Abstract
Background:
Non-motor symptoms (NMS) are common in late stage Parkinson’s disease (PD), as the frequency and severity of most of these symptoms increase with advancing disease.
Objective:
To assess effect of dopaminergic therapy on NMS in late stage PD and to investigate relationships between dopaminergic effect on NMS and on motor function.
Method:
Thirty PD patients in Hoehn and Yahr (HY) stages IV and V in “on” were included. Dopaminergic effect on non-motor symptomatology was assessed by the modified version of the Non-Motor Symptoms Scale (NMSS) in the “off” and the “on” state during a standardized L-dopa test, in parallel also assessing motor function.
Results:
NMS were common and many of the symptoms occurred in >80% of the individuals. The highest NMSS scores were seen within the NMSS domains 3: mood/apathy and 7: urinary in both the “off” and the “on” state. There was a statistically significant (p < 0.001) improvement in the modified NMSS total score (median) from 79 in “off” to 64 in “on”. There were statistically significant differences between the “off” and the “on” state for domains 2: sleep/fatigue, 3: mood/apathy, 5: attention/memory, 6: gastrointestinal and 7: urinary. The differences in the NMSS score between the “off” and the “on” state were in general larger for motor responders than for motor non-responders. In motor non-responders, differences of the NMSS score between the “off” and the “on” state were found for the total score, domain 3: mood/apathy and its item 11-flat moods.
Conclusion:
There is an effect of dopaminergic medication on NMS in late stage PD, to some extent also for those with a non-significant response on motor function during L-dopa test. It is therefore of importance to optimize dopaminergic therapy in order to give the most effective symptomatic treatment possible.
Keywords
INTRODUCTION
Parkinson’s disease (PD) is not only a movement disorder, but a disease that involves a large number of non-motor symptoms (NMS) as well [1]. Common NMS include cognitive impairment, mood-related symptoms (e.g., depressive symptoms, apathy), sleep/daytime sleepiness, fatigue as well as autonomic dysfunction [1–3]. The frequency and severity of most of these symptoms increase with advancing disease [2–5]. Previous research has shown that the frequency and severity of NMS as a whole is the most important predictor of health-related quality of life (HRQoL) in patients with PD [6].
In the late and most severe stage of PD, i.e., Hoehn and Yahr (HY) [7] stages IV and V, both motor- and non-motor symptomatology are pronounced [8]. Patients in the late stage of the disease have previously often lost contact with specialist care and are often excluded from clinical studies. Therefore, little is known about their clinical picture, including the effect of dopaminergic treatment [9].
It has been observed that the frequency and severity of NMS may vary with motor “off” and “on” states. Depressive symptoms, anxiety and bladder urgency have been reported as the NMS that have fluctuated the most between the “off” and the “on” state, with a more frequent occurrence of NMS in the motor “off” state [10].
The levodopa (L-dopa) motor effect seems to be somewhat less pronounced in late stage PD compared to earlier disease stages [11, 12]. In a study on L-dopa effect and motor function of the current sample [12], half of the participants had an L-dopa response of≥15% improvement on the motor score (part III) of the Unified PD Rating Scale (UPDRS) [13].
The effect of dopaminergic therapy on NMS in late stage PD has previously hardly been studied [5]. The aim of this study was to describe the NMS burden and to assess responsiveness of NMS to dopaminergic medication in a group of patients with late stage PD. Moreover, to investigate to which degree patients who have a motor effect of L-dopa also have an effect of dopaminergic therapy on NMS and to which degree patients who do not have a significant motor effect of L-dopa have an NMS response to dopaminergic therapy.
