Abstract
Background
Parkinson's disease (PD) is often accompanied by gastrointestinal symptoms. While elevated inflammatory biomarkers have been reported in PD patients compared to controls, the role of intestinal dysmotility and inflammation in disease manifestation is not fully understood.
Objective
This study sought to determine if fecal biomarkers and genetic predisposition to intestinal inflammation could help identify PD subtypes for future targeted therapies.
Methods
The association of disease activity, assessed through United Parkinson's disease Rating Scale (UPDRS) and Non-Motor Symptoms Questionnaire (NMSQ), with constipation severity, fecal calprotectin and six short-chain fatty acid (SCFA) levels, polygenic risk scores (PRS) for inflammatory bowel disease (IBD) and PD, and microbiota diversity were investigated in 95 participants with established PD using regression analyses. Unsupervised k-means clustering was applied to stratify PD patients based on inflammatory biomarkers.
Results
Having constipation was linked to worse mentation (UPDRSI, adj.p = 0.03) and more limited daily living activities (UPDRSII, adj.p = 0.03), with symptom severity linearly associated with higher disease activity (UPDRSI, adj.p = 0.002; NMSQ-total, adj.p = 0.02). Fecal calprotectin was elevated in those with constipation (p = 0.02) and associated with longer disease duration irrespective of the age (adj.p = 0.02). Cluster analysis demonstrated that PD patients with a higher non-motor symptom UPDRSII score were more likely to have more severe constipation, lower fecal SCFA levels, lower bacterial diversity, and higher PRS-CD and PRS-IBD.
Conclusions
Gut dysmotility, along with pro-inflammatory intestinal profiles, and greater genetic predisposition to IBD were observed in PD patients with worse non-motor symptoms. Monitoring intestinal biomarkers may help identify PD patients for targeted interventions.
Plain language summary
Parkinson's disease (PD) is a brain disorder that mostly affects movement. However, problems with smell, mood, sleep, and speech are also common. In addition, patients with PD often have gut-related issues including constipation, characterized by fewer than three bowel movements a week with stools that are difficult or painful to pass. Previous studies have shown that constipation may emerge years or even decades prior to PD diagnosis and appear in up to 80% of PD patients. The goal of our study was to test if gut inflammation, changes in the gut bacteria richness, and genetic risk factors, is related to how severe disease symptoms are in PD patients. We included 95 people with PD diagnosis who provided stool samples to measure inflammatory markers and the types of bacteria residing in the gut. We also used blood samples to assess genetic risk for inflammatory bowel disease (IBD) and obtained information about their PD symptoms. We found that PD patients with more severe constipation had worse symptoms that are not movement related. They also tended to have higher levels of inflammation and poorer bacteria richness in the gut. Patients with more severe PD symptoms in daily activities were also more likely to have a higher genetic risk for IBD. Our results suggest that monitoring biomarkers in the gut may help identify PD patients who could benefit from treatments that reduce intestinal inflammation and improve bacterial richness. This research could eventually help develop new ways to treat or slow down PD.
Keywords
Introduction
Current treatment for Parkinson's disease (PD) primarily focuses on addressing dopamine deficiency in the brain to alleviate symptoms. However, no available therapeutic approach alters the underlying neurodegenerative process in PD and most experimental strategies that aim to modify disease progression attempt to target alpha-synuclein. 1 This may be in part attributable to the fact that PD is a heterogeneous condition with different subtypes that require distinct therapies, indicating a substantial unmet need for novel biomarkers of disease pathogenesis and progression.
PD has been shown to be often preceded by gastrointestinal symptoms with multiple lines of evidence linking PD to intestinal dysmotility and inflammation. 2 Specifically, constipation, a condition characterized by difficult and infrequent bowel movements, may precede the motor symptoms of PD by over 10 years 3 and double the risk of developing PD when compared to those without constipation. 4 In established PD, constipation is one of the most frequent non-motor symptoms, with a prevalence reaching 50% to 80%, which negatively impacts patient's quality of life. 4 Also, elevated inflammatory biomarkers have been reported in individuals with PD, including fecal calprotectin, a sensitive and validated marker of intestinal inflammation.5–9 Additionally, decreased levels of fecal short-chain fatty acid (SCFAs), important metabolites of bacterial fiber fermentation in the gut, known to control inflammation, regulate epithelial barrier function, and provide energy source for enterocytes, have been reported in PD patients compared to control subjects. 10 This is in line with recent reports demonstrating depletion of bacterial taxa involved in SCFA production as the most reproducible gut microbiome alteration in PD patients, potentially promoting a pro-inflammatory status, which could be associated with the recurrent gastrointestinal symptoms. Also, enrichment in immunogenic bacterial components, as well as metagenomic pathways involved in degradation of neuroprotective molecules, and methanogenesis has been detected in PD compared to control stool samples.11–13
Moreover, multiple reports demonstrated an increased incidence of PD in patients with inflammatory bowel disease (IBD), a chronic immune-mediated disease of the gastrointestinal tract,14–17 suggesting shared pathogenesis between the two diseases. This observation, at least in part, can be explained by strong genetic pleiotropy reported between PD and IBD, implicating PD-related LRRK2 mutations in IBD risk,18,19 IBD-related NOD2 variants in PD risk,20,21 and other loci across the genome in the risk of both diseases.22–24 However, the roles of intestinal dysfunction and inflammation in PD activity have not been fully characterized.
Therefore, we assessed genetic predisposition to intestinal inflammation, fecal biomarkers and microbiota composition to determine their association with the PD manifestation in a cohort of patients with well-established disease. Stratifying PD patients by the degree of intestinal inflammation may offer an avenue toward dissecting disease subtypes and identifying those who would benefit from targeted therapies.
Methods
Cohort description
Study participants were recruited between 2019 and 2022 into the Saarland University Medical Center cohort. All enrolled subjects had a diagnosis of clinically established PD confirmed by a neurologist specialized in movement disorders and examined by one of three investigators at the Department of Neurology (inpatient and outpatient) throughout the whole recruitment period. The study participants were > 18 years old, showed willingness and ability to comply with the study-related procedures, and provided written informed consent to participate. Exclusion criteria included incapacity, use of antibiotics or steroids during the previous 12 weeks, acute disorders of the gastrointestinal tract (including IBD), or history of gastrointestinal surgery (other than appendectomy). This study was approved by the Ethics Committee of the Medical Association of Saarland (registration number 119/18).
Clinical assessment
Data on medication, comorbidities, and PD-related clinical scales were obtained from each subject during their research visit. Each subject received a thorough physical and neurological examination. In addition, each subject was evaluated using established clinical scales. Unified Parkinson's Disease Rating Scale (UPDRS), containing 3 parts: UPDRSI (mentation, behavior, and mood), UPDRSII (self-evaluation of the activities of daily life, including speech, swallowing, handwriting, dressing, hygiene, falling, salivating, turning in bed, walking, and cutting food), and UPDRSIII (motor examination), 25 were assessed by individual questions scored between 0 (normal) to 4 (most severe dysfunction) each. All UPDRS scores were assessed in the ON state (i.e., when motor symptoms are controlled by the subject's Parkinson's disease-directed medication). Non-Motor Symptoms Questionnaire (NMSQ) recorded 30 non-motor symptoms, each with yes/no responses with NMSQ_total ranging between 0 (no non-motor symptoms) and 30 (worst non-motor symptoms). One of the questions, NMSQ-5, records constipation symptoms. 26 When assessing NMSQ-5, we explicitly asked the patient to refer to the past month and explained to the patient that constipation is present in case of either less than three bowel movements a week or regularly having to strain to pass a stool. Additional tests included the Mini-Mental State Exam (MMSE) to evaluate cognitive function 27 ranging from 0 (worst cognitive status) to 30 (perfect cognitive status), and the Constipation Scoring System (CSS) questionnaire quantifying gut dysmotility symptoms 28 with the range of 0 (no constipation) to 30 (severe constipation). L-dopa equivalent daily dose (LEDD) was calculated as a sum of each Parkinsonian medication with respective conversion factors as previously described. 29 Disease duration was defined as the time between onset of typical PD motor symptoms (reported by the patient) and the time of the biospecimen collection.
