Abstract
Background
In addition to its cardinal motor symptoms, Parkinson’s Disease (PD) is also associated with a variety of neuropsychiatric symptoms, including both cognitive impairment and affective dysfunction, which affect up to 85% of people with PD (PwP).1,2 Mild cognitive impairment (MCI) is common in PD (PD-MCI), affecting 40% of PwP, 3 with 20–33% meeting criteria for PD-MCI at time of PD diagnosis.4,5 Notably, a significant number of PwP also develop Parkinson’s Disease dementia (PD-D), which is characterised by declines in more than two cognitive domains, severe enough to impair daily life. 6 PD-D is estimated to affect up to 78% of PwP within eight years from diagnosis. 7 In line with this, a meta-analysis of 39 studies showed that, over a three-year follow-up period, 25% of PwP with normal cognition progressed to PD-MCI (with 2% progressing to PD-D), while 20% of PD-MCI cases progressed to PD-D (although, of note, 28% also reverted back to normal cognitive function). 8 These conversion rates were even greater when looking at a follow-up period of longer than three years. Similarly, affective dysfunction has also been widely reported in people PwP, with approximately 30–40% of PwP displaying clinically significant depressive symptoms9,10 and up to 40% experiencing elevated anxiety.11,12
Cognitive impairment in PwP is a major determinant of both quality of life (QoL) and ability to carry out daily tasks,13,14 and significantly increases burden for carers of PwP. 15 Likewise, affective dysfunction in PwP is also a major contributor to reduced QoL and is associated with a doubled risk of mortality. 16 Despite their prevalence and profound effects, however, understanding of factors that contribute to the progression of cognition and affective impairment in PD is limited. 17 To address this gap, it is first necessary to characterise the specific subtypes of cognitive and affective impairment that may present in PD. Unlike the relatively well-accepted motor subtypes (most commonly the tremor- and postural instability gait disturbance (PIGD)-dominant subtypes 18 ), however, subtypes of cognitive impairment and affective dysfunction in PD are not as well-established. 19 One attempt to define such cognitive clusters is the dual syndrome hypothesis, which presents an amnestic vs non-amnestic subtyping of either posterior-dependent memory or more anterior-dependent executive function-related impairment.20,21 While this classification has received support in the literature, 22 key questions still remain, as such impairments are difficult to interpret clinically, face low sensitivity due to the use of screening tools, rather than measures sensitive to subtle individual differences, and may lead to underestimation of the extent of cognitive impairment in practice. 23 Attempts to subtype based on neuropsychiatric symptoms have also failed to find widespread support, due to similar methodological limitations. 24
The incorporation of biomarkers to help inform prediction of cognitive and affective trajectory in PD may offer a potential path forward. Of note, multiple pathophysiological markers have been proposed to play a role in driving the cognitive and affective impairments seen in PD, with particular focus placed on the role of pathological protein aggregates. In line with this, prior studies have suggested that decreased levels of amyloid beta 25 and alpha-synuclein 26 and increased levels of phosphorylated-tau 27 in the cerebrospinal fluid (CSF) are associated with impairments in attention and memory function, as well as increased depression, in PwP. Neuroimaging-derived measures of dopamine integrity may also have utility. For example, T2-weighted MRI scans of the substantia nigra (SN), a key DA-producing nucleus of the midbrain, along with Dopamine Transporter (DaT) imaging, a form of single-photon emission computed tomography (SPECT), 28 have both been shown to correlate with memory impairment, deficits in attention and clinically relevant rates of anxiety and depression in PwP.29-31 Thus, incorporating such multimodal markers may improve accuracy of prediction of long-term cognitive and affective outcomes in PwP.
Machine-learning-based studies have recently begun to investigate this, demonstrating that overall prediction accuracy of longitudinal cognitive decline increased by as much as 44% following inclusion of multimodal variables, such as CSF biofluid markers (e.g. alpha-synuclein and neurofilament light chain) and prodromal symptom presentation.32,33 These prior studies, however, did not attempt to cluster individuals based on cognitive performance. Additionally, to the best of our knowledge, it appears that no studies conducted to date have assessed longitudinal affective change in PD using such a multimodal approach. To address these gaps, our study categorised de novo PD patients based on their cognitive ability and affective function at a 5-year follow-up using a fuzzy C-means data-driven clustering algorithm. We then explored whether a combination of multimodal markers at time of diagnosis better predicted cluster membership compared to baseline cognition and affective measures alone.
Methodology
Participants
Extracted PPMI Variables.

Extracted variables, clinical assessments, and biomarkers from the PPMI database, including some supporting explanations and rationale for their use.
Principal Component Analysis to Derive Overall Cognition and Affective Scores
To create overall cognition and overall affective scores at both baseline (time of diagnosis) and at 5-year follow-up, four separate principal component analyses (PCA) were conducted. The baseline cognition score was derived from a PCA on the five cognition assessment scores at baseline, represented by the individual test loadings on the first unrotated component aggregated into a single score. This method of combining cognitive measures has been shown to be an adequate representation of general cognitive function. 54 As the GDS and STAI have been shown to correlate highly, 55 these affective measures were also combined. The baseline affective score was similarly derived from a PCA on the two affective measures at baseline.
