Abstract
People with Parkinson's disease (PD) are prescribed a variety of medications to mitigate symptoms and improve their quality of life. These symptomatic therapies cover a range of pharmacological classes, including classical dopaminergic treatments, other antiparkinsonian agents, and pharmacotherapies for non-PD conditions. Often, medications are prescribed for concomitant use and in increasing doses, particularly as the disease progresses. Documentation of these interventions in clinical trials is necessary to accurately capture medication usage, compare medication utilization across different studies, understand factors contributing to experimental therapeutic response, and analyze clinical trial data in a precise manner. At the present time, there is no current international standard for how these medications are documented within clinical trials. As a case example, we will highlight medication use documentation in a large international multicenter observational study commonly used as a reference for design of clinical trials. This review aims to raise awareness within the scientific community of the importance of proper medication documentation and the need for standardization to harmonize prescriptive practices, improve treatment interpretability, and perform consistently robust analyses from clinical trials data.
Plain Language Summary
Currently approved medications for the treatment of Parkinson's have been shown to improve motor symptoms, overall disability, and quality of life. There is an urgent need for novel treatments that delay, halt, or reverse the relentless progression of Parkinson's. New disease-modifying treatments are advancing rapidly, and integration of data across different trials is ideally needed to gain insights from every study. It is important to accurately capture the medications that are prescribed to people enrolled in clinical trials. Dopaminergic medications are known to have distinct effects in different patients and at varying stages of disease. This study carefully examines the classification of dopaminergic medications in the PPMI study, a flagship observational study that is used often by sponsors to plan and design clinical trials. The results demonstrate that there are some inconsistencies across sites and study cohorts in terms of classifying medication use in PPMI. There is an urgent need to align on consensus standards to document medications, including both prescribed Parkinson's medications and any other drugs. This would help to accurately identify potential beneficial and/or adverse effects of drugs being evaluated in clinical trials. Several initiatives aimed at standardization are underway with a need for unified consensus and implementation by sponsors of clinical trials.
Introduction
Antiparkinsonian medications have a significant impact on improving the quality of life for people living with Parkinson's disease (PD). Highly effective treatment of motor symptoms offers excellent motor responses; however, over time, efficacy begins to fluctuate across the course of the day and overall treatment effectiveness diminishes as the disease progresses.
Medications prescribed for PD can elicit a wide array of benefits as well as adverse effects that vary by individual across the spectrum of the disease. Overall, the risk-benefit is favorable for symptomatic therapies in that those taking dopaminergic medications have improved functionality and quality of life, reduced risk of premature mortality, and reduced symptom burden.1,2 Yet, some medications commonly prescribed for improving motor function are associated with undesired consequences such as impulse control disorders, 3 motor fluctuations and dyskinesias, and psychiatric symptoms including hallucinations and delusions. 4 Moreover, a significant increase in mortality has been reported in PD due to antipsychotic use. 4 Furthermore, the effectiveness of antiparkinsonian medications reduces as the disease progresses such that patients experience increased pill burden at moderate to advanced stages, thus putting patients at an increased risk of adverse health events due to drug-drug interactions.1–3,5
Since many medication classes have significant effects on quality of life and can be associated with various side effects, it is crucial that they are thoroughly documented in PD clinical settings and captured accurately in clinical research. This issue becomes particularly relevant during clinical trials. For example, when comparing the effects of novel investigational agents to the placebo arm, it is important to ensure that confounding effects on safety or efficacy are attributed to the therapeutic candidate itself as opposed to other medications taken by study participants. Furthermore, the inclusion/exclusion criteria for clinical studies typically define specific medications permitted or excluded from study participants; documentation of this adherence is integral. Concurrent medication use is common in the majority of PD clinical trials, yet many challenges exist in how this information is captured in both clinical trials and in observational research studies.
Assessment of dopaminergic therapies
It is well known that current symptomatic treatments of PD can mask putative beneficial effects of novel therapies being evaluated in clinical trials, 6 yet masking is partly due to crude clinical measures of disease progression. In order to mitigate the impact of symptomatic medications on the read out of presumed efficacy of tested intervention, many trials are carried out at early stages of PD, prior to when symptomatic drug treatment is necessitated. Clinical trials are commonly carried out in drug-naïve participants or under conditions where symptomatic dopaminergic therapy is withheld for an extended period of time prior to clinical evaluation. Examples of recent PD clinical trials investigating the potential effects of disease modifying targets serve to illustrate this concept.7,8 The pharmacology of dopaminergic medications is complex and varies according to the pharmacological class of drug and to the specific medical product itself.