Fecal biomarkers measurement
Fecal samples were collected as previously described. 30 Quantitative analyses of fecal SCFAs and calprotectin were carried out by the Institute of Microoecology, Herborn, Germany. All personnel involved in these analyses were blinded to clinical data and the diagnosis of the subjects. Fecal SCFAs were measured by gas chromatography. Specifically, fecal samples were first lyophilized and dissolved in 1:4 5M HCOOH and aceton before centrifugation. Concentrations of SCFA were determined using a GC-2010 Plus gas chromatograph (Shimadzu Deutschland GmbH, Duisburg, Germany) equipped with a flame ionization detection with a thin-film capillary column Stabilwax®-DA (Restek, Bad Homburg, Germany). The samples were spread out by split injection using the auto-sampler AOC-20s/I (Shimadzu Deutschland GmbH). GCsolution Chromatography Data System (Shimadzu Deutschland GmbH) was used for data processing. An external standard (SupelcoTM WSFA-1 Mix, Supelco Sigma-Aldrich Co., Bellefonte PA) was used for SCFA quantification. 31 Fecal calprotectin was measured using an enzyme-linked immunosorbent assay (ELISA) kit (Immundiagnostik AG, Bensheim, Germany) as previously described. 32 Briefly, the assay used the two-site sandwich technique in which the intensity of the color was directly related to the calprotectin concentration in the sample. Samples were read at 450 nm, and the 4-parameter algorithm was used to form the standard curve for calculation. All tests were carried out in duplicate. 32
Generation and analysis of genetic data
DNA was isolated from peripheral blood mononuclear cells (PBMCs) using the FlexiGene DNA kit (Qiagen, #51206). All individuals underwent genotyping with the Illumina high-density Infinium Multiethnic Global Array as described previously. 33 For each participant, we calculated a series of polygenic risk scores (PRS) for IBD and PD, based on the GWAS summary statistics for Crohn's disease (CD) and ulcerative colitis (UC), two main types of IBD, 34 IBD, 35 and PD 36 using the sum of risk allele dosages (0, 1, or 2) corresponding to a phenotype of interest, weighted by the effect size estimate of the respective GWAS as employed in PLINK. 37 In addition, genotype status was extracted for selected coding variants in the genes linked to both PD and IBD: NOD2 (R702W, G908R, L1007fs)21,38 and LRRK2 (R1398H, G2019S, N2081D).18,19,39
Quantification of abundance of gut microbiota
Total DNA was extracted from each fecal sample as previously described. 30 Library was prepared for the 16S region V3-V4 amplicon and sequenced with MiSeq. Sequencing data was processed with QIIME2 pipeline 40 coupled with R package DADA2. 41 Microbiota composition was characterized using alpha-diversity with Chao1 and Shannon indices,42,43 reflecting bacterial richness within each sample, and beta-diversity with Bray-Curtis distance matrices, 44 representing the similarity between samples, calculated using R packages phyloseq and vegan 45 as previously described. 46 Permutational multivariate ANOVA (PerMANOVA) tests were performed to compare the microbiota composition between groups. Genera with mean abundance <0.1% or with >80% zero values were removed 46 and differential taxa were derived from linear discriminant analysis (LDA) effect size (LEfSe) method. 47 PICRUSt (phylogenetic investigation of communities by reconstruction of unobserved states) was used to predict the pathways according to KEGG (Kyoto Encyclopedia of Genes and Genomes) associated with particular microbial abundances, 48 which were further subjected to group comparison using STAMP (STatistical Analyses of Metagenomic Profiles) under White's non-parametric t-test. 49
Statistical analysis
We used Spearman correlation to assess the bivariate relationship between clinical and biomarker variables. To account for potential confounding, we used multiple linear regression analysis adjusting for age, sex, disease duration, and laxative usage, unless otherwise specified. Fecal calprotectin and SCFA levels were natural log transformed prior to further analysis.7,50 False discovery rate (FDR) was used to account for multiple testing using Benjamini-Hochberg method. 51 Both adjusted (adj.p) and unadjusted p-values were derived. Statistical significance was set at p < 0.05.
Additionally, we used k-means clustering algorithm, an unsupervised method to group subjects based on the biomarkers of intestinal inflammation. 52 Variables used for clustering were PRS-CD, Chao1, and fecal calprotectin, representing genetic, microbial, and biochemical markers of intestinal inflammation. Subjects with missing values for those variables were excluded. To assess internal cluster quality, cluster stability of the optimal solution was computed using Jaccard bootstrap values with 1000 runs using R packages cluster and NbClust. 53 Resulting clusters were used as comparison groups to determine differences in biomarkers levels and disease phenotypes using Student's t-test or Wilcoxon rank-sum test for continuous outcomes and Chi-square test or Fisher's exact test for categorical variables.
Results
Cohort characteristics
A total of 95 PD cases were included in the analyses. The mean age was 69.0 ± 9.0 years, 37.9% were female, the median disease duration was 82 months (25%–75%, 43–132), and median age of disease onset was 62 years (25%–75%, 54–69). The PD-related symptoms were consistent with a mild mentation, behavior, and mood dysfunction (median UPDRSI of 2; 25%–75%, 1–3), mild impairment of activities of daily living (median UPDRSII of 10; 25%–75%, 6–15), and mild motor impairments in ON state (median UPDRSIII of 20; 25%–75%, 12–29). The majority had normal cognitive function (median MMSE of 28; 25%–75%, 27–30). Nearly half of the study subjects had constipation (48.4%; median CSS of 1; 25%–75%, 0–3), 12.5% reported a history of diabetes mellitus and 4.8% of autoimmune diseases (Table 1).
Patient characteristics by constipation status.
