Abstract
Background
Continuous subcutaneous apomorphine infusion (CSAI) is a standard of care treatment in advanced Parkinson's disease (PD) to treat motor fluctuations. However, literature about its long-term data is scarce.
Objective
The aim of this study was to report about CSAI tolerance and discontinuation predictors in a large monocentric cohort.
Methods
Consecutive PD patients who had CSAI were included. CSAI duration, discontinuation rates at 3, 12, 24, 36, 48, and 60 months, demographic data, MDS-UPDRS motor score, adverse events (AEs), discontinuation reasons and predictive factors were analyzed with logistic regression.
Results
A total of 208 patients were included from 1999 to 2023 (51% male; age: 67.4 ± 8.3 years; PD duration: 11.2 ± 5.0 years). In the overall group, CSAI duration was 25.0 ± 32.9 months (median: 13.0, range: 0.1–260.0). Ninety-five patients (45.7%) discontinued CSAI after 12.7 ± 15.3 months (median: 8.0). Main discontinuation causes were switching to deep brain stimulation (44.2%) and low efficacy (15.8%). Sixty% of discontinuations occurred within the first year. CSAI duration was the only significant difference between ongoing CSAI (116) and discontinued patients (35.4 ± 39.7 vs. 11.1 ± 18.4 months;
Conclusions
These results may help clinicians better select patients, anticipate and manage AEs, and predict CSAI discontinuation.
Plain language summary
Continuous subcutaneous administration of apomorphine using pumps is an established treatment available to individuals with Parkinson's disease (PD) at an advanced stage of disease. Although its short-term efficacy and tolerance are known, more information about its long-term tolerance is needed. Therefore, we studied the apomorphine long-term tolerance, adverse effects, and discontinuation causes in individuals with PD who were treated in our hospital between 1999 and 2023. We also tried to find clinical factors that could predict apomorphine withdrawal. Of the 208 people included, 51% were male, they had a mean age of 67.4 years, and 95 (45.7%) stopped apomorphine. Overall, apomorphine pump was kept during a mean duration of 25 months. People with PD who stopped apomorphine often did so within 12 months (60%). CSAI duration was the only significant difference between ongoing CSAI individuals (35.4 months) and discontinued CSAI individuals who did not switch to deep brain stimulation (11.1 months). Discontinuations mainly occurred because of people switching from apomorphine to deep brain stimulation (44.2%), and low apomorphine efficacy (15.8%). Adverse events were common in our population since 79.8% of them had at least one. The most frequent adverse events were hallucinations, which happened in 41.3% of people, followed by skin nodules (24%). The best characteristics that could predict apomorphine withdrawal were lower initial motor score in the
Introduction
Parkinson's disease (PD) is a complex neurodegenerative disease characterized by a constellation of motor and non-motor features.
1
Motor fluctuations appear on a variable time onset after dopaminergic treatment is initiated, and are mainly characterized by
Tolerance and efficacy patterns of invasive treatments have been studied and compared in several works.
CSAI has been used for decades to treat PD motor complications.3–5 Apomorphine is a dopamine agonist with primary affinity for D1 and D2 dopamine receptors like dopamine, which explains their pharmacological similarities. 6 Apomorphine is usually titrated to a dose that provides an equivalent antiparkinsonian response to that provided by levodopa, and its subcutaneous delivery allows a rapid onset of action. 7 CSAI has shown to be effective in reducing off time and dyskinesia, with a good tolerance in the short- (11% of patients discontinued) and long-term follow-up (16.7% discontinuation).5,8 The effects of apomorphine on some non-motor symptoms in PD have also been assessed. CSAI may improve urinary dysfunction, pain, and neuropsychiatric symptoms. 9 Nighttime CSAI may improve sleep disturbances in PD patients. 10 On the other hand, CSAI might cause or exacerbate impulsive compulsive behaviors to a lesser extent compared with oral dopamine agonists. 11 In two reviews, CSAI has showed overall positive effects on executive functions and emotion recognition, although some studies report on cognitive slowing and long-term global cognitive deterioration.12,13 Discontinuation at 52 weeks was reported in 51.5% of the patients, mostly due to skin issues, dyskinesias, nausea, and somnolence. 14 For longer follow-up, CSAI discontinuation varied from 53% (the main reasons being hallucinations, skin reactions, and confusion) to 68.6%.15,16
Compared to the other available invasive treatments, CSAI seems to be less effective. From the European Academy of Neurology/Movement Disorders Society-European Section guidelines on invasive therapies, CSAI and LCIG had a moderate effect on daily OFF and ON time with troublesome dyskinesias, but with a lower certainty effect for CSAI. 2 CSAI showed no relevant effect on quality of life, nor on the motor score in the ON condition. LCIG had a large effect on quality of life and activities of daily living, whereas STN-DBS yielded a large improvement of motor impairment, and a moderate increase in daily ON time and decrease in daily OFF time. Cognition and depression were likely unchanged by STN-DBS and LCIG, but were not assessed in CSAI. 2 The EuroInf2 real-life prospective study found distinct non-motor profiles between subthalamic (STN)-DBS, CSAI and LCIG, although all of them improved quality of life, motor fluctuations and non-motor symptoms. STN-DBS improved urinary functions, depression, cognition and fatigue. LCIG improved the three latter domains and gastrointestinal symptoms. CSAI improved depression and cognition, hallucinations and attention. 17
However, studies focusing on CSAI long-term efficacy and tolerance have been limited by a follow-up duration of only a few years, and a small number of subjects included.18,19 Consequently, although CSAI efficacy and tolerance have been established, there is still a dearth of evidence concerning long-term effects, AEs, and reasons for discontinuation. 2
The movement disorders center of the Centre Hospitalier Universitaire (CHU) of Grenoble (France) has been treating PD patients with CSAI for more than 25 years.20–22 Over the years, patients’ indications and pump devices have changed, making easier the access to CSAI, and more comfortable and manageable the treatment.
The main aim of this study was to retrospectively assess the long-term tolerance and AEs of CSAI in individuals with advanced PD, to find possible predictive factors of CSAI discontinuation. Moreover, we analyzed the possible impact of CSAI recent advances in indication and technology on CSAI discontinuation and AEs.
Methods
Patients
All consecutive individuals with advanced PD who could no longer be satisfactorily treated with oral medication,2,23 and thus underwent CSAI at the CHU of Grenoble, were retrieved from the hospital database. Individuals whose initial PD diagnosis was modified during follow-up (i.e., corrected to atypical parkinsonian syndrome), people who previously had DBS surgery, and people who had CSAI for less than one week were excluded.
Protocol approval and patients consent
The study was approved by a local ethics committee according to French regulation. The Clinical Research and Innovation Directory of the Grenoble University Hospital approved the study. Patients were informed that their medical charts could be used for research purposes after anonymization of the data. No individual written consent was required by the local ethics committee.