Solutions of the four PCAs.
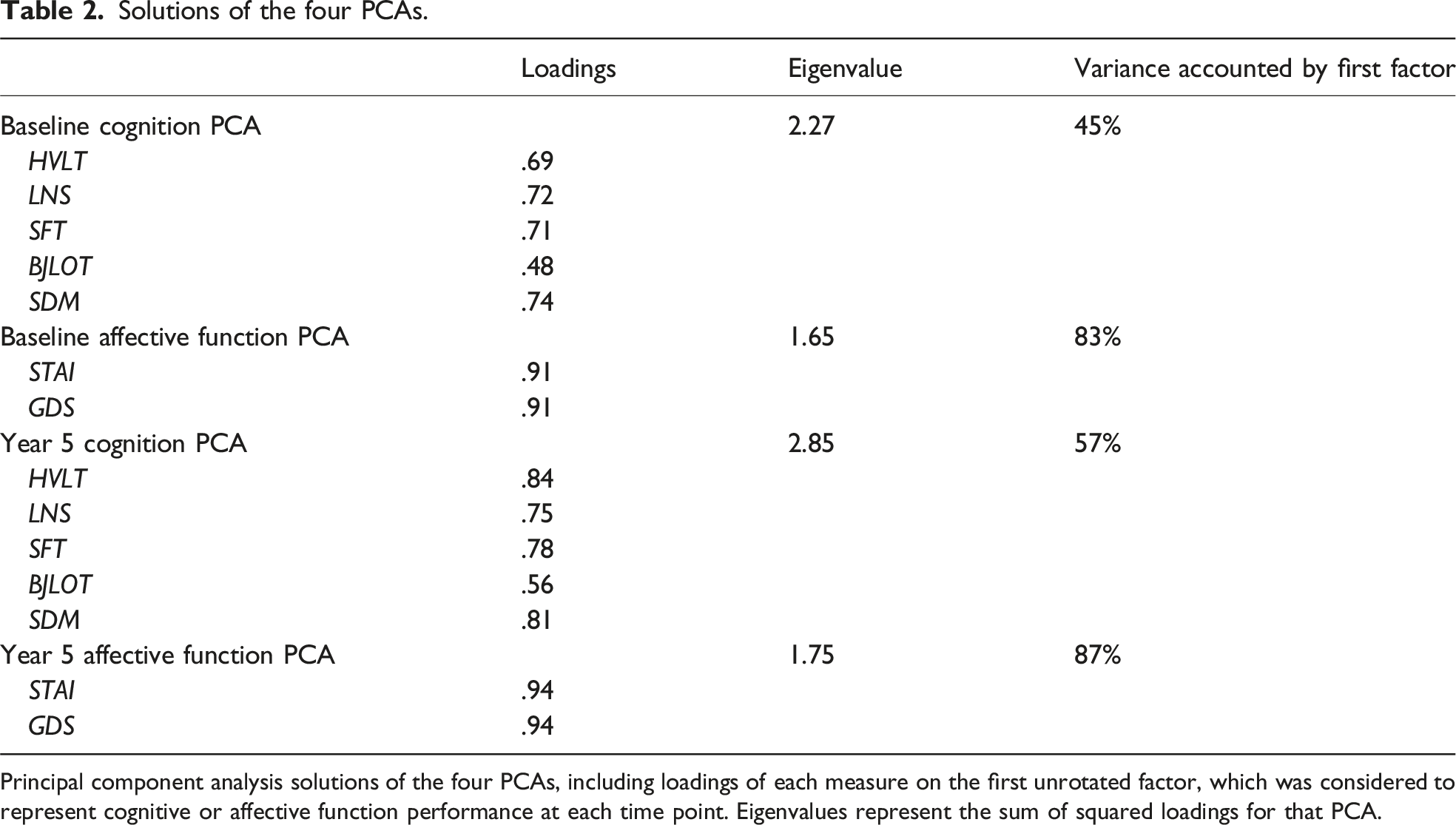
Principal component analysis solutions of the four PCAs, including loadings of each measure on the first unrotated factor, which was considered to represent cognitive or affective function performance at each time point. Eigenvalues represent the sum of squared loadings for that PCA.
Fuzzy C-Means Clustering
Fuzzy C-means clustering was performed on the Year 5 cognitive and affective function scores using the R package fclust.
