Abstract
Individuals with isolated REM sleep behavior disorder (iRBD) are at high risk of developing α-synucleinopathies, particularly Parkinson's disease (PD) and dementia with Lewy bodies (DLB). With the development of potential neuroprotective treatments for synucleinopathies, including PD, identifying clinical features that can allow for tracking subtle changes in prodromal disease and thereby monitoring risk of phenoconversion in iRBD is paramount. Subtle motor deficits have been suggested to be present in iRBD, making them potentially important clinical markers for predicting future phenoconversion. This review aims to summarize existing literature that has investigated differences in motor function between iRBD and healthy individuals, as well as progression of motor decline in iRBD. 39 eligible studies were included in this review. The results suggest that quantitative motor assessments may be more sensitive to motor impairments in this population than clinical scales. Moreover, dual-tasking tended to unmask subtle motor deficits in individuals with iRBD, particularly in gait, balance, and tapping assessments. Longitudinal studies demonstrate that motor function worsens over time in iRBD, with earliest signs of motor deficits and clear progression in tapping assessments in particular. Larger longitudinal studies that use quantitative methods of motor assessments are needed to better characterize motor progression in iRBD, and confirm the reliability of different motor markers for predicting phenoconversion of iRBD into PD and other synucleinopathies.
Plain language summary
People with isolated REM sleep behavior disorder (iRBD) have a higher risk of developing Parkinson's disease (PD) and dementia with Lewy bodies (DLB). Currently, researchers are investigating different treatments to protect against the risk of developing these diseases in those with iRBD. To do this, it is important to identify markers that can help clinicians monitor progression of iRBD and assess the risk of developing PD or DLB. Studies have shown that individuals with iRBD exhibit subtle motor deficits before developing PD or DLB, which could suggest changes in motor function may be important signs to predict disease progression. This review aims to summarize existing research on motor deficits in iRBD compared to healthy individuals, and how motor function changes over time in iRBD. We found 39 relevant studies and included them in this review. Our findings suggest that tests that measure motor skills precisely, for example, with wearable sensors or motion capture camera, are better at capturing motor deficits in iRBD compared to tests commonly used in clinics. We also found that performing motor tests by adding a secondary task, for example, counting backwards by sevens while walking, revealed motor deficits in iRBD, particularly during walking, balance and finger and keyboard tapping tasks. The longitudinal studies in this review showcased that motor function declines over time in iRBD, but some of the earliest changes in motor function are in tapping assessments. Future research should involve larger study samples with longitudinal follow-up to improve our understanding of how motor function changes over time in iRBD and confirm whether these motor features can predict conversion of iRBD into PD and DLB.
Keywords
Introduction
Isolated REM sleep behavior disorder (iRBD) is a parasomnia characterized by a loss of muscle atonia during REM sleep and dream enactment, with an estimated prevalence ranging from 0.5% to 2.01% in the general population1–3. Polysomnography (PSG) is currently the gold standard for identifying iRBD. 4 Individuals with iRBD have a high risk of developing α-synucleinopathies, particularly Parkinson's disease (PD) and dementia with Lewy bodies (DLB).5,6 The estimated risk of phenoconversion is 33.5% after 5 years, 82.4% after 10 years, and 96.6% after 14 years. 6
For this reason, iRBD represents an important target for potential neuroprotective therapies, and an important model for understanding disease progression of prodromal PD. 7 Therefore, it is of paramount importance to establish biomarkers that can reliably identify and monitor subjects at high risk of rapid phenoconversion to PD. Imaging studies have demonstrated decreased striatal dopamine transporter uptake and substantia nigra hyperechogenicity as potential markers to identify individuals with iRBD at risk of phenoconversion within three years. 8 However, these imaging methods are relatively expensive and time-consuming, and may not be readily available in all healthcare settings. 4
Subtle motor deficits have been reported to be present in iRBD, and therefore, may be helpful markers for predicting future phenoconversion. Therefore, motor assessment methods may be relatively inexpensive tools that can aid in screening for iRBD and monitoring disease progression, particularly risk of phenoconversion. Several studies have aimed to characterize the motor impairment seen in iRBD using various motor assessment methods, however, to our knowledge, no review has aimed to summarize the existing evidence.
Therefore, the purpose of this systematic review was to a) understand what motor features best distinguish individuals with iRBD from healthy controls (HCs), and b) explore how motor features progress over time in iRBD compared to HCs.
Methods
This systematic review protocol was a priori published on PROSPERO (registration number: CRD42024509669) and guided by the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement and checklist. 9 The protocol has deviated from the a priori published protocol, specifically with regard to the inclusion criteria, as we have included studies that do not include healthy controls if they have a longitudinal design that measures motor function in participants with iRBD over time. This was done to appropriately address our second aim.
Search strategy and study selection
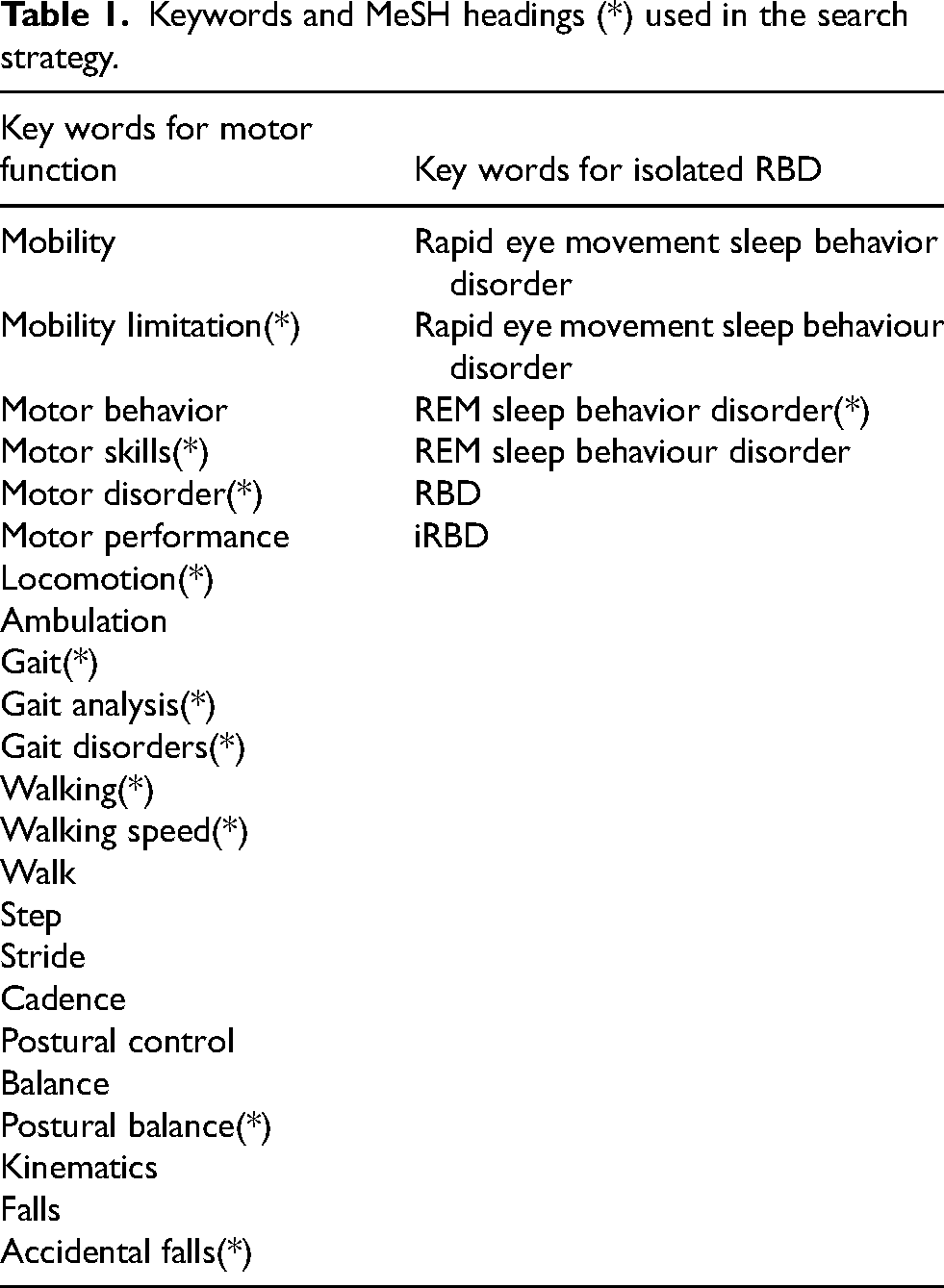
A detailed systematic search of the existing literature was performed in PubMed, Scopus, EMBASE, and CINAHL between February 2024 and April 2024. Keywords and Medical Subject Headings used to search the databases are listed in Table 1. There was no date limitation. All articles were imported into Covidence, an online systematic review software, and duplicates were removed. 10
Keywords and MeSH headings (*) used in the search strategy.
