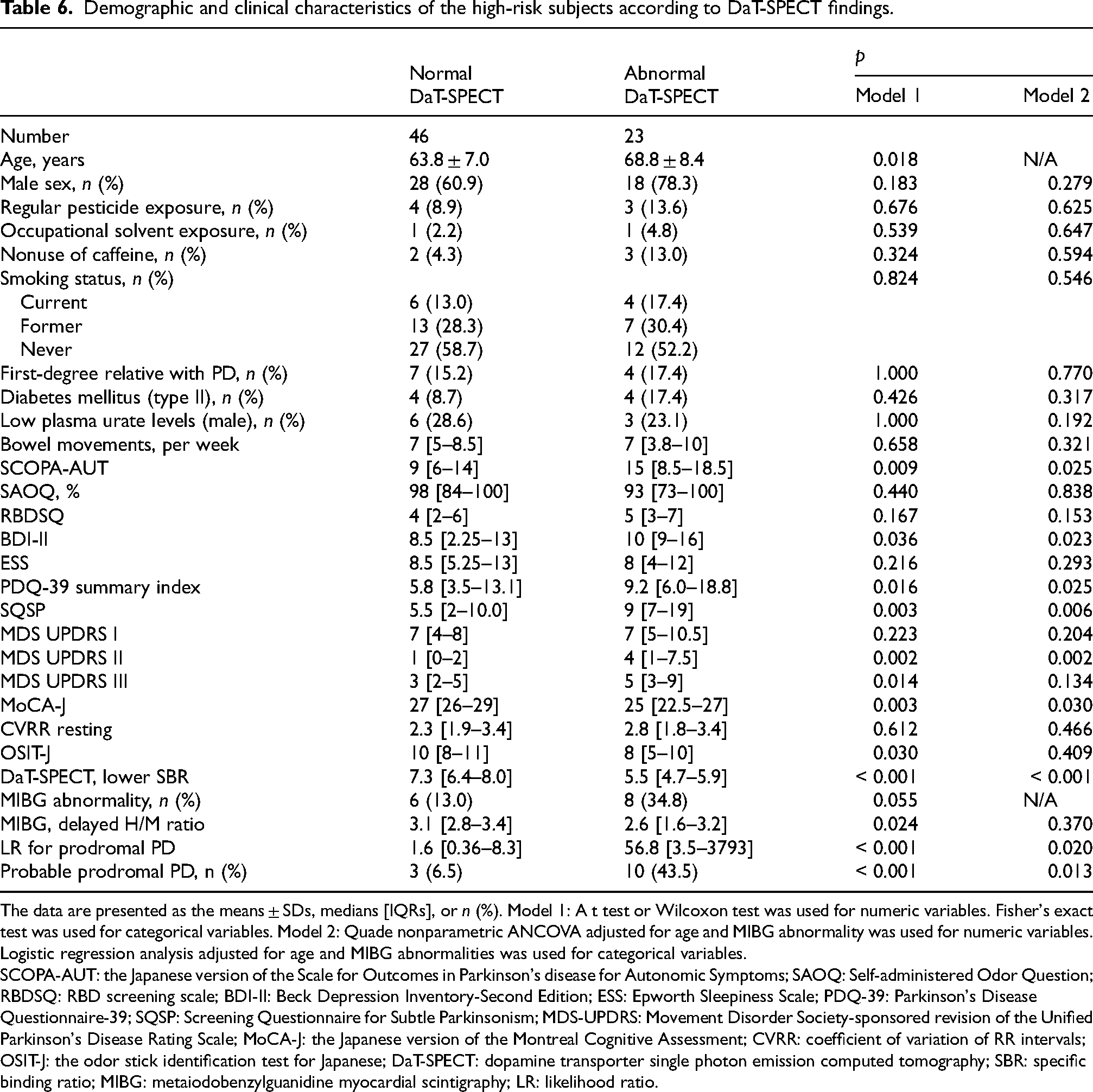
The data are presented as the means ± SDs, medians [IQRs], or n (%). Model 1: A t test or Wilcoxon test was used for numeric variables. Fisher's exact test was used for categorical variables. Model 2: Quade nonparametric ANCOVA adjusted for age and MIBG abnormality was used for numeric variables. Logistic regression analysis adjusted for age and MIBG abnormalities was used for categorical variables.