MATERIAL AND METHODS
Participants and recruitment
Participants were recruited from the southern region of Sweden through neurology departments and the municipality based health care system. Late stage PD was here defined as, inclusion criteria: HY stages IV and V in “on” and/or having a substantial need of help with activities of daily living, ADL (defined here as ≤ 50% on the Schwab and England Scale) [14], as well as having been diagnosed with PD for a minimum of seven years. All participants had been treated by a neurologist or geriatrician (two cases) and all had the diagnosis idiopathic PD. Exclusion criteria were: cognitive symptoms that started before the PD diagnosis, a score of <15 on Mini Mental State Examination (MMSE, score range 0–30, higher = better) [15]. Thirty-three consecutive patients from the baseline assessment of the Swedish part of the European multicenter study Care for Late Stage Parkinsonism (CLaSP) [16], fulfilled the inclusion criteria and were asked to participate in the present L-dopa test substudy, including assessments of both motor and NMS. One patient declined participation, while two were excluded due to not being able to fully participate. The final sample consisted of thirty participants.
The study was approved by the Regional Ethical Review Board in Lund, Sweden (JPND HC-559-002). Written informed consent was obtained by all participants.
Baseline assessments (background clinical data)
Baseline assessments were performed in the “practically defined on” state (i.e., during the patients’ ordinary medication) about a week prior to the L-dopa test and included demographic data, disease stage by HY [7], cognitive function assessed by the MMSE [15] and PD clinical data assessed by the UPDRS parts I: mentation, behavior and mood (score range 0–16, higher = worse); II: ADL (score range 0–52, higher = worse); III: motor function (score range 0–108, higher = worse) and IV: complications of therapy (score range 0–23, higher = worse) [13]. Depressive symptoms were assessed by the Geriatric Depression Scale (GDS-30; score range 0–30, higher = worse) [17]. Depression = scores ≥10, cut-off validated for PD [18]. PD specific HRQoL was assessed by the PD questionnaire-8 (PDQ-8; score range 0–32, higher = worse) [19]. Non-motor symptomatology was assessed by the Non-Motor-Symptoms Scale (NMSS; score range 0–360, higher = worse) [20]. The NMSS has 30 items divided into nine domains; cardiovascular including falls (two items), sleep/fatigue (four items), mood/cognition (six items), perceptual problems/hallucinations (three items), attention/memory (three items), gastrointestinal tract (three items), urinary function (three items), sexual function (two items) and miscellaneous (four items). Each item is scored according to both severity (0–3, higher = worse) and frequency (1–4, higher = worse) over the past four weeks, which are then multiplied, resulting in item/domain scores. For frequency analyses, items/domains were dichotomized into 0 = symptom not present and ≥1 = symptom present.
A full medication list was obtained from each participant and the L-dopa equivalent daily dose (LEDD) was calculated according to a standardized formula [21].
Assessment of dopaminergic effect
Two health care professionals (PD nurse, physiotherapist), with long experience in the field of PD, carried out the L-dopa test in the homes of the participants, using a standardized test protocol according to CAPSIT-PD [22, 23], to assess motor and NMS when the patient was in the defined “off” and “on” states. In most cases, a family member of the participant was present. The participants were first assessed in the morning in the defined “off” state; at least 12 hours after having taken the previous dose of dopaminergic medication [22, 23], before their morning L-dopa medication, in a fasting condition. They were then given a standardized dose of dissolvable L-dopa (200 mg soluble L-dopa + benserazid dissolved in a glass of water) and assessed again in the defined “on” state [22, 23]. This occurred when the patient and the investigator agreed that the best therapeutic L-dopa effect had been reached, typically after approximately 60 minutes. Each patient was observed clinically for about three hours. Two patients with known severe orthostatic hypotension received domperidone prior to testing (10 mg three times per day, starting 12 hours before the test). Non-motor symptomatology was assessed by the modified version of the NMSS to evaluate differences in NMS between the “off” and the “on” state [24]. The NMS were assessed during an interview with the patient in the defined “off” and “on” state, respectively, during a standardized L-dopa test. The patient was asked to report frequency and severity of the respective NMS during “off” and “on” periods occurring during the past four weeks. The interview concerning “off” symptoms was carried out in the defined “off” state and concerning “on” symptoms in the defined “on” state [24]. In the modified NMSS, the following domains/items are not used: domain 1: item 2-fainting; domain 4: perceptual problems/hallucinations (whole domain); domain 6: item 2-constipation and domain 9: item 28-taste or smell and item 29-weight change. The modified NMSS score ranges from 0 to 276 points (higher = worse).