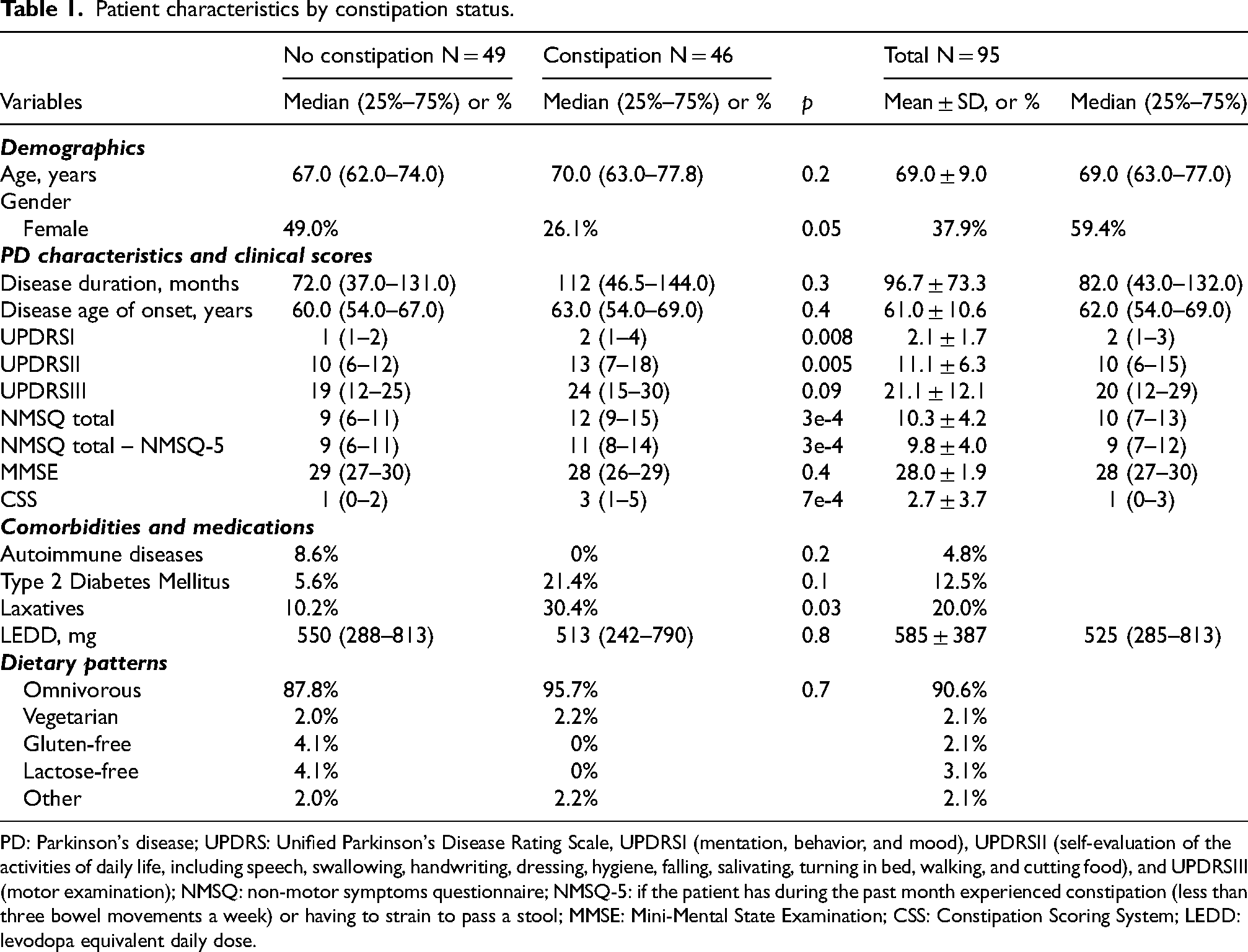
PD: Parkinson's disease; UPDRS: Unified Parkinson's Disease Rating Scale, UPDRSI (mentation, behavior, and mood), UPDRSII (self-evaluation of the activities of daily life, including speech, swallowing, handwriting, dressing, hygiene, falling, salivating, turning in bed, walking, and cutting food), and UPDRSIII (motor examination); NMSQ: non-motor symptoms questionnaire; NMSQ-5: if the patient has during the past month experienced constipation (less than three bowel movements a week) or having to strain to pass a stool; MMSE: Mini-Mental State Examination; CSS: Constipation Scoring System; LEDD: levodopa equivalent daily dose.
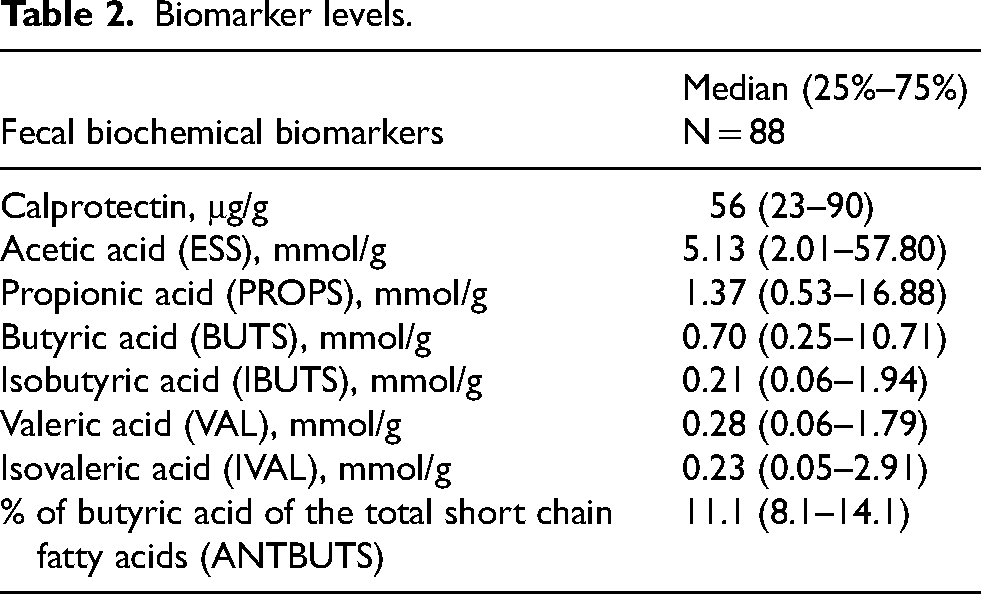
Fecal biomarker levels are summarized in Table 2. Moreover, the study participants were more likely to carry NOD2 R702W or G908R risk alleles, and LRRK2 R1398H protective alleles, less likely to carry NOD2 L1007fs variants, and had LRRK2 N2081D frequency similar to the general population of European ancestry. None of our PD cases carried LRRK2 G2019S (Supplemental Table 1).
Biomarker levels.
Relationship between fecal biomarkers, polygenic risk scores and PD activity
In univariate analysis, we detected strong relationship among the motor and non-motor scores (Figure 1). Specifically, higher UPDRSII was linked to older age and longer disease duration, whereas higher UPDRSIII was related to lower MMSE. Moreover, UPDRSI, UPDRSII, and NMSQ_total positively correlated with CSS, Also, we found significant correlations between PD-related symptoms and gut-related inflammatory biomarkers. UPDRSI was positively associated with various fecal SCFA levels, while worse UPDRSII was associated with higher PRS-UC and PRS-CD, and worse UPDRSIII with higher PRS-CD. Additionally, individuals with higher NMSQ_total, higher CSS, or longer disease duration had lower microbiota alpha-diversity Chao1 index. Higher fecal calprotectin levels were associated with older age, longer disease duration, and more severe constipation, whereas higher SCFA levels significantly correlated with later disease onset, shorter disease duration, and lower LEDD. SCFA levels also positively correlated with microbiota alpha-diversity assessed by Chao1 and Shannon indices. PRS-PD positively correlated with LEDD, and negatively with Shannon index, whereas PRS-CD positively correlated with age and disease onset and negatively with alpha-diversity (Figure 1(a)). The connectivity among the most strongly associated variables (q-value < 0.05 and |Rho|>0.2) is shown in Figure 1(b).