Treatment initiation protocol and follow-up
Individuals with PD were assessed before CSAI (baseline) with the Unified Parkinson's Disease rating scale (UPDRS) before 2011, 24 the Movement Disorders Society (MDS)-UPDRS after 2011, 25 the Montreal Cognitive Assessment (MoCA), 26 or Mini-Mental State Examination (MMSE), 27 and a composite frontal score (rated over 50, lower score indicating worse frontal cognitive syndrome, including the Wisconsin Card Sorting Test, verbal fluency, graphic series, gestural series). 28
Domperidone (10 mg TID, orally) was prescribed three days before starting apomorphine to ensure better tolerance.
Apomorphine infusion was started at 0.5–1 mg/h flow, and progressively increased until reaching a good clinical response (i.e., reduction of motor fluctuations). Meanwhile, oral and/or transdermal dopaminergic medications were progressively decreased. Patients with disabling nighttime akinesia or morning akinesia could have nighttime apomorphine infusion. The standard management included a short hospitalization (five days) three months after CSAI initiation, and regular follow-ups afterwards. Patients were assessed once or twice a year as outpatients, but treatment changes could be decided in the meantime if needed. Motor MDS-UPDRS score, levodopa equivalent daily dose (LEDD),29,30 apomorphine daily dose (mg), CSAI flow (mg/h), use of nighttime CSAI, disease duration at CSAI initiation, previous hallucinations, AEs, and treatment modifications were systematically collected at each visit. Medication changes, including apomorphine dosage and other antiparkinsonian medications, could be performed by the CHU movement disorders specialists depending on the patient's clinical conditions, and during all the follow-up time. CSAI duration and cause of withdrawal were also retrieved.
To note, the practice of starting CSAI changed in our hospital within the years. For example, before 2020, patients were seldom hospitalized before starting the CSAI whereas it has been often the case recently. Moreover, pump devices have evolved to become smaller and easier to manage (SoConnect, Crono Par type 50, 3 or 4 - Canespa).31,32
Since our center started to use the MDS-UPDRS after 2011, the UPDRS-III total scores were regressed to the corresponding MDS-UPDRS scores using the available conversion formula to standardize the entire cohort to the MDS-UPDRS. 33
Statistical analysis
Descriptive analysis included the following variables: age, sex, age at PD onset, disease duration, apomorphine initiation, discontinuation date and reason, AEs, previous hallucinations, MoCA and/or MMSE score, frontal score at apomorphine initiation, presence of oral PD treatments at baseline, LEDD at initiation and discontinuation, total apomorphine daily dose at initiation and discontinuation, daytime and nighttime apomorphine flows at initiation and discontinuation, period of CSAI initiation, and pump device type. Many patients awaiting DBS could have CSAI in our center for several months as a surrogate treatment, that is why we performed separate analyses that excluded CSAI-to-DBS patients, and other analyses in the entire cohort.