Complex pharmacological features of dopaminergic therapy form the basis of motor fluctuations. The pathophysiology of motor fluctuations is strictly related to the pharmacokinetics and pharmacodynamics of Levodopa (L-Dopa) which are dominated by the short plasma half-life of the drug and the short-duration response. This ON/OFF pattern represents the portion of the antiparkinsonian response that parallels the plasma L-Dopa levels. Several pharmaceutical companies have aimed to address ON/OFF states by varying formulations and routes of administration for a more consistent therapeutic effect. These include controlled-release pump levodopa 9 and intravenous or transdermal delivery of lipid-based nanoparticle loaded levodopa. 10 These medications emphasize the importance of documenting the route of administration and drug formulation within clinical trials.
Accurate documentation of ON and OFF medication status is important in order to evaluate effects on disease progression, yet the precise definition of ON and OFF dopaminergic medication is not consistent across different PD clinical studies. Furthermore, the time course of loss of full clinical effect may take significantly longer than typical defined OFF states and washout times based on pharmacokinetic half-lives. For instance, it has been estimated that more than a month of washout would be needed to eliminate the effects of levodopa, despite a pharmacokinetic half-life of approximately 90 min. The concept of practically defined OFF states is defined strictly based on the time since the last dose. 11 The duration of time needed to achieve undetectable levels of dopamine in the circulation after administration of L-Dopa can be long and may not reflect true brain tissue levels.12–14 As a result, it is not possible to accurately quantify PK/PD at the site of action. The variability of medications administered across different PD research studies and clinical trials leads to challenges in the quantification of disease progression, accurate prognosis, and the interpretation of putative new treatment effects. To estimate the total daily dose of L-Dopa, it has been proposed to utilize a conversion factor to generate a total L-dopa equivalent daily dose (LEDD), calculated as a sum of each parkinsonian medication. Therefore, LEDD provides an artificial summary of the total daily medication a patient is receiving. 15 Without accurate documentation, it is challenging to successfully decipher which results are truly due to the investigational therapeutic agent. A recent publication carried out by the Movement Disorders Society developed an updated LEDD conversion formula to serve as a tool to compare the relative dose intensities of novel and longstanding drugs used to treat PD in clinical studies. 16
Variability in assessments of antiparkinsonian therapies
Many technical issues exist beyond the problems of accuracy and completeness of medication logs. When all medications are logged properly, it can still be hard to interpret the actual dose of medications and the duration of pharmaceutical treatment, particularly for medications that have been prescribed for long durations of time. Off-label prescription of medications further complicates this interpretation. For example, exenatide is a GLP-1 agonist approved for use to increase insulin release in patients with Type II diabetes mellitus. More recently, this medication has been prescribed for off-label usage for patients with PD, as some studies have shown GLP-1 agonists can play a neuroprotective role. 17 Medications prescribed off-label can elicit unwanted side effects and unanticipated drug-drug interactions, which can have an adverse effect on patient safety, such as risk for falls, which is particularly common in the advanced stages of PD. 18
Natural history studies benefit disease drug development because they provide an opportunity for researchers to take a range of physiological measurements that could allow for identification of biomarkers and new endpoints for the disease. A growing number of natural history studies are being carried out in PD.19,20 Observational cohort studies serve to enhance broader understanding of the risk, onset, and manifestation of disease progression throughout the continuum and have been key to elucidate novel discoveries related to genetics and biomarkers. Analysis of existing and new PD cohorts is underway aiming to carry out unbiased strategies for enabling precision medicine. 21 The true benefit of natural history observational cohorts would be enhanced by integration across different cohorts to improve sample size, predictive accuracy, and reproducibility of novel findings. Cataloging of medication use across the different cohorts is key to accurate standardization, harmonization, and evaluation across distinct PD clinical studies.
Flagship case example: PPMI
Parkinson's Progression Markers Initiative (PPMI) is a flagship multisite global clinical study that aims to discover biomarkers of PD onset and disease progression. 22 Significant progress has resulted from this seminal study over the past decade to inform progression trajectories of distinct biologically defined subtypes in ways that are informing clinical trials.
