Abstract
Background
While sleep disturbances are among the most frequent non-motor symptoms of Parkinson's disease (PD), there is a lack of evidence to support its treatment.
Objective
To evaluate the efficacy of opicapone 50 mg in treating sleep disturbances in patients with PD and end-of-dose motor fluctuations.
Methods
OASIS was an exploratory, open-label, single-arm clinical trial in PD patients with end-of-dose motor fluctuations and associated sleep disturbances. The primary endpoint was change from baseline to week 6 in Parkinson's Disease Sleep Scale 2 (PDSS-2). Secondary endpoints included functional motor and non-motor assessments (Movement Disorder Society [MDS]-Unified Parkinson's Disease Rating Scale [UPDRS], MDS-Non-motor Scale [NMS], 8-item PD Questionnaire [PDQ-8], 16-item PD Fatigue Scale [PFS-16], ON/OFF home diary), Clinical and Patient Global Impression of Change (CGI-C; PGI-C) and adverse events.
Results
At week 6, there was a significant reduction of −7.9 points (95%CI −13.6, −2.2; p = 0.0099) in PDSS-2 total score, with a significant mean change of −4.7 in the PDSS-2 domain of disturbed sleep (95%CI: −7.2, −2.3; p = 0.0009). Significant reductions were also observed in PFS-16 (−9.6; p = 0.0211), MDS-NMS total score (−28.9; p = 0.0015), MDS-UPDRS-III (−6.3; p = 0.0253), MDS-UPDRS-IV (−1.2; p = 0.0044) and PDQ-8 (−14.2; p = 0.0051). Absolute OFF-time was reduced (−142.1 min). Most patients (93.3%) and most clinicians (80.0%) reported improvements on PGI-C and CGI-C, respectively. Opicapone was well tolerated.
Conclusions
Adding opicapone 50 mg to levodopa/DDCI therapy in patients with PD and motor fluctuations and sleep disturbances improved both sleep disturbances and OFF time in these patients.
Plain language summary
Patients with Parkinson's disease (PD) commonly experience a range of sleep problems, including insomnia (difficulty falling or staying asleep), difficulties turning over in bed, excessive daytime sleepiness, acting out while sleeping, and other disruptions that affect their sleep quality and quantity, with an overall impact on their quality of life. However, there is no clear guidance on how to manage these problems. Opicapone is a PD medication that can be added to standard levodopa therapy to manage ON-OFF episodes in patients with PD. The OASIS study evaluated whether adding opicapone to a previous stable regimen of levodopa can be beneficial for sleep problems in patients with PD and ON-OFF episodes. During the OASIS study, 16 patients with a diagnosis of sleep problems associated with PD received opicapone in addition to their standard levodopa therapy for 6 weeks. At the end of the 6-week treatment period with opicapone, patients demonstrated significant improvements in overall sleep, particularly in their sleep disturbances. Moreover, patients reported improvements in motor and non-motor symptoms, including fatigue (another common symptom in patients with PD) and in their quality of life. Overall, improvements in health status and their clinical conditions were noted by both patients and clinicians following the addition of opicapone. This study suggests that opicapone can improve sleep problems in patients with PD and ON/OFF episodes but larger studies should be conducted to confirm these benefits.
Keywords
Introduction
Clinical Parkinson's disease (PD) is primarily characterized by motor impairment caused by loss of dopaminergic neurons and the consequent decrease in dopamine levels.1,2 However, non-motor symptoms, including neuropsychiatric symptoms, autonomous disorders, sleep disorders, fatigue, and sensory symptoms, feature in approximately 90% of patients with PD, with a substantial negative impact on health-related quality of life.3,4 Sleep disorders are amongst the most frequent non-motor symptoms of PD after psychiatric symptoms, affecting up to 80% of patients with PD, from early to advanced stages; for example, in the PRIAMO study, ∼41% of treatment-naïve patients reported sleep disturbances, but the percentage was >78% in complicated patients (defined as those patients who had developed motor fluctuations and/or dyskinesia under dopaminergic treatment).1,5,6 Sleep disorders include difficulties falling asleep with restlessness, frequent awakenings, nocturnal cramping, painful dystonia, and most frequently nocturnal akinesia with difficulties turning in bed and getting out of bed. Nocturnal urinary problems and confusion or hallucinations can also worsen patients’ condition.5,7 Patients with PD may also report daytime sleepiness and sudden sleep attacks as a consequence of the disease itself, disturbed sleep or side effects of medications.5,7
Symptomatic treatment of PD focusses on the stimulation of the dopaminergic system, such as replacement of endogenous dopamine via oral administration of the dopamine precursor levodopa.8,9 In the early stages of PD, levodopa is highly effective at controlling motor symptoms; however, with the progression of PD and the loss of dopaminergic neurons causing a reduction of the storage of dopamine, its therapeutic window of effect narrows, leading to the development of motor complications, such as end-of-dose motor fluctuations.10–12 These motor fluctuations are commonly associated with non-motor fluctuations, such as pain, sleep disorders, and anxiety, all of which result in lower quality of life and reduced well-being.1,11,13,14 Strategies to optimize levodopa therapy, such as changing the total daily dose of levodopa or its dose frequency, or adding catechol-O-methyl transferase (COMT) inhibitors, are therefore required to manage these complications. COMT inhibitors prevent peripheral degradation of levodopa, allowing a higher concentration to cross the blood-brain barrier. 15
Opicapone is a third-generation, once-daily COMT inhibitor approved in the European Union, USA, Japan, South Korea, Australia and other countries as adjunctive therapy to preparations of levodopa/dopa decarboxylase inhibitor (DDCI) in adult patients with PD and end-of-dose motor fluctuations who cannot be stabilized on those combinations.16,17 In two pivotal Phase III clinical trials, BIPARK-I and BIPARK-II, opicapone was efficacious as add-on to levodopa/DDCI therapy in reducing OFF-time in patients with PD and motor fluctuations, thereby improving patients’ overall health perception.18,19 These findings were further supported by the results of a Phase IV real-world study (OPTIPARK) conducted in routine clinical practice across the UK and Germany. 20 In this study, improvements in patients’ quality of life and non-motor symptoms were observed after 3 months of treatment with opicapone, further suggesting that its addition to levodopa therapy might also have a positive effect on sleep.