The primary exploratory endpoint was the CSAI duration dropout rate at 3, 12, 24, 36, 48, and 60 months after treatment initiation, and the reason for discontinuation. Patients who kept CSAI until their death were not considered as having discontinued it. The secondary endpoints were the occurrence and frequency of AEs in all patients, and their comparison between patients with ongoing CSAI and discontinued CSAI. All patients were allocated according to their duration follow-up groups and assessed for the CSAI discontinuation reasons. Discontinuation rates were compared according to CSAI initiation date, and arbitrarily divided in three periods in order to have comparable sample sizes (1999–2011, 2012–2019, 2020–2023). Predictive factors of discontinuation were also investigated.
Pearson's χ² test for categorical variables or Fisher's exact test in case of small sample size, and the Student's t test and the Mann-Whitney test for quantitative variables were used. The latter was used if the normality of variables or the homogeneity of variances were not met. Normality was assessed thanks to the Shapiro-Wilk test, and the homogeneity of variances was controlled with Levene's test. The Benjamini-Hochberg correction with a false discovery rate set at 0.05 was used to avoid false positive results because of multiple comparisons. Finally, CSAI discontinuation predictors were analyzed for exploratory purposes thanks to binomial logistic regression (bidirectional elimination approach), with Nagelkerke's R² and Akaike Information Criteria (AIC) used to assess the goodness-of-fit of our models.
All statistical analyses were run with Jamovi 2.5, running on R (RStudio 2024.09.0) and the Clinicopath module (2022).34–36
Results
From December 1999 to December 2023, 227 individuals with PD underwent CSAI at the CHU of Grenoble. From these, eight individuals were excluded because data was not available, seven were excluded because they had active DBS, and four were excluded because CSAI was used for only a few days while awaiting emergency DBS replacement (due to hardware infection). Therefore, a total of 208 patients were analyzed.
Main outcomes
Discontinuation rates were significantly different between discontinuation periods (
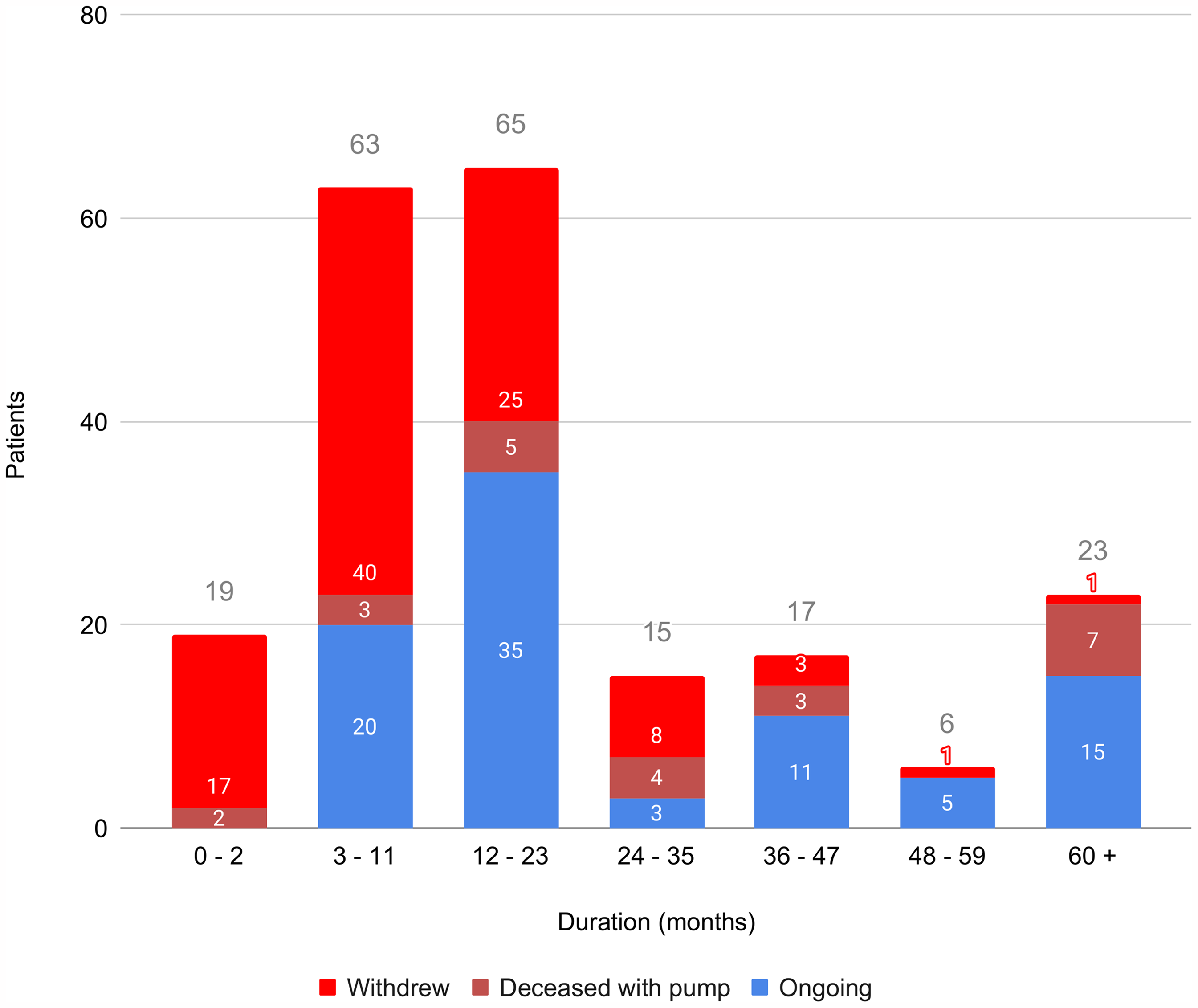
Patients’ status per follow-up duration. All patients were allocated in a duration period (0–2, 3–11, 12–23, 24–35, 36–47, 48–59, 60 and more months) according to their effective CSAI duration. On top of every column there is the total number of patients per duration period.
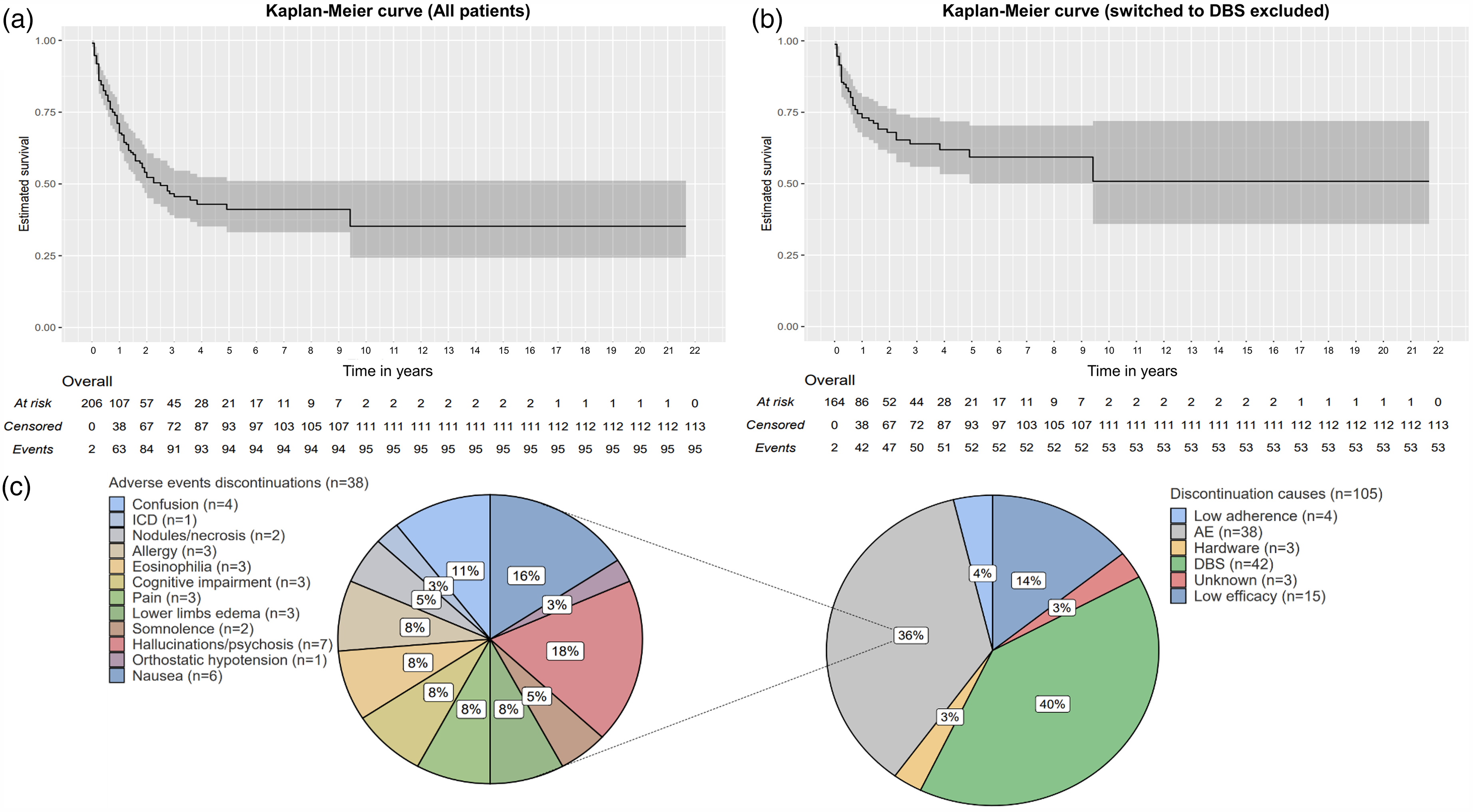
(a) Kaplan-Meier curve (all patients) displaying the remaining patients on CSAI over the years. (b) Kaplan-Meier curve that excluded the patients who switched CSAI to DBS. (c) Pie charts showing detailed discontinuation causes due to adverse events (left), and all other causes (right). A patient could have more than a single discontinuation cause; the 95 patients had a total of 105 discontinuation causes. AE: adverse events; DBS: deep brain stimulation; ICD: impulse control disorder.
Ninety-five (45.7%) patients had their CSAI discontinued, among whom 42 (20.2%) switched to DBS; 57 out of 95 patients (60%) discontinued CSAI within twelve months after treatment initiation. Detailed discontinuation reasons’ rates are shown in Table 1. In the overall group, mean CSAI duration was 25.0 ± 32.9 months (median: 13; range: 0–260). Discontinued patients (CSAI-to-DBS included) had a shorter mean duration (12.7 ± 15.3 months: median: 8,
CSAI discontinuation reasons for each follow-up duration group and for the total CSAI discontinuation patients. A patient could have more than a single discontinuation cause; the 95 patients had a total of 105 discontinuation causes.