Two independent reviewers (SE and HH) conducted the process of study selection, starting with title and abstract screening to identify relevant studies, followed by full text review of relevant studies to identify studies that met the following eligibility criteria:
Study was conducted with human participants. Study includes participants with confirmed or probable isolated REM sleep behavior disorder, without PD or other neurodegenerative conditions. Study includes a healthy control group (for cross-sectional studies only). Longitudinal studies without a control group that follow individuals with RBD over time were included. Study includes a clearly described motor assessment and clearly defined motor outcomes.
Studies were excluded if they were not written in English or not published in peer-reviewed journals. Reviews, case studies, case series, conference proceedings, books, and dissertations were also excluded. Disagreement during the screening process was resolved by another reviewer (KEM). Finally, reference lists of relevant studies that met the eligibility criteria were manually screened to identify articles that were not captured in the electronic database searches.
Data extraction
Data extraction was performed in Covidence. The following information was extracted from eligible studies by SE and HH: authors, country, date of publication, journal of publication, inclusion and exclusion criteria, methods for diagnosing iRBD, sample size (% Female), mean age, follow-up duration (if applicable), cognitive test scores, motor assessment method(s), motor assessment outcome(s), and covariate adjustments. We additionally extracted mean and standard deviation (or median and range if mean and standard deviation were not reported) of the motor outcomes, as well as the results of the statistical analyses (e.g., p-values for group comparisons, or AUC, sensitivity, and specificity for discriminatory analyses). Data extracted were compared between both reviewers to ensure accuracy. Any disagreements were resolved by checking the study for the correct data.
Risk of bias/quality assessment
A quality assessment of each eligible study was conducted independently by two reviewers (SE and HH) using a modified Downs and Black checklist, which assesses reporting, external validity, internal validity, and power. 11 This form of risk of bias assessment has been previously used in systematic reviews focusing on motor assessments in various populations, including PD.12–15
The checklist was modified to exclude items that applied to intervention studies, and only items relevant for observational studies were used. 11 The items included were numbered 1, 2, 3, 5, 6, 7, 10, 11, 12, 16, 18, 20, 21, 22, 25, and 27, making the maximum possible score 16. Item 5 was modified so that principal confounders included age, gender, and cognitive function, which can impact motor behavior. Item 17 was also used for studies with longitudinal follow-up. 11 Items 17, 21, and 22 were not applicable to longitudinal studies without a control group. Item 25 was assessed in terms of statistical analyses for the motor function outcomes only, as this is the focus of our review. Disagreements were resolved through consensus between the two reviewers and inter-rater reliability was calculated. Scores were converted into percentages, and the quality of each study was classified as high (>66.8%), medium (33.4–66.7%) or low (<33.3%).
Results
Search strategy and study selection results
The results of the study selection process are summarized in Figure 1. Our electronic database search resulted in a total of 3363 articles. After duplicates were removed, 1905 studies remained for title and abstract screening. From these, we identified 77 relevant studies for full text review, five of which were identified from manual citation searching. After applying the eligibility criteria, 38 studies were excluded: 26 were excluded because they were conference abstracts (four of which reported on preliminary results from longitudinal studies), two were excluded for not including the results of their motor assessments, one for not clearly describing their motor assessment, and three for conducting the motor assessments in the iRBD group only, not the control group. Three studies had the wrong study design/aims for our review, for example, two compared iRBD groups that have converted to DLB or PD, while another subdivided and compared the iRBD group by cognitive performance. Two studies were excluded for the wrong patient population (one was a study in PD, and one recruited participants that did not have isolated RBD). Finally, one study was excluded as it was not written in English. Therefore, we identified 39 studies that were included in this review.16–54
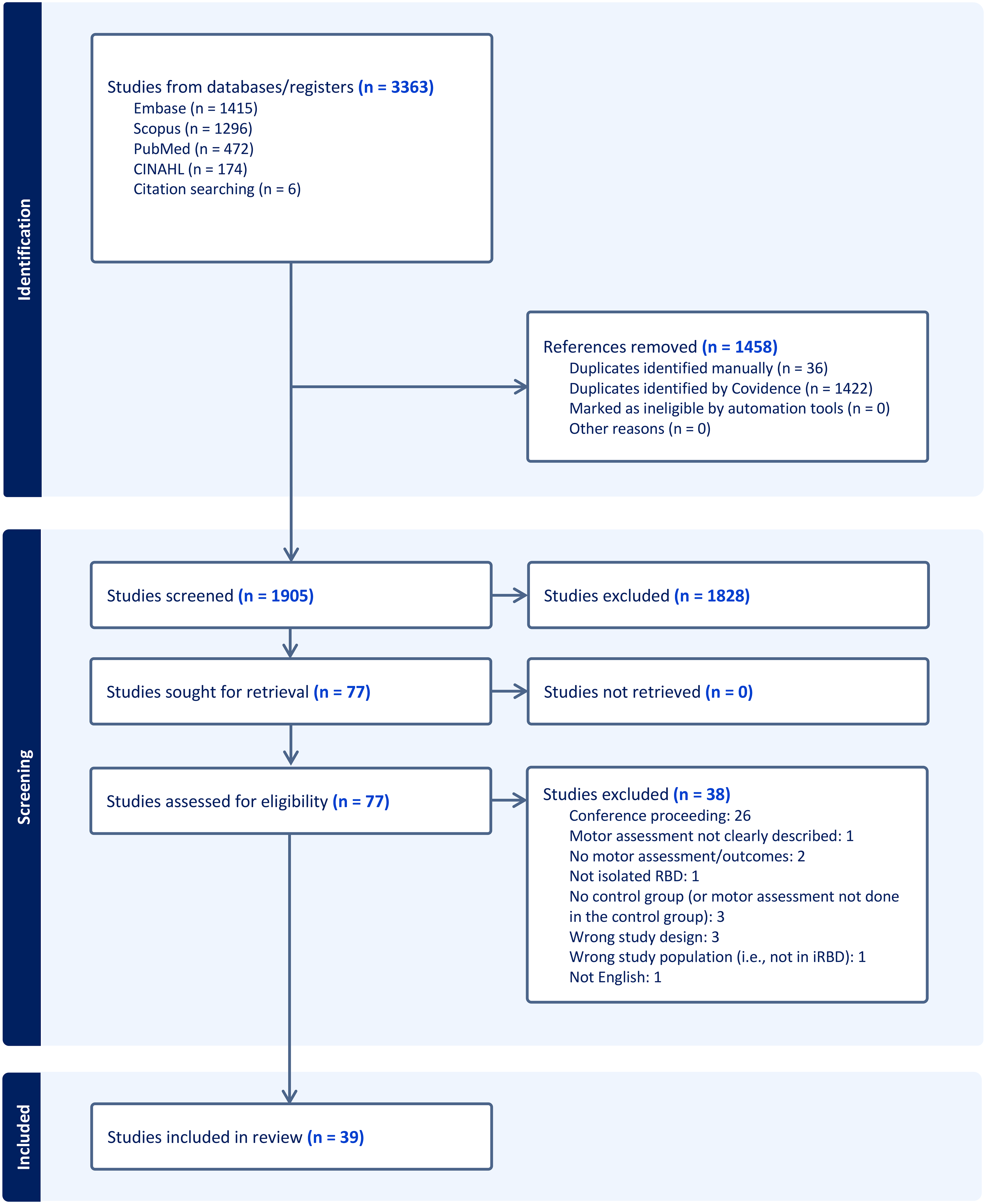
Flow diagram of search strategy and study selection results.
Cross-sectional results
All studies included in this review are summarized in Table 2 in terms of authors, year of publication, country, inclusion/exclusion criteria, method of iRBD diagnosis, motor assessment methods and outcomes, and main findings. Table 2 summarizes the results of all cross-sectional studies by motor assessments and whether group differences between iRBD and HCs were found or not. We have also included supplemental tables, which summarize study results by the type of motor assessment conducted.
Full summaries for included studies.


