Abstract
Identifying the coexistence of Lewy body (LB) pathology with Alzheimer’s disease (AD) in clinical practice is important in the era of anti-amyloid-β antibody therapy. However, few studies have predicted the presence of comorbid LB pathology with AD using indicative biomarkers of dementia with Lewy bodies or by collecting detailed clinical symptoms. We report the clinical progression of a 67-year-old patient diagnosed with AD who developed rapid eye movement sleep disorder-like symptoms and transient visual hallucinations 10 years after AD onset and was considered to have comorbid LB pathology based on imaging indicative biomarkers of dementia with Lewy bodies.
Keywords
INTRODUCTION
Alzheimer’s disease (AD) is the most common neurodegenerative disease and is pathologically characterized by the accumulation of senile plaques and neurofibrillary tangles in the brain. Patients with AD present with amnesia, executive function impairment, and visuospatial dysfunction [1]. In contrast, dementia with Lewy bodies (DLB) is pathologically characterized by the accumulation of Lewy bodies (LBs) in the cerebral cortex, brain stem, and autonomic nervous system [2]. Patients with DLB present with cognitive fluctuations, visual hallucinations, rapid eye movement sleep behavior disorder (RBD), and idiopathic parkinsonism [2].
AD-type pathology and LB pathology often coexist, and patients with this dual pathology experience increased visual and auditory hallucinations, delusions, and RBD compared to patients with pathological changes consistent with pure AD [3]. However, patients with dual pathology show fewer visual hallucinations and RBD than those with pure DLB, suggesting that when AD pathology and DLB pathology coexist, DLB-specific symptoms are less noticeable [3, 4]. However, there is currently no consensus on whether it is possible to predict co-occurring AD-type pathology and LB pathology based on clinical symptoms [5, 6], and thus, there is a possibility of false-positive or false-negative diagnoses [7].
The coexistence of pathological changes characteristic of both conditions is common in late-onset dementia; however, a recent study revealed that even in sporadic early-onset AD (EOAD) with an onset before the age of 65, more than half of the cases had coexisting LB pathology [8]. This coexistence of LB pathology with AD might affect the efficacy of anti-amyloid-β antibody therapy for these patients compared to that in patients with pure AD. Therefore, understanding both the clinical course and dynamics of indicative biomarkers of DLB in patients with AD and comorbid LB pathology is crucial. However, previous studies have been limited to retrospective pathological cohort studies with limited methods to capture clinical symptoms and no adaptation of biomarkers of DLB [5, 6]. Here, we report the clinical progression of a patient diagnosed with AD at the age of 67 years. This patient developed RBD-like symptoms and transient visual hallucinations 10 years after AD onset and was considered to have comorbid LB pathology based on imaging for indicative biomarkers of DLB.
CASE PRESENTATION
The patient was a 67-year-old, right-handed woman with an education of 16 years. She had a medical history of dyslipidemia and no familial history of dementia. In 2013, the patient developed amnesia. In 2014, she visited our hospital because of worsening amnesia and executive dysfunction, including the inability to drive a car or cook. Her Mini-Mental State Examination (MMSE) score was 25 points, Wechsler Memory Scale-Revised derived Verbal Memory Index score was 64, Visual Memory Index score was 86, General Memory Index score was 68, Attention/Concentration Index score was 72, and Delayed Recall Index score was <50, indicating amnesia, attention deficits, and visuospatial cognitive dysfunction. Magnetic resonance imaging (MRI) showed mild left medial temporal lobe atrophy (Fig. 1A). The degree of atrophy in the medial temporal lobes (Z-score), quantified using the voxel-based specific regional analysis system for AD (VSRAD) software (Eisai, Tokyo, Japan), was 1.33, indicating mild medial temporal lobe atrophy [9]. Technetium-99-ethyl cysteinate dimer ([99mTc]ECD) single-photon emission computed tomography (SPECT) revealed mild hypoperfusion in the right parietal lobe, bilateral posterior cingulate gyri, precuneus, and occipital lobes (Fig. 1B, C). The three indicators calculated using eZIS (PDRadiopharma Inc., Tokyo, Japan) that distinguish AD patients from healthy controls—severity, extent, and ratio—were 1.44, 20.36, and 6.62, respectively, and the three indicators were above the cut-off values, indicating that AD was likely [10]. In addition, although, the indicator suggesting the cingulate island sign (CIS), a characteristic of DLB, calculated using eZIS, was 0.22 (cut-off value: 0.28) and likely suggested CIS [11], there was no obvious CIS on the original images of brain perfusion SPECT. [11C]Pittsburgh Compound-B (PiB) positron emission tomography (PET) showed amyloid positivity (Centiloid scale, 45.4) [12] (Fig. 1D). The patient’s APOE genotype was ɛ3/ɛ3. She was diagnosed with probable AD dementia based on the diagnostic criteria [1]. The patient was administered donepezil (5 mg/day). In 2015, her cognitive impairment gradually progressed (MMSE score, 23 points), and she developed delusions of theft. In 2020, her cognitive impairment progressed further (MMSE score, 14 points), and she displayed notable apathy. She began to get lost in her home. In 2023, her husband reported that she had visual hallucinations of people outside the window and illusions, such as temporarily mistaking a streetlight for a person. The attending physician suspected comorbid LB pathology and asked about loud sleep-talking or constipation, confirming their occurrence. Her MMSE score was 4, which indicated severe cognitive impairment. She had no apparent parkinsonism (Movement Disorder Society-United Parkinson’s Disease Rating Scale part III score, 0 points). At this time, the patient was taking donepezil (5 mg/day) and memantine (20 mg/day). An MRI showed atrophy of the bilateral medial temporal lobes, including the hippocampus, predominantly in the left medial frontal lobes (Fig. 1E). The bilateral hippocampal heads showed lateral and downward slopes (i.e., “sloping shoulders sign”). This finding is characteristic of advanced-stage AD [13]. The Z-score quantified using the VSRAD software was 4.58, indicating severe medial temporal lobe atrophy [9]. [99mTc]ECD-SPECT revealed hypoperfusion in the left parietal lobe, bilateral posterior cingulate gyri, precuneus, left frontal lobes, and left medial and lateral temporal lobes (Fig. 1F, 1G). The three indicators calculated using eZIS that distinguish AD patients from healthy controls—severity, extent, and ratio—were 2.04, 50.86, and 3.28, respectively, and the three indicators were above the cut-off values, indicating that AD was more likely [10]. In addition, the indicator suggesting the CIS calculated using eZIS was 0.61 (cut-off value: 0.28) and suggested that CIS was unlikely [11]. PiB-PET revealed increased amyloid accumulation (Centiloid scale, 98.1) [12] (Fig. 1H). These neuroradiological findings indicated the progression of AD. In addition, as the development of visual hallucinations, RBD-like symptoms, and dysautonomia was suggestive of the coexistence of DLB, she underwent dopamine transporter (DAT) SPECT and 123I-metaiodobenzylguanidine (MIBG) myocardial scintigraphy to detect the indicative biomarkers of DLB. DAT-SPECT showed a marked decrease in DAT availability throughout the striatum (Fig. 1I). In addition, MIBG myocardial scintigraphy showed markedly reduced MIBG uptake (Fig. 1J). Although the visual hallucinations were transient, the RBD-like symptoms and positivity for two imaging biomarkers indicative of DLB fulfilled the diagnostic criteria for probable DLB [2]. This study was approved by the Ethical Review Committee of the Yamagata University Faculty of Medicine (Approval number: 2023-35). Written consent for the publication of this report was obtained from the patient and her family.