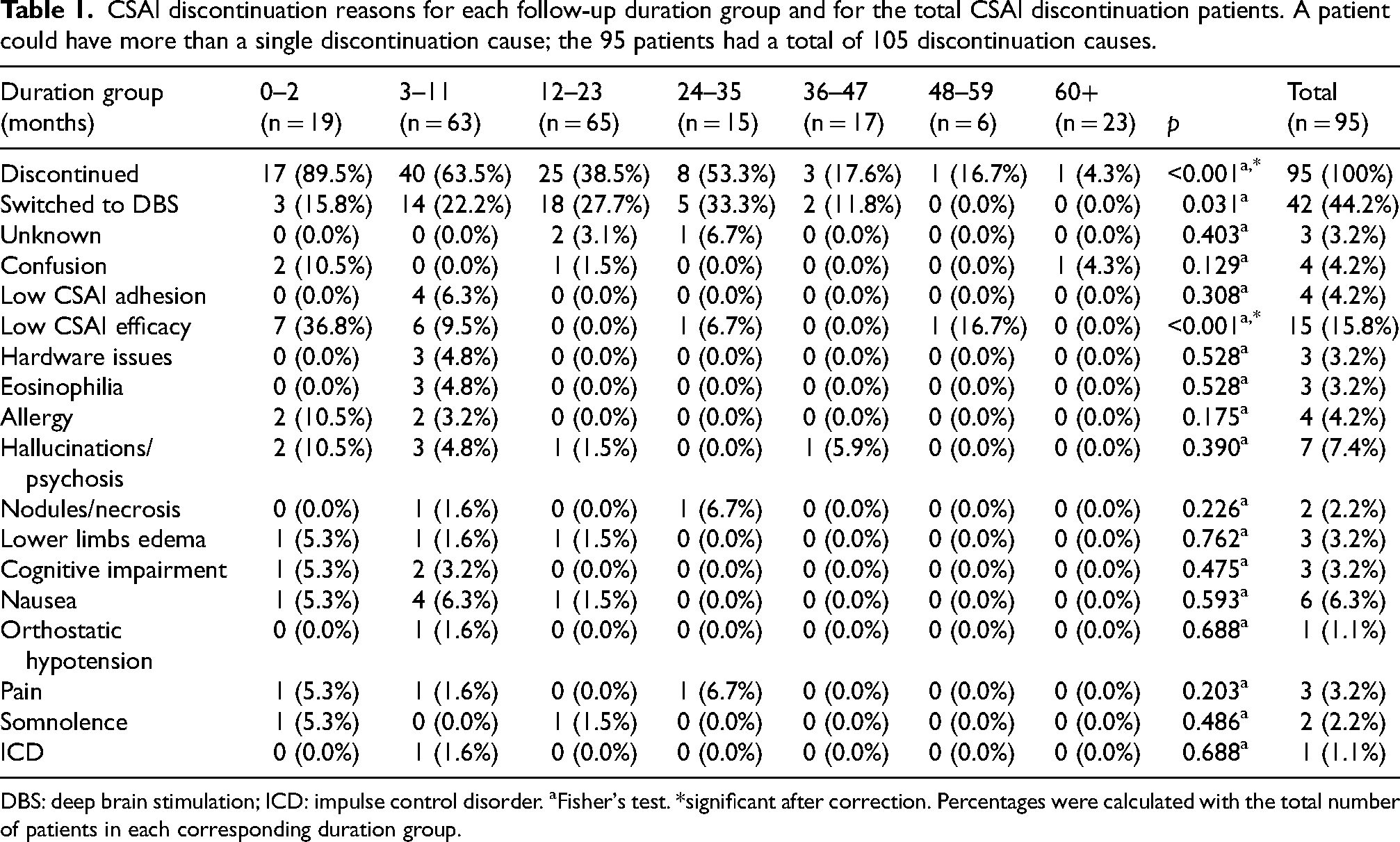
DBS: deep brain stimulation; ICD: impulse control disorder. aFisher's test. *significant after correction. Percentages were calculated with the total number of patients in each corresponding duration group.
Analyses between continuing and discontinued patients (CSAI-to-DBS patients excluded) did not find significant differences, besides the finding that CSAI duration was shorter in discontinued patients (35.4 ± 39.7 m vs. 11.1 ± 18.3 m;
Apomorphine doses, daytime and nighttime flows, and LEDDs were not different between groups. Table 2 summarizes patients’ characteristics and treatments aside from CSAI. Table 3 reports baseline and final CSAI data (CSAI-to-DBS patients excluded).
Patient's demographic and pre-CSAI PD characteristics.