56
The optimal number of clusters for the data was determined using the R package NbClust,
57
which determines the optimal number of clusters and highlights the best solution by majority vote. Fuzzy C-means clustering best classified the PD patients into two clusters. In addition to the binary cluster membership, fuzzy C-means clustering also calculates the probability that each participant belongs to each cluster, which could then be analysed as a continuous variable (Figure 1). We chose this approach for our main regression and machine learning analyses, given that the two clusters were not well differentiated from one another. Clustering plot of PD patients based on Year 5 Cognition and Affective scores, with their probability of membership for the more impaired group (cluster 2) indicated by the shade of the dots. Note: For a plot depicting the binary cluster membership for all participants, see Figure S1.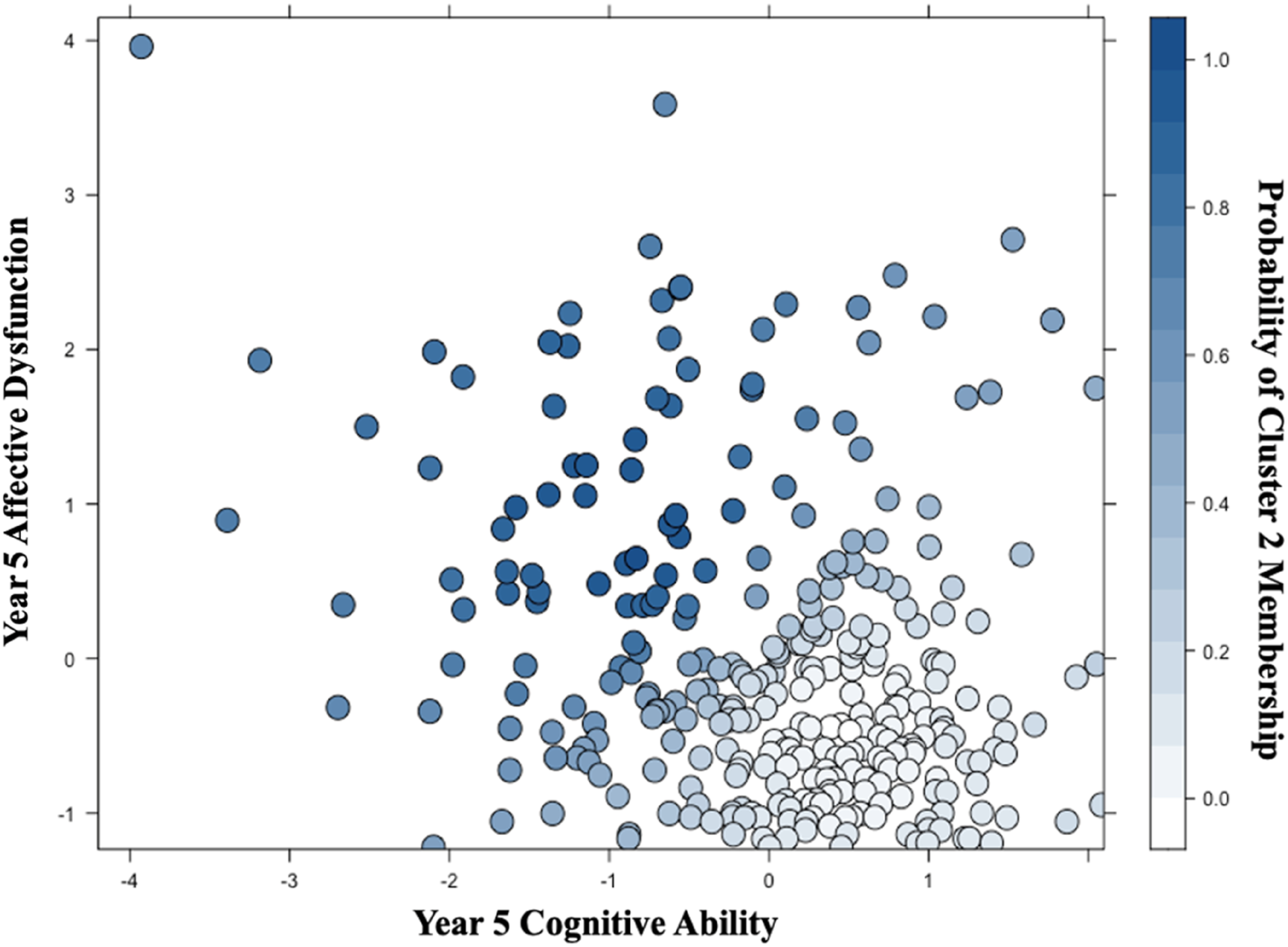
SN Proxy Marker
A subset of PwP (n = 136; median age 61, IQR 53-68; 64% males) had baseline axial T2-weighted (T2w) brain MRI scans acquired on a Siemens 3T trim trio scanner using a FLAIR sequence with the following parameters: 12-channel matrix-head coil, TR/TE of 3000.0/11.0 ms with a flip angle of 150.0°, phase encoding direction left to right with an acquired matrix size of 228 × 256 × 48 and voxel dimensions 0.93 × 0.93 × 3 mm3.
The SN was evaluated using a 2D proxy marker, the hypointense region comprised of crural fibres and anterosuperior portions of the SN. 58 Note that despite the three-dimensional nature of the scans, this proxy was two-dimensional because this region was typically clearly visible in only one axial slice. We have adopted the term “SN-related T2 hypointensity” to describe this marker. The RN was also selected for segmentation, as A10 dopaminergic projections from the RN to the cortical regions are spared in PD59,60 and, hence, this may act as a control area to account for individual differences in brain size.