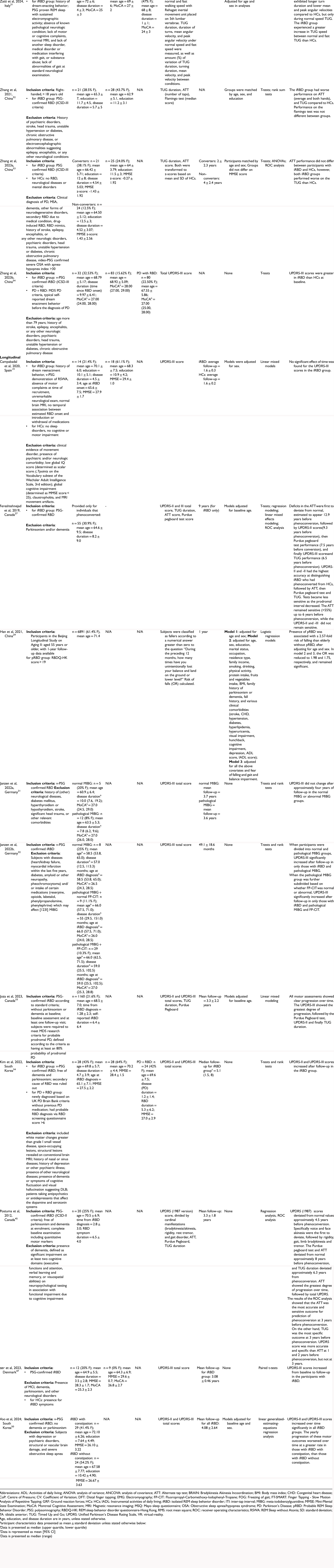
Abbreviations: ADL: Activities of daily living; ANOVA: analysis of variance; ANCOVA: analysis of covariance; ATT: Alternate tap test; BRAIN: Bradykinesia Akinesia Incoordination; BMI: Body mass index; CHD: Congenital heart disease; CoP: Centre of Pressure; CV: Coefficient of Variation; DFT: Distal finger tapping; EMG: Electromyography; FP-CIT: Fluoropropyl-Carbomethoxy-Iodophenyl-Tropane; FOG: Freezing of gait; FT-SMART: Finger Tapping - Slow Motion Analysis of Repetitive Tapping; GRF: Ground reaction forces; HCs: HCs; IADL: Instrumental activities of daily living; iRBD: isolated REM sleep behavior disorder; ITI: inter-tap interval; MIBG: meta-iodobenzylguanidine; MMSE: Mini-Mental State Examination; MoCA: Montreal Cognitive Assessment; MRI: Magnetic resonance imaging; MSQ: Mayo sleep questionnaire; OSA: Obstructive sleep apnea/hypopnea syndrome; PD: Parkinson's Disease; pRBD: Probable REM Sleep Behavior Disorder; PSG: polysomnography; RBDQ-HK: REM sleep behavior disorder questionnaire-Hong Kong, RMS: root mean square; ROC: receiver operating characteristics; RSWA: REM Sleep without Atonia; SD: standard deviation; TA: tibialis anterior; TUG: Timed Up and Go; UPDRS: Unified Parkinson's Disease Rating Scale, VR: virtual-reality.
Age, education, and disease duration are in years, unless stated otherwise.
Participant characteristics data presented as mean ± standard deviation unless stated otherwise below:
Data is presented as median (upper quartile, lower quartile)
Data is represented as mean [95% CI]
Data is presented as median (range)
UPDRS
Four studies assessed motor complications in daily living using the UPDRS-II.21,22,40,44 Of these studies, two studies found that UPDRS-II scores were greater in iRBD compared to HCs,40,44 and two did not find any group differences.21,22
The UPDRS-III was assessed by 20 studies.16,18,20–24,28,30,35–38,40,42–44,46,49,54 Of these studies, 14 reported that UPDRS-III scores were greater in iRBD compared to HCs,18,23,24,28,30,35,36,38,40,42,43,46,49,54 and five reported that UPDRS-III scores were not different between participants with iRBD and HCs.16,20–22,44 It is worth noting that in the studies that did not detect differences, motor abnormalities were detected through other motor assessments.16,20–22,44 Results of these studies are discussed in greater detail in later sections.
Some studies also performed discriminatory analyses to investigate the diagnostic accuracy of the UPDRS-II and III at discriminating iRBD from HCs, or iRBD that phenoconverted from HCs.36,37,46 One study reported that the UPDRS-III had good discrimination accuracy for iRBD from HCs. 37 Other studies showed the UPDRS-III has good accuracy at discriminating iRBD from HCs, however, it was less accurate than other quantitative motor assessments.36,46
Details of all studies that assessed UPDRS-II and UPDRS-III are summarized in Supplemental Table 1.
Tremor
Tremor was assessed and compared in three studies, which are summarized in Supplemental Table 2.17,21,22 One study used a smartphone with inertial measurement units to measure postural and rest tremor, which was found to be the most salient features that could discriminate iRBD from HCs. 17 Two other studies also found tremor, measured by the UPDRS-III, to be greater in iRBD than HCs.21,22
Tapping assessments
Several different tapping assessments were performed, namely the alternate tap test, finger-tapping assessments, and keyboard tapping tests, in a total of 11 studies.17,21,35,36,41,43,44,46,49,52,53 We subdivided tapping assessments into clinical assessments of tapping (e.g., UPDRS-III finger-tapping item, alternate tap test) and quantitative assessments (e.g., motion capture analysis). These studies are summarized in Supplemental Table 3.
Two studies assessed finger-tapping using the UPDRS-III finger-tapping item, with both finding that scores did not differ between groups.35,36 Additionally, six studies used the alternate tap test,41,43,44,49,52,53 four of which reported that iRBD performed worse than HCs,43,44,49,52 and two did not.41,53
Four studies assessed tapping quantitatively.17,21,36,46 One study used an alternate finger-tapping test on a smartphone, finding that finger-tapping task outcomes were among the most salient features that discriminated iRBD from HCs. 17 Another study found participants demonstrated more arrhythmic tapping during unpaced and paced tapping, as well as faster tapping in the unpaced test. 21 Another study used the UPDRS-III finger-tapping test instructions but assessed finger-tapping with a motion capture system, reporting that participants with iRBD exhibited amplitude decrements in their finger-tapping compared to HCs, which were not captured in the UPDRS-III finger-tapping item subscore. 36 It is also worth noting that the finger-tapping outcomes from the motion capture analysis exhibited better accuracy than the UPDRS-III. 36
Finally, another study also used motion capture to assess finger-tapping using the Slow-Motion Analysis of Repetitive Tapping (SMART), and two keyboard tests, namely the BRadykinesia Akinesia INcoordination test (BRAIN) and Distal Finger Tapping test (DFT). SMART refers to a video-based tool which tracks repetitive finger tapping that follows the UPDRS-III instructions. 46 This was done alone and with a secondary task of counting backwards by 3s. BRAIN requires participants to alternate between tapping the “s” and “;” keys on a keyboard with one index finger for 30 seconds, whereas DFT requires participants to repeatedly tap the down arrow key with their left index finger while depressing the left arrow key with their left middle finger for 20 seconds. Worse performance was found in iRBD compared to HCs under dual-tasking conditions, specifically participants with iRBD exhibited slower and more erratic tapping with lower amplitude than controls. 46 This study also found that participants with iRBD performed worse on the keyboard tapping tests as they had lower kinesia (less keystrokes), greater akinesia (greater dwell time when keys were depressed), and more incoordination in their keyboard tapping. 46
Reaction time
Reaction time was assessed and compared in two studies, which are summarized in Supplemental Table 4.17,41 One study used a smartphone application and found that reaction time outcomes were not among the most salient features that could discriminate iRBD from HCs. 17 On the other hand, another study evaluated reaction time using the falling stick test, and found it was significantly different between groups and demonstrated strong discrimination accuracy. 41 The falling stick test involves the participant catching a plunging stick as it was dropped by the examiner without any notice. The difference between the initial position of the stick and the position after griping the stick is measured and averaged across three trials for each hand.
Purdue pegboard test
The Purdue pegboard test was assessed and compared by seven studies, one of which used the Grooved pegboard test.18,23,37,41,43,44,49 These studies are summarized in Supplemental Table 5. Three studies found that participants with iRBD performed worse than HCs,23,44,49 three found that performance did not differ,18,41,43 and one reported the Purdue Pegboard test had poor discrimination accuracy between iRBD and HCs. 37
Balance assessments
Balance was assessed by eight studies total, and assessments included the Flamingo test,18,23,52 quantitative analyses of balance on a smartphone, 17 force plates, 41 a pressure sensor carpet, 24 and with wearable sensors. 20 One study used a dynamic balance test. 41 All studies are summarized in Supplemental Table 6.