SCOPA-AUT: the Japanese version of the Scale for Outcomes in Parkinson's disease for Autonomic Symptoms; SAOQ: Self-administered Odor Question; RBDSQ: RBD screening scale; BDI-II: Beck Depression Inventory-Second Edition; ESS: Epworth Sleepiness Scale; PDQ-39: Parkinson's Disease Questionnaire-39; SQSP: Screening Questionnaire for Subtle Parkinsonism; MDS-UPDRS: Movement Disorder Society-sponsored revision of the Unified Parkinson's Disease Rating Scale; MoCA-J: the Japanese version of the Montreal Cognitive Assessment; CVRR: coefficient of variation of RR intervals; OSIT-J: the odor stick identification test for Japanese; DaT-SPECT: dopamine transporter single photon emission computed tomography; SBR: specific binding ratio; MIBG: metaiodobenzylguanidine myocardial scintigraphy; LR: likelihood ratio.
We also obtained family assessments of the SQSP for 12 of 24 low-risk and 55 of 71 high-risk subjects, using the same set of questions. Family assessments of the SQSP moderately strongly correlated with the self-assessments in both the low-risk and the high-risk subjects, regardless of the presence/absence of DaT SPECT deficit (Figure 3(a), (b)). The correlations between the SQSP assessed by family and indices of motor symptoms were weaker than those between self-assessed SQSP and the indices, though the family-assessed SQSP weakly correlated with MDS-UPRDS II scores in the low-risk and high-risk subjects (Figure 3(c)–(g)).
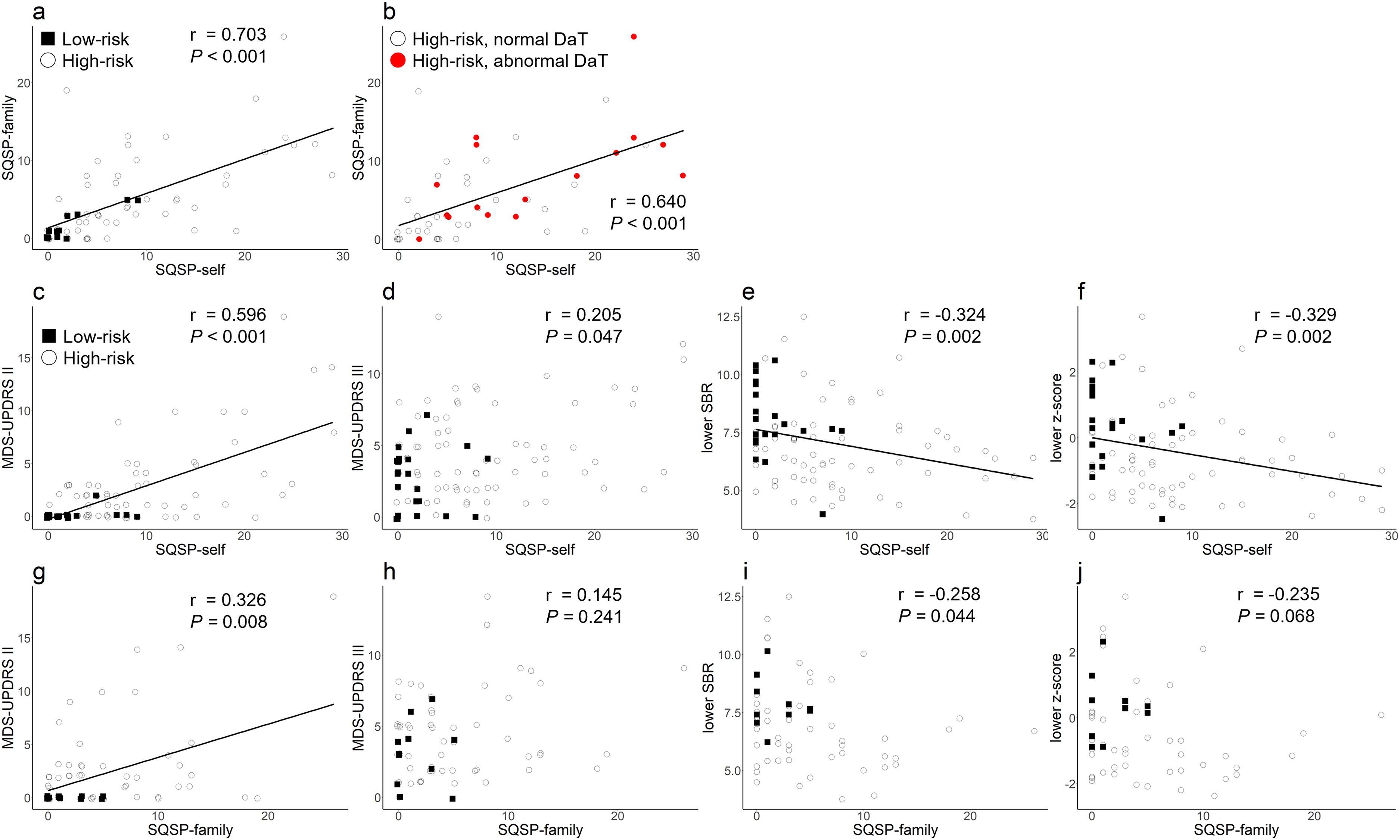
Family assessment of SQSP. Correlation between self-assessments and family assessments of the SQSP in low-risk and high-risk subjects (a) and only in high-risk subjects (b). Correlation between self-assessments of the SQSP and the MDS-UPDRS II (c) and the MDS-UPDRS III (d) in low-risk and high-risk subjects. Correlation between self-assessments of the SQSP and lower SBR of DaT-SPECT (e) and lower z-score of DaT-SPECT (f). Correlation between family assessments of the SQSP and the MDS-UPDRS II (c) and the MDS-UPDRS III (d) in low-risk and high-risk subjects. Correlation between family assessments of the SQSP and lower SBR of DaT-SPECT (e) and lower z-score of DaT-SPECT (f). SQSP: Screening Questionnaire for Subtle Parkinsonism; MDS-UPDRS: Movement Disorder Society-sponsored revision of the Unified Parkinson’s Disease Rating Scale; SBR: specific binding ratio; DaT-SPECT: dopamine transporter single photon emission computed tomography.
To investigate the relationship between each domain of SQSP and dopaminergic neuron degeneration, we defined the sum of Q1–4 of the SQSP as Voice & Face subscore, the sum of Q5–10 as Limb subscore, and the sum of Q12–17 as the PIGD subscore. The total score of the SQSP as well as all subscores but the Voice & Face were significantly higher in the high-risk subjects with abnormal DaT-SPECT compared to those without DaT-SPECT deficit, both in self-assessments and family assessments. The Voice & Face subscore was significantly higher in the high-risk subjects with abnormal DaT-SPECT only in the family assessments (Table 7).
Subscores of the SQSP by self-assessments and family assessments in high-risk subjects according to DaT-SPECT findings.