Motor function was assessed [22] by the UPDRS part III [13]. For part of the analyses, the sample was divided into motor responders (≥15% improvement on the UPDRS III during L-dopa test) and motor non-responders (<15% improvement on the UPDRS III during L-dopa test) [12, 25].
Statistical analyses
Descriptive data are given by median and first and third quartiles (q1–q3) and frequencies and percentages, as appropriate. Dopaminergic responsiveness was calculated as the absolute differences (“off”-“on”) in scores. Differences between motor states and frequency pattern analyses were tested statistically with the Wilcoxon sign-rank test, χ2 test, Fisher’s exact test or McNemar test, as appropriate. The exact P-values (2-tailed) were used. P-values of <0.05 were considered significant. Due to the explorative character of the study and the high number of statistical tests, P-values were not adjusted for multiple testing. All analyses were performed using IBM SPSS version 22.0 (IBM 211 Corporation, Armonk, NY, USA).
RESULTS
Demographic and clinical data
Thirty individuals, 18 (60%) men, were included in the study (Table 1). The median age was 83 and the median PD duration was 12 years. Nineteen (63%) of the participants were in HY stage IV and 11 (37%) in stage V. The median (q1–q3) LEDD was 774 (519–925) mg. All of the patients had a history of motor fluctuations according to their medical records. Twenty-one (70%) of the participants reported that they experienced motor fluctuations (≥1 on item 36 of the UPDRS IV), all reported some part of the day in “off” (≥1 on item 39 of the UPDRS IV) and 14 (47%) reported dyskinesias (≥1 on item 32 of the UPDRS IV). The median (q1–q3) total NMSS score in the “practically defined on” state during baseline assessment was 94 (56–111) with a range from 35 to 164. The most frequent NMS were observed within the domains 3: mood/apathy (93%) (in particular item 11-flat moods), 6: gastrointestinal (93%), 2: sleep/fatigue (90%), 7: urinary (87%) and 5: attention/memory (80%). The most pronounced (severity×frequency) NMS were seen within the domains 3: mood/apathy (in particular item 11-flat moods) and 6: gastrointestinal (items 19-saliva and 21-constipation). Problems having sex (item 26), change in ability to taste or smell (item 28), nocturia (item 24), pain (item 27) and memory problems (items 17–18) were other areas where pronounced (severity×frequency) symptoms were commonly reported (Table 2).
Participant demographic and clinical data, N = 30
Q1-q3, first and third quartiles; PD, Parkinson’s disease; HY, Hoehn and Yahr staging scale (I-V, higher = worse); Schwab and England, Schwab and England activities of daily living scale (0–100, higher = better); MMSE, Mini-mental state examination (0–30, higher = better); NMSS, Non-Motor Symptoms Scale (0–360, higher = worse); UPDRS, Unified PD Rating Scale, part I = mentation, behaviour and mood (0–16, higher = worse); part II = activities in daily living (0–52; higher = worse); part III = motor examination (0–108, higher = worse); part IV = complications of therapy (0–23, higher = worse); GDS-30, Geriatric Depression Scale; PDQ-8, PD questionnaire-8 (0–32, higher = worse); LEDD, Levodopa equivalent daily dose. * For information on NMSS domains and single items, see Table 2. ** Responders are defined as patients with≥15% improvement in UPDRS part III motor score during L-dopa test.
Non-motor symptoms at baseline in the “practically defined on” state (n = 30)
NMSS, Non-Motor Symptoms Scale (0–360, higher = worse); D, domain; Pts, patients. Symptom present = score ≥ 1 on each item/domain. NMSS score = severity × frequency.