Two individual raters performed the manual delineation. Using the imaging visualization program FSLeyes (v1.3.0), available through the FSL package,
61
overlay masks were created over the top of the T2w images and edited through manual tracing and filling of the regions of interest (Figure 2). In cases where the chosen axial slices did not match between the raters, they mutually agreed on the most appropriate slice and individually re-masked the regions. In cases where differences between masks exceeded 20 voxels, masks were re-traced independently and compared again. The area (voxel count) of these filled masks was recorded and averaged. An SN:RN area ratio was then calculated for each participant, to account for the global effect of individual head size. Larger ratios corresponded to a larger SN-related T2 hypointensity and, consequently, more advanced disease. (A) T2w MRI of the brainstem (B) The same T2w MRI with the regions delineated: red nucleus (red) and the hypointense region associated with the substantia nigra (green).
Statistical Analysis
All data were analysed via R (version 1.4.1717). 62 A Shapiro-Wilk test was conducted to determine if the variable distributions deviated significantly from normality. For non-normally distributed variables, averages are reported as median (interquartile range) and summarised as counts (percentage).
As cluster membership was determined via cognitive ability and affective function scores at 5-year follow-up, retrospective assessment was conducted to determine whether differences between clusters were present at baseline using Mann-Whitney U tests for non-normally distributed variables, or t-tests for those normally distributed. Bonferroni corrections for multiple comparisons were implemented for all comparisons of variables between clusters.
Multiple linear regression was used to statistically assess the utility of baseline variables of interest to predict the probability of belonging to the more impaired cluster 2. Using R’s stats package, 62 two regression models were generated and compared. The first model comprised only baseline cognitive and affective function scores (the same measures used as inputs for the clustering algorithm, though clustering was performed on Year 5 data). Thus, the first model tested the extent to which baseline cognition and affective function could predict Year 5 cognition and affective function. Conversely, the second model contained these baseline cognition and affective function scores, as well as demographic data, motor scores, prodromal symptom profiles and biomarkers measured at baseline. An ANOVA was run to compare models to assess whether the inclusion of these additional variables significantly improved predictive accuracy. Both models only included participants with complete data (n = 113). A random forest relative importance plot of each predictor was also calculated using R’s caret package 63 for both models, which ranks the predictors in terms of their relative contribution to the model’s explained variance from most to least important (see Figure 4). All regression models used mean absolute error (MAE) and R2 as statistical parameters to gauge accuracy.
In addition to the main multiple linear regression models, nested individual linear regression models based on grouped variables of interest as their only predictors (i.e. demographics, behavioural measures, neuroimaging, prodromal symptom profile and biofluid-based biomarkers) were also run. This was done to further investigate which variables, or groups of variables, showed greater utility in predicting membership. Each individual model was compared against the full multimodal model, as shown in Table S4.
Though 309 participants were included in the final cluster analysis, we initially extracted data from 422 participants from PPMI, with 113 participants lost to follow-up at 5 years. To explore if this attrition impacted our data, Mann-Whitney U tests were run comparing depression scores (from the GDS), state and trait levels of anxiety scores (from the STAI) and cognitive impairment (from the Montreal Cognitive Assessment (MoCA)) between those who returned for 5-year follow-up and those who did not, to assess whether a significant portion of more impaired PwP were lost to attrition (Table 4).
Supervised Machine Learning
The Python library Scikit-learn was utilised for the machine learning analysis (https://www.scikit-learn.org). The target outcome was the probability of belonging to the more impaired cluster 2, and the predictive features included all baseline variables that has also been included in the regression models. The dataset was divided into a training set and a hold-out test set, comprising 80% and 20% of the cases, respectively. Three machine learning models were compared: Support Vector Regressor, K Nearest Regressor, and Random Forest Regressor. We optimized the hyperparameters for each model through grid search, employing a 5-fold cross-validation. Support Vector Regressor was determined to be the most effective model, based on the validation performance (obtained from the 5-fold cross validation), and we subsequently used it for further machine learning tasks. For final model evaluation, we employed MAE and R2 as statistical parameters to gauge accuracy. Before training any of the machine learning methods, we calculated a baseline MAE score on the 80% training set to measure the average absolute differences (MAE) between a constant prediction of 0.5 and true probability values in that set. Following this, we trained the best model, with the optimized parameters, and then calculated the MAE on the 20% test subset.
Results
Comparisons of Variables Between Fuzzy Clusters
Descriptive statistics and Wilcoxon test comparisons of each baseline variable between the two fuzzy clusters.