This international study consists of several sub-study cohorts and collects longitudinal data that are used to inform the impact of factors on the rate of disease progression. PPMI is unique in that all data are made available to the public, consensus standards are employed, and all data are carefully collected to allow data to be integrated across all sites. 22 This study protocol requests that sites collect data using a standardized, comprehensive medication logging system, with detailed recommendations in the operations manual. This manual requires clinical researchers to document reasons behind any missing data.
There has been significant progress in the understanding of disease progression based on PPMI to date, including novel discoveries of biomarkers, genetics, and different clinical subtypes of the disease.23–25 In fact, a majority of PD clinical trials have relied on data from PPMI for design and selection of target populations. 22 The medication status of participants in PPMI has been a topic of research evaluating drugs for motor26,27 and non-motor manifestations. 28 For example, Javidnia M. et al., (2020) found that non-motor medication use was reported by 73% of PPMI participants, most commonly for depression, constipation, and anxiety.
Table 1A illustrates the PD classes of medications that are reported for participants in PPMI.
Medication classes and drug names for PPMI.
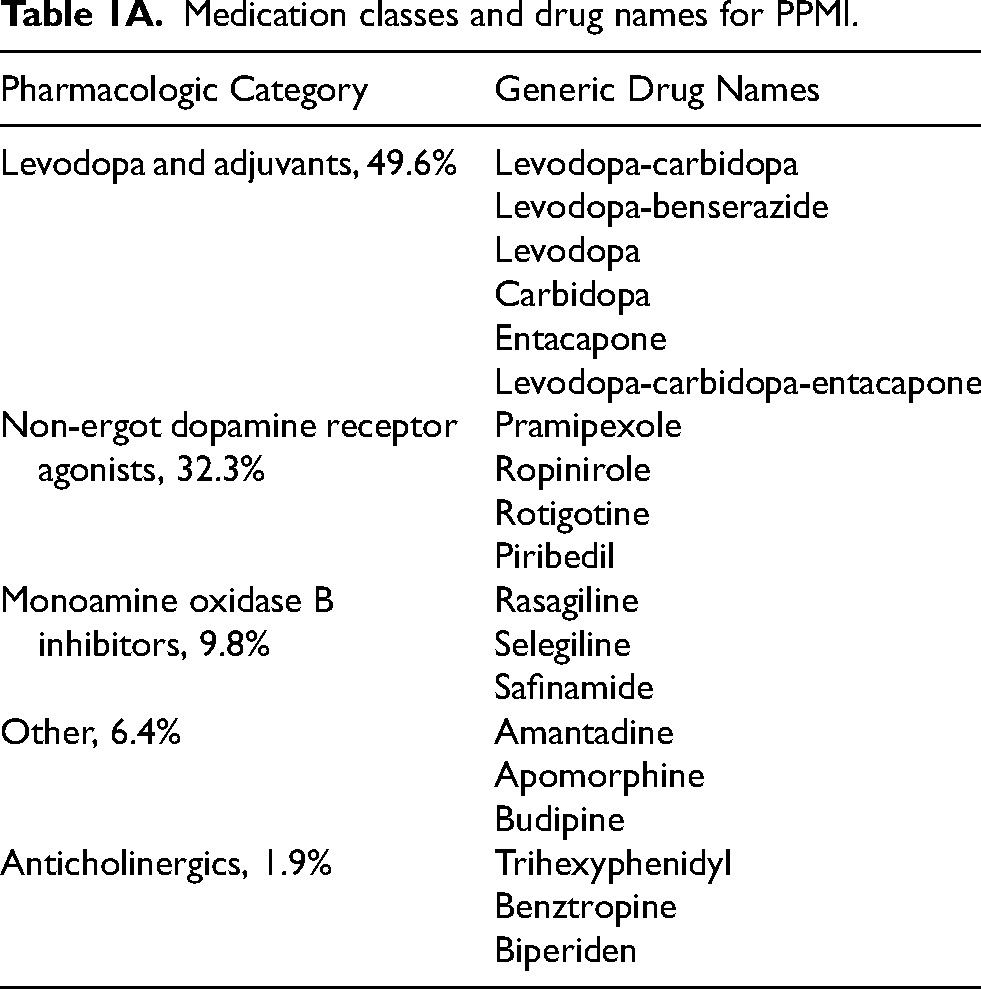
List of drug class monotherapy and combination therapies in PPMI.