The objective of the OpicApone in Sleep dISorder (OASIS) study was to evaluate the potential benefits of opicapone 50 mg on sleep when added to existing levodopa/DDCI treatment in patients with PD and end-of-dose motor fluctuations and associated sleep disturbances, not previously treated with COMT inhibitors.
Methods
Study design
OASIS was an exploratory, open-label, single-arm, multicenter, interventional clinical trial in PD patients with end-of-dose motor fluctuations and associated sleep disturbances, conducted between June 2021 and June 2023 (NCT04986995). The study consisted of a 1-week screening period, 6-week treatment period, and 2 weeks of follow-up (Supplemental Figure S1). Following screening, patients received opicapone 50 mg once daily in addition to their current levodopa/DDCI treatment (3 to 8 daily doses) during the 6-week treatment period. The dose of levodopa/DDCI could be decreased (without changing the number of daily intakes) up to day 15 of the study if the patient experienced enhanced effects of levodopa due to opicapone. The levodopa/DDCI dose could be increased back to the baseline level, as required. Changes to levodopa/DDCI dose were not permitted after day 15 to the end of the study. No new anti-PD drugs or new medications for the treatment of sleep disturbances could be started during the study and any that were ongoing at baseline were to remain at a stable dose throughout the study. Concomitant medications for the treatment of sleep disturbances (e.g., sedatives, hypnotics, anti-depressants, anxiolytics or other) were allowed but these medications could not be newly started during the study. The regimen of these sleep medications had to remain stable for at least four weeks prior to study initiation, and during the study, their dose could only be reduced due to occurrence of medication-related adverse events (AEs).
Study population
Eligible participants were male and female patients aged ≥30 years with idiopathic PD, as defined by the UK Parkinson's Disease Society Brain Bank Clinical Diagnostic Criteria (2006) 21 or the Movement Disorder Society (MDS) Clinical Diagnostic Criteria (2015), 22 and with disease severity Stages I-III at ON (according to modified Hoehn and Yahr [H&Y] staging). In addition, patients were required to be experiencing wearing off despite optimal anti-PD therapy, with average total daily OFF-time while awake of ≥1.5 h (excluding the early morning pre-first dose OFF), and PD-associated sleep disturbances, confirmed clinically by the investigator via the Parkinson's Disease Sleep Scale 2 (PDSS-2) with a score of ≥18. 23 PD-associated sleep disturbances had to be present for ≥4 weeks prior to screening. Patients were also required to be receiving 3 to 8 intakes per day of levodopa/DDCI, which may include a slow-release formulation, on a stable regimen for ≥4 weeks prior to screening. Patients could be receiving other treatment for PD if the regimen had been stable for ≥4 weeks prior to screening and was likely to stay stable until the end of opicapone treatment.
Patients were excluded from the study if they had severe or unpredictable OFF periods, according to the investigator's judgement, or if they had major or prominent non-PD-related sleep disturbances, such as sleep apnea or narcolepsy. Other exclusion criteria included treatment with prohibited medications, including history or current intake of entacapone, tolcapone, monoamine oxidase inhibitors (except selegiline, rasagiline or safinamide), or any antidopaminergic agents. In addition, patients were excluded if they had previous levodopa/carbidopa intestinal gel infusion, deep brain stimulation or stereotactic surgery, including pallidotomy and thalamotomy, or if these procedures were planned before the end of the study.
All patients who had been enrolled in the study but discontinued the study before allocation to treatment at baseline due to any reason (withdrawal of consent, non-fulfilment of eligibility criteria, investigator's decision, etc.) were considered screening failures.