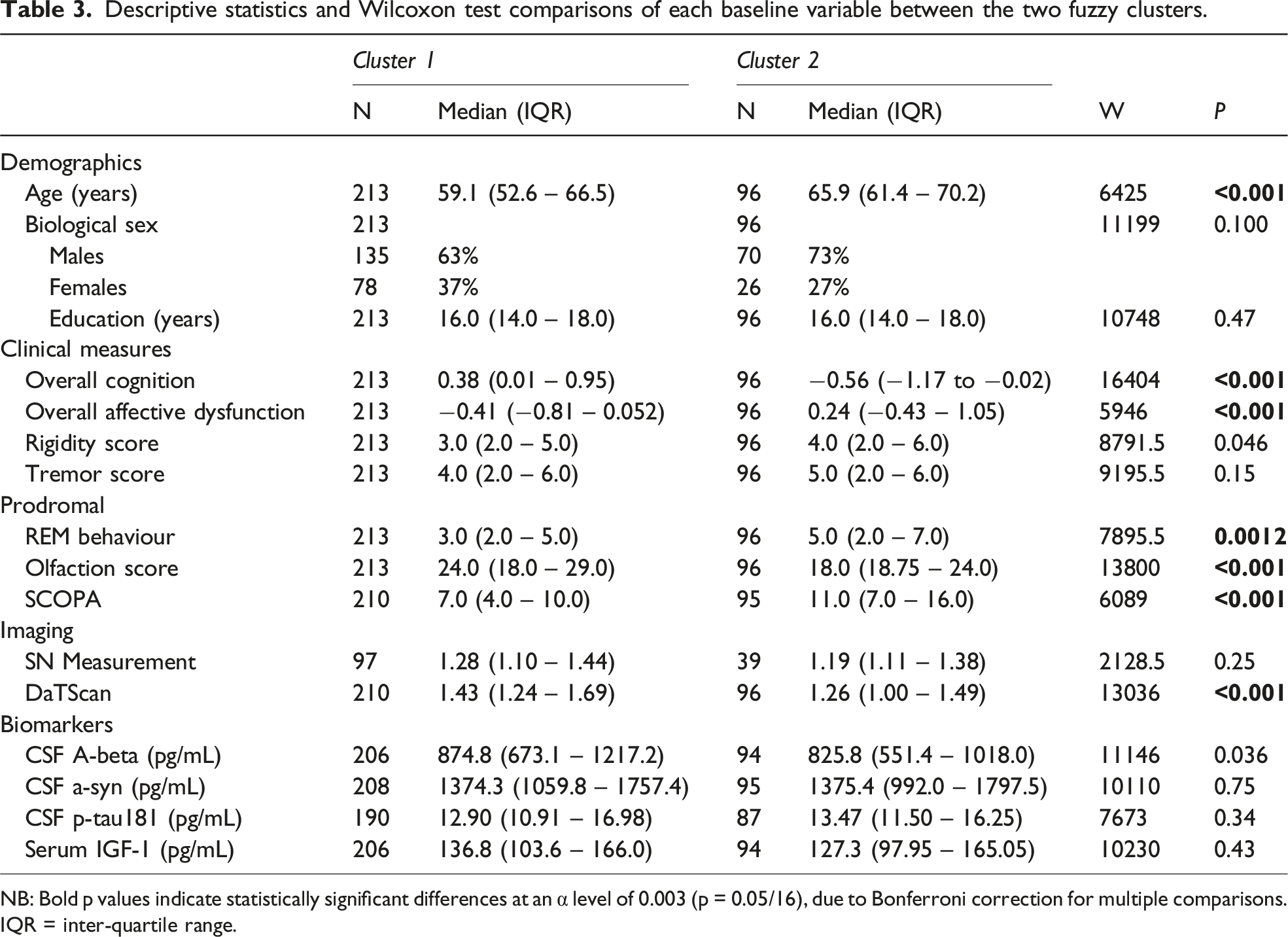
NB: Bold p values indicate statistically significant differences at an α level of 0.003 (p = 0.05/16), due to Bonferroni correction for multiple comparisons. IQR = inter-quartile range.