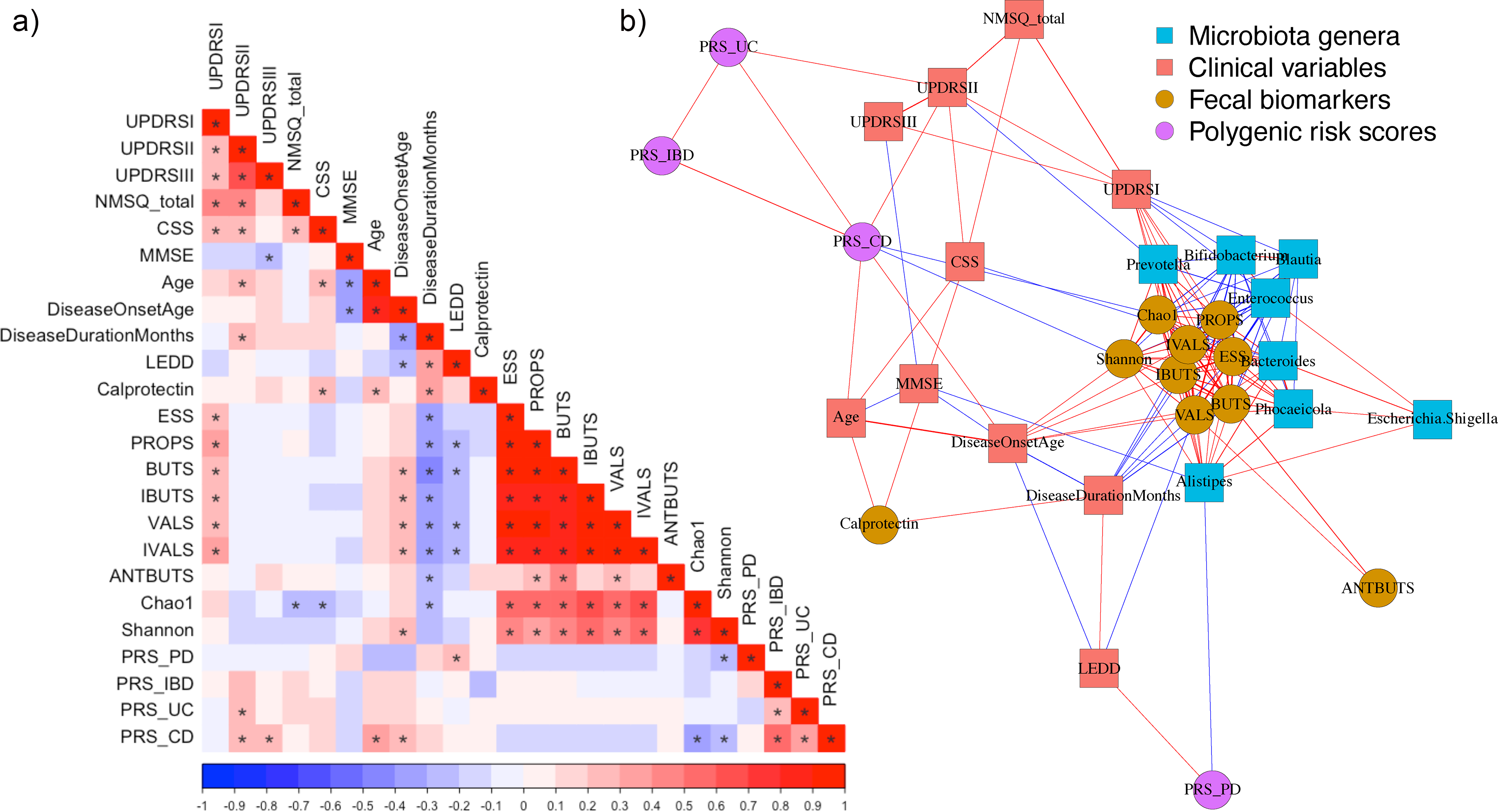
Heatmap and network diagram of univariate correlations between the intestinal inflammatory biomarkers and Parkinson's disease phenotypes. (a) Heatmap of Spearman correlation. * p < 0.05. Color intensity reflects magnitude of correlation coefficient Rho. (b) Network visualization of correlations with |Rho|> 0.2, q < 0.05 (after adjustment for multiple testing), and microbiome genera mean abundance > 1%. UPDRS: Unified Parkinson's Disease Rating Scale; NMSQ: non-motor symptoms questionnaire; CSS: Constipation Scoring System; MMSE: Mini Mental State Examination; LEDD: levodopa equivalent daily dose; short chain fatty acids (SCFAs): ESS: acetic acid; PROPS: propionic acid; BUTS: butyric acid; IBUTS: isobutyric acid; VALS: valeric acid; IVALS: isovaleric acid; ANTBUTS: % of butyric acid (of all SCFA), Chao1 and Shannon: microbiome alpha-diversity estimates; PRS: polygenic risk score; PD: Parkinson's disease; IBD: inflammatory bowel disease; UC: ulcerative colitis, and CD: Crohn's disease.
We next followed up on the most significant univariate associations using multiple regression models. After adjusting for age, sex, and laxative usage, we detected that individuals with longer PD duration had higher fecal calprotectin (adj.p = 0.02, Supplemental Figure 1(a)), lower fecal SCFAs (adj.p ≤ 0.03, Supplemental Figure 1(b)–(g)), and lower microbial alpha-diversity (adj.p = 0.02, Supplemental Figure 1(h)).
Moreover, after adjustment for age, sex, disease duration, and laxative usage, we found that individuals with constipation had significantly worse non-motor UPDRSI (adj.p = 0.03), UPDRSII (adj.p = 0.03) and NMSQ_total scores (adj.p = 0.03), but similar motor function, UPDRSIII (adj.p = 0.5; Figure 2(a)–(d)). CSS, the degree of constipation symptom severity, linearly associated with UPDRSI (adj.p = 0.002) and NMSQ_total scores (adj.p = 0.02), but not with UPDRSII (adj.p = 0.3) or UPDRSIII (adj.p = 0.6; Figure 2(e)–(h)). Moreover, PD patients with constipation had higher fecal calprotectin levels compared to those with no constipation (Figure 3(a), p = 0.02) with patients in the first quartile of fecal calprotectin showing significantly lower CSS than any one of the other quartiles (Figure 3(b), all adj.p ≤ 0.04). Additionally, lower microbiota alpha-diversity Chao1 was associated with more severe constipation symptoms (Figure 3(c), adj.p = 0.03), and worse NMSQ_total score (Figure 3(d), adj.p = 0.03). Furthermore, PD patients with worse UPDRSII score had significantly higher PRS-IBD and PRS-UC (adj.p = 0.02 for both, Figure 4(a) and (b)). No statistical significance association between UPDRSII and PRS-CD or PRS-PD was observed (Figure 4(c) and (d)). Subjects carrying LRRK2 or NOD2 IBD-associated risk alleles showed no statistically significant associations with PD severity (Supplemental Table 2).