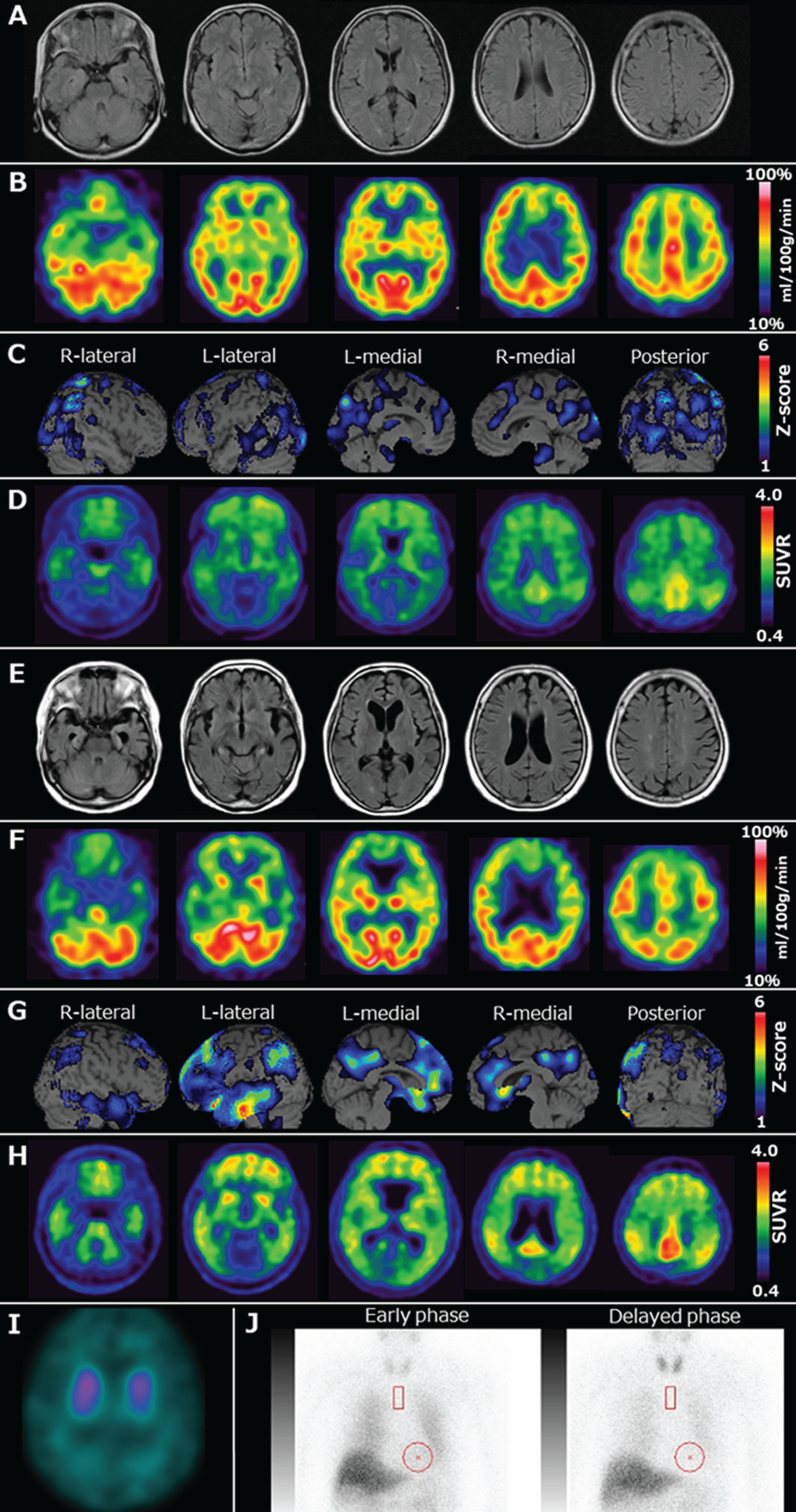
Neuroradiological findings. A) Magnetic resonance fluid-attenuated inversion recovery imaging performed in 2014 showing mild atrophy of the left medial temporal lobes including the hippocampus. B, C) Technetium-99-ethyl cysteinate dimer ([99mTc]ECD) single-photon emission computed tomography (SPECT) and brain perfusion SPECT easy Z-score imaging system (eZIS) analysis (PDRadiopharma Inc., Tokyo, Japan) performed in 2014 showing hypoperfusion in the right parietal lobe, bilateral posterior cingulate gyri, precuneus, and occipital lobes. The color scale for the Z-score is shown in the right part of the figure. Colored areas indicate a Z-score >1. D) Pittsburgh compound-B (PiB) positron emission tomography (PET) standardized uptake value ratio (SUVR) imaging performed in 2014 showing amyloid deposition in the frontal lobes, posterior cingulate gyri, and parietal lobes. The Centiloid scale, calculated using software developed by Matsuda et al. [10], was 45.4. E) Magnetic resonance fluid-attenuated inversion recovery imaging taken in 2023 showing atrophy of the bilateral medial temporal lobes, including the hippocampus, predominantly on the left, and, medial frontal lobes. F, G) [99mTc]ECD SPECT and brain perfusion SPECT eZIS analysis (PDRadiopharma Inc., Tokyo, Japan) performed in 2023 showing hypoperfusion in the left parietal lobe, bilateral posterior cingulate gyri, precuneus, left frontal lobes, and left medial and lateral temporal lobes. The color scale for the Z-score is shown in the right part of the figure. Colored areas indicate a Z-score >1. H) PiB-PET SUVR image taken in 2023 showing more amyloid deposition in the frontal lobes, posterior cingulate gyri, and parietal lobes, compared to that in 2014. The Centiloid scale was 98.1. I) 123I-N-omega-fluoropropyl-2-beta-carbomethoxy-3-beta-(4-iodophenyl) nortropane SPECT revealing a marked reduction in dopamine transporter (DAT) availability in