Classes of dopaminergic medication administered to Parkinson's disease participants enrolled in PPMI evaluated at baseline at each visit up to year 5 following enrollment of the de novo cohort. COMT, catechol-O-meth.
Table 1B illustrates the list of PD medications prescribed to treat motor symptoms among PPMI study participants. The category defined as ‘other’ includes a diverse array of medications that do not fall under typical pharmacological categories for antiparkinsonian agents.
Figure 1 illustrates a graphical representation of the trajectory of medication onset in PPMI in conjunction with the reduction in the number of treatment naïve participants as a function of time. This diagram illustrates an inflection point at nine months following baseline, a time point at which it becomes a challenge for current clinical trials to remain treatment naïve.
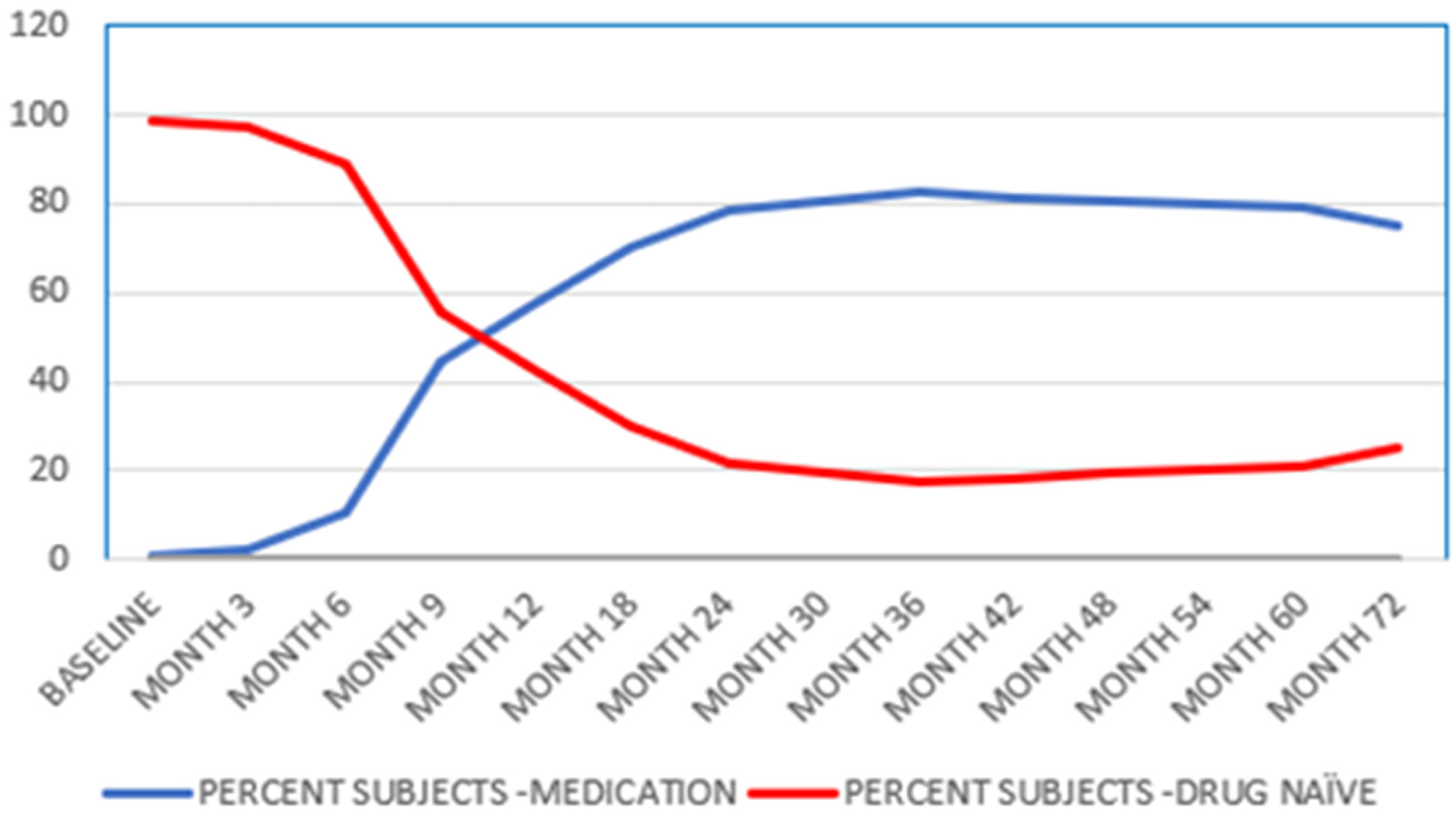
Graphical illustration of the time course of medication use in participants enrolled in the PPMI De Novo Cohort. The values represent percentages of total subjects at each timepoint over five years. The medications that are included in this figure are primarily PD medications (Table 1).