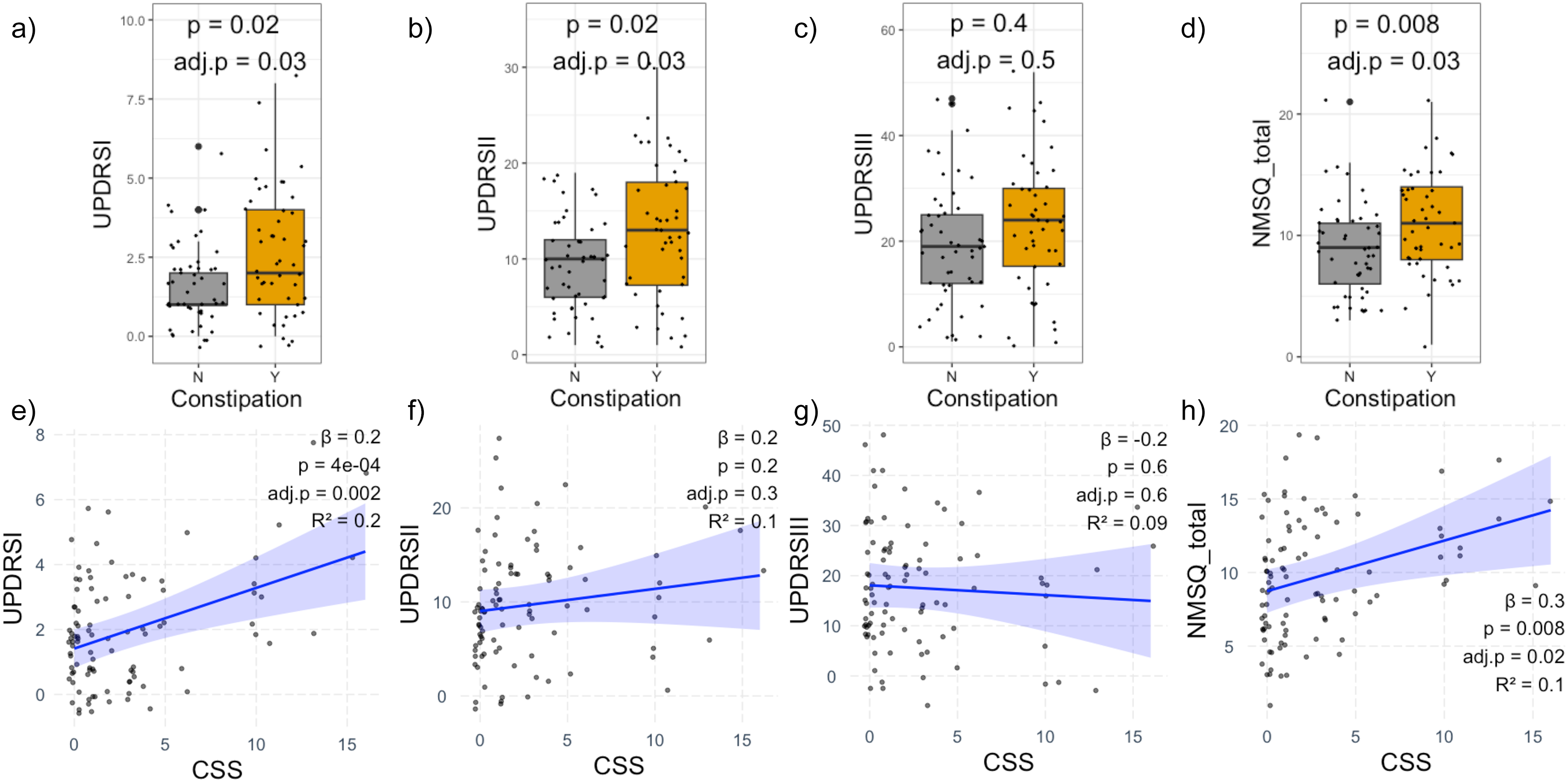
Association between Parkinson's disease-related symptoms and constipation. (a) Unified Parkinson's Disease Rating Scale I, UPDRSI. (b) UPDRSII. (c) UPDRSIII. (d) Total score across the non-motor symptoms questionnaire, NMSQ_total calculated excluding item 5 for constipation status. (e) UPDRSI and constipation severity using constipation scoring system, CSS. (f) UPDRSII and CSS. (g) UPDRSIII and CSS. (h) NMSQ_total and CSS. (e-h), scatterplots of mean-centered values showing linear fit line taken from linear regression with 95% confidence interval. All analyses adjusted for age, sex, disease duration, and laxative usage. False discovery rate-adjusted p-value (adj.p) was calculated to account for multiple testing.
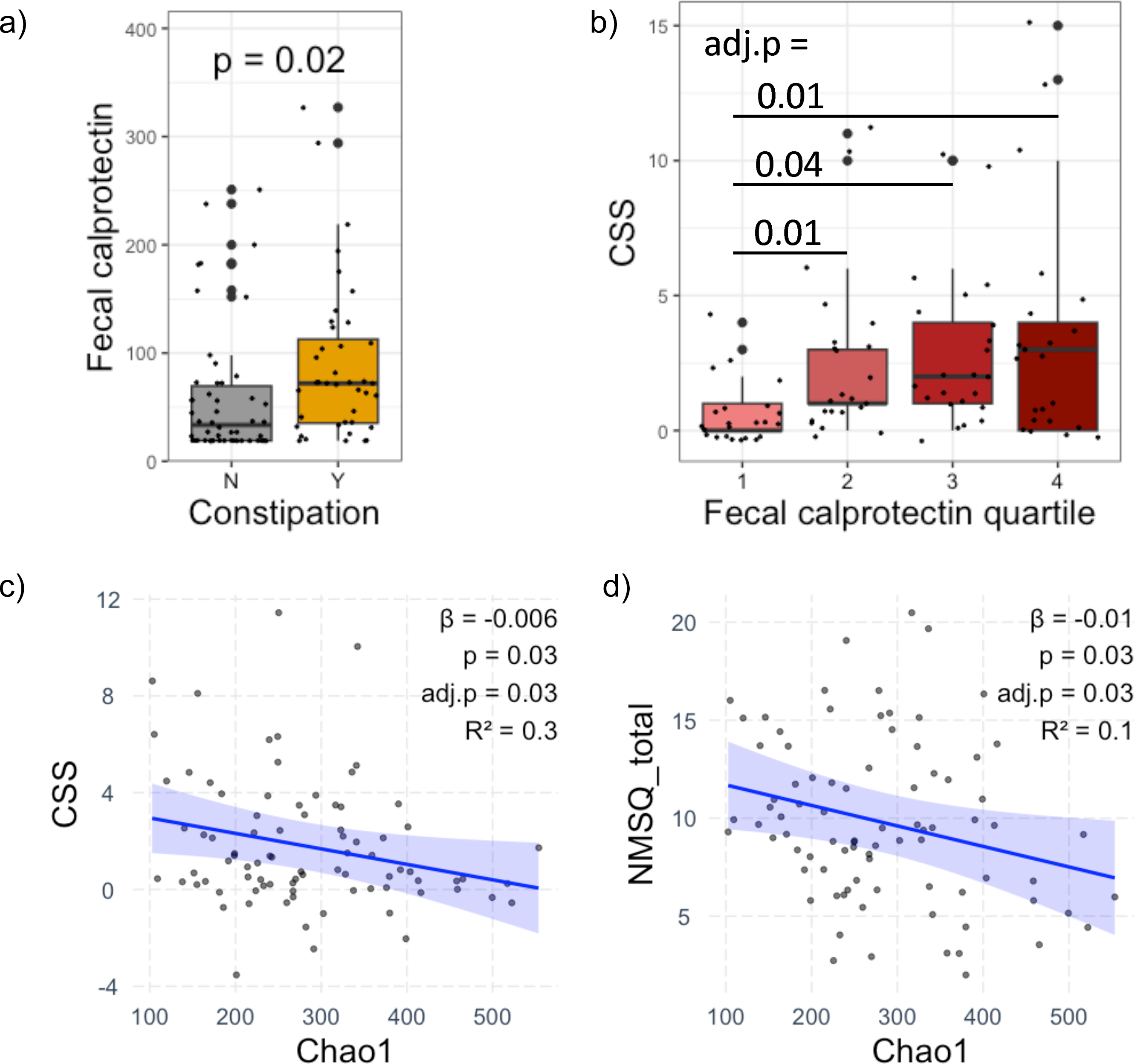
Association between Parkinson's disease clinical features and intestinal biomarkers. (a) Fecal calprotectin by constipation symptoms. (b) Constipation severity using constipation scoring system, CSS, by fecal calprotectin quartiles. (c) CSS versus fecal microbiota alpha-diversity index Chao1. (d) Total score on the non-motor symptoms questionnaire, NMSQ_total, versus Chao1. (c-d) Scatterplots of mean-centered values showing linear fit line derived from linear regression with 95% confidence interval. All analyses adjusted for age, sex, disease duration, and laxative usage. False discovery rate-adjusted p- value (adj.p) was calculated to account for multiple testing.