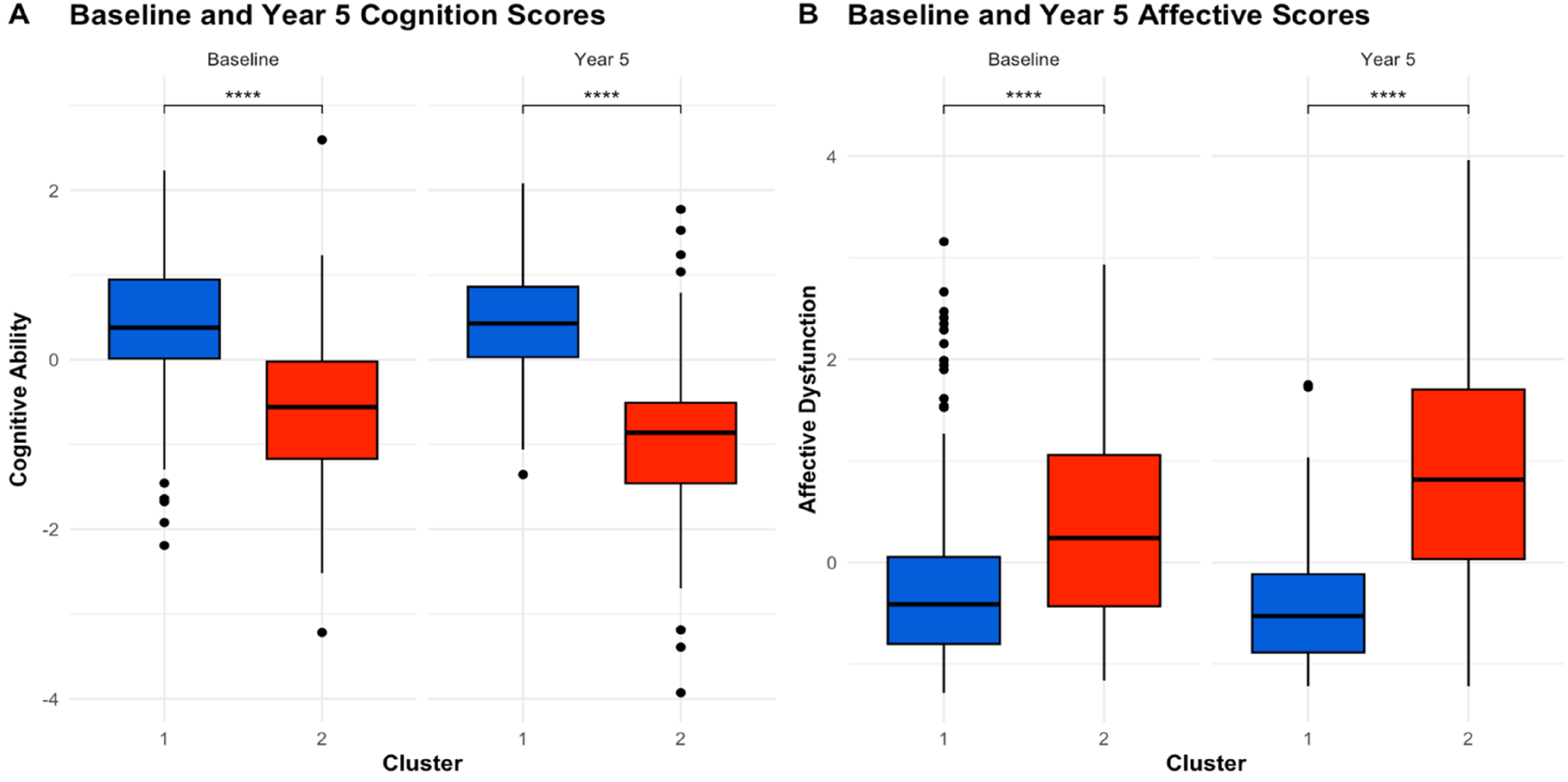
Post-hoc comparison of baseline and Year 5 cognition and affective scores at each time point for each cluster. (A) Cluster 2 showed worse cognitive performance at baseline (W = 16404, P < .001) and Year 5 (W = 18150, P < .001). (B) Cluster 2 showed greater affective impairments at baseline (W = 5946, P < .001) and Year 5 (W = 2664, P < .001).
Cognitive and Affective Function Profiles of Each Cluster
Individuals in cluster 1 had significantly better performance on all 5 cognitive tests than members of cluster 2 at baseline, suggesting that these individuals had more preserved executive functioning, working memory and visuospatial abilities at time of PD diagnosis and these trends continued at 5-year follow-up (Table S3). This was further supported by a significantly greater MoCA score, a measure of global cognitive function that was not included in the cognition PCA and hence had not been used for the purpose of clustering, at baseline (W = 12819, P < .001). Similarly, individuals in cluster 1 had significantly lower rates of both depression and anxiety than members of cluster 2 both at baseline and at 5-year follow-up (Table S3).
Regression Analyses
Demographic comparisons and number of people who met clinically relevant cut-off scores in both the retained group included for analysis, and those that had only baseline assessments and did not return for the Year 5 follow-up.