the bilateral whole striatum based on a visual assessment. The quantitative analysis also indicated DAT reduction. The average specific binding ratio (SBR) for the whole striatum, calculated using the DaTView software (AZE Corp., Tokyo, Japan), was 2.50 (right striatal SBR = 2.86, left striatal SBR = 2.14). J) 123I-metaiodobenzylguanidine myocardial scintigraphy showing a decrease in the heart-to-mediastinum ratio in both the early and delayed phase and an increased washout ratio (early phase: 1.63 = delayed phase = 1.22; washout rate = 41.4%).
DISCUSSION
The present case exhibited RBD-like symptoms and transient visual hallucinations, which are characteristic of DLB, 10 years after the onset of AD. Additionally, abnormal findings of DAT-SPECT and MIBG myocardial scintigraphy indicated the presence of DLB. Therefore, the coexistence of LB pathology during the progression of AD may have caused the appearance of DLB-like symptoms in the present case.
A previous retrospective pathological study identified visual hallucinations and bradykinesia as predictors of LB pathology in AD [5]. In contrast, another retrospective pathological study reported that the antemortem clinical diagnosis of AD with LB pathology is difficult [6]. In our patient, RBD-like symptoms, transient visual hallucinations, and dysautonomia, which are characteristic of DLB, were noted. In particular, RBD is a core clinical feature of DLB [2] and has improved the diagnostic accuracy of autopsy-confirmed DLB. Thus, the development of RBD-like symptoms during AD progression may strongly indicate coexisting LB pathology. Even if RBD-like symptoms are transient, the coexistence of LB pathology may be predicted in patients with AD pathology [4]. In addition, the application of DAT-SPECT and MIBG myocardial scintigraphy, which are indicative biomarkers of DLB in cases with DLB-like symptoms, especially RBD, may be useful for diagnosing patients with both AD and LB pathologies [4].