Many trials are aimed at therapeutic intervention in de novo participants to avoid the confounding effect of standard of care medications. 29 Overall, the total medications prescribed increases maximally at 18 months and then appears to stabilize over a five-year time since onset of PD clinical diagnosis. These results emphasize the point that medication usage is complex and varies significantly across the disease continuum, further complicating consistent documentation within and between different participants and studies.
PPMI is recognized for collecting data with a high-level of standardization as compared to other observational natural history studies. All global sites have agreed to collect data with the same specifications, protocols and preanalytical methodologies. This rigor has been key to generating novel findings, including what is now being recognized as the first in vivo assessment of alpha-synuclein that predicts onset of PD up to eight years prior to onset of clinical symptoms. 30 Analysis of the PPMI data requires diverse experts to work together to analyze and interpret the data successfully.
There is a need to prospectively identify ways to optimize future data collection approaches for accurate collection of medications. Categorization of PD medications is needed in order to successfully identify findings at the patient level. The historic approach to capture medication use and lack of standardization can lead to a wide variety of issues. To illustrate one example, Figure 1 shows an analysis of the differences of medications within the PPMI database. These include differences in capturing medications, including misspellings, using the brand name, and including dose details (Figure 2).

Illustration of medication entry variations observed in PPMI. Bubbles are color-coded based on the medication's mechanism of action. Each cluster within a color group represents a medication. The subgroups within each cluster represent all the variations (e.g., brand names, misspellings, and dose details) within each medication's name. The size of each bubble correlates to the frequency of which the variation occurred within the data set. Results are from evaluation of PPMI concomitant medication data, downloaded on 01/07/2021. For example, in the green bubble group, the cluster on the left represents all the logged variations for the MAO-B inhibitor Rasagiline. This included misspellings such as “Rasagilin” and “Rasageline”.
Additional issues that are apparent from evaluation of medication use at the patient level in PPMI to date cover a range of themes. For example, time of drug administration prior to clinical examination and dose of medications are frequently not recorded. Furthermore, data regarding medication history is often difficult to interpret properly. For example, medications prescribed for off-label use further complicate interpretation of results and records of both supplements and recreational drugs are rarely noted. Additionally, as expected in studies with international cohorts, there is a lack of uniformity of medication names within the source data set, given there are different brand names for the same drug throughout different countries.
The above issues have been observed in other observational cohorts, including the Parkinson's Disease Biomarker Program (PDBP). 31 Such challenges are specifically apparent in combinatorial integrated data from observational cohorts and PD clinical trials (Critical Path for Parkinson's unpublished observations, Critical Path for Parkinson's (CPP) Integrated Parkinson's Database) (https://c-path.org/programs/cpp/#section-24786). Lack of standardization of PD medications has additionally made it challenging to harmonize and integrate data across different clinical studies. Overall, these issues pose barriers to achieving the goal of quantifying the impact of different medication classes on the rate of disease progression in defined subtypes of PD, a gap highlighted by regulatory agencies. 32
Existing data standards
The concept of harmonization of data elements to enable meta-analysis and collaboration has been advanced in the U.S. by the Common Data Elements (CDE) initiative, which defines standards for data collection for all US government-funded research. 33 There are numerous neurological diseases with CDEs, including stroke, epilepsy, PD, and traumatic brain injury (TBI), in addition to new initiatives that are being launched outside of the U.S. to align CDEs across brain diseases (BrainCODE, Canada). 34 The National Institute of Neurological Disorders and Stroke (NINDS) recently released an updated revised collection of CDEs for PD that includes several sections related to medication use. This initiative, led by clinical trial experts and academic PD clinical leads, was published after public review and feedback (https://www.commondataelements.ninds.nih.gov/cde-catalog).