The study was conducted in accordance with the Declaration of Helsinki on Ethical Principles for Medical Research Involving Human Subjects, as well as with the applicable regulatory requirements of the participating countries. The clinical study protocol and the informed consent form were reviewed and approved by the respective independent ethics committee. All patients included in the study signed a patient consent form.
Study assessments
Change from baseline to week 6 (end of treatment) was evaluated for several measures of sleep, motor function and PD symptoms. Questionnaires and assessments were completed by patients, caregivers, or investigators, as appropriate. The primary efficacy endpoint was PDSS-2 total score. The PDSS-2 measures 15 characteristics of nocturnal sleep, in which patients with PD rate symptom severity on a 5-point scale (very often, often, sometimes, occasionally, never) during the previous 7 days, with higher scores indicating greater severity. Change from baseline in PDSS-2 for the domains of disturbed sleep, motor problems at night, and PD symptoms at night were also assessed.
Fatigue was assessed using the Parkinson's Disease Fatigue Scale 16 (PFS-16), a 16-item self-reported questionnaire measuring the presence of fatigue (7 items) and its impact on daily functioning (9 items) in patients with PD. 24 Patients rated PD-related fatigue symptoms within the previous 2 weeks on a 5-point scale (from strongly disagree = 1 to strongly agree = 5), with the PFS-16 total score ranging from 16 to 80, with higher scores indicating greater fatigue severity.
Other efficacy outcomes included the Movement Disorder Society-sponsored Non-motor Rating Scale (MDS-NMS),25,26 the Movement Disorder Society-sponsored Unified Parkinson's Disease Rating Scale (MDS-UPDRS) Parts III and IV 27 and the Parkinson's Disease Questionnaire 8 (PDQ-8). 28
Functional status was assessed via review of the Hauser's PD diary 29 for changes in absolute OFF-time, absolute ON-time, and ON-time without dyskinesia. Improvements or worsening of patients’ overall status was assessed using the Clinical and Patient's Global Impression of Change (CGI-C; PGI-C).
Safety and tolerability were assessed throughout the study by evaluating the incidence of treatment-emergent adverse events (TEAEs).
Statistical analyses
The study population for analyses of efficacy measures was the full analysis set (FAS), defined as all patients who were allocated to treatment and underwent at least one key efficacy assessment. The population for assessment of safety was the safety set, defined as all patients who took at least one dose of opicapone. The per protocol set was defined as all patients who were included in the FAS and had no major protocol deviations that could have an influence on the key efficacy endpoint. Efficacy results are presented for the FAS. The exploratory efficacy endpoints were analyzed using paired t-tests and descriptive statistics, including 95% confidence intervals (CI). Safety assessments were summarized by number and percent of patients. TEAEs were summarized by the Medical Dictionary for Regulatory Activities (MedDRA, version 23.1) preferred term and system organ class. TEAEs were also summarized by severity and relationship to treatment. Number and percent of patients with serious TEAEs, related serious TEAEs and TEAEs leading to discontinuation from the study were also reported.
Results
Study population
Of the 22 patients who were screened, six did not meet the eligibility criteria, therefore, a total of 16 entered the study and received opicapone 50 mg (Supplemental Figure S2). These patients were recruited from three study centers in Portugal (n = 12) and two study centers (n = 4) in Germany. One patient discontinued the study due to TEAEs and 15 patients completed the study.
Demographic and clinical characteristics at baseline are shown in Table 1. The mean age of patients was 65.4 years, half were male, and all were Caucasian. The mean duration of PD was 6.0 years, with PD-related sleep disturbances experienced for a mean duration of 3.2 years. Patients also reported psychiatric disorders at baseline (Table 1). The majority of patients (87.5%) received 4 or 5 intakes of levodopa per day, with most patients (93.8%) taking an additional anti-PD medication, most commonly pramipexole (43.8%). Patients had been experiencing motor fluctuations for an average of 1.5 years and reported a mean OFF time of 6.1 h per day.
Demographic and clinical characteristics at baseline (safety set).

*In the full analysis set (same population as safety set)
BMI: body mass index; MDS-UPDRS: Movement Disorder Society-sponsored Unified Parkinson's Disease Rating Scale; MF: motor fluctuations; PD: Parkinson's disease; PDSS: Parkinson's Disease Sleep Scale; PFS: Parkinson's Disease Fatigue Scale; SD: standard deviation
The dosage of sleep medications was maintained stable in all patients for the duration of the study, as no adverse events in relation to these medications were reported.
Overall, the levodopa dose was maintained stable during the study, with the mean (standard deviation [SD]) total daily dose being 536.72 (202.1) mg/day at baseline and 533.59 (208.7) mg/day at the end of the study.