Three studies used the Flamingo test, with only one finding that the iRBD group performed worse than HCs, 18 while the other two studies did not report a difference.23,52 The flamingo test evaluates the participant's ability to stand on one leg for a given amount of time. In two of these studies, participants were asked to stand on one leg for 30 seconds, while in the other study, participants were asked to stand on one leg for one minute.
Quantitative balance assessments were conducted by four studies.17,20,24,41 One study used a smartphone application and found that balance outcomes were among the most salient features that could distinguish iRBD from HCs. 17 One study used wearable sensors to measure postural sway parameters, finding that these measures were worse in iRBD compared to HCs under dual-tasking conditions (subtracting from 100 by 3s) with eyes open and eyes closed, as well as during tandem standing. 20 Another study assessed balance on a pressure sensor carpet and found only marginal differences between groups in root mean square (RMS) when standing with eyes closed. 24 Finally, a study used force plates and measured total excursion of center of pressure when standing in bipedal or unipedal stances, but no differences were identified between groups. 41
One study used a dynamic balance test where participants walked forwards and backwards on a straight line marked on the floor and the number of missteps were recorded. 41 Researchers found worse dynamic balance in iRBD when walking backwards, and that this test had good discrimination accuracy for iRBD from HCs. 41
Trunk mobility/axial signs
Trunk mobility and axial signs were assessed and compared in three studies,22,38,41 which are summarized in Supplemental Table 7. One study used the UPDRS-III to measure axial signs, reporting that participants with iRBD had significantly more axial signs than HCs. 22 Another study assessed trunk mobility during gait with wearable sensors, reporting that iRBD exhibited decreased trunk motion (reduced peak angular velocity and range of motion of the trunk) while walking at normal pace, fast pace, and while dual-tasking. 38 Finally, another study used a bend, twist, and touch test to assess trunk mobility, which required participants to stand with their back to the wall before bending forward to touch a mark on the floor, then standing up and touching a mark on the wall behind them for 20 seconds. The total number of taps were counted. Individuals with iRBD performed worse on this task than HCs. 41
Walking assessments
We subdivided walking assessments into clinical assessments (e.g., TUG, or other timed walk tests), and quantitative walking assessments (e.g., using pressure sensor walkways, or wearable sensors). All studies are summarized in Supplemental Table 8.
Twelve studies conducted clinical assessments of walking.18,23,35,37,41,43,44,46,49,51–53 Ten of these studies used the TUG.18,23,37,41,43,44,49,51–53 Of the studies that assessed TUG duration, seven reported that participants with iRBD took longer compared to HCs.18,23,43,44,49,52,53 Two studies did not find a difference in TUG duration.41,51 Additionally, two studies reported the TUG had poor discrimination accuracy for iRBD and HCs.37,41
Viteckova et al. had participants complete the TUG on a pressure sensor walkway under single-task, as well as cognitive (serially subtracting by 3s from 100) and motor (carrying a glass of water) dual-task conditions. They assessed gait characteristics in the pre-turn segment of the TUG, and did not find any group differences between iRBD and HCs in any gait outcomes. 48 They did not compare TUG duration. Zatti et al. measured TUG duration, as well as characteristics of turning in the TUG using wearable sensors. They found that participants with iRBD exhibited decreased step time before the turn, as well as decreased peak and mean angular velocity when completing the TUG at their fastest pace. 51
The remaining two studies that used clinical walking assessments used a 5-m walking test and a 10-m walking test (with single and dual-task conditions).35,46 The first study reported that participants with iRBD took longer to complete the 5-m walking test with less steps. 35 The second study found that participants with iRBD took longer to complete the 10-m walking test only when dual-tasking. 46
Eleven studies completed a quantitative gait assessment using either pressure sensor walkways, wearable sensors, or a smartphone.16,17,22–26,38,39,48,51 One of these studies focused on gait initiation, reporting deficits in anticipatory postural adjustments, the posterior shift of the center of pressure during the propulsive phase of gait initiation. 16
Four studies measured gait outcomes during single-task overground walking, either on a pressure sensor-walkway24,39 or with wearable sensors.22,38 Two studies assessed gait with a pressure-sensor walkway. 24 39 The first study did not find differences when walking at a normal pace, however, step length asymmetry was greater in iRBD compared to HCs in fast-paced walking. 24 The second study found that a diagnosis of probable RBD, based on the Mayo Sleep Questionnaire, was associated with reduced cadence, velocity, and stride length, and increased stride time variability, swing time variability, and double support time. 39 Cochen de Cock et al. found that gait outcomes measured using six wearable sensors discriminated iRBD from HCs with excellent accuracy, particularly range of motion and asymmetry in the limbs, peak swing velocity and asymmetry in the limbs, trunk range of motion, trunk peak velocity, and gait phase difference and cadence. 22 Ma et al. also measured gait using six wearable sensors, reporting that participants with iRBD exhibited decreased trunk motion (reduced peak angular velocity and range of motion) while walking and increased step time before turning compared to HCs. 38 Therefore, changes in gait that have been reported in iRBD compared to HCs under single-task conditions included decreased velocity and stride length, increased step time, decreased trunk motion (reduced peak angular velocity and range of motion), decreased cadence, increased swing time, and increased variability in swing time, stride time and double support time.
Five studies used dual-tasking with a quantitative gait assessment.22,24,25,38,48 Dual-tasking revealed additional subtle gait deficits in one other study, namely increased step width variability. 24 One study found decreased trunk motion (reduced peak angular velocity and range of motion), which was also seen in single-task walking. 38 One study did not find impaired gait under dual-tasking conditions, 48 and one study reported that dual-tasking did not improve discrimination accuracy of gait for iRBD and HCs. 22 It is worth noting that these studies used an easier secondary cognitive task (subtracting by 3s) compared to the secondary cognitive tasks used in the studies that did detect gait differences when dual-tasking (e.g., subtracting by 7 s, or the VR Stroop task).22,24,48
Ehgoetz Martens et al. utilized a virtual-reality (VR) gait paradigm while participants laid supine inside a magnetic resonance imaging (MRI) machine and navigated a virtual environment using foot pedals. In the environment, participants navigated an environment under a single-task condition, and a dual-task condition that was simple (reacting to a simple “STOP” or “WALK” cue) or complex (a Stroop task), while step time and step time variability were measured from the foot pedals. HCs increased their step time in response to the complex dual-task, but individuals with iRBD did not. 25
In another study, Ehgoetz Martens at al utilized a similar VR gait paradigm, but in this environment, participants navigated through wide and narrow doorways, Individuals with iRBD had longer step time and exhibited a greater increase in step time in response to HCs when navigating narrow doorways. They also had increased step time variability when navigating narrow doorways than HCs. 26
Finally, Del Din et al., used a free-living gait protocol whereby gait characteristics were measured remotely with wearable sensors over a seven-day period, and they found that participants with iRBD walked slower with decreased gait velocity variability and reduced cadence (increased step time, swing time, and stance time) compared to HCs. 23 Gait characteristics significantly discriminated iRBD from HCs, with swing time demonstrating the strongest accuracy. 23 iRBD also had shorter ambulatory bouts on average and a greater ratio of short ambulatory bouts to long bouts (i.e., decreased alpha). 23
One study reported that gait characteristics have very high accuracy at discriminating iRBD from HCs, particularly characteristics that represented limb range of motion and asymmetry, limb peak swing velocity and asymmetry, trunk range of motion, trunk peak velocity, and gait phase difference and cadence. 22 On the other hand, Del Din et al found that micro gait measures of pace, variability and rhythm collected from free-living gait distinguished individuals with iRBD from HCs, with swing time as the best discriminator. 23 In one study that used a smartphone to assess gait, gait features were among the most salient features that could discriminate iRBD from HCs, although they did not contribute the most, compared to other motor features. 17
Falls
Cross-sectionally, one study compared falls rate, which was measured by asking participants about the number of falls they had in the past 6 months, reporting it was greater in iRBD than HCs. 23
Multivariate analyses
Few of the studies also included analysis of the accuracy of a composite motor score or a motor battery at distinguishing iRBD from healthy controls.37,41,46 The first study calculated and validated a composite motor score from the scores of the UPDRS-III, TUG, and Purdue Pegboard test. 37 Their results demonstrated that the composite score had greater discrimination accuracy for iRBD from controls than the TUG and Purdue Pegboard test, however the UPDRS-III score alone was more accurate. 37
Two other studies conducted multivariate logistic regressions to assess the accuracy of a combined motor test battery.41,46 Nisser et al. included the falling stick test, the bend, twist and touch test, and the backwards line walking task in their model (as they significantly different between groups), but found that the combined model did not increase diagnostic accuracy. 41 Additionally the falling stick test was the only significant predictor of group status in the combined model. 41
Finally, Simonet et al. assessed the accuracy of a combined model that included their keyboard tapping test outcomes (BRAIN and DFT), change in 10-m walking time between single and dual task conditions, and variability in finger tapping amplitude during finger-tapping while dual-tasking. 46 This combined model demonstrated greater accuracy, sensitivity and specificity at distinguishing iRBD from HCs than the UPDRS-III alone. 46
Longitudinal results
Table 2 also summarizes longitudinal studies included in this review in terms of authors, year of publication, country, inclusion/exclusion criteria, method of iRBD diagnosis, motor assessment methods and outcomes, and main findings. Note that if a study had a longitudinal design but motor function was only investigated at baseline or a single time point, we summarized this result under cross-sectional results as any conclusions from these studies are about cross-sectional differences. Supplemental tables also summarize longitudinal result by motor assessment method.