Dopaminergic effect on non-motor symptoms
First analyzes concerned the frequencies of reported items/domains of the modified NMSS in the “off” and the “on” state. As shown in Fig. 1 and Table 3, neither the frequencies of the items nor of the domains differed significantly between the “off” and the “on” state during the L-dopa test (P > 0.05, McNemar test). This was the case also for the complete modified NMSS with frequencies of 100% of patients with symptoms present in both states. NMSS frequency patterns of the four possible distributions of NMSS items and domains (no symptom; symptom in “off” only; symptom in “on” only; symptom present in “off” and “on”) are displayed in Fig. 2. The most frequently occurring domain was 2: sleep/fatigue, where 100% of the patients reported symptoms, the vast majority in both the “off” and the “on” state. There was a small number of patients with symptoms present in the “on” state only, while there were a few more with symptoms present in the “off” state only. When a NMS was present, it was typically present in both the “off” and the “on” state (Fig. 2). We did not detect differences of the frequency patterns between domains (χ2 test or Fisher’s exact test as appropriate), except for the comparison of the pattern of domain 2: sleep/fatigue with that of domain 9: miscellaneous (P = 0.007, Fisher’s exact test).
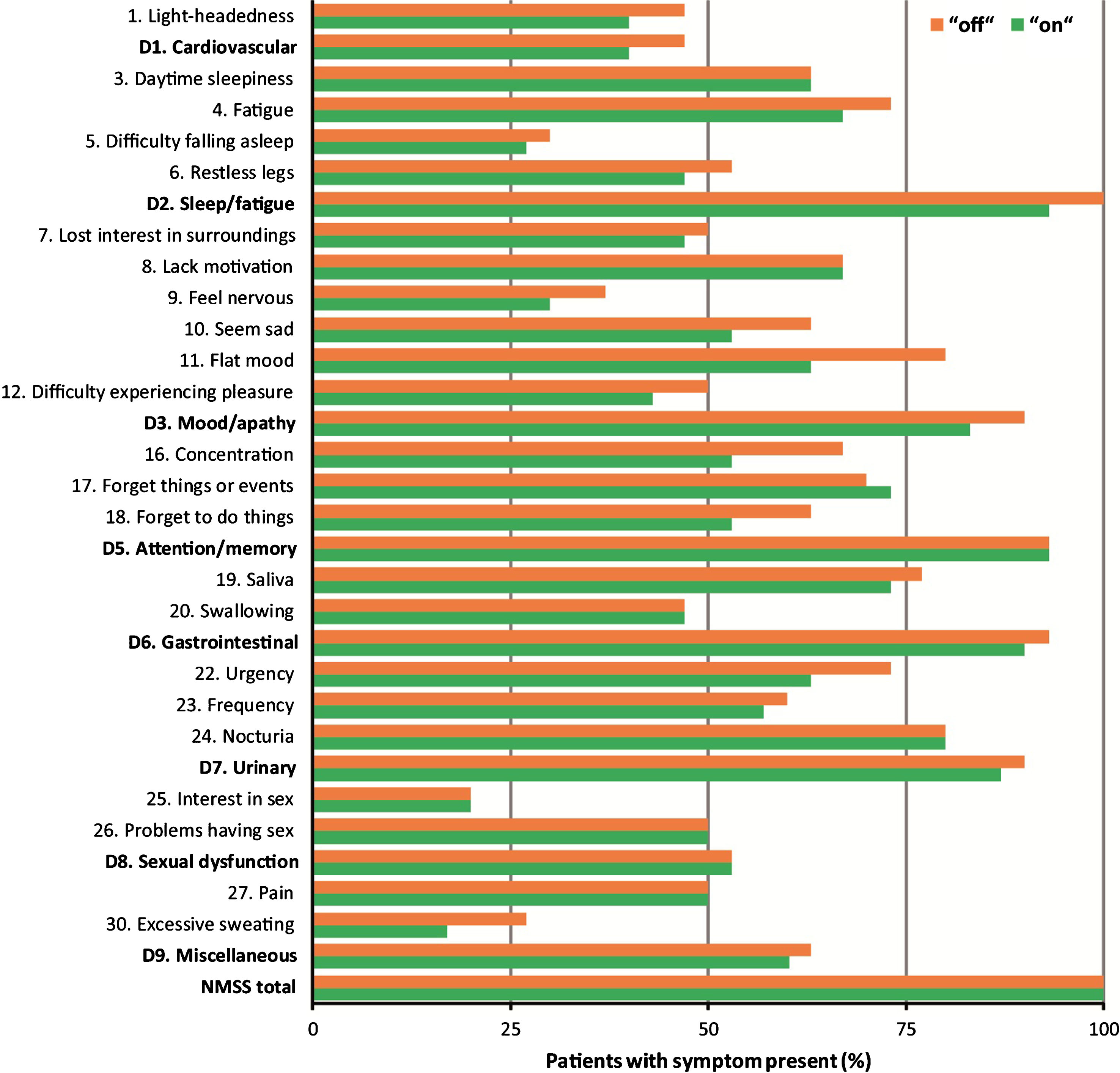
NMSS, Non-Motor Symptoms Scale (modified; 0–276, higher = worse); D, domain. Frequencies of the modified NMSS in the “off” and the “on” state.