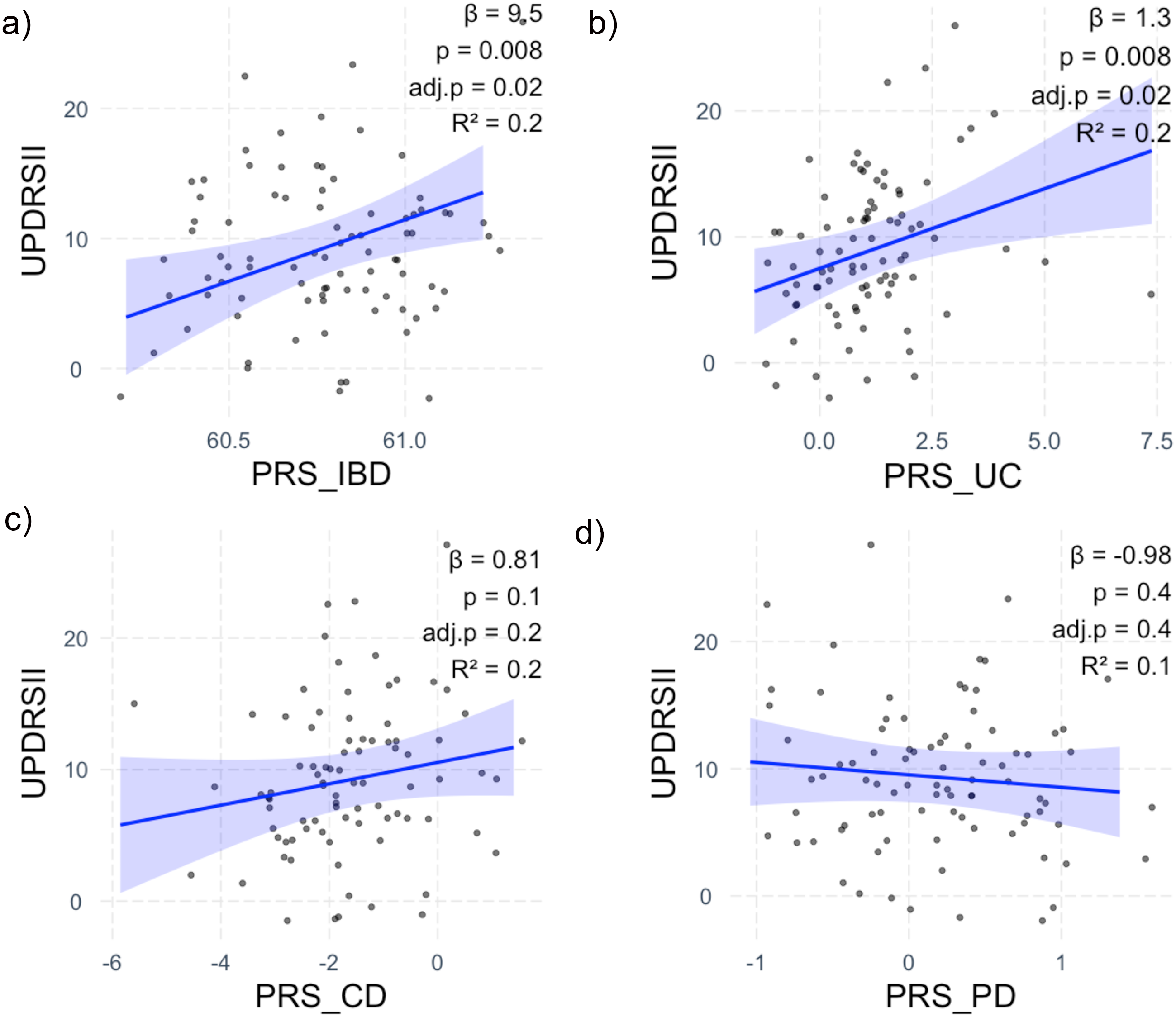
Association between genetic predisposition to inflammatory bowel disease and Parkinson's disease risk and Parkinson's disease symptoms. (a) Unified Parkinson's Disease Rating Scale II, UPDRSII and polygenic risk score for (PRS) for inflammatory bowel disease, PRS-IBD. (b) UPDRSII and PRS for ulcerative colitis, PRS-UC. (c) UPDRSII and PRS for Crohn's disease, PRS-CD. (d) UPDRSII and PRS for Parkinson's disease, PRS-PD. (a-d) Scatterplots of mean-centered values show linear fit line derived from linear regression with 95% confidence interval. All analyses adjusted for age, sex, disease duration, and laxative usage. False discovery rate-adjusted p-value (adj.p) was calculated to account for multiple testing.
Unsupervised cluster analysis
77 subjects with complete data on PRS-CD, Chao1, and fecal calprotectin were included in the clustering analysis. Average Jaccard bootstrap values were ≥0.85, suggesting high cluster stability. Overall, 47 PD patients were assigned to Cluster 1, while the remaining 30 assigned to Cluster 2. The first two principal components representing the cluster separation accounted for 75.9% of the total variance in the dataset (Supplemental Figure 2(a)). In post-hoc comparison (Figure 5(a)), subjects assigned to Cluster 1 showed more severe non-motor symptoms, including higher UPDRSII and CSS (adj.p = 0.049 and 0.046, respectively), and marginally higher NMSQ-total (p = 0.09), but not UPDRSI, UPDRIII, or disease duration (adj.p = 0.7, 0.4, and 0.2, respectively). As expected, those in Cluster 1 also had significantly higher PRS-CD (adj.p = 7e-6) and PRS-IBD (adj.p = 6e-4) as well as lower Chao1 (adj.p = 2e-8), Shannon (adj.p = 1e-5) and fecal SCFA levels (adj.p ≤ 0.002). No significant difference between the two clusters was detected with regard to the age, sex, age at disease onset, or fecal calprotectin.