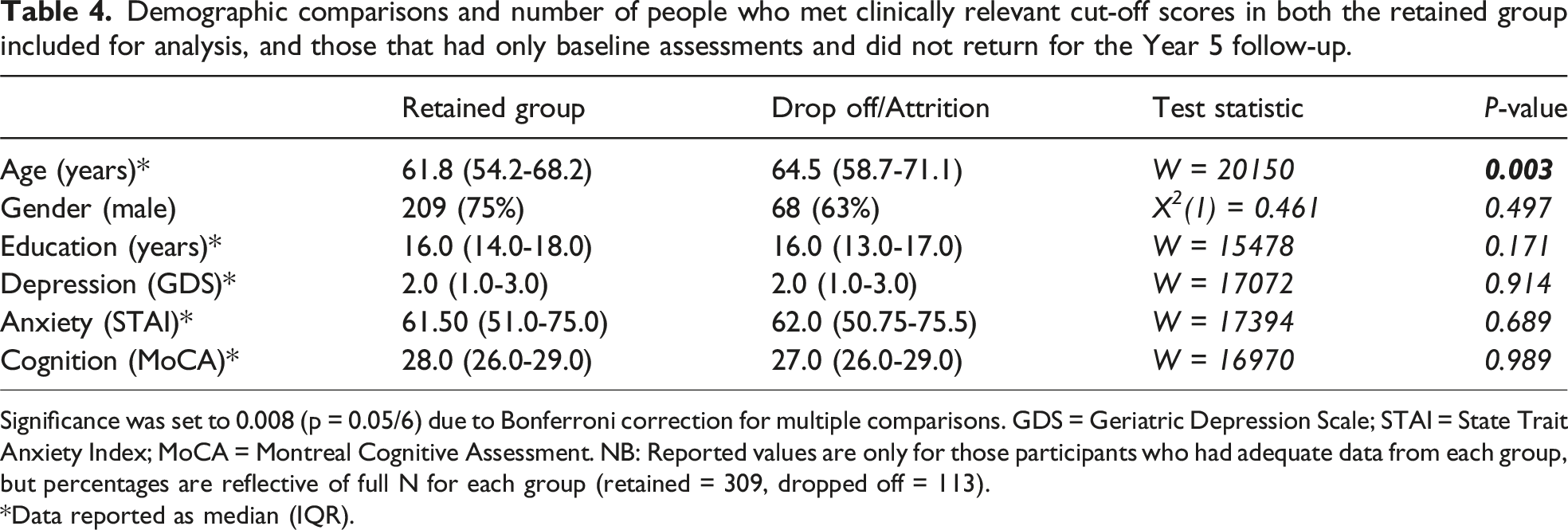
Significance was set to 0.008 (p = 0.05/6) due to Bonferroni correction for multiple comparisons. GDS = Geriatric Depression Scale; STAI = State Trait Anxiety Index; MoCA = Montreal Cognitive Assessment. NB: Reported values are only for those participants who had adequate data from each group, but percentages are reflective of full N for each group (retained = 309, dropped off = 113). *Data reported as median (IQR).
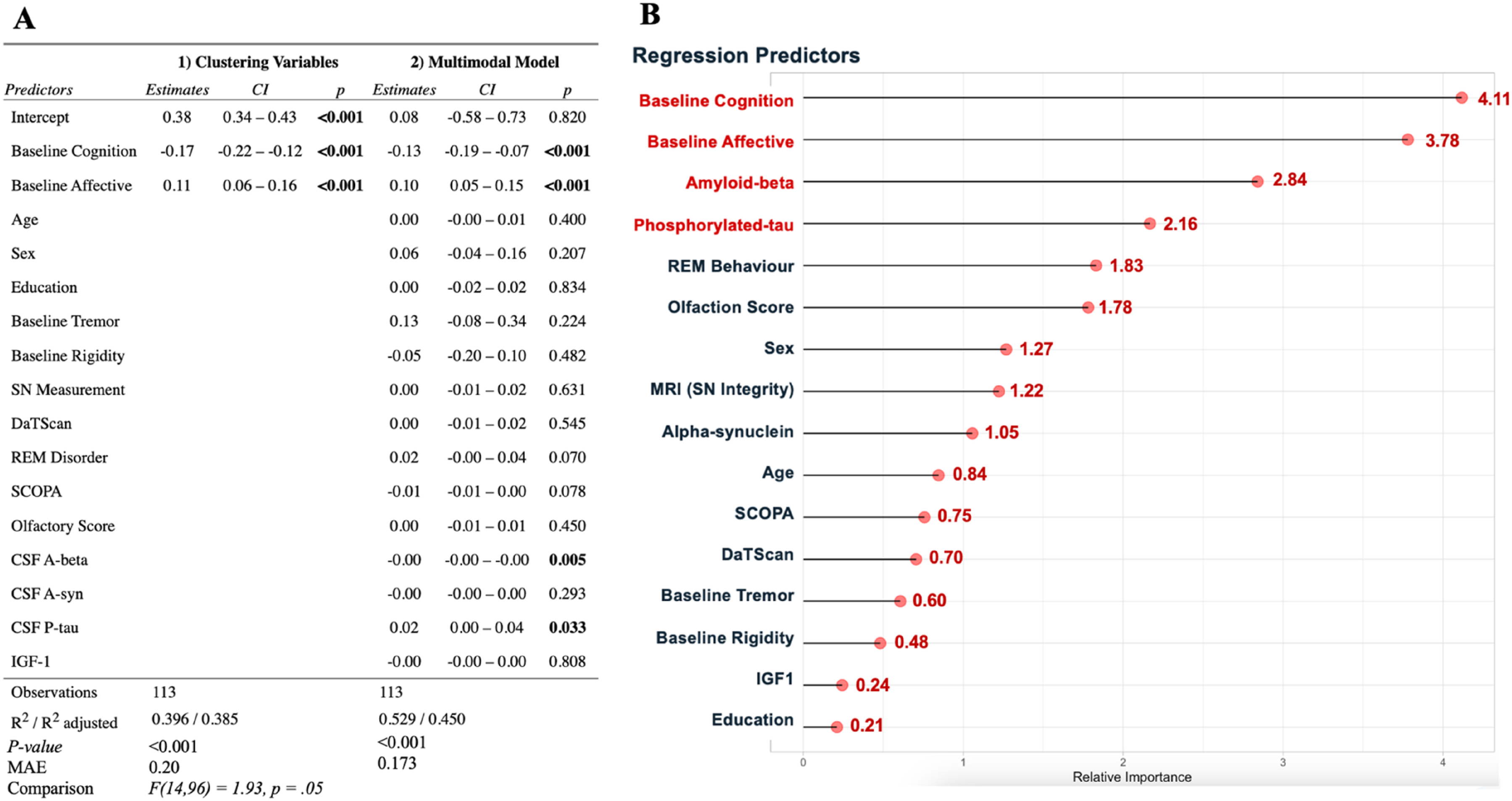
(A) Output of the two linear models, both with the probability of belonging to cluster 2 as the outcome variable. The first model solely included, as predictors, the baseline values of the variables used to derive the clustering at Year 5 (i.e. baseline cognition and affective function), while the second included the additional multimodal baseline variables. Significant predictors are denoted at P < .05 and are bolded, as R compute internal correction for multiple comparisons. MAE = Mean absolute error; CI = Confidence interval, Comparison = Statistical summary of an ANOVA comparing the two models. Estimates and CIs are reported to two decimal places (B) Plot of the random forest relative importance of each of the predictors in the linear regression in order of their influence of explaining variance between fuzzy C-means clusters. Red variables indicate significant predictor in multimodal regression.