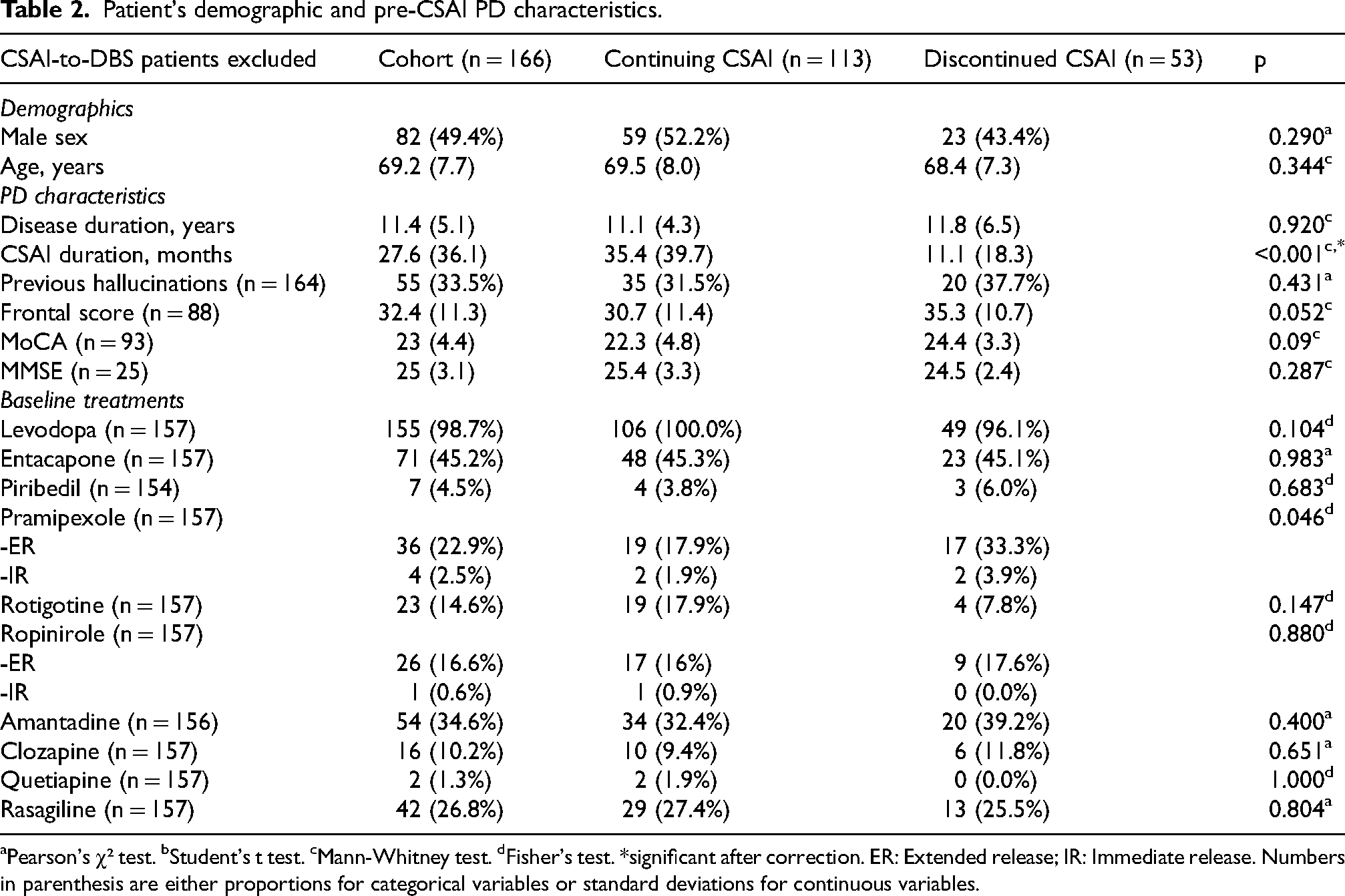
aPearson's χ² test. bStudent's t test. cMann-Whitney test. dFisher's test. *significant after correction. ER: Extended release; IR: Immediate release. Numbers in parenthesis are either proportions for categorical variables or standard deviations for continuous variables.
Patient's CSAI characteristics.
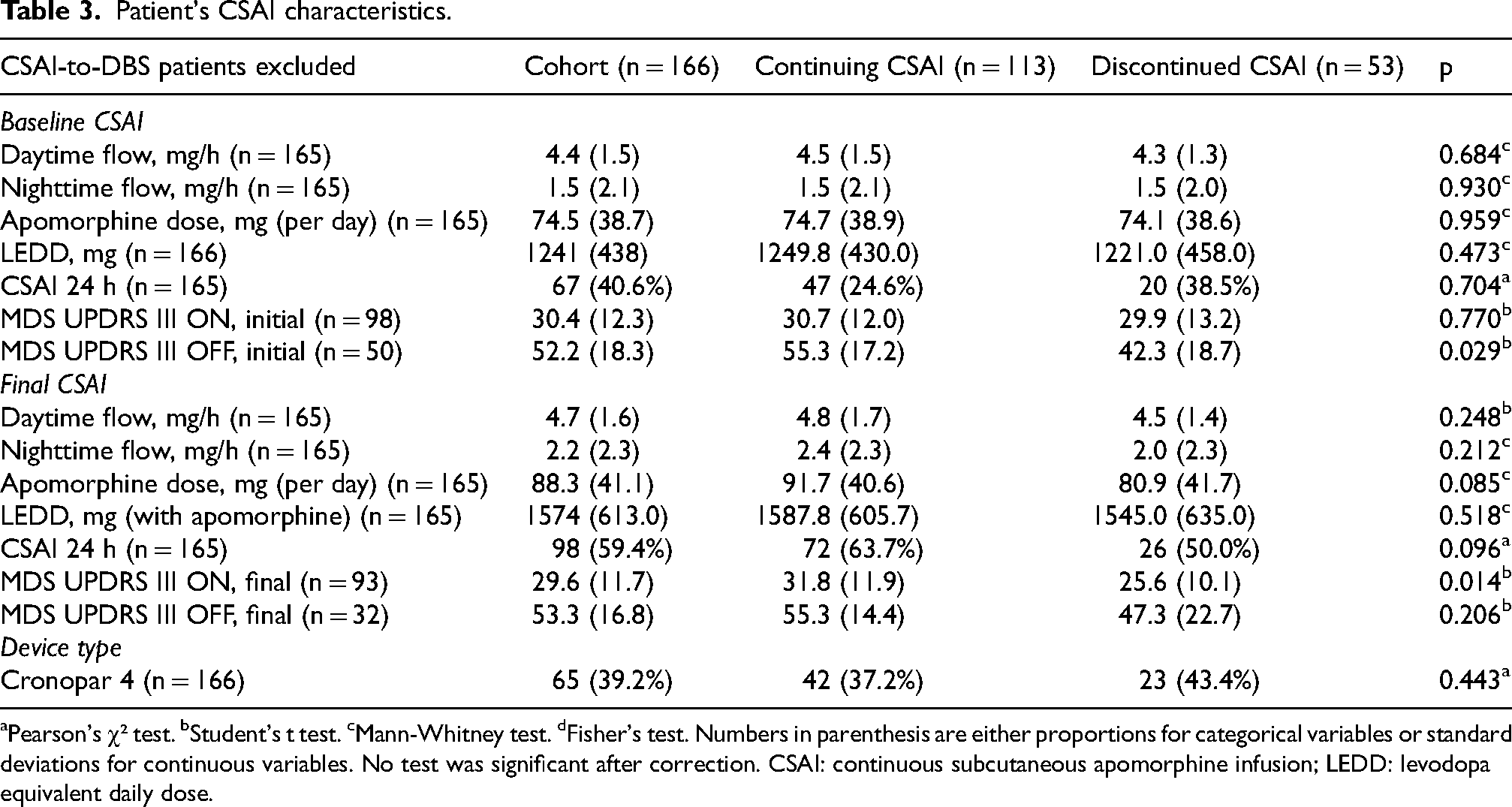
aPearson's χ² test. bStudent's t test. cMann-Whitney test. dFisher's test. Numbers in parenthesis are either proportions for categorical variables or standard deviations for continuous variables. No test was significant after correction. CSAI: continuous subcutaneous apomorphine infusion; LEDD: levodopa equivalent daily dose.
When analyzing all patients from the entire cohort (i.e., CSAI-to-DBS patients included), continuing patients were older (69.5 ± 7.9 years) than the others (64.8 ± 7.9 years,
Ninety-one (44%) patients had 24h CSAI when it was started, while 132 (63.8%) had it at the final assessment. Mean initial total daily apomorphine dose was 78.1 mg, which increased to 94.2 mg at final assessment. Apomorphine doses, daytime and nighttime flows, and LEDDs were not different between groups.