Comprehensive case report forms (CRFs) have been developed for Parkinson's-specific medications and non-Parkinson's medications as two distinct forms in the new PD CDEs developed by NINDS. Specific examples of variables to collect in the new CRFs include the following:
Text describing reason for administration of a prior/concomitant (non-study) agent or measure. Total daily dose of the prior or concomitant medication taken by the participant/subject. Indicator of whether the participant/subject reported taking any medications during the time period relevant to the study protocol. Frequency of use of a prior/concomitant medication. Code that represents the dosage unit of measure of the prior or concomitant medication administered. Unified Code for Units of Measure (UCUM). The date (and time, if applicable and known) on which the prior/concomitant medication usage began. Dosage unit of measure of the prior or concomitant medication administered.
This level of detail and consistency of medication use may not be available in PD research studies and in PD clinical trials carried out in the past; however, the new CDEs will be required for all NIH-funded prospective studies. These standards are focused on U.S.-funded studies, yet global impact may be possible as well. An international CDE project for TBI research has been endorsed by numerous federal agencies and professional organizations to enable standardization to take place in other countries and may be a model to explore for the future.
35
Several groups have launched initiatives with recommendations for a more comprehensive system of cataloging medication use for all participants with standardization for ongoing and future collections of data (Table 2). For example, PPMI study investigators have recently launched a new version of the PPMI database that has more granular information to be collected for medications.
Initiatives for generating universal medication documentation resources and standardization in Parkinson's and related disorders.
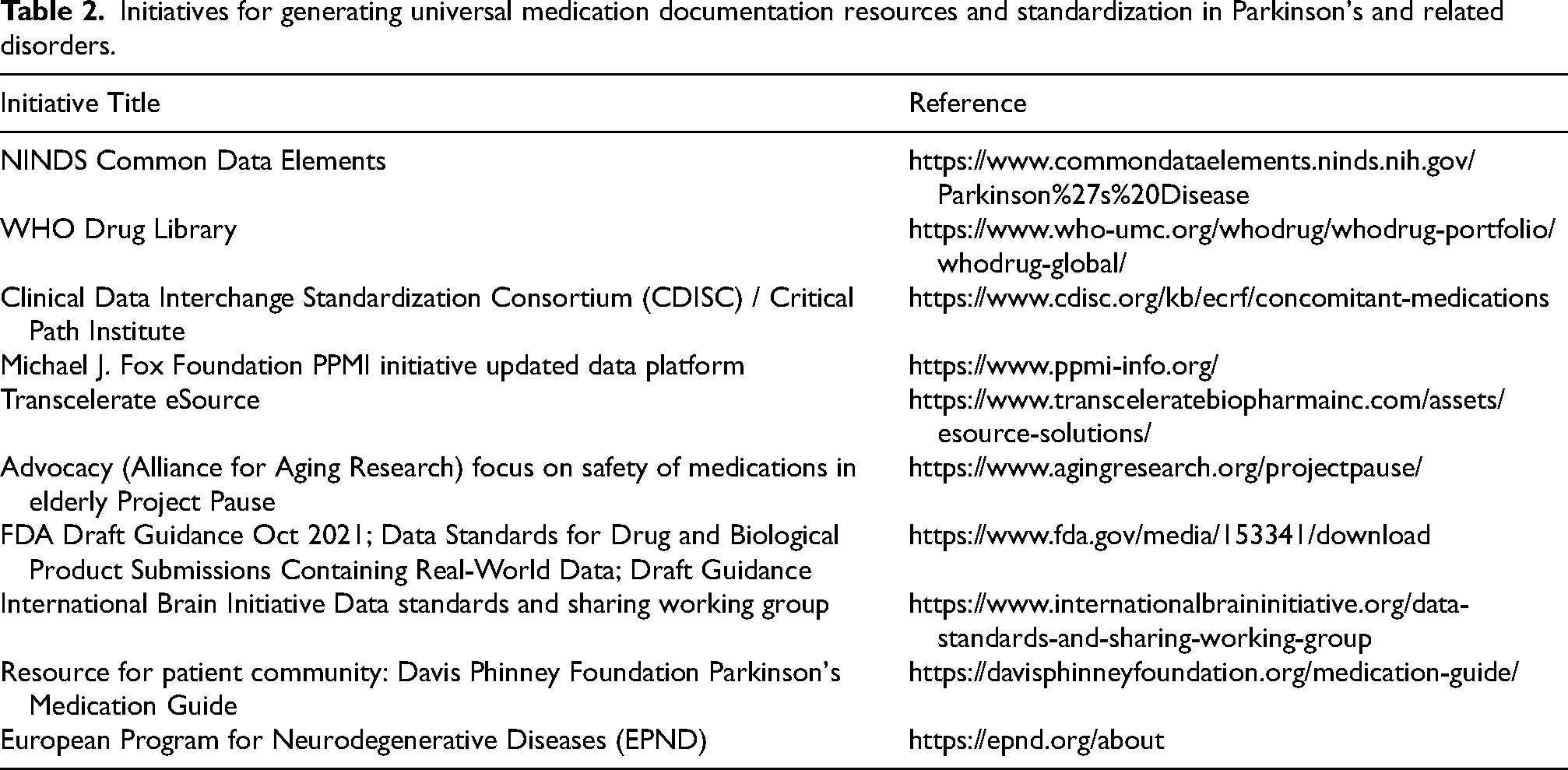
These examples cover a wide range of approaches; some are targeted for different stakeholders including clinicians, patient community, and clinical trialists. In order to achieve a desired level of standardization, the first step is to initiate this conversation about the difficulties of medication logging within the scientific community and to raise awareness of the consequences of inaction.
From challenges to solutions
The recommendation to embrace a global medication documentation scheme for PD clinical trials would have significant impact for future PD clinical research and drug development. By streamlining this process and agreeing on a consensus minimal data set for collection of medications with distinct terminology, more efficiencies will be gained in interpreting analysis of investigational products. A standardized consensus methodology that can be used for all clinical trials would also make meta-analysis more efficient and accurate.