Efficacy
After 6 weeks of opicapone 50 mg treatment, the primary exploratory efficacy endpoint of PDSS-2 total score was significantly reduced, with a mean change from baseline of −7.9 (95% CI: −13.6, −2.2; p = 0.0099) (Figure 1(a)). Similarly, there was a mean change of −4.7 from baseline at week 6 in the PDSS-2 domain of disturbed sleep, which was a significant reduction (95% CI: −7.2, −2.3; p = 0.0009) (Figure 1(b)). In addition, there were numerical reductions from baseline at week 6 in the PDSS-2 domains of motor problems at night (−1.9; p = not significant) and PD symptoms at night (−1.3; p = not significant; Figure 1(b)). Fatigue, as measured by the PFS-16, was significantly improved from baseline at week 6, with a mean reduction of −9.6 (95% CI: −17.5, −1.7; p = 0.0211) (Table 2). The mean change from baseline in MDS-NMS total score of −28.9 demonstrated a significant reduction in the non-motor symptoms of PD (95% CI: −44.7, −13.2; p = 0.0015) (Table 2). In the sleep and wakefulness domain of MDS-NMS, the mean change from baseline at week 6 was significantly reduced by −6.4 (95% CI: −11.9, −0.9; p = 0.0253) (Table 2).
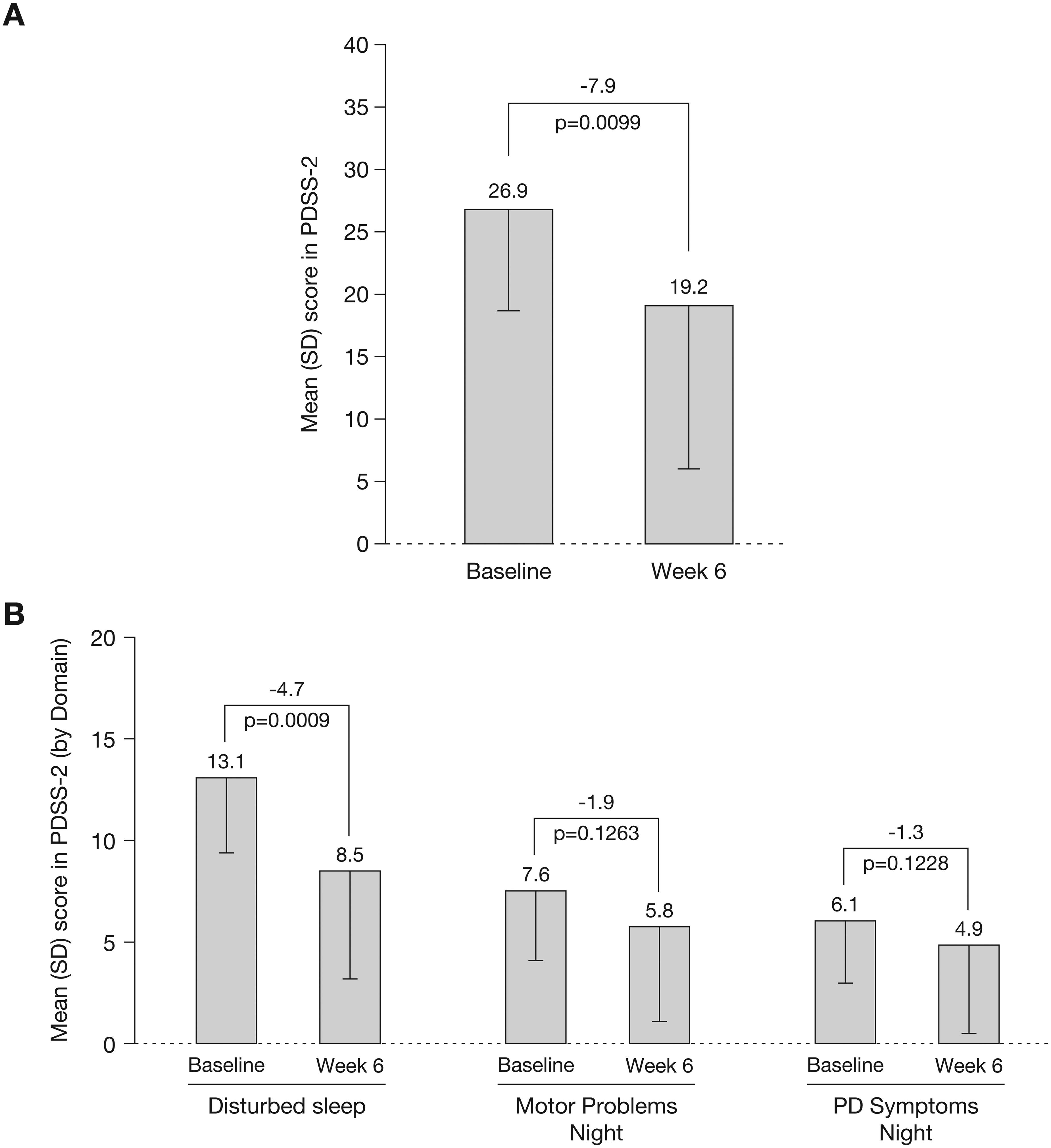
Change from baseline to week 6 in PDSS-2 (a) total score and (b) by domain (full analysis set). PDSS-2: Parkinson's Disease Sleep Scale-2; SD: standard deviation.