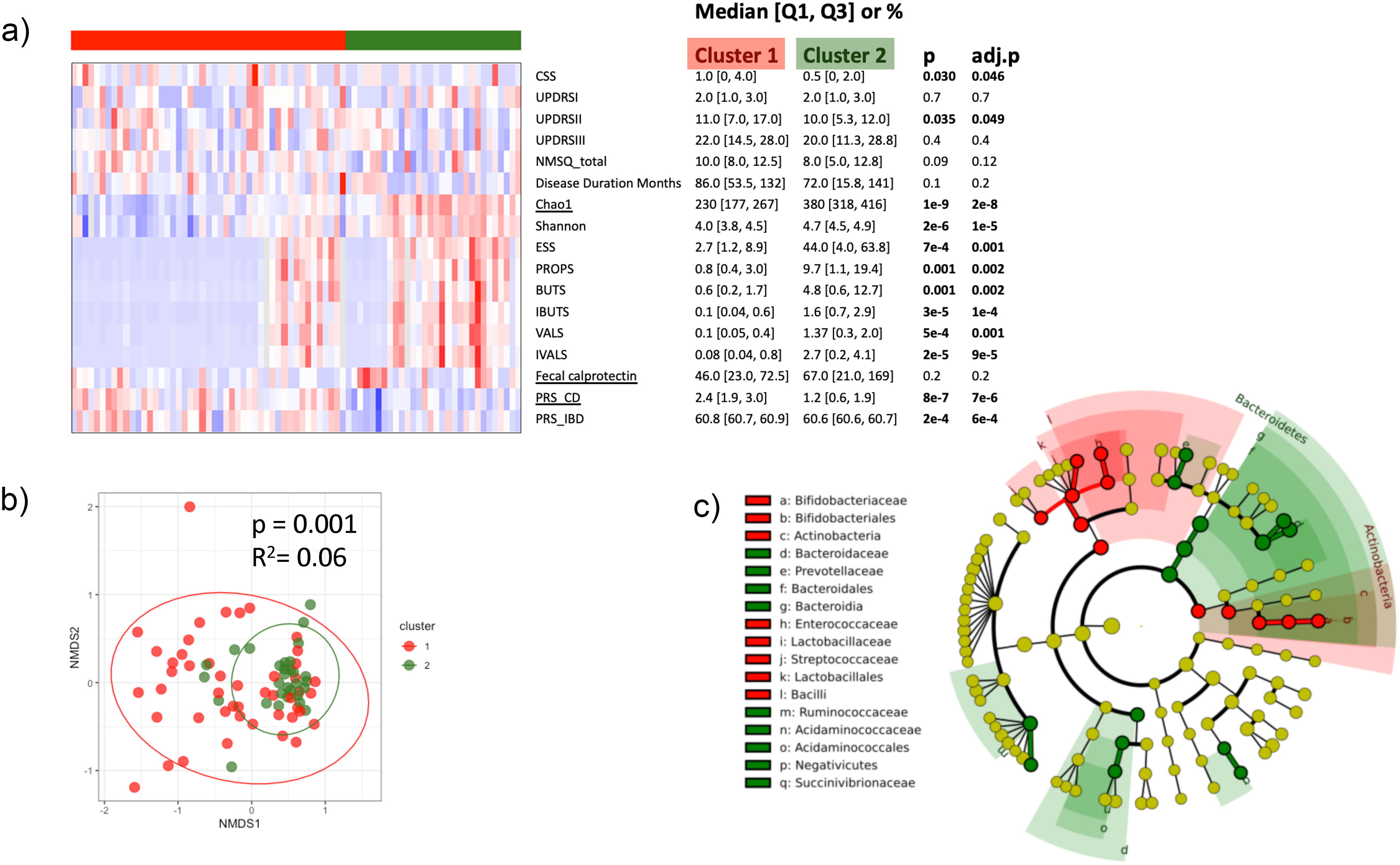
Unsupervised clustering of patients with Parkinson's disease based on biomarkers of intestinal inflammation. (a) Heatmap of post-hoc group comparisons. Variables used for clustering (Chao1, fecal calprotectin, and PRS-CD) are underlined. CSS, constipation scoring system; UPDRS: Unified Parkinson's Disease Rating Scale; NMSQ: non-motor symptoms questionnaire; Chao1 and Shannon, fecal microbiota alpha-diversity indices; short chain fatty acids (SCFA): ESS: acetic acid; PROPS: propionic acid; BUTS: butyric acid; IBUTS: isobutyric acid; VALS: valeric acid; IVALS: isovaleric acid; PRS-CD: polygenic risk score for Crohn's disease; PRS-IBD: PRS for inflammatory bowel diseases. False discovery rate-adjusted p-value (adj.p) was calculated to account for multiple testing. (b) Differences in fecal microbiota beta-diversity between the 2 clusters based on Bray-Curtis Distance Matrix. (c) Cladogram reflecting taxa differences between the 2 clusters after removing genera with mean abundance <0.1% or with >80% zero values. LDA (linear discriminant analysis) >4 was used as cutoff.
We next further explored the microbiota composition between the two clusters and demonstrated significant differences in beta-diversity (Figure 5(b), p = 0.001). After filtering out the low abundance genera, 71 out of 390 detectable genera were included in the differential abundance analysis. 19 taxa demonstrated differential abundance (LDA > 4), with the phylum Actinobacteria, family Bifidobacteriacea, and family Lactobacillaceae being significantly more abundant in Cluster 1, whereas phylum Bacteroidetes and family Prevotellaceae more abundant in Cluster 2 (Figure 5(c), and Supplemental Figure 2(b)). 328 KEGG pathways were predicted by PICRUSt, among which 7 showed significant differences between the two clusters (FDR < 0.05; Supplemental Figure 2(c)). Notch signaling pathway, chronic myeloid leukemia, Wnt signaling pathway, and phosphotransferase system pathways were significantly enriched in Cluster 1, whereas NOD-like receptor signaling pathway and protein digestion and absorption were enriched in Cluster 2.
Discussion
In this study, we demonstrated that in a cohort of patients with prolonged PD, disease manifestation was associated with various biomarkers of intestinal health. Specifically, we showed that having constipation was linked to more limited activities of daily living, and other non-motor dysfunctions, with the constipation symptom severity linearly associated to higher non-motor disease severity scores. Fecal calprotectin, an established biomarker of intestinal inflammation, was elevated in those with constipation and linked to longer disease duration irrespective of the age. Also, PD patients with worse non-motor symptoms had lower microbiota alpha-diversity and higher genetic predisposition to IBD. Importantly, unsupervised clustering based on intestinal inflammatory biomarkers identified two distinct groups of PD patients; those with more severe non-motor dysfunctions (reflected by higher UPDRSII, CSS, and NMSQ-total scores) were more likely to have greater genetic predisposition to IBD, as well as disadvantaged lower gut microbiota diversity and fecal SCFA levels.
In our cohort, nearly half of the PD patients reported having constipation, which is consistent with previous reports.4,54 We found positive relationship between the presence/severity of constipation and all major non-motor PD scores (UPDRSI, UPDRSII, and NMSQ_total), further strengthening the notion that gut dysmotility is not only an early biomarker of prodromal PD3,4 but may also be associated with disease manifestation. 55 Non-motor PD symptoms include diverse dysfunctions, such as sexual and urinary problems, orthostatic hypotension, cognitive decline, hyposmia, and sleep disorders. However, we did not observe a significant effect of constipation on the motor dysfunction score. Constipation has been reportedly associated with worse motor symptoms in a previous study, though in the cohorts with a significantly shorter disease duration (median <12 months), 55 suggesting that while constipation could facilitate accumulation and spreading of the misfolded alpha-synuclein to substantia nigra in the brain to initiate PD, it may not affect motor symptom progression in patients with a prolonged (median 82 months) well-managed disease or this association could be masked by medications and other exposures. However, it may still contribute to progressive seeding and spreading of the misfolded alpha-synuclein throughout the brain over time, thereby worsening the non-motor symptoms.
Also, we detected elevated levels of fecal calprotectin in PD patients with constipation, which further increased with disease duration and constipation severity independent of age. Several previous studies have shown higher fecal calprotectin levels in PD cases, compared to age-matched control subjects,5–9 and that fecal calprotectin correlates with disease duration and depression. 56 Fecal calprotectin is a sensitive biomarker of intestinal health known to accurately identify inflammatory conditions, 57 but less so the constipation.58–60 Our findings may indicate that PD-associated constipation has unique features compared to other types of constipation including elevated fecal calprotectin. At the same time, bacterial or parasitic infections, colorectal cancer, diverticulitis, and certain drugs, such as non-steroidal anti-inflammatory drugs (NSAIDs) like ibuprofen associated with mildly elevated fecal calprotectin have first to be ruled out. 61 Molecular and histological profiling of the intestinal samples from individuals with different types of constipation is warranted to distinguish between PD-related and -unrelated constipation. Moreover, future prospective studies should determine whether elevated fecal calprotectin could be a potential biomarker of PD initiation and development, especially in the patients with constipation, given that higher fecal calprotectin levels have been shown to precede the development of CD in unaffected first-degree relatives of CD patients. 62
Furthermore, we detected the inverse association between microbiota alpha-diversity and severity of constipation and overall non-motor PD symptoms. Previous studies have reported altered gut microbiota diversity and composition negatively affecting intestinal health, 63 and PD risk and progression. 64 Dysbiotic gut microbiota may modulate the colonic motility, secretion, and absorption by altering microbial metabolic activities involving bile acids, SCFAs, 5-hydroxytryptamine, and methane, leading to functional constipation.65,66 Compositional and functional changes in the gut microbiota following treatment with probiotics, prebiotics, and synbiotics have often resulted in the improvement of the symptoms, 67 suggesting a causal link between the microbiota and constipation. However, the relationship between the microbiota and the gut transit is likely bidirectional, especially in the setting of PD where lack of dopamine production in the brain and dysfunction of the peripheral nervous system may impair control of bowel muscles leading to bacterial overgrowth in gastrointestinal tract. 68 Prospective studies are warranted to better characterize the causal relationship between intestinal dysmotility and altered microbiota in order to identify early disease biomarkers and novel therapeutic interventions for patients with PD.