UPDRS
The 1987 version of the UPDRS was assessed over time in iRBD by one study in this review. 45 The results demonstrated that this score deviated from normal approximately 4.5 years from phenoconversion. Researchers also divided the test by cardinal manifestations and location, finding that voice and face akinesia were the first to deviate, followed by rigidity, gait, limb bradykinesia and tremor. 45
UPDRS-II was assessed longitudinally in four studies in this review. All four studies showed that UPDRS-II scores increase over time in iRBD.27,33,34,50 One of the longitudinal studies in this review estimated that UPDRS-II scores deviated from normal 9.3 years before phenoconversion and detected significant differences from HCs three years before. 27
UPDRS-III was assessed longitudinally in eight studies in this review.19,27,31–34,47,50 Six of these studies demonstrated that UPDRS-III scores increase over time in iRBD,27,32,33,35,47,50 while two studies did not find an effect of time on UPDRS-III scores.19,31 Of the two studies that did not find a difference, one reported a mean follow-up duration of less than two years, which was the shortest of the eight studies, and the other had the smallest sample size.19,31
One longitudinal study reported that UPDRS-III scores in iRBD deviated from scores for healthy controls 5.6 years prior to phenoconversion. In this study, the UPDRS-II and III exhibited the highest accuracy at discriminating participants with iRBD who phenoconverted from HCs at phenoconversion, however these measures did not remain sensitive six years before phenoconversion (<50%). 27
These studies are summarized in Supplemental Table 10.
Tapping assessments
Two studies measured the alternate tap test longitudinally in iRBD.27,45 They are summarized in Supplemental Table 11. One study found that deficits in the alternate tap test were the earliest motor deficits in iRBD, estimated to deviate from normal 12.9 years before phenoconversion and significantly differing from HCs at 6 years before phenoconversion. 27 This study additionally found that the alternate tap test demonstrated excellent accuracy at phenoconversion and remained sensitive (>55%) up to 6 years before phenoconversion in discriminating between iRBD and HCs. 27 In line with this, the alternate tap test was also the earliest motor outcome to deviate from normal (8 years prior to phenoconversion) in the second study, showed the greatest degree of progression over time, and was the most accurate and sensitive outcome at distinguishing iRBD from HCs at 3 years before phenoconversion. 45
Purdue pegboard
The Purdue Pegboard test was measured longitudinally in iRBD by three studies in this review.27,33,45 These studies are summarized in Supplemental Table 12. All studies demonstrated that Purdue Pegboard performance worsens over time in iRBD.27,33,45 One study estimated that scores deviated from normal approximately 8 years before phenoconversion, while the other reported deviation from normal at 7.5 years prior to phenoconversion, with significant differences at four years prior to phenoconversion.27,45
Walking assessments
Three studies measured TUG performance longitudinally.27,33,45 They are summarized in Supplemental Table 13. All three studies demonstrated that TUG performance worsens over time in iRBD.27,33,45 However, one study noted that compared to the Purdue Pegboard and UPDRS-II and -III, the TUG had the smallest degree of progression in iRBD. 33 Two of the studies estimated that TUG performance in iRBD deviated from HCs 6.5 and 6.3 years prior to phenoconversion.27,45
Falls
One longitudinal study assessed fall risk over one year between people with pRBD (diagnosed using the REM Sleep Behavior Disorder Questionnaire-Hong Kong) and without pRBD, reporting a 2.57-fold increase in risk of falling compared to HCs, after adjusting for age and sex. This remained significant after adjusting for several demographic covariates, medical history, lifestyle factors, and previous history of falls (See Table 2 for full list of covariates), although the odds ratio was reduced to 1.57. 29
Results of quality assessment
The results of the quality assessment for each study are demonstrated in Figures 2 and 3, for cross-sectional and longitudinal findings, respectively. The intraclass correlation coefficient was calculated to be 0.6 (95% CI: 0.549, 0.647, p < 0.001), indicating moderate inter-rater reliability.
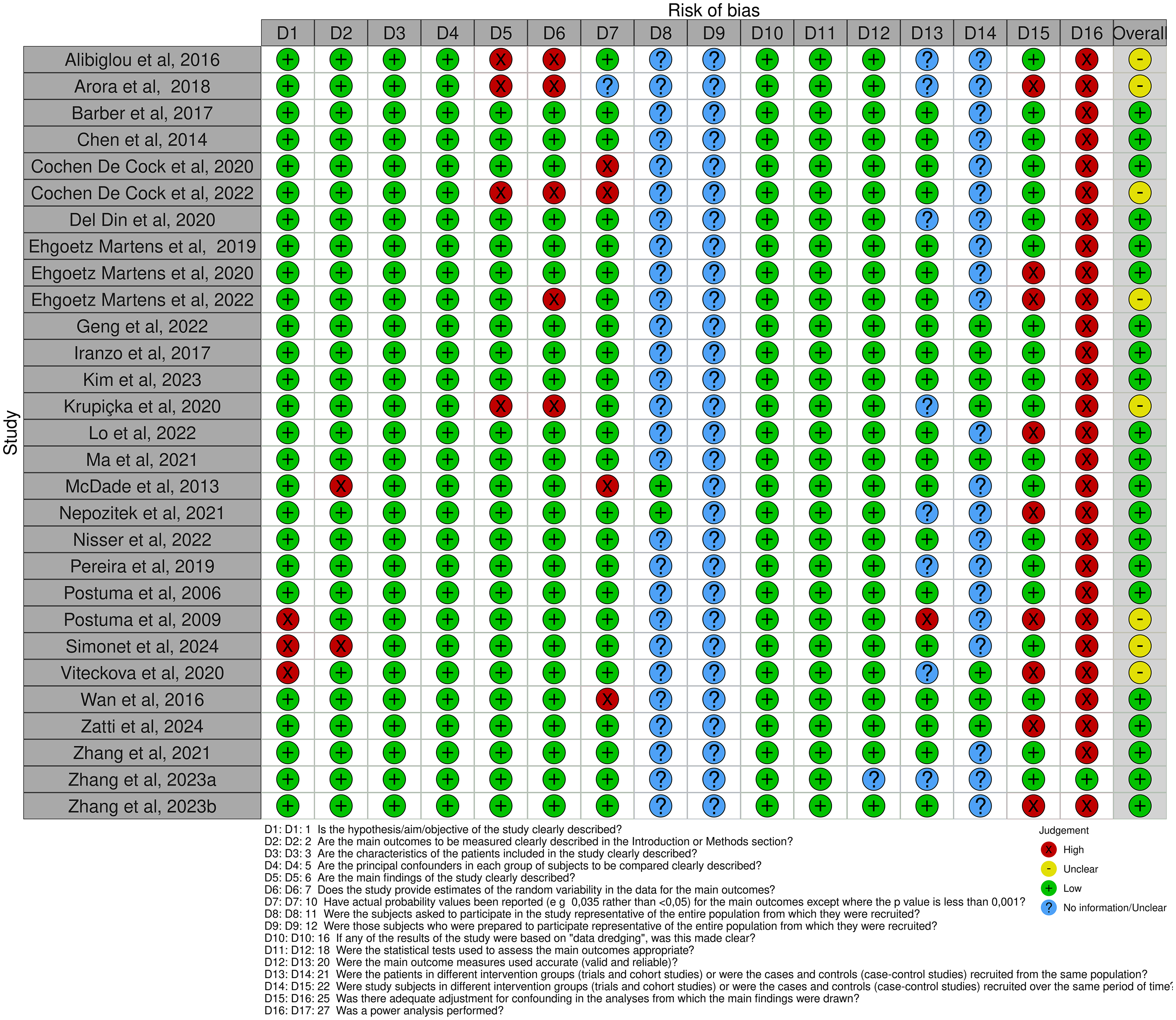
Summary of risk of bias assessment for cross-sectional studies, conducted using a modified Downs and Black checklist.