Change from baseline to week 6 in PFS-16, MDS-NMS, MDS-UPDRS, and PDQ-8 (full analysis set).

CI: confidence interval; MDS-NMS: Movement Disorder Society-sponsored Non-motor Rating Scale; MDS-UPDRS: Movement Disorder Society-sponsored Unified Parkinson's Disease Rating Scale; PDQ-8: Parkinson's Disease Questionnaire; PFS: Parkinson's Disease Fatigue Scale; SD: standard deviation; SE: standard error
Opicapone 50 mg treatment also led to significant improvements in motor function and motor complications at week 6, as measured by MDS-UPDRS Parts III and IV, respectively, with a mean reduction from baseline of −6.3 (95% CI: −11.6, −0.9; p = 0.0253) in motor function and −1.2 (95% CI: −2.0, −0.4; p = 0.0044) in motor complications (Table 2). Results for the PDQ-8 questionnaire also indicated that there was a significant improvement in quality of life at week 6, with a mean change from baseline of −14.2 (95% CI: −23.3, −5.0; p = 0.0051) (Table 2).
Absolute OFF-time was reduced after 6 weeks of opicapone treatment (change from baseline: −142.1 min), mirrored by an increase in ON-time and ON-time without dyskinesia (Figure 2). Most patients (64.3%) reported a reduction in OFF-time of ≥1 h at the end of the 6-week treatment period. The great majority of patients (93.3%) and most clinicians (80.0%) reported an improvement from baseline in their clinical condition at week 6, as evaluated by the PGI-C and CGI-C, respectively (Figure 3).
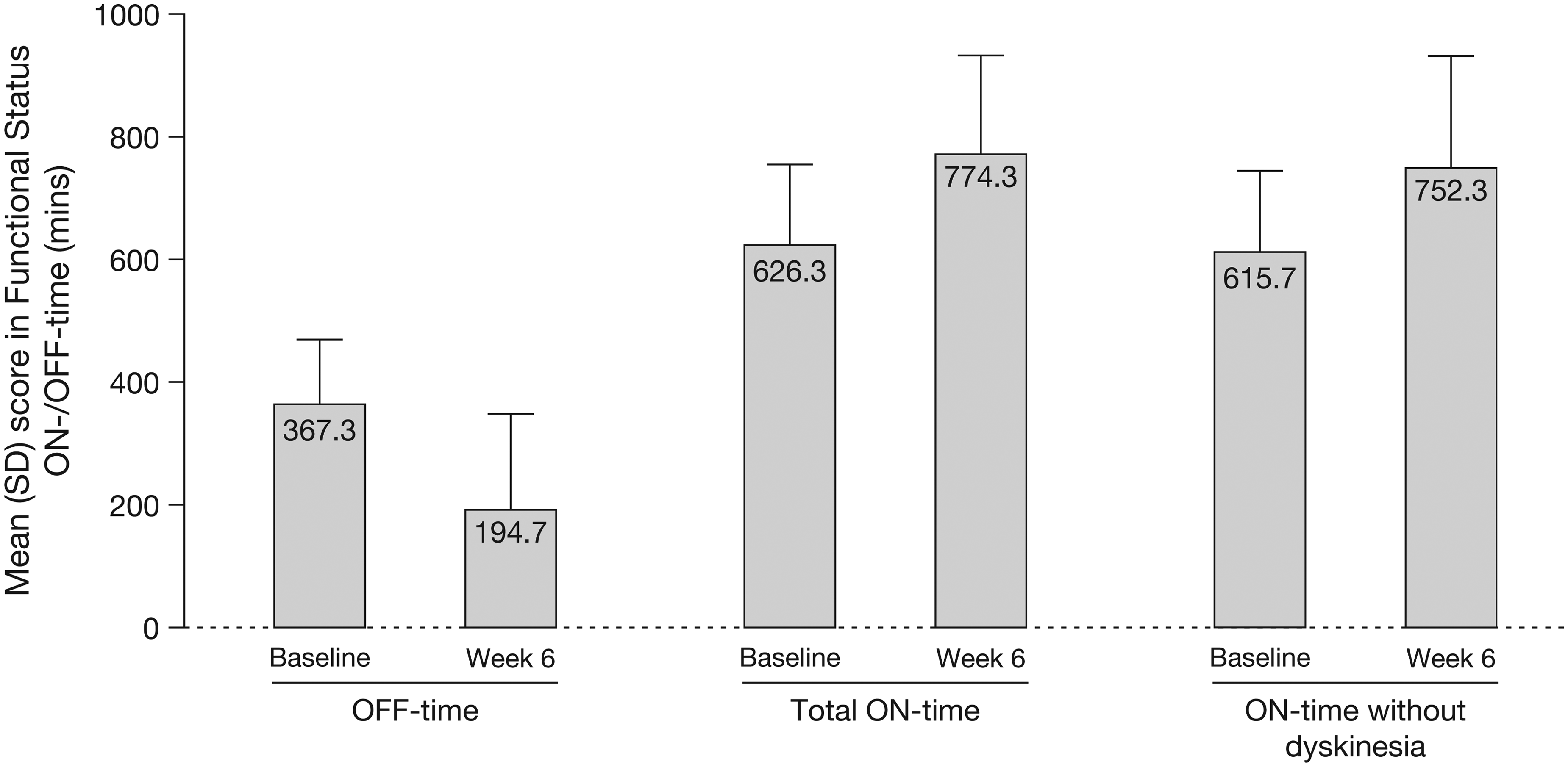
Change from baseline to week 6 in functional status (full analysis set). SD: standard deviation.