Non-Motor Symptoms in the “off” and the “on” states during L-dopa test as assessed with the modified NMSS (N = 30)
NMSS, Non-Motor Symptoms Scale (modified; 0–276, higher = worse); q1–q3, first and third quartiles; D, domain; Pts, patients. Symptom present = score ≥ 1 on each item/domain. NMSS score = severity × frequency. *Differences between defined “off” and “on” states; number of patients who reported worse symptoms in the “off” than in the “on” state. P-values are from Wilcoxon sign-rank test for the comparisons of NMSS score between the defined “off” and “on” states. P-values were not adjusted for multiple testing. Bold P-values are statistically significant at P < 0.05.
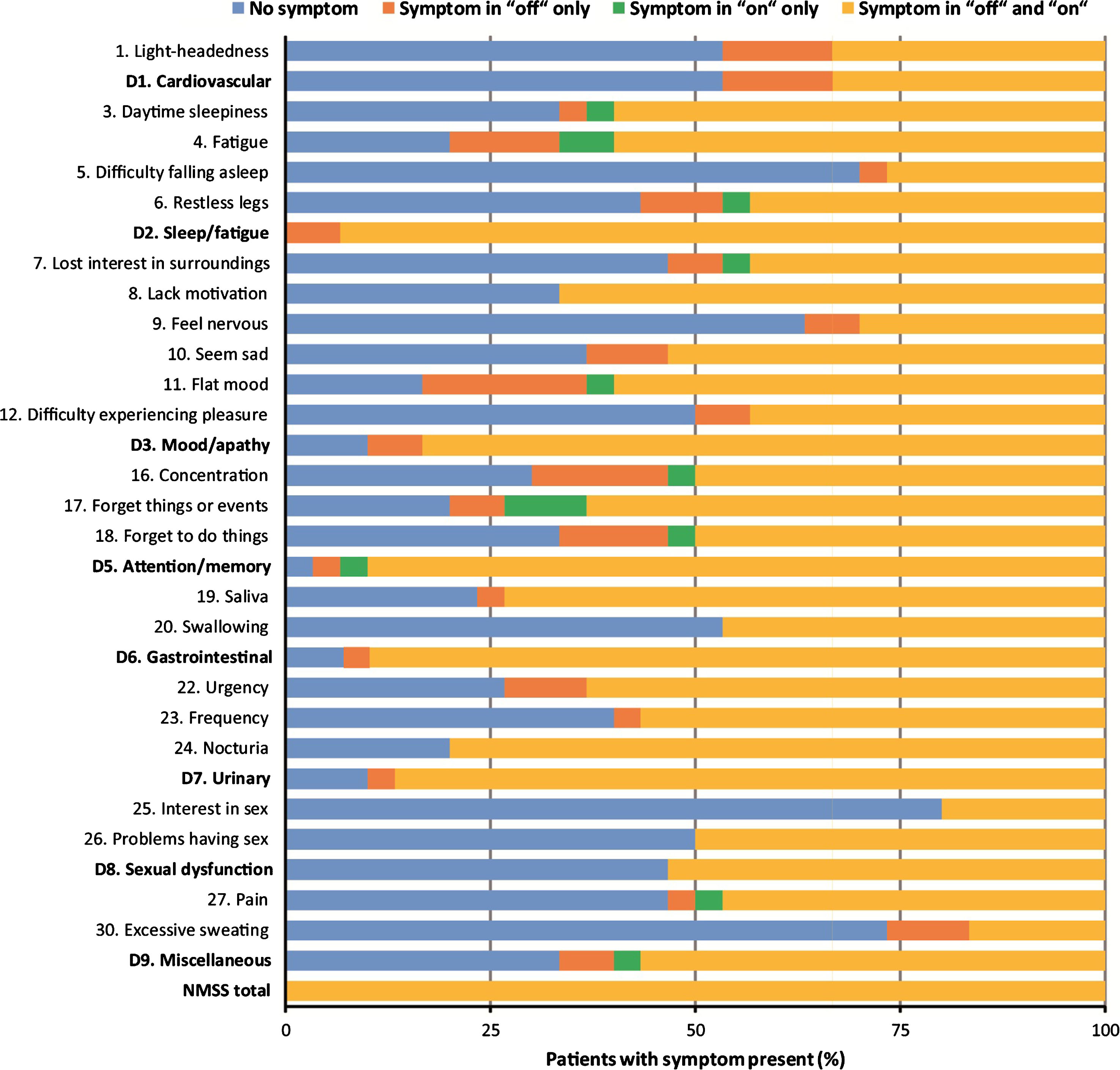
NMSS, Non-Motor Symptoms Scale (modified; 0–276, higher = worse); D, domain. Frequency patterns of the modified NMSS in the “off” and the “on” state. When a NMS was present, it was present in both the “off” and the “on” state in most cases. No differences of the frequency patterns between domains were detected (χ2 test or Fisher’s exact test as appropriate), except for the comparison of the pattern of domain D2: Sleep/fatigue with that of domain D9: Miscellaneous (P = 0.007, Fisher’s exact test).
Second analyzes concerned the NMSS scores (severity×frequency) with respect to the two motor states using the modified NMSS (Table 3). The median (q1–q3) total NMSS score in the “off” state was 79 (48–115). The median (q1–q3) total NMSS score in the “on” state (64; 45–96) was significantly lower compared to the NMSS score in the “off” state (P < 0.001; Wilcoxon sign-rank test). When comparing the NMSS in the “off” and the “on” state at the domain level, the domains that had higher score in the “off” compared to the “on” state were: 2: sleep/fatigue, 3: mood/apathy, 5: attention/memory, 7: urinary and 9: miscellaneous (Table 3). Regarding NMSS items, eight items showed significantly higher scores in the “off” compared to the “on” state: domain 2: sleep/fatigue, item 4-fatigue; domain 3: mood/apathy, items 9-feel nervous, 10-seem sad, 11-flat moods and 12-difficulty experiencing pleasure; domain 5: attention/memory, item 16-concentration; domain 6: gastrointestinal, item 20-swallowing and domain 7: urinary, item 22-urgency. The highest NMSS scores were seen within the domains 3: mood/apathy and 7: urinary in both the “off” and the “on” state.
Non-motor symptoms in motor responders versus non-responders
Results regarding L-dopa effect on motor function, including more in-depth analyses, have been reported separately [12]. Sixteen participants (53%) were classified as motor responders (≥15% improvement on the UPDRS III score during L-dopa test) and 14 (47%) as motor non-responders (<15% improvement on the UPDRS III score during L-dopa test).
When dividing the sample into motor responders and motor non-responders, there were in general larger differences in the NMSS scores between the “off” and the “on” state for motor responders than for motor non-responders (Table 4). The total NMSS score was higher in the “off” state compared to the “on” state for both subgroups. In motor responders, there was a statistically significant difference of NMSS scores for the domains 2: sleep/fatigue, 3: mood/apathy, 5: attention/memory and 6: gastrointestinal with higher scores in the “off” compared to the “on” state. In motor non-responders, only domain 3: mood/apathy with item 11-flat moods showed a significant difference between the two motor states.