Initial
Supplemental Table 1 summarizes all patients’ characteristics and treatments aside from CSAI. Supplemental Table 2 reports baseline and final CSAI data in all patients.
There were no discontinuation rate differences between the 23 patients who started CSAI between 1999 and 2011 (43.5% discontinued), the 94 who started CSAI between 2012 and 2019 (46.8% discontinued), and the 91 who did between 2020 and 2023 (45.1% discontinued,
Secondary outcomes
The only discontinuation reason that was different among discontinuation groups over time was the low CSAI efficacy, which mostly occurred between 0 and 2 months (36.8%), and 3 and 11 months (9.5%,
Hallucinations were the most frequent AEs in the entire cohort (41.3%), and were more frequent in continuing CSAI patients, although this result was not significant after correction. Skin nodules (24%) and somnolence (23.1%) were the second and third most frequent AEs in our cohort, ICDs were found in 11.5% of patients. Only 20.2% of the entire cohort patients had no AEs. All AEs are displayed in Table 4.
Adverse events frequency among all patients. A patient could have more than one. Percentages were calculated with the number of patients in each column.
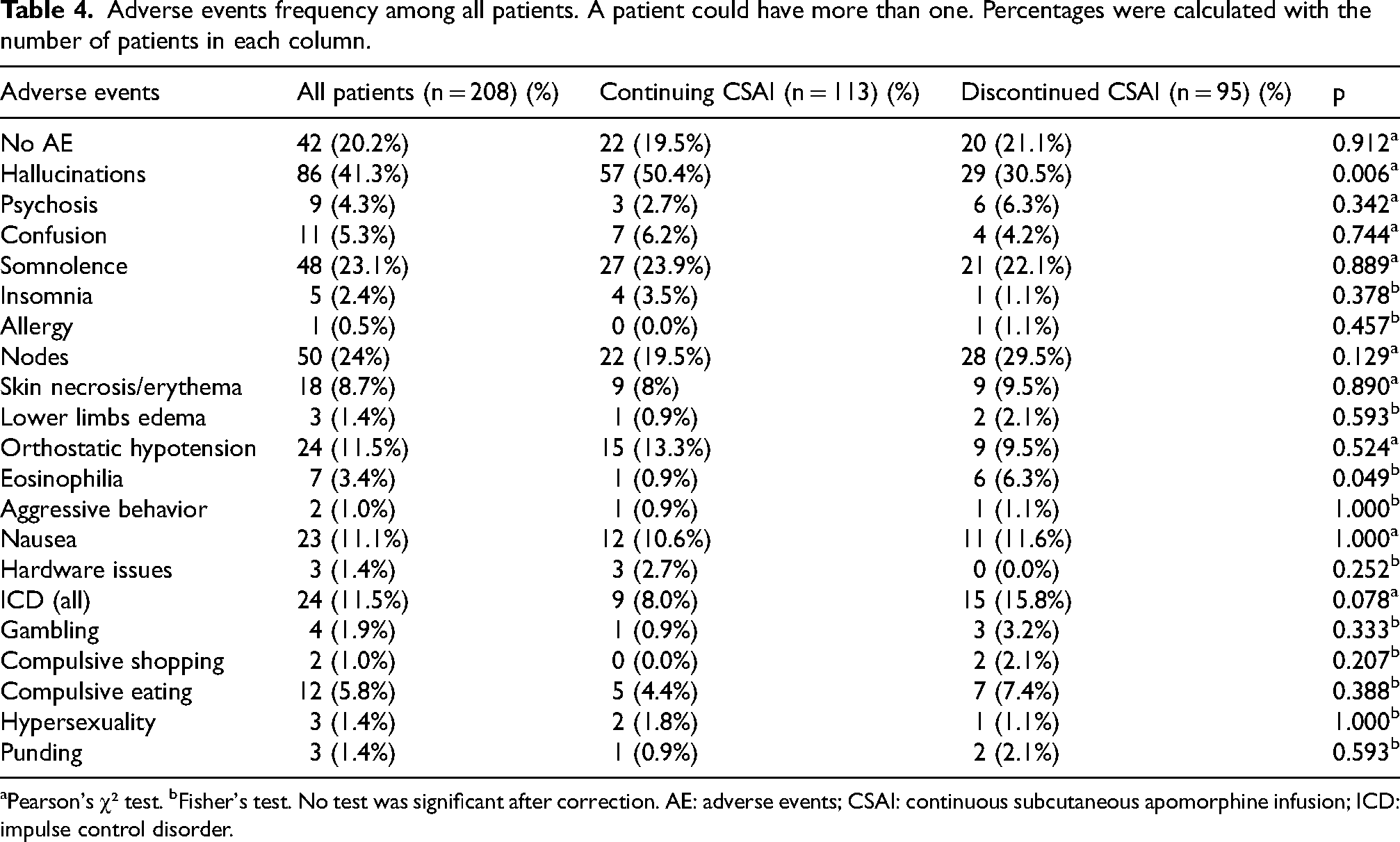
aPearson's χ² test. bFisher's test. No test was significant after correction. AE: adverse events; CSAI: continuous subcutaneous apomorphine infusion; ICD: impulse control disorder.
Comparisons between patients who discontinued CSAI before and after twelve months, and between those who switched to DBS and those who discontinued CSAI for other reasons are available in the Supplemental Material.
The first predictive factors analysis excluded the patients who discontinued CSAI for DBS, and the best predictive model which was a significant predictor of CSAI discontinuation in these patients had two variables (Nagelkerke's R2 = 0.47; AIC = 42.3; χ2 = 18.8;
The second analysis included all patients, and found a model that was a significant predictor of CSAI discontinuation. This model had the highest fitness assessed with the lowest AIC and the highest R² value among those found in our analyses: Nagelkerke's R2 = 0.61; AIC = 36.1; χ2 = 22.5;
Other significant variables are in Table 5. All ROC curves and models’ predictive measures are in Supplemental Material.
Binomial logistic regressions’ significant variables and their predictive measures.