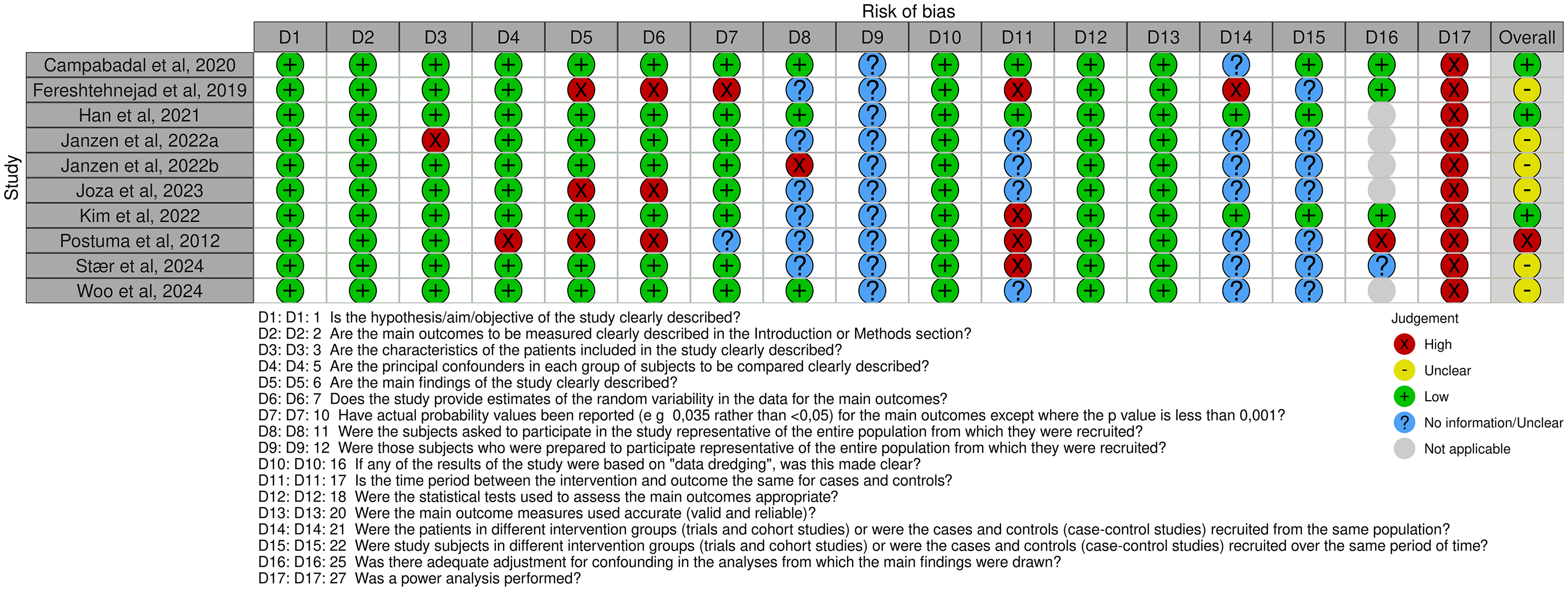
Summary of risk of bias assessment for longitudinal studies, conducted using a modified Downs and Black checklist.
Of the 29 cross-sectional studies in this review, 21 studies had low risk of bias and eight had medium risk of bias. Scores ranged from 68% to 94% for the 21 studies with low risk of bias, and 44% to 63% for the eight studies with medium risk of bias. Of the ten longitudinal studies, three had low risk of bias, six had medium risk of bias, and one had high risk of bias. Scores ranged from 70% to 81% for the studies with low risk of bias, and 41% to 56% for the studies with medium risk of bias. The study with high risk of bias scored 29%.
Prominent deficits in reporting included sample size justifications through power calculations, describing sampling methods, as well as proper identification of when and from where participants were recruited. 11 Longitudinal studies also often only followed the patient population (iRBD group) over time, and not the healthy controls. Item 12 of the checklist, which probes the representativeness of the study sample to the source population, was unclear for all the studies in this review as it required researchers validate that distribution of confounding variables was the same in the study's sample and the source population, which was not reported by any of the included studies. 11
Discussion
Interpretation of findings
In order to understand which motor features can distinguish individuals with iRBD from HCs and how motor function changes over time in iRBD, we identified 39 studies that compared motor function between iRBD and HCs, or in iRBD over time. We characterized these studies by the type of motor assessment conducted. In this section, we will interpret our findings and discuss clinical implications of this research, risk of bias in the included studies, and finally future directions.
Overall, the results support the presence of subtle motor impairment in iRBD, even when parkinsonian signs are absent or very mild. Similarly, longitudinal studies showcase that motor function deteriorates over time in individuals with iRBD. These studies also demonstrate that the alternate tap test is one of the earliest motor deficits in iRBD. 27 Most studies used clinical assessments, such as the UPDRS-III and the TUG, however, there is some inconsistency in the results, as some studies report they are different while other studies fail to detect a difference between participants with iRBD and HCs. Longitudinal studies, although fewer total, have more consistent results. This is not surprising, as cross-sectional studies may be affected by heterogeneity of iRBD so motor function may be variable across participants.
The UPDRS-II was assessed in fewer studies than the UPDRS-III and demonstrated more inconsistent results. The UPDRS-II focuses on activities of daily living and involves questions about daily tasks, self-care and social activities that are answered by the participant and/or their caregiver, whereas the UPDRS-III involves a detailed examination of motor features, including rigidity, tremor, bradykinesia, gait and postural instability, performed by the clinician. Individuals with iRBD more consistently performed worse on the UPDRS-III than the UPDRS-II, suggesting perhaps motor deficits are detected clinically but do not yet impact activities of daily living in participants with iRBD. On the other hand, in one study, UPDRS-II score was greater in iRBD than HCs, but not UPDRS-III. 44 Fereshtehnejad et al. also estimated that UPDRS-II scores deviate from normal earlier than UPDRS-III scores in participants with iRBD who later phenoconverted. 27 One possible explanation for this is because deficits in activities of daily living may also be impacted by non-motor symptoms of iRBD, which may not be detectable in motor functions evaluated by the UPDRS-III. Individuals with iRBD exhibit sleep dysfunction, but they also are at risk of mood disturbances, including anxiety and depression, which may impact daily tasks, and therefore UPDRS-II scores. 55
We also found that in several studies that reported that clinical assessments failed to detect motor deficits, participants with iRBD did exhibit motor deficits detected through quantitative motor assessments. Furthermore, studies that assess the diagnostic value of these clinical assessments find that they are often not the most accurate or sensitive at distinguishing these groups, particularly when compared to other quantitative motor assessments, such as the alternate tap test, or finger-tapping assessments.23,27,36,41
In two longitudinal studies, the alternate tap test was found to detect the earliest motor manifestation in iRBD, and the most accurate at distinguishing iRBD individuals who phenoconverted from HCs.27,45 The alternate tap test also remained sensitive up to six years before phenoconversion, while other clinical tests, namely the UPDRS-II and -III did not. 27 Similarly, the UPDRS finger-tapping performance was not different between iRBD and HCs in one study, but motion-capture of finger-tapping revealed deficits in finger-tapping, particularly in the amplitude of finger taps. 36 This research also found that the finger-tapping outcomes were more accurate at distinguishing iRBD from HCs. 36 This disconnect between clinical rating and quantitative measurement was also reflected in one study that assessed the TUG using wearable sensors to probe at deficits in turning, whereby they found that performance on the TUG (i.e., duration, as it is assessed clinically) did not differ between iRBD and HCs, however, deficits in turning in iRBD could be detected by the sensors. 51 Overall, this suggests that iRBD may exhibit subtle motor deficits that are not detected by clinical assessments, particularly ther UPDRS, potentially due to floor effects as they are designed for use in PD, and not in prodromal disease. The inconsistency in study results using clinical assessments may also be due to heterogeneity in disease severity and duration in the iRBD samples, whereby these tests may be better able to detect motor deficits when participants are close to phenoconversion, particularly to PD. This is further supported by the finding that the UPDRS-II and -III did not remain sensitive at six years before phenoconversion, compared to the alternate tap test. 27 This finding demonstrates that clinical scales developed for characterizing PD may be less able to capture motor deficits than quantitative methods, like the alternate tap test. Furthermore, it suggests that disease duration is an important confounder for these findings, as proximity to phenoconversion will impact changes in motor function and accuracy of different tests.