The future holds tremendous potential for rapid advances given the innovative progress in development of digital health technologies. Medical devices, when combined with advanced analytics, are poised to transform our ability to assess patients in their natural environments. New methodologies in this area are evolving rapidly including the ability to track and monitor medication 36 in ways that may impact both drug development and clinical trial practice. 37 Collaborations are essential for success in advancing digital health technologies with efficiencies as advised by regulatory agencies.38–40
There are a growing number of case examples that highlight the critical impact of data standardization in advancing drug development tools for clinical trials and research for diseases of high unmet medical need. Critical Path Institute's success in regulatory endorsement of biomarkers and modeling tools in T1D41,42 and kidney transplantation 43 were achieved based on integration of diverse data sources across observational and clinical trial data. In the neurosciences, successes in Alzheimer's disease and Huntington's disease biological staging frameworks were enabled by data standardization and integration.
In PD, the global GP2 consortium collects diverse cohorts from around the world through collaboration and openly sharing data, processes, and results whose mission is to further understand the genetic architecture of PD through genotyping diverse participant groups and studying highly suspected monogenic forms of PD. 44 This effort has resulted in discovery of novel risk genes in diverse underrepresented populations. The potential response to medications of diverse populations is not known and represents a high profile need for why the timing for medication standards is needed. Such efforts were enabled by the ability to integrate diverse sources of clinical data to harmonize to a common data standard. The US FDA has pioneered data standardization policies and have generously funded many disease specific standards including those for disorders of the nervous system including Alzheimer's disease.45,46
With the rapid evolving progress and recent FDA drug approvals to treat disorders of the nervous system such as Friedreich's Ataxia, 47 amyotrophic lateral sclerosis, and Alzheimer's disease, the opportunities that lie ahead hold tremendous potential. The field of PD is at an inflection point given the rich portfolio of candidates in the pipeline, 48 novel biomarker discoveries, 30 and the new advances in biological staging of disease. 49 There is a sense of urgency to advance collective and harmonized strategies to advance data-driven solutions transnationally. Adoption of consensus standards and methodologies in capturing medication use with attention to rigor and harmonization will serve to accelerate progress across the globe to address the urgent needs of people with lived experience of PD.