Of note, several prior reports have demonstrated the association of constipation symptoms with cognitive decline,55,69–71 or even rapidly progressive dementia55,72 in PD patients. While these reports are in line with our findings demonstrating an association between constipation presence/severity and UPDRSI, which captures intellectual impairment and memory loss, in our study, no differences in MMSE, a direct measurement of cognitive function, was observed between PD patients with vs without constipation. The plausible explanation is that despite the long disease duration in this cohort, the vast majority of study participants had normal cognitive status (MMSE > 25), limiting our ability to characterize the degree of intestinal dysmotility in PD patients with cognitive decline or dementia. As the latter patients are more likely to be left out of the research studies like ours, it is imperative to include individuals with cognitive impairment in future studies to better understand the effect of intestinal inflammation on cognitive impairment.
We showed that higher SCFA levels were correlated with later age of onset, shorter disease duration, greater microbiota alpha-diversity, and lower levodopa intake. This is consistent with previous studies that have shown negative correlation between fecal SCFAs and PD severity, further supporting the beneficial effects of SCFA.50,73,74 Also, later disease onset and lower LEDD have been previously reported in PD patients with higher SCFA levels. 7 Interestingly, we detected higher SCFAs levels in PD patients with more severe UPDRSI score, which is counterintuitive given the beneficial effects of SCFA. This observation can be at least in part explained by inverse correlation between LEDD and SCFA, as medications such as levodopa are known to affect microbiota composition, and thus SCFA production.75,76 A previous in vitro fecal fiber fermentation study has shown that levodopa therapy suppressed the production of certain SCFAs. 77 Levodopa has also been reported to be related to the decrease in SCFA-producing genera, such as Faecalibacterium 78 and 6 other genera. 79 We speculate that, while levodopa improves PD symptoms, at the same time it may suppress the microbiota SCFA production, 80 especially in those with prolonged disease duration. These findings may support a supplementation of SCFA-producing bacteria as a means to restore the microbiota composition balance in PD patients, which may help mitigate PD severity.
Furthermore, we report positive associations between genetic predisposition to IBD and its subtype UC, assessed by PRS measuring the cumulative effects of millions of genetic variants on disease risk,35,81 and PD-associated non-motor symptoms. PD patients with higher PRS-IBD and PRS-UC had more limited activities of daily living, independent of patient age and disease duration, whereas those with higher PRS-PD or PRS-CD did not. Also, a significant proportion of PD cases carried the IBD-associated NOD2 R702W and G908R risk variants, which may at least in part explain a high burden of intestinal inflammation in this cohort. Previously, an increased incidence of PD in patients with IBD has been reported globally,14–17 supporting a strong connection between intestinal inflammation and neurodegeneration. 82 This is also consistent with extensive genetic pleiotropy observed between IBD and PD.18,23,24 Yet, Mendelian randomization studies have not found significant evidence of the causal effect of IBD on PD,83,84 suggesting that it is the degree of intestinal inflammation rather than an established IBD diagnosis that could drive PD risk. Of note, we also observed positive correlation between PRS-PD and LEDD, suggesting PD patients with higher genetic predisposition to PD may progress faster and require a higher medication dose. Both PRS-PD and PRS-CD were also negatively associated with microbiota alpha-diversity, supporting microbiota manipulation as a potential target in individuals with high genetic risk.
Finally, we demonstrated that PD cases could be separated into two distinct clusters based on the intestinal biomarkers. PD patients with worse intestinal inflammatory profiles, including higher genetic predisposition to CD and IBD, lower bacterial alpha-diversity and lower beneficial SCFA levels, who were assigned to Cluster 1, presented with more severe non-motor symptoms of PD (UPDRSII, CSS, and NMSQ_total), compared to Cluster 2, with no difference in age or age of disease onset between the clusters. Furthermore, phylum Actinobacteria, family Bifidobacteriaceae, and family Lactobacillaceae were more abundant in those assigned to Cluster 1 than Cluster 2, which is in line with previous reports showing the abundance of those taxa to be significantly higher in PD cases compared to age-matched controls. 85 Similarly, phylum Bacteroidetes, and family Prevotellaceae were less abundant in Cluster 1 than in Cluster 2, agreeing with previous reports that the abundance of those taxa was significantly lower in PD cases compared to their healthy counterparts. 86
The strengths of our study include the availability of genetic, microbial, and biochemical biomarkers of intestinal dysfunction alongside clinical information in well-characterized PD cases with prolonged disease. The limitations include the fact that, by requiring study participant to be capable and willing to provide an informed consent and participate in study activities, we inadvertently excluded PD cases with more severe disease manifestation, thereby biasing the results towards null, especially regarding cognitive dysfunction, a common limitation87–89 that needs to be addressed in the future. Moreover, our cohort consisted exclusively of individuals of European ancestry and therefore caution is necessary when generalizing our findings to other populations. Additionally to fecal calprotectin, other fecal biomarkers of intestinal inflammation (beta-defensin 2 and lactoferrin 56 ) or intestinal permeability (alpha-1-antitrypsin 5 and zonulin5,9,56) have been shown to be significantly elevated in PD patients compared to age-matched controls. Prospective studies measuring those biomarker levels at early stages of the disease are warranted to better understand the causal relationship between the changes in intestinal biomarkers and PD risk and manifestation. 82 Also, the cross-sectional nature of our study limits our ability to infer causality and directionality of the observed associations. Prospective studies are warranted to determine if any of the inflammatory biomarkers precede the disease onset or are its consequence. Importantly, our study did not record detailed information on diet, water intake, or physical activity, which are essential determinants of general constipation, 90 nor was anti-TNF-alpha therapy recorded, which could affect PD progression. 15 Those factors need to be considered in future studies. Lastly, none of the study participants carried the strong genetic risk variant for PD, LRRK2 G2019S, reportedly associated with a more benign phenotype,91,92 and could potentially modify the relationship between the PD manifestation and the inflammatory biomarkers. Future studies should determine the impact of intestinal dysfunction on the disease course in G2019S carriers.
In summary, PD patients with worse non-motor symptoms presented with more severe constipation, lesser bacterial diversity, lower fecal SCFAs, and greater genetic predisposition to intestinal inflammation, independent of age and disease duration, suggesting that intestinal health could be a valuable target for controlling PD manifestation and progression.
Footnotes
Acknowledgments
The authors have no acknowledgments to report.
ORCID iDs
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Michael J Fox Foundation grant #010200 (to I.P., M.M.U.), National Institutes of Health's National Institute of Neurological Disorders and Stroke P20NS123220 (to I.P.). This work was supported in part through the computational and data resources and staff expertise provided by Scientific Computing and Data at the Icahn School of Medicine at Mount Sinai and supported by the Clinical and Translational Science Awards (CTSA) grant UL1TR004419 from the National Center for Advancing Translational Sciences. Research reported in this publication was also supported by the Office of Research Infrastructure of the National Institutes of Health under award number S10OD026880 and S10OD030463. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
Sequences generated and analyzed for this study are accessible at the NCBI Sequencing Read Archive (SRA) under BioProject ID PRJNA1106422 and submission ID SUB14410308. Other datasets are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