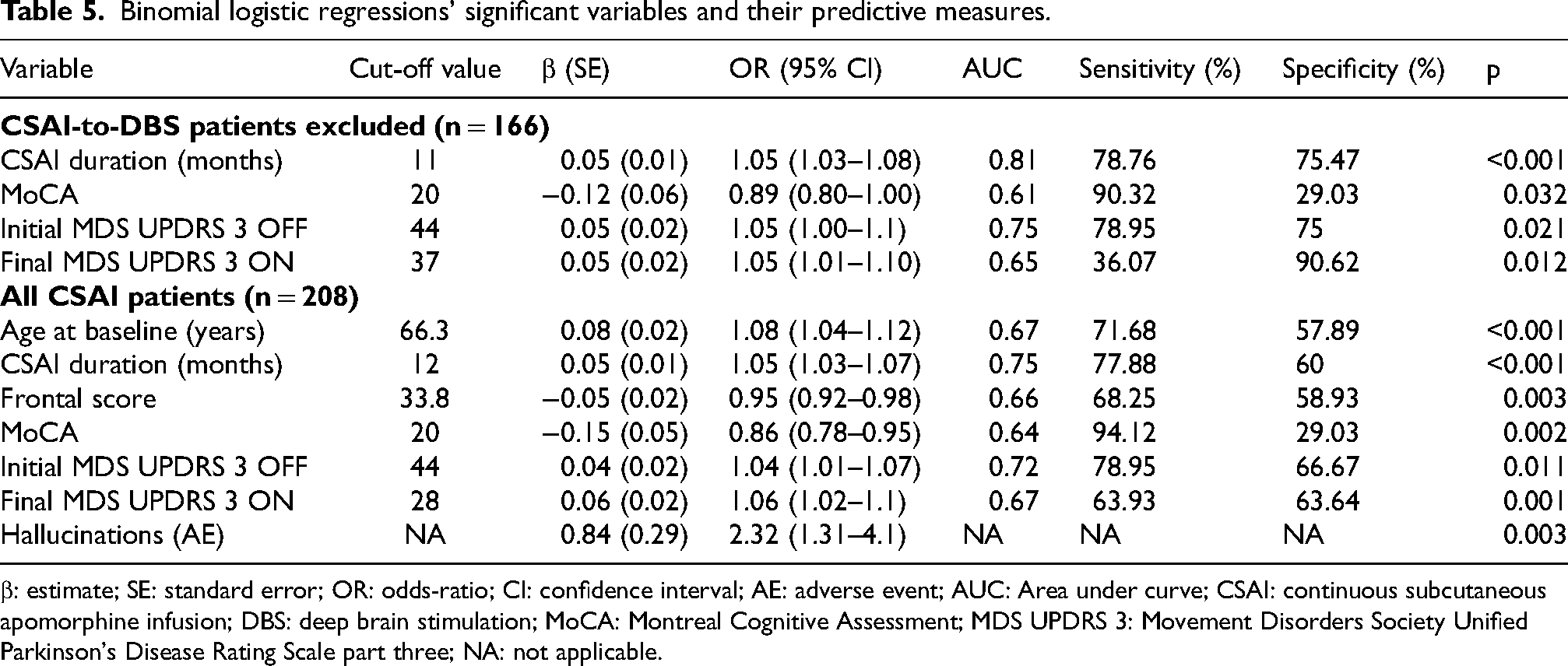
β: estimate; SE: standard error; OR: odds-ratio; CI: confidence interval; AE: adverse event; AUC: Area under curve; CSAI: continuous subcutaneous apomorphine infusion; DBS: deep brain stimulation; MoCA: Montreal Cognitive Assessment; MDS UPDRS 3: Movement Disorders Society Unified Parkinson's Disease Rating Scale part three; NA: not applicable.
Discussion
Although apomorphine use has a longstanding history,3,37,38 and CSAI has been recognized as one of the effective treatments in advanced PD patients, 2 long-term data are still relatively scarce. Our study adds several new findings to the available literature. 39
In our cohort, 95 (45.7%) patients stopped CSAI. Compared to the available literature, our discontinuation rate is overall lower or similar. For example, a 59.6% discontinuation rate was reported in 230 patients followed for ten years, among which 50% stopped at 36 months, 18 whereas in another study with 279 patients discontinuation was reported in 53%. 16 Another study in 45 patients found a similar discontinuation rate (42.7%) compared to our study. 40
In our population, the mean CSAI duration (27.6 months, CSAI-to-DBS patients excluded) was comparable to others. A study had a similar mean duration (32.3 months), while two other studies reported shorter durations, 26.3 and 21.6 months, respectively.18,41 A more recent retrospective study found a longer duration (34.1 months, and 22 months for discontinued patients). 16 Our shorter median duration may be caused by more patients having DBS earlier than in other studies.
Most of our patients discontinued CSAI within two years after treatment initiation (39.4%). This percentage is variable in other studies. A prospective study reported 34% discontinuations, and their cohort had less patients who switched to DBS (6 out of 39, 15.4%), but in a 24-month analysis. 19 Another cohort had 36.5% of patients stopping CSAI at one year, whereas our cohort reported 27.4% discontinuations at this time. 18 Our discontinuation rates were probably higher because AEs mainly happened during CSAI titration that can take several months. 8
After removing CSAI-to-DBS patients, we found that only CSAI duration was different between ongoing and discontinued patients. In our entire cohort, discontinued patients were younger, had a higher frontal score, worse baseline
Low efficacy, low adherence, hardware issues, nausea, hallucinations, and psychosis mostly occurred during the first twelve months, although only low efficacy remained significant. This is consistent with the TOLEDO trial in which the overall incidence of AEs tended to decrease with continued use, and less commonly after patients achieved a stable apomorphine dose. 8 Nevertheless, a study reported that discontinuation due to low efficacy mainly occurred after 36 months in their patients. 16 Discontinuation caused by low efficacy may either happen early because of poor response to apomorphine titration (like in our cohort), or later, because fluctuations’ worsening may no longer be effectively addressed by CSAI.
In our study, the most frequent AE-related discontinuation causes were hallucinations, nausea, and confusion. Among the possible reasons for CSAI discontinuation, a retrospective 10-year study found that hallucinations were the first cause, tied with lack of efficacy on motor fluctuations. 41 Similarly, another retrospective work has shown that the main reason for discontinuation was the lack of dyskinesia reduction. 42
These patterns are somewhat like others found in the literature, 41 albeit our patients discontinued because of skin AE (nodules, necrosis) less often than in other centers.15,16 It may be due to higher tolerance to these AEs in our patients or different nurse practices leading to better infusion management.
Our patients had a different AE profile from those in other long-term follow-up works. We found less nodules, more hallucinations, less orthostatic hypotension, and somnolence than in a real-life study in which CSAI had a shorter duration (24 months), and patients were younger and less cognitively impaired. 19 A 6-month study on 142 patients reported more frequent nodules, somnolence, behavioral disorders. 4 ICDs were found in 11.5% of patients in our cohort, mostly compulsive eating. This varies in the literature. A Thai study reported a similar rate (15.7%), 15 whereas a real-life study reported more ICDs (29.3% at 3 months, 18.9% at 2 years), but it included several patients with previous ICDs. 19 CSAI may not increase ICDs.11,12
AEs were frequent in our patients like in the TOLEDO study which reported that 92.6% of patients had at least one AE, 5 while another work found 58% AEs which remained relatively stable from 12 to 48 months. 16 The frequency of AEs may be variable. 39
Our long inclusion time (25 years), along with changing practices, AE management, and patient inclusion criteria could have led to CSAI discontinuation or AE discrepancies over the years. Nevertheless, we did not find differences in discontinuation rates or causes over time. Since our center is the birthplace of modern DBS,43,44 our patients could have access to this treatment (if they matched our criteria) before 1999, the year when the first patient in this study was included. However, DBS implantation rates may not have changed CSAI practices in our cohort.