SQSP: Screening Questionnaire for Subtle Parkinsonism; DaT-SPECT: dopamine transporter single photon emission computed tomography; PIGD: postural instability and gait difficulty
Discussion
We developed a self-administered questionnaire, the Screening Questionnaire for Subtle Parkinsonism (SQSP), to assess subtle motor symptoms at the prodromal stage of PD. First, we showed that health checkup examinees experiencing non-motor symptoms of PD had higher SQSP scores than those without non-motor symptoms in Study 1. This finding is consistent with a previous report showing that subjective motor symptoms are associated with a greater probability of prodromal PD in older adults. 13 Further analysis in Study 2 revealed that the patients with PD had the highest SQSP and MDS-UPDRS II scores, followed by the high-risk subjects with ≥2 non-motor symptoms and then the low-risk subjects without non-motor symptoms. SQSP scores were correlated with MDS-UPDRS II and III scores as well as with SBRs and z-scores of DaT-SPECT. Additionally, among the high-risk subjects, those with abnormal DaT-SPECT presented higher SQSP and MDS-UPDRS II scores than those with normal DaT-SPECT. These findings from Study 1 and Study 2 complemented each other and suggested that the SQSP had potential as a tool for assessing subtle motor symptoms at the prodromal stage of PD and identifying prodromal PD cases in the general population.
Several studies have reported different measures to assess motor symptoms at the prodromal stage of PD: the MDS-UPDRS III via neurological examinations; 26 evaluation of walking and arm swinging via wearable devices;27,28 and remote analysis of tremor, finger tapping, walking, and vocalization via smartphone apps. 29 However, these methods are unsuitable for screening the general population, for which evaluating subjective motor symptoms via a self-administered questionnaire is considered more practical. Furthermore, no quantitative assessment tool has been developed to assess subjective subtle motor symptoms during the prodromal phase of PD. The SQSP is a short, easy questionnaire that can be used to screen subjects at risk of developing PD.
We also revealed that the high-risk subjects with abnormal DaT-SPECT had significantly higher SQSP and MDS-UPDRS II scores than those with normal DaT-SPECT. These findings indicated that both the SQSP, which was designed to detect subtle motor symptoms, and the MDS-UPDRS II, which has been used for evaluating motor experiences of daily living of patients with PD, were capable of assessing motor symptoms at the prodromal stage of PD. However, the MDS-UPDRS II, but not the SQSP, exhibited a floor effect among the high-risk subjects. Therefore, the SQSP may be a more sensitive tool for assessing motor symptoms at the prodromal stage of PD. Furthermore, we demonstrated that the assessments by family members were consistent with self-assessments. The DaT-SPECT deficit was associated with the subscore of SQSP for voice and facial expressions only in family assessments but not in self-assessments. Combining self-assessments with family assessments may enable more sensitive and earlier detection of prodromal PD cases, but longitudinal evaluations and larger-scale studies are required.