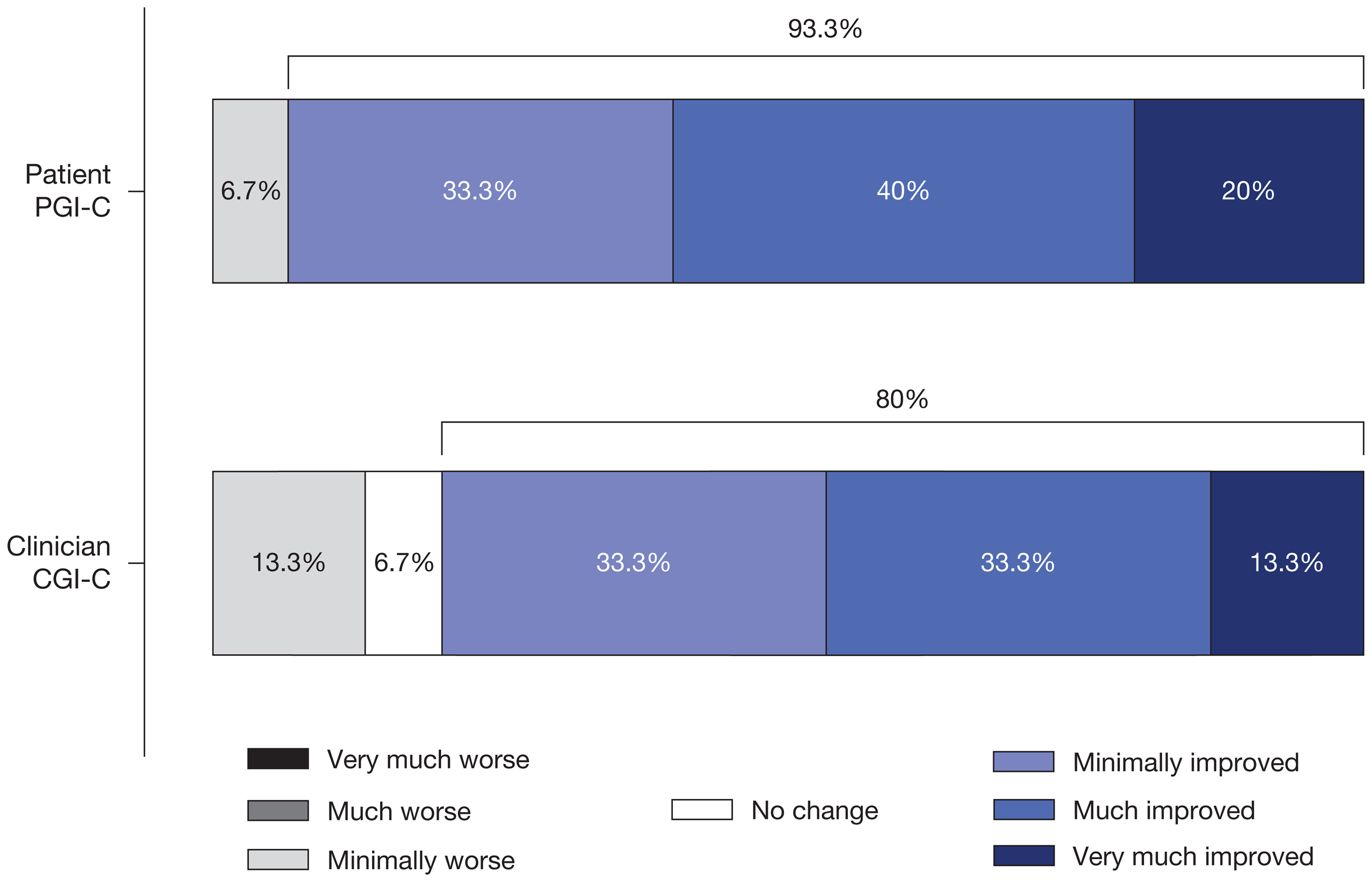
Patient and clinician global impression of change at week 6 (full analysis set). CGI-C: Clinical Impression of Change; PGI-C: Patient Impression of Change.