Interestingly, rhythm impairments in iRBD were consistently reported in studies that used tapping and gait assessments. These studies showed that iRBD have impaired cadence and rhythm during walking and arrhythmic finger-tapping, as well as increased incoordination in keyboard tapping tasks.21–23,46 Similar deficits in rhythm production have been reported previously in PD patients and may be linked to impairment in basal-ganglia-cortical circuitry that is involved in rhythm and timing.22,56,57 Therefore, it is possible that motor dysfunction in rhythm may be an early manifestation of degeneration of the basal ganglia prior to onset of parkinsonism.
Impairments in gait variability and asymmetry were also reported in people with iRBD compared to HCs. 22 ,23–26 To our knowledge, variability and asymmetry in finger-tapping behavior has not previously been investigated, but it would be of interest to explore if individuals with iRBD who have asymmetrical and variable gait exhibit similar deficits in their upper limb mobility or finger dexterity. Since motor asymmetry is a cardinal feature of PD, it may also be the case that asymmetrical impairment appears as iRBD duration and severity increases. Researchers have suggested that rhythm impairments precede motor impairment that is characteristic of PD, particularly asymmetry.21,22 It is also a possibility that asymmetrical impairment in iRBD precedes PD phenoconversion, as opposed to other synucleinopathies. The pattern of motor deficits in iRBD leading to phenoconversion should be characterized in future studies in order to elucidate the significance of different types of motor deficits.
There is also a trend of an additional value in using dual-tasking with motor assessments, particularly because this may reveal motor impairment that is not apparent during single-task walking.24,25,38 For example, one study revealed that postural sway parameters increased in iRBD when performing a dual-task during quiet standing, compared to quiet standing alone. 24 Consistent with this, two studies that assessed gait under single- and dual- task conditions found that dual-tasking uncovered impairments in gait that they did not detect when participants were performing motor tasks alone, without a dual-task.24,25 Similar findings were reported in finger-tapping. 46 The type and difficulty of dual-tasking appears to be important, as studies using less difficult dual tasks (e.g., serially subtracting by 3s, instead of by 7s) did not report differences in motor outcomes, or an added predictive value.22,48 It is now understood that walking is not a fully automatic task, rather gait and balance control involve cognition and have attentional requirements. During dual-tasking, attentional resources are divided between gait control and the secondary task. This divided attention may lead to changes in walking behavior as resources available for motor control and coordination are compromised. 58 On the other hand, it also appears that simply increasing task complexity can reveal impairments in motor function, for example, by walking at a fast pace or by walking through a narrow doorway.24,26 One study in particular reported that tasks that are more complex and may not be common in daily life (e.g., walking backwards) demonstrated the strongest accuracy at distinguishing iRBD from HCs when compared to clinical assessments or motor assessments that reflect common daily activities. 41 Future research should explore the type and difficulty of secondary tasks in dual-task paradigms, or complexity of tasks that best uncover the subtle motor deficits in iRBD, and their potential utility as tools to identify RBD and estimate risk of phenoconversion. This work is particularly important considering that individuals with iRBD that experience cognitive deficits are at higher risk of developing DLB, which is characterized by deficits in cognition, particularly attention and executive function. 59 Motor deficits being exacerbated during dual-tasking is important as it highlights that participants may be compensating for motor dysfunction using cognitive resources, which is impeded when a cognitive dual-task is introduced. Incorporating dual-tasking into motor assessments in clinical settings may be helpful for identifying early motor deficits in this group.
Another interesting finding in a few studies in this review is the potential for motor assessments in iRBD to be related to particular symptoms or subtypes of PD. For example, several studies found evidence of increased axial symptoms and reduced trunk mobility in iRBD compared to HCs, which suggests rigidity that is similar to what is seen in PD patients with the postural instability and gait disorder (PIGD) subtype of PD.21,38,41 Research has indicated individuals with RBD are more likely to develop the PIGD subtype. Similarly, other studies have found evidence of increased step time prior to turning, and gait initiation impairments that are similar to those seen in PD patients who experience freezing of gait (FOG).16,38 This may be an area of research that is worth investigating in future studies as this will help improve prediction of disease prognosis.
Finally, only a few studies investigated the accuracy of several motor tests or a motor battery at identifying iRBD from healthy controls. Although only one study found improved accuracy from a motor battery of tests, direct comparison of these results is difficult as different motor tests were combined in each study. Combining multiple motor measures can capture different aspects of motor impairment that are not easily captured in one motor test, highlighting the importance of considering a combination of motor assessments. Additionally, integrating motor assessments with non-motor markers, such as assessments of cognition, olfaction, or autonomic function, may further improve identification of iRBD and prediction of future phenoconversion. For example, Del Din et al. found that including the Sniffin' Sticks score, which is a measure of olfactory function, with their gait outcomes improved discrimination of iRBD from HCs, compared to the gait outcomes alone. 23
Strength of evidence
Based on the number of studies and consistency of results for each motor assessment, we organized studies from strongest evidence of motor deficits in iRBD to weakest evidence. We concluded there was strongest evidence of gait deficits, as walking was assessed in the greatest number of studies with relatively consistent findings. Clinical assessments such as the TUG, for example, did not always reveal motor deficits in iRBD and exhibited poor discrimination accuracy,37,41 however, studies using quantitative methods of evaluating gait, for example, wearable sensors, demonstrated more consistent findings of gait deficits in iRBD. In two studies, gait outcomes measured using wearable sensors demonstrated excellent discrimination accuracy between iRBD and HCs.22,23
We concluded that there was also strong evidence for tapping and balance assessments, as studies assessing these motor functions consistently reported motor deficits in iRBD compared to HCs. As mentioned previously, in one longitudinal study, the alternate tapping test was found to be the earliest motor deficit in iRBD, suggesting it may be an important screening tool for iRBD and monitoring proximity to phenoconversion. 27 Studies assessing balance quantitatively using force platforms and wearable sensors have demonstrated that individuals with iRBD exhibit deficits in balance that were not demonstrated by studies that using clinical assessments of balance, namely the Flamingo test.17,20,24,41
There was relatively weak evidence for other clinical assessments, particularly the UPDRS-III. While in many studies it was shown that individuals with iRBD have greater scores, it discriminated iRBD from HCs with reduced accuracy compared to other quantitative motor assessments.27,36,41
Falls, tremor, and trunk mobility or axial signs were only measured in few studies, however, they consistently demonstrated greater deficits in iRBD than HCs in all studies that measured them. We concluded there is also relatively weak evidence for these deficits due to the small number of studies that have assessed them. Additional research is needed to confirm that these are motor deficits consistently found in iRBD compared to HCs.
Finally, we concluded there was the weakest evidence for the UPDRS-II, reaction time and Purdue pegboard for identifying motor deficits in iRBD, as the existing studies have inconsistent findings, with an equal number of studies reporting deficits are present and absent. Additional research is needed to clarify and explain these inconsistencies.
Quality assessment
The results of our quality assessment revealed that most of the eligible studies had low risk of bias, while some had medium risk, and one had high risk. Overall, studies had good reporting of their aims, outcomes, and main findings. In the external validity section of the Downs and Black checklist, we consistently found that most studies did not adequately describe their sampling method or the source of the populations in the study, making it difficult to make conclusions about how well the sample in each study represented the source population.
Most studies were cross-sectional, limiting any conclusions that can be made about the progression of motor impairment in iRBD. One common source of risk of bias in the longitudinal studies included in this study is a different follow-up length between groups, whereby only the iRBD group had longitudinal follow-up, not the control group. Following both individuals with iRBD and HCs over time is important to distinguish changes in motor function due to aging, from those due to the neurodegenerative progression of iRBD. Most studies described and adjusted for potential confounders adequately, however, there were a few studies that reported differences in sex, or cognitive function, that were not adjusted for in their analyses. Few studies included RBD disease duration as a confounder, which is a limitation as this may affect results, considering longer disease durations may suggest greater proximity to phenoconversion, and therefore motor differences are more likely to be detected. Finally, some of the included studies did not assess cognitive function, which is important as this could potentially confound study findings, particularly if the subjects in the iRBD or HCs groups exhibit impaired cognition which may be indicative of latent neurodegeneration.