In addition to the SQSP and the MDS-UPDRS II scores, the high-risk subjects with abnormal DaT-SPECT presented higher scores on the SCOPA-AUT and the BDI-II compared with those with normal DaT-SPECT after adjusting for the MIBG myocardial scintigraphy H/M ratio, although RBDSQ, SAOQ, and OSIT-J scores were equivalent between the groups. Furthermore, among health checkup examinees, SQSP scores were moderately strongly correlated with SCOPA-AUT and BDI-II scores. These results were not entirely in line with the “body- and brain-first” theory. 6 Instead, our study suggested an alternative pattern in which motor symptoms and DaT-SPECT deficits appeared alongside peripheral autonomic dysfunction. These results seemed to be similar to a previous study reporting that prodromal PD cases increasingly experience anxiety and depressive symptoms, as well as laxative use, in addition to motor symptoms. 12 Depression symptoms in patients with PD are considered to arise from several mechanisms, such as an impairment of norepinephrine neurons in the locus coeruleus and subcoeruleus, serotonin neurons in the caudate nucleus, and limbic system structures, including the amygdala. 30 The results of the present study support the view that depressive symptoms at the prodromal stage stem from diverse pathophysiologies.
The following limitations should be considered in this study. We asked all the participants in Study 1 about their medical history and current diseases under treatment as part of the questionnaire study on health checkups and excluded patients with neurological disorders from the analysis. However, it was still possible that patients with undiagnosed PD may have been included. RBD was evaluated only with the RBDSQ, and polysomnography was not performed. The questionnaires were subjective and might be influenced by various biases. The study was cross-sectional and did not include longitudinal data from high-risk and low-risk subjects. Additionally, high-risk subjects may develop prodromal atypical parkinsonism, such as multiple system atrophy or progressive supranuclear palsy. Owing to the limited sample size of the low-risk group, it was difficult to identify “brain-first” prodromal subjects, who had high SQSP scores without non-motor symptoms, in our cohort. Genetic evaluation was not performed, so cases with different genetic backgrounds may exist. Therefore, further studies with longitudinal follow-up are warranted to establish the validity of the SQSP as a screening tool for prodromal PD.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X251361506 - Supplemental material for Utility of a newly created questionnaire on subtle motor symptoms in high-risk subjects with Parkinson's disease
Supplemental material, sj-docx-1-pkn-10.1177_1877718X251361506 for Utility of a newly created questionnaire on subtle motor symptoms in high-risk subjects with Parkinson's disease by Daigo Tamakoshi, Keita Hiraga, Taiki Fukushima, Takashi Uematsu, Takashi Tsuboi, Maki Sato, Makoto Hattori, Yuki Satake, Atsushi Hashizume, Masayuki Yamamoto, Hideaki Shimizu, Masakazu Wakai and Masahisa Katsuno in Journal of Parkinson's Disease
Footnotes
Acknowledgements
The authors thank Akihiro Murata at Nihon Medi Physics and radiological technologists at Nagoya University Hospital, Kumiai Kosei Hospital, and Chutoen General Medical Center. The authors also thank the Springer Nature Author Services team for English-language editing and the Simul International, Inc for the English translation of the SQSP.
ORCID iDs
Ethical considerations
This study was conducted in accordance with the 1964 Declaration of Helsinki and its later amendments, and the Ethical Guidelines for Medical and Health Research Involving Human Subjects endorsed by the Japanese government. The study protocol was approved by the Ethics Review Committee of Nagoya University Graduate School of Medicine (Nos. 2016-0238, 2016–0328, and 2017-0521).
Consent to participate
All participants provided written informed consent before participation in the study.
Consent for publication
All participants provided written informed consent for the use of their individual data collected in the study and for publication.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by AMED under Grant Numbers JP23ek0109652, JP 23lk0201124 and JP24wm0625104; JSPS KAKENHI Grant Number JP23H00420; and the Research Funding for Longevity Sciences (Nos. 22-26) from the National Centre for Geriatrics and Gerontology (NCGG), Japan.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data used to support the findings of this study are available from the corresponding author upon reasonable request, which includes the specification of a clear research question and confirmation of the approval from the Ethics Review Committee of Nagoya University Graduate School of Medicine.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