Safety
Opicapone 50 mg was generally well tolerated. Overall, 11 (68.8%) patients experienced a total of 20 TEAEs during the study, of whom four (25.0%) patients experienced TEAEs considered related to study treatment (somnolence, tremor, dry mouth, muscle spasm, and hypotension) (Table 3 and Supplemental Table S1). The majority of TEAEs (18/20) were mild and only two events were moderate. There were no serious TEAEs or deaths during the study. One (6.3%) patient had a dose interruption due to a TEAE and one (6.3%) patient discontinued due to TEAEs (freezing phenomenon and motor dysfunction; both considered unrelated to opicapone) (Table 3). All TEAEs occurred in only one (6.3%) patient each, except somnolence that was reported in two (12.5%) patients (Supplemental Table S1).
Summary of TEAEs (safety set).

TEAE: treatment-emergent adverse event
At least possibly related TEAEs.
Patients could experience more than one TEAE.
Discussion
The aim of the OASIS study was to evaluate the potential benefits of opicapone 50 mg on sleep and nocturnal disturbances when added to standard levodopa/DDCI therapy in patients with PD and end-of-dose motor fluctuations and associated sleep disturbances. The study also evaluated the effects of opicapone therapy on daytime motor and non-motor symptoms, quality of life and the overall health status of these patients. Adding opicapone 50 mg to levodopa therapy resulted in a significant decrease in the total PDSS-2 score, which measures both the general sleep quality and disease specific nocturnal disturbances in PD. The significant decrease in the score for the ‘disturbed sleep’ domain of the PDSS-2 suggests that opicapone can improve the quality of sleep of patients with PD. Improvements were also reported in the scores of the other PDSS-2 domains (motor problems and PD symptoms at night), but the changes were not statistically significant. The beneficial effects of opicapone therapy on sleep were also confirmed by the significant reduction observed for the sleep and wakefulness domain of the MDS-NMSS.
A key concern in clinical trial result analyses is how to interpretate the treatment effect, as statistical significance does not equate to efficacy or clinical relevance, 30 especially in pilot trials with a small number of participants. The PDSS-2 has been formulated to help both clinicians and patients understand and quantify the level of sleep disruption and it has been validated and translated into different languages.23,31 However, it is necessary to establish if changes in scores over time and/or following an intervention can be considered clinically relevant. In order to address this, a study of 413 patients with PD utilized an anchor-based method to determine the minimal clinically important difference for the PDSS-2 score, using the Patient Global Impression of Improvement (PGI-I) as the anchor. 32 The study concluded that any improvement larger than −3.44 points and any worsening larger than +2.07 points from baseline to endpoint should be considered a clinically important change for patients. 32 In the current study, the mean decrease in PDSS-2 total score was −7.9 points, well above the established threshold for a clinically significant change, indicating that opicapone might have substantial sleep benefits for patients with PD.
The correlation between the occurrence and the severity of sleep disturbances and the stages of PD was recently evaluated in a study of 154 PD patients with different H&Y stages. 33 The prevalence of sleep disturbances was over 80% in the subgroup of PD patients with H&Y stage 1 but increased up to 100% in the subgroups of patients with later stages of disease. Moreover, the PDSS-2 total score significantly increased with the H&Y stages, reaching a plateau at H&Y stage 3 and 4, indicating that sleep problems tend to worsen with the progression of PD. 33 Indeed, the mean PDSS-2 total score was 8.91 for H&Y stage 1, 15.68 for H&Y stage 2, 22.34 for H&Y stage 3 and 23.50 for H&Y stage 4. 33 In the current study, all patients had H&Y stage 2 or 3 at baseline with a mean PDSS-2 score of 26.9, supporting that the patients suffered from moderate to severe sleep problems (a PDSS-2 score ≥18 was an inclusion criterium). However, despite the severity of these sleep problems, 6 weeks of treatment with opicapone 50 mg was sufficient to induce clinically significant improvements.
Our findings support the results of a post-hoc analysis of the pivotal opicapone trials BIPARK-I and II, 34 which suggested that opicapone could be efficacious in improving sleep by reducing nighttime wakefulness. Hauser and colleagues analyzed pooled diary data to assess OFF-time, nighttime sleep duration and awake periods, demonstrating that adding opicapone to a levodopa regimen significantly reduced total time spent awake at night in patients with night-time “OFF” when compared to placebo. 34
The current study also suggests that opicapone's effect on non-motor symptoms is not limited to sleep but might extend to a wider range of symptoms, as indicated by the significant decreases in the MDS-NMSS and PFS-16 scores. Fatigue is a common non-motor symptom of PD, and it presents both as an independent symptom and as a consequence of the other conditions frequently recorded in patients with PD, such as anxiety, depression, and sleep disturbances. 35 Our results indicate that adding opicapone can significantly improve fatigue. The observed significant reduction in PDQ-8 score also suggests that the overall quality of life of patients improved following the addition of opicapone to standard levodopa therapy. Several studies have already demonstrated a correlation between the presence of comorbid sleep disturbances and other non-motor symptoms, including depressive symptoms, lower quality of life, poorer cognition, and fatigue,4,33,36 it is perhaps unsurprising that improvements in sleep disturbances were accompanied by improvements in other non-motor symptoms and overall quality of life.