Finally, only one study adequately reported a sample size justification through a power calculation, which is important to allow readers to make conclusions about non-significant outcomes.
Clinical implications
Clinically, the findings from this review suggest that clinical scales such as the UPDRS-III are inconsistent when it comes to distinguishing individuals with iRBD from HCs, potentially due to floor effects. On the other hand, quantitative motor assessments may provide more useful tools that can reveal subtle motor deficits in iRBD, prior to the onset of parkinsonian signs. Additionally, incorporating dual-tasking in motor assessments can aid in revealing motor impairment in this population. Quantitative motor assessments may therefore be useful as a first line of assessment for phenoconversion risk, as they are relatively cost-effective, non-invasive, and easy to implement. This can be followed with more specific screening, such as neuroimaging (e.g., dopamine transporter imaging) assess neurodegeneration and phenoconversion risk. It is worth noting that not all methods of motor assessment are inexpensive or easy to implement, for example using gait walkways or force plates to investigate gait and balance, would require additional costs and complex set up. This highlights an opportunity for future research to investigate methods to assess motor function in this population that are more accessible for clinicians.
The presence of motor deficits in iRBD prior to phenoconversion suggest motor function may be a helpful marker of phenoconversion risk; however, further research is needed to characterize changes in motor function over time in iRBD, and how well specific motor deficits predict phenoconversion risk.
Recently, a new biological staging system for PD has been proposed, which incorporates subtle motor deficits, which do not involve functional impairment, in stage 2B of the disease. This further underscores the importance of motor markers in the prodromal stage of the disease, before clinical symptoms of PD are present. This makes them potentially important outcomes in clinical trials for disease-modifying treatments to monitor disease progression in early stages of the disease and in iRBD.
Future directions
Of the 39 studies in this review, we identified only ten studies that included longitudinal follow-up. One of our study aims was to characterize how motor deficits change over time in iRBD, however, due to the limited research, we are not able to make firm conclusions for this aim. It is clear that motor functions deteriorates over time in iRBD, but the existing longitudinal research has largely used clinical scales, most frequently the UPDRS-III, to measure motor functoin only. A variety of quantitative motor assessments should be included in future research to better capture the subtleties of motor progression in iRBD. Future studies should use repeated motor assessments in iRBD over time to uncover the pattern by which motor manifestations emerge as prodromal disease progresses, which will improve our ability to monitor phenoconversion risk. More longitudinal research is also needed to better assess how well motor assessments can predict future phenoconversion, as well as if they can distinguish PD and DLB converters. Recently, research by Postuma et al. highlighted that motor assessments such as the UPDRS-III, and quantitative motor assessments including the alternate tap test, Purdue Pegboard test, and TUG were predictive of phenoconversion in a large, multicenter cohort study of individuals with iRBD. This underscores the potential for motor deficits as biomarkers for identifying individuals at high risk of converting to overt α-synucleinopathies and highlights the importance of incorporating motor assessments in clinical practice and in the design of future neuroprotective trials in iRBD. 5
Another possibility that is worth investigating in future research is whether these motor assessment outcomes can allow prediction of specific subtypes or symptoms of PD. As mentioned above, some studies identified rigidity in the trunk in iRBD, which may develop into the PIGD subtype of PD.38,41 Other studies identified impairments in gait and turning that are similar to impairments seen in PD patients who experience FOG.16,38 Longitudinal research is needed to confirm whether these motor deficits develop into these subtypes or symptoms of PD. This would also help improve our prediction of phenoconversion and disease prognosis, which can have important implications for early intervention strategies.
Furthermore, as mentioned previously, only few studies conducted multivariate analyses to investigate the accuracy of composite scores or motor batteries, as opposed to individual tests. This is an important future direction to help identify combinations of motor features that could be useful predictors of progression towards phenoconversion.
Finally, it is likely that the variability in results between studies can be explained by heterogeneity between iRBD samples, meaning it is possible that samples in one study where motor deficits are found are closer to phenoconversion than samples in studies that do not detect any motor deficits in the same or similar motor outcomes. Currently, there is no method to estimate proximity to phenoconversion in iRBD. Therefore, an important next step in iRBD research is the need to characterize heterogeneity in this population, particularly by estimating disease severity or proximity to phenoconversion. While the RBD Symptom Severity Scale is a novel tool that has been validated for characterizing severity of RBD symptoms, it is not clear how well this correlates to risk of phenoconversion, making it an important next step for future research. 60 It is also important to note that because of the heterogeneity of iRBD, a comprehensive phenotyping approach is needed to better understand and predict phenoconversion risk. This should include not include only motor features, but also considerations of PSG findings, iRBD symptom duration, and other prodromal features, such as cognitive and autonomic function, neuroimaging, etc. This approach would allow for better risk estimation for phenoconversion, as well as predicting which specific synucleinopathy a given individual is at risk for.
It is also worth noting that motor deficits also exist in DLB, which indicates that subtle motor deficits detected in iRBD may still precede phenoconversion to DLB, and not PD alone. This may be another source of heterogeneity in iRBD samples, as they can convert to either PD or DLB. Future work should investigate whether motor deficits in iRBD can differentiate participants that will advance to PD and DLB, as well characterize the progression of these motor deficits to phenoconversion.
Limitations
A key limitation of our review is the absence of a meta-analysis, which would have clarified consistency of results and magnitude of effect sizes between studies. To our knowledge, this is the first review to summarize existing literature. Because of this, we intentionally included a wide range of motor assessments used in previous studies. This broader inclusion allowed us to capture the diversity of motor features in iRBD but hindered our ability to conduct a meta-analysis due to the heterogeneity in study design and motor outcomes assessed in each study. This further highlights a need to standardize motor assessment methods across different studies to improve generalizability of findings.
Furthermore, while this review aimed to characterize motor features in individuals with iRBD, we acknowledge that other motor features, namely speech and facial expression, were not included. Although several studies have examined speech changes in iRBD, we opted to exclude them from our review as they rely on different assessment methods than motor assessments focused on gait or limb movements, which would have made direct comparisons of findings difficult. Given that speech and facial expression have potentially important relevance for early disease detection in iRBD, future research should continue to characterize these changes, as well as how changes in speech relate to broader motor deficits in iRBD.
Conclusion
In conclusion, the existing literature supports the existence of subtle motor deficits in iRBD prior to phenoconversion, with the strongest evidence for walking, tapping, and balance deficits. There is inconsistency in the results of existing studies, which may be explained by heterogeneity in participants with iRBD. For this reason, an important next step is to develop a method of characterizing this heterogeneity and staging iRBD severity and proximity to phenoconversion. Longitudinal studies are needed to characterize the progression of motor impairment in iRBD to improve our understanding of prodromal disease progression and assess which motor assessments can best predict future phenoconversion. Gaining an understanding of which motor impairments can distinguish iRBD from HCs, and how motor function deteriorates in iRBD can reveal the utility of motor deficits as markers of disease progression and phenoconversion, which is particularly relevant for identifying useful biomarkers for clinical trials of neuroprotective therapies in iRBD.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X251359225 - Supplemental material for Motor features that distinguish isolated REM sleep behavior disorder patients from healthy controls: A systematic review
Supplemental material, sj-docx-1-pkn-10.1177_1877718X251359225 for Motor features that distinguish isolated REM sleep behavior disorder patients from healthy controls: A systematic review by Salma Elasfar, Hajr Hameed and Kaylena Ehgoetz Martens in Journal of Parkinson's Disease
Footnotes
Funding
This work was supported by Parkinson's Canada, the Canada Research Chairs program, and the National Science and Engineering Research Council.
Declaration of conflicting interests
The authors declare the following potential conflicts of interest: KEM has previously received funding from Weston Family Foundation, Michael J Fox Foundation, and Movement Disorders Society. KEM has also received support for speaking at the 2023 World Parkinson’s Disease and attending the 2022 International Society for Posture and Gait (ISPGR) and serves on the University of Waterloo Clinical Research Ethics Board, Executive Committee for International consortium for Freezing of Gait, and the ISPGR board. Remaining authors declare no conflicts of interest.
Data availability statement
Data sharing is not applicable to this article as no datasets were generated or analyzed in the current review.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