Discontinuation rates and causes vary according to the different types of infusion therapies. 45 A recent 3-year analysis on 195 patients having LCIG found that 54.4% discontinued prematurely during the follow-up, of which 27.2% because of AEs. 46 The GLORIA registry on 375 LCIG patients followed over two years showed a global 31.2% discontinuation, which is similar to our discontinuation rate at 2 years (39.4%). 47 About 8.2% of patients stopped LCIG because of AEs (device dislocation, device issues, intestinal perforation). Concerning LCEIG, long-term studies are still missing. A study involving 27 patients with LCEIG found a 26% discontinuation rate at 6 months, mostly because of hallucinations or difficulties in finding a suitable infusion rate. 48 Lastly, in a group of 24 patients followed during a mean of 305 days, three (12.5%) patients discontinued because of diarrhea and one because of hallucinations. 49 The available few CSFF studies have shown more AEs and AEs-related discontinuations than in our study. The 12-week randomized controlled phase 3 trial reported 35% discontinuations in 74 patients receiving CSFF. The most frequent AEs were infusion site reactions in 72% of patients, but serious AEs led to discontinuations in 22% of cases. 50 This dropout rate matches the overall discontinuation rate at 12 weeks in the recent CSAI randomized trial (30.3%). 14 A 12-month CSFF open-label study reported 43.9% discontinuations. 51 It is more than in our study (27.4%) and in another CSAI trial (36.5%). 18 26% of patients had AEs causing discontinuation, mostly hallucinations (4.1%), infusion site erythema (3.7%), cellulitis (3.7%), nodules (2%). To note, 4.5% of patients stopped CSFF because of lack of efficacy, similar to our 6.3% of patients. 51 The higher rate of skin AEs in CSFF studies compared to CSAI may be related to a shorter experience in pump and skin management, and to a longer time using each subcutaneous injection site.
To summarize, aside from disease progression and deaths unrelated to treatment, LCIG patients mostly discontinued because of device issues, stoma infections, worsening of dyskinesias. LCEIG patients especially discontinued because of low efficacy or difficulties in finding the optimal infusion flow, hallucinations, device issues. CSFF patients primarily discontinued due to hallucinations, skin issues, and difficulties handling the device. Finally, CSAI patients showed a similar pattern, but with less skin issues.
Our study found several exploratory predictors of CSAI discontinuation. After excluding CSAI-to-DBS patients in the analysis, longer CSAI duration may predict discontinuation, which could be related to a global discontinuation risk increasing over time due to a long-term worsening of patients’ cognitive or behavioral symptoms which can prompt physicians to stop CSAI. This is consistent with lower baseline MoCA being a predictor in our study (cutoff value: 20/30). It may also be related to cognitively impaired patients being more frequently addressed to nursing homes, where CSAI is probably harder to keep for practical reasons. Nevertheless, CSAI effects on cognition are still debated. 39 A recent review showed no obvious effects of CSAI on cognition, whereas another one reported some decreases in visuospatial memory and executive functions.12,13 Cognitive deterioration in CSAI patients may be more linked to disease progression but more studies are needed.
In our study, the baseline
Our study has several limitations. Due to its retrospective design, we had a non-negligible amount of missing data, despite fully screening all CSAI patients’ records. Patients had a visit in our hospital three months after initiation to assess CSAI parameters but there was no obligation for some of them, who were followed elsewhere, to keep regular appointments in our center. AEs were collected in a binary manner, lacking specific scales or severity data, and no follow-up was conducted on these AEs. Besides, systematic use of standardized scales such as the MDS-UPDRS part IV at baseline was implemented only recently in our center, which prompted us not to use them in this study. Similarly, we did not focus on medication doses at baseline for each medication type (levodopa, agonists, entacapone, etc.) or medications during follow-up. How AEs and discontinuation causes were noted in our database may be problematic since there were some discrepancies in a few definitions that could not be retrospectively corrected. For instance, hallucinations and psychosis were merged in discontinuation causes and not in AEs.
Nevertheless, our study found that CSAI was a safe and globally well-tolerated PD treatment for at least two years in most patients. CSAI was withdrawn in a fifth of all patients because they underwent DBS surgery. PD duration, CSAI and other treatments’ characteristics such as LEDD, apomorphine dose, apomorphine flows, use of a recent pump device, or nighttime use do not seem to be linked to pump withdrawal. Discontinuation predictive factors, aside from CSAI duration itself, may be more severe motor symptoms at baseline and cognitive impairment (low MoCA). Altogether, these results may help clinicians select patients for CSAI among the multiple advanced treatments, anticipate, and better manage CSAI-related AEs.
Supplemental Material
sj-pdf-1-pkn-10.1177_1877718X251344896 - Supplemental material for Twenty-five-year experience with apomorphine pump in Parkinson's disease: A real-life long-term retrospective tolerance study
Supplemental material, sj-pdf-1-pkn-10.1177_1877718X251344896 for Twenty-five-year experience with apomorphine pump in Parkinson's disease: A real-life long-term retrospective tolerance study by Sina R Potel, Maria Chondrogiorgi, Andrea Gozzi, Sandrine Correia, Anna Castrioto, Sara Meoni, Pierre Pelissier, Emmanuelle Schmitt, Valérie Fraix and Elena Moro in Journal of Parkinson's Disease
Footnotes
Acknowledgements
Dr Sina R. Potel extends his warmest thanks and utmost affection to Napoléon F.C. Potel for useful discussions and continuous support during writing and revising the draft.
ORCID iDs
Ethical considerations
The study was approved by a local ethics committee according to French regulation. The Clinical Research and Innovation Directory of the Grenoble University Hospital approved the study.
Consent to participate
Patients were informed that their medical charts could be used for research purposes after anonymization of the data. No individual written consent was required by the local ethics committee.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
SRP discloses travel grants by Adelia Medical, Ever Pharma and Asten, honoraria for speeches for Adelia Medical.
EM has received honoraria from Medtronic for consulting. She has received research grant support from France Parkinson and Abbott.
SM has received research grants from Grenoble Alpes University, and Abbott.
VF has received consultancy fees from AbbVie France, and honoraria from Boston Scientific and The International Movement Disorders Society.
Data availability
The data supporting the findings of this study are available on request from the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