A retrospective study of a pathological cohort of patients with early-onset dementia showed that approximately 55% of the patients with AD had coexisting LB pathology [8]. Nevertheless, over the clinical course, only 2.5% of these patients had been initially diagnosed with AD, later developed visual hallucinations and parkinsonism, and were subsequently diagnosed with DLB [8]. This result may be due to the limitations of retrospective studies and the fact that the symptoms of RBD are not easily recognized and reported by patients and caregivers unless requested by the attending physician [14]. Our patient’s husband reported only visual hallucinations and illusions, and the presence of RBD-like symptoms was only confirmed through questioning. Therefore, considering the high prevalence of LB pathology, not only in patients with late-onset AD but also EOAD [8], clinicians should be vigilant regarding the development of RBD-like symptoms during the progression of AD. In addition, clinicians should also consider that comorbid AD pathology and LB pathology make the core clinical features of DLB, such as visual hallucinations and RBD, less noticeable [3]. In fact, her visual hallucinations were transient, and RBD-like symptoms were only loud sleep-talking without limb movements. Similarly, parkinsonism may be less noticeable due to the comorbidity of this dual pathology [15]; however, it may be related to the discrepancy between the reduced DAT findings and the absence of parkinsonism, which is frequently found in DLB [16] and was also observed in our patients.
This study had several limitations. First, the presence of LB pathology was not confirmed postmortem. However, markedly abnormal findings on both MIBG myocardial scintigraphy and DAT-SPECT supported the coexistence of LB pathology. Second, polysomnography was not performed. However, patients with RBD confirmed through polysomnography are highly likely to have abnormal findings on MIBG myocardial scintigraphy [17], which may support the diagnosis of RBD in this patient. Third, information regarding phosphorylated tau was lacking for this patient, as cerebrospinal fluid analysis and tau PET were not performed, although this does not confirm that AD was the main pathology. Finally, we cannot deny the possibility that this patient had simultaneous LB pathology and AD pathology from an early stage. The indicator suggesting CIS calculated using eZIS and the longitudinal course of increased amyloid accumulation on PiB-PET may also support this hypothesis. However, a previous study has suggested that amyloid-β deposition is an ongoing process that occurs even at the AD stage, rather than reaching a plateau when cognitive impairment develops [18]. In addition, the first Centiloid scale (45.4) in this case exceeded the Centiloid scale (45.0) that is associated with a clinicopathological diagnosis of AD in 94% of patients in a pathological cohort [19]. Additionally, one particular physician continued to observe that the patient progressed for 10 years with only typical AD symptoms and no DLB-like symptoms. These findings may suggest that AD was the main pathology in the early stages and that later, the LB pathology became concomitant. Despite these limitations, this case report provides important information for clinicians to predict the coexistence of LB pathology with AD and to perform the appropriate imaging studies for detecting indicative biomarkers of DLB. Identifying the coexistence of LB pathology with AD is crucial in the era of anti-amyloid-β antibody therapy and may influence treatment strategies and care management for patients with AD and LB pathology.
AUTHOR CONTRIBUTIONS
Ryota Kobayashi (Conceptualization; Data curation; Formal analysis; Funding acquisition; Investigation; Methodology; Writing – review & editing); Kenichi Hashiguchi (Data curation; Investigation; Writing – original draft); Daichi Morioka (Data curation; Investigation; Writing – review & editing); Shinobu Kawakatsu (Data curation; Formal analysis; Funding acquisition; Writing – review & editing); Kazutaka Sakamoto (Data curation; Investigation; Writing – review & editing); Hiroshi Matsuda (Data curation; Formal analysis; Writing – review & editing); Akihito Suzuki (Formal analysis; Supervision; Writing – review & editing).
Footnotes
FUNDING
This work was partially supported by grants from the Japanese Ministry of Education, Culture, Sports, Science and Technology (KAKENHI Grant No. 22K07552) and a Health Labour Sciences Research Grant (Grant No: 23GB1003).
CONFLICT OF INTEREST
Ryota Kobayashi received honoraria for lectures from PDRadiopharma Inc. and Nihon Medi-physics Co., Ltd., and research support from Nihon Medi-physics Co., Ltd. All other authors have no conflicts of interest to report.
DATA AVAILABILITY
All data generated or analyzed during this study are included in this published article.