There is a lack of data from robust clinical trials on the pharmacological treatment options for sleep disturbances. Lack of evidence for treating sleep disturbances in PD was found for the majority of treatments (controlled-released levodopa/carbidopa, pergolide, piribedil, eszopiclone, melatonin, modafinil and caffeine [modafinil and caffeine were evaluated for excessive daytime somnolence and sudden onset of sleep]) in a review study published in 2019. 37 Eszopiclone and melatonin were considered ‘possibly useful’ despite a lack of evidence. Based on a low-quality, positive study, rotigotine was considered ‘likely efficacious’ and ‘possibly useful’ in improving sleep, as it has been shown to have significant effects on sleep quality and maintenance in patients with PD; modafinil was considered ‘possibly useful’ for daytime sleepiness despite a lack of evidence. 37 This review highlighted the need for high-quality evidence and well-designed clinical trials in order to define the most appropriate strategies to improve sleep disturbances. To our knowledge, the current study is the first clinical trial evaluating the efficacy of opicapone in PD-related sleep disturbances as a primary outcome. Our findings indicate that opicapone might be efficacious in the treatment of these disturbances, despite the presence of limitations, such as the short duration of the trial and the lack of a placebo arm. Additional limitations of this trial include the open-label design and the low number of patients included. The observed magnitude of treatment effect with opicapone supports the need for larger studies to further assess its benefits and corroborate our findings. Notably, this was an exploratory study design to inform potential larger randomized trials.
Overall, adding opicapone to levodopa therapy improved both motor and non-motor symptoms and the general health status of the patients, as reflected by the CGI-C and PGI-C results. There is limited evidenced on the effects of COMT inhibitors on sleep disturbances, but our results are broadly in line with the ones obtained for tolcapone. The effects of tolcapone on sleep were evaluated in 61 patients with advanced PD in a prospective, open-label, multicenter, non-interventional study. The mean PDSS scores significantly improved from 21.6 at baseline to 16.3 at the 4-week endpoint. In addition, GCI-C, daily OFF time, activities of daily living, and quality of life were also significantly improved following treatment with tolcapone. 38
In summary, adding opicapone 50 mg to standard levodopa/DDCI therapy in patients with PD and motor fluctuations and sleep disturbances improved sleep disturbances and OFF time in these patients.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X241306711 - Supplemental material for Opicapone for Parkinson's disease-related sleep disturbances: The OASIS clinical trial
Supplemental material, sj-docx-1-pkn-10.1177_1877718X241306711 for Opicapone for Parkinson's disease-related sleep disturbances: The OASIS clinical trial by Joaquim J Ferreira, Miguel F Gago, Raquel Costa, Miguel M Fonseca, Joana Almeida, José Francisco Rocha, Joerg Holenz and Claudia Trenkwalder in Journal of Parkinson's Disease
Supplemental Material
sj-docx-2-pkn-10.1177_1877718X241306711 - Supplemental material for Opicapone for Parkinson's disease-related sleep disturbances: The OASIS clinical trial
Supplemental material, sj-docx-2-pkn-10.1177_1877718X241306711 for Opicapone for Parkinson's disease-related sleep disturbances: The OASIS clinical trial by Joaquim J Ferreira, Miguel F Gago, Raquel Costa, Miguel M Fonseca, Joana Almeida, José Francisco Rocha, Joerg Holenz and Claudia Trenkwalder in Journal of Parkinson's Disease
Footnotes
Acknowledgments
Editorial assistance for this manuscript was provided by mXm Medical Communications. Support for this assistance was funded by BIAL–Portela & Cª, S.A.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by BIAL–Portela & Cª, S.A.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JJF has received grants from GlaxoSmithKline, Grunenthal, Fundação MSD (Portugal), TEVA, MSD, Allergan, Novartis, Medtronic. Received consultancy and speaker fees and participated in advisory boards for GlaxoSmithKline, Novartis, TEVA, Lundbeck, Solvay, BIAL, Merck-Serono, Merz, Ipsen, Biogen, Acadia, Allergan, Abbvie, Sunovion Pharmaceuticals, Zambon, Affiris and Angelini.
MFG has received payment/honoraria for lectures from Zambon; Bial Portugal; Takeda; Amicus Therapeutics and payment/honoraria for advisory boards from Abbvie and Bial Portugal
MMF, RC, and JH are employees of Bial.
JFR and JA are former employees of Bial.
CT has received consulting/independent contractor fees from AbbVie, UCB, Roche, Bial, Ono, and Boehringer, speakers honoraria from AvvVie, STADA, Bial and Alexion; receives royalities from Thieme Publisher, License fee: PDSS-2; receives grant and contracted research support from The Michale J. Fox Foundation, EU: Era-Net program, BRAVA Projext; and is an employee (full or part-time) of Paracelsus-Elena Hospital, Kassel.
Data availability
The data supporting the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