Non-Motor Symptoms in the “off” and the “on” states in motor responders and motor non-responders as assessed with the modified NMSS, N = 30
NMSS, Non-Motor Symptoms Scale (modified; 0–276, higher = worse); q1–q3, first and third quartiles; D, domain. Motor responders, ≥15% improvement in the UPDRS III score during L-dopa test; motor non-responders, < 15% improvement in the UPDRS III score during L-dopa test. NMSS score = severity × frequency. P-values are from Wilcoxon sign-rank test for the comparisons of NMSS score between the defined “off” and “on” states. P-values were not adjusted for multiple testing. Bold P-values are statistically significant at P < 0.05.
DISCUSSION
It has often been questioned whether there is an L-dopa effect in late stage PD. In a previous study on the current sample, we showed that about half of patients with late stage PD appear to exhibit a significant L-dopa response of≥15% on motor function as measured by the UPDRS III during L-dopa test [12]. In a similar way, we here aimed to survey dopaminergic effect on NMS in the same sample. The results show that NMS are common in late stage PD, which has been indicated in previous studies [3, 26].
In some NMS areas, such as mood/apathy, gastrointestinal, sleep/fatigue, urinary and attention/memory, >80% of the participants experienced NMS. The NMSS domain D3: mood/apathy (and particularly item 11-flat moods) was the area with the most pronounced symptoms as well as where the greatest dopaminergic effect was observed, also in patients who did not have a significant L-dopa effect on motor function during L-dopa test.
Fluctuations in mood or “mood swings” have previously been described to occur in up to 75% of PD patients who experience motor fluctuations [24, 28]. Neuropsychiatric NMS such as depressive symptoms and fatigue as well as anxiety, have been shown to have great impact on a person’s life satisfaction [29] and HRQoL [3, 31] and the domain 3: mood/apathy has been pointed out as having the most negative impact on HRQoL [6]. Further, depression and cognitive impairment have been shown to be associated with disability in PD, indicating an effect of NMS also on ADL function [32].
There seems to be a dopaminergic contribution to several NMS and a response to dopaminergic treatment for NMS such as neuropsychiatric symptoms (e.g., depressive symptoms, apathy, anxiety, anhedonia, panic attacks); sleep disorders; autonomic symptoms (e.g., urinary); gastrointestinal symptoms (e.g., constipation); sensory symptoms (e.g., pain) and other symptoms such as non-motor fluctuations and fatigue [33]. For some NMS (e.g., sleep disturbance, depressive symptoms and cognitive impairment) also other neurotransmitter systems than the dopaminergic, e.g., serotonergic or noradrenergic, are involved [24, 34–36]. In addition to dopaminergic therapy, more specific treatment for various NMS (via other neurotransmitter systems) may be beneficial.
The most frequent NMSS domains in late stage PD were 2: sleep/fatigue, which was the most frequent (present in 100% in “off” and 93% in “on”), closely followed by 6: gastrointestinal (present in 93% in “off” and 90% in “on”), 7: urinary (present in 90% in “off” and 87% in “on”) and 3: mood/apathy (present in 90% in “off” and 83% in “on”). These results are in line with those of a previous study including patients in earlier disease stages (HY stages I–IV) [24] were the most frequent domains were 2: sleep/fatigue and 7: urinary, where about 95% reported symptoms.
The results showed more frequent occurrence of NMS in motor “off” state compared to motor “on” state, which previous research has shown also for younger patients in earlier disease stages [10]. Compared to patients in earlier disease stages [10, 24], the late stage PD patients seem to have somewhat less pronounced differences in NMS between the “off” and the “on” state. However, the groups cannot be directly compared due to various differences in the study samples as well as the testing procedures.
In the present study, HY stages IV and V have been studied as one group. When splitting the group into motor responders and motor non-responders and further dividing these groups into HY stages IV and V, it becomes clear that in the group motor responders 12 individuals (75%) were in HY stage IV and four (25%) in stage V. In the group motor non-responders, 7 of the individuals (50%) were in HY stage IV and 7 (50%) in stage V. The total score of the modified NMSS was higher among motor non-responders than motor responders, which could indicate a higher NMS burden for motor non-responders than for motor responders. This is in line with previous research implying that NMS symptomatology increase with advancing disease [2–5].