Conclusion
The current pipeline of PD therapeutics is rich and filled with promising candidates aimed at halting or slowing disease progression. 50 Alignment of all stakeholders on consensus recommendations for medication documentation standards holds promise for improving the outcome of ongoing and future PD clinical investigations. While this issue is especially a challenge within PD research, this is a necessary conversation that will yield fruitful discussions within other disease areas. The ultimate, long-term goal is to achieve a standardized medication documentation system that will be adopted in ongoing and future PD clinical studies and trials. This new system would allow for improved treatment interpretability and lead to more accurate meta-analysis between clinical trials and across participant cohorts.
Footnotes
Acknowledgments
The authors acknowledge the dedicated efforts of Critical Path Institute's data managers Bob Stafford, Diane Corey and Quantitative Medicine scientist Kim Collins. We acknowledge Michael Lawton for his role in contributing to the completion of the manuscript. Charles Venuto and Jamie Adams (University of Rochester) are key leaders that have contributed to the conceptual framework and review of this manuscript. We thank the data expert leaders of PPMI including Leslie Kirsch, Dave Alonso, Alyssa Reimer, and Samantha Hutten of the Michael J. Fox Foundation. The authors appreciate Erin Lowry and her assistance with the references. The authors acknowledge the contributions and leadership of Dr Ira Shoulson and Dr Karl Kieburtz as scientific advisors of CPP who shaped the conceptual framework for this effort.
Funding
The Critical Path for Parkinson's is one of many public private partnerships of Critical Path Institute and is supported financially by industry and nonprofit organizations:
CPP acknowledges all its members and advisors (please see list of collaborators here: ![]() ) for driving success in furthering accelerated treatments for Parkinson's. CPP also gratefully acknowledges the Michael J. Fox Foundation and Parkinson's UK for generously funding. Critical Path Institute is supported by the Food and Drug Administration (FDA) of the Department of Health and Human Services (HHS) and is 54% funded by the FDA/HHS, totaling $19,436,549, and 46% funded by non-government source(s), totaling $16,373,368. The contents are those of the author(s) and do not necessarily represent the official views of, nor an endorsement by, FDA/HHS or the U.S. Government.
) for driving success in furthering accelerated treatments for Parkinson's. CPP also gratefully acknowledges the Michael J. Fox Foundation and Parkinson's UK for generously funding. Critical Path Institute is supported by the Food and Drug Administration (FDA) of the Department of Health and Human Services (HHS) and is 54% funded by the FDA/HHS, totaling $19,436,549, and 46% funded by non-government source(s), totaling $16,373,368. The contents are those of the author(s) and do not necessarily represent the official views of, nor an endorsement by, FDA/HHS or the U.S. Government.
PPMI – a public-private partnership – is funded by the Michael J. Fox Foundation for Parkinson's Research and funding partners, including 4D Pharma, Abbvie, AcureX, Allergan, Amathus Therapeutics, Aligning Science Across Parkinson's, AskBio, Avid Radiopharmaceuticals, BIAL, Biogen, Biohaven, BioLegend, BlueRock Therapeutics, Bristol-Myers Squibb, Calico Labs, Celgene, Cerevel Therapeutics, Coave Therapeutics, DaCapo Brainscience, Denali, Edmond J. Safra Foundation, Eli Lilly, Gain Therapeutics, GE HealthCare, Genentech, GSK, Golub Capital, Handl Therapeutics, Insitro, Janssen Neuroscience, Lundbeck, Merck, Meso Scale Discovery, Mission Therapeutics, Neurocrine Biosciences, Pfizer, Piramal, Prevail Therapeutics, Roche, Sanofi, Servier, Sun Pharma Advanced Research Company, Takeda, Teva, UCB, Vanqua Bio, Verily, Voyager Therapeutics, the Weston Family Foundation and Yumanity Therapeutics.
Declaration of conflicting interests
Tanya Simuni is an Editorial Board Member of this journal but was not involved in the peer- review process of this article nor had access to any information regarding its peer-review.
The remaining authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
Data used in the preparation of this article were obtained on January 2019 from the Parkinson's Progression Markers Initiative (PPMI) database (www.ppmi-info.org/access-data- specimens/download-data), RRID:SCR_006431. For up-to-date information on the study, visit ![]() . This analysis used data openly available from PPMI.
. This analysis used data openly available from PPMI.