Strengths, limitations, and future perspectives
Our study has several limitations: One limitation in this study is the small number of participants. Furthermore, there are sometimes difficulties in collecting adequate data due to cognitive difficulties and problems in communicating, that to various degrees are part of the picture of late stage PD. However, the participants of this study fulfilled the inclusion criteria of MMSE ≥15 and were all able to participate in all parts of the study. In most cases, a family member or formal caregiver was present during testing to support the participant. On the other hand, a major strength is that we were able to recruit and conduct L-dopa tests with in many cases severely ill patients, as the occurrence of NMS and the dopaminergic effect on NMS as measured by the NMSS have not previously been studied in late stage PD. The present results could become an important base for future improvement of therapy and care.
NMS fluctuations during patients’ ordinary medication have not been monitored in this group, as diaries would often not be possible to obtain in late stage PD. The results of this study are based on assessments of NMS in the “off” and the “on” state during an L-dopa test, with a supramaximal dose of L-dopa and not during the patients’ ordinary dopaminergic medication. Whether NMS fluctuations during the patients’ ordinary medication occur in these patients is not shown in this study. However, it seems likely as there is a significant L-dopa motor effect in at least half of the sample and the occurrence of motor fluctuations in about 70% as measured by the UPDRS IV and further; signs of motor fluctuations as measured by a mobile movement recording system, the Parkinson’s KinetiGraph (PKG) [12, 37].
There are various ways of collecting information about the frequency and severity of NMS; patient reported questionnaires [38], patient diaries and assessment instruments administered by clinicians. In the late stage of PD, where both physical and cognitive difficulties are common, a clinician rated assessment of frequency and severity of NMS [20] in dialogue with the patient may be the preferred approach. Only a few NMS during the patients’ ordinary medication would be possible to measure objectively in this patient group; e.g., sleep/daytime sleepiness [39–42] or impulse control disorders [43]. Diaries would most probably be too difficult to obtain in late stage PD due to physical and cognitive difficulties to perform the task of keeping a diary. Previous studies described that patients with the best ability to comply with keeping a diary are younger with a shorter disease duration and mild PD symptoms [10]. The assessment instrument NMSS may be suboptimal for assessing NMS during actual “off” and “on” states, though is likely the best overall evaluation instrument for NMS at this time. The character of most NMS, in contrast to that of motor symptoms, does usually not allow for an assessment at one time point only such as during the defined “off” or “on” states but is an assessment over time, i.e., during the past month as in the case with the NMSS. In order to help the patient to give an estimation of NMS severity in the two motor states, the assessment of NMS was carried out during the actual, defined “off” and “on” states, as has been done previously with this instrument [24]. However, the medication effect on NMS might come also from other dopaminergic drugs than L-dopa.
Further studies on the effect of therapy on NMS in this group are needed, both regarding dopaminergic and non-dopaminergic therapy, in order to be able to give patients the most effective symptomatic treatment possible. This could also include shifting to advanced dopaminergic therapy, such as L-dopa-carbidopa intestinal gel (LCIG) [44] and apomorphine subcutaneous infusion [45], which potentially can give additional improvement of motor and non-motor symptomatology compared to peroral/transdermal dopaminergic therapy.
Conclusions
While based on a small sample of patients, this study shows that there is a dopaminergic response on NMS in late stage PD with pronounced effects particularly on mood/apathy, to some extent also for those with a non-significant response on motor function during L-dopa test. It is therefore of importance to optimize dopaminergic therapy also in the late stage of the disease, even in patients who do not show significant motor response.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
The CLaSP consortium. The main funding of this project: a JPND grant provided by the Swedish Research Council (VR), CLaSP/JPND HC-559-002; MultiPark, the strategic research area for neuroscience at Lund University; the Swedish Parkinson Foundation; the Swedish Parkinson Academy, the Faculty of Medicine at Lund University.