Supervised Machine Learning
In the machine learning analysis, the simple constant baseline prediction showed an MAE score of 24% in predicting the probability of fuzzy class 2 membership (defined from Year 5 follow-up data). The machine learning model, trained according to the optimal parameters chosen via 5-fold cross validation, resulted in an MAE that was reduced to 19%. Hence, this model increased accuracy and reduced errors through supervised training by 5%, with a total proportion of the variance explained of 38% (R 2 of 0.38).
Attrition
Table 4 summarises the comparisons of baseline demographic variables between those returning to PPMI for follow-up assessments and those who did not. While 422 were initially enrolled, only those with both baseline and Year 5 follow-up data for cognition and affective function (n = 309) were included in this study. The only significant difference between the retained group and those assessed at baseline was age, with those lost to drop off being older at time of diagnosis (W = 20150, P = .003). These groups did not significantly differ in either their depression scores (W = 17072, P = .914), anxiety scores (W = 17394, P = .689) or degree of cognitive impairment (W = 16970, P = .989), suggesting that attrition was unlikely to have greatly influenced our findings.
Discussion
To date, the prediction of cognitive and affective function outcomes in PwP using a multimodal approach remains largely unexplored. Thus, this study aimed to use data-driven clustering techniques to firstly categorise individuals based on performance in these domains at five-year follow-up and then assess baseline factors which are predictive of this cluster membership. At baseline, those classified within the second cluster by the fuzzy clustering algorithm were older and demonstrated more severe cognitive impairment and a higher presence of affective dysfunction. They also had a greater presentation of prodromal symptoms and lower striatal DaT binding, perhaps indicating more established underlying pathology at time of diagnosis. This is consistent with previous reports, with a recent systematic review of PD subtypes derived from global functional outcome studies (including cognition and mood) indicating that PwP generally fall into either younger, less impaired groups or older, more impaired groups. 64
Utility of a Multimodal Approach for Predicting Progression of Cognitive and Affective Function Changes in PD
The most significant finding of the current work was that it highlighted the benefit of incorporating a multimodal prognostic panel of baseline predictors when considering the prediction of disease progression, rather than relying on clinical assessment of cognitive or affective function alone. Membership in the more impaired cluster (cluster 2) was predicted not only by lower levels of cognitive function and greater affective dysfunction at baseline, but also by baseline CSF levels of several biomarkers, including lower levels of amyloid beta and higher levels of phosphorylated-tau181. The total explained variance was ∼7% greater in the multimodal modal (45%) than in the model containing only the clustering variables at baseline (38.5%). Further, the supervised machine learning analysis also supported these findings- with sufficient training pipelines, the model itself could predict the probability of cluster 2 membership at 5-year follow-up with a mean absolute error of 19%. These findings are in line with those of other studies focused on the prediction of cognitive trajectory in PD, which also saw improvements when incorporating additional multimodal variables, such as the Modified Schwab & England Activities of Daily Living Score, above clinical variables alone.32,33
Of note, however, these previous studies have noted larger improvements (up to 40%) when incorporating these additional variables, compared to the more modest improvement found in the current work. This may indicate that the inclusion of additional variables, not considered in our models, may have utility for predicting the progression of cognitive and affective change in PD. In support of this, the Unified Parkinson’s Disease Ranking Scale Part III (UPDRS-III) 33 and the Epworth Sleepiness Scale (ESS) 32 have both been shown to be significant predictors of cognitive change up to 10 years following PD diagnosis. In contrast, our study assessed cognitive and affective function change only up to a 5-year follow-up in de novo PD. Given that more severe impairments in cognitive and affective function, including the presentation of PD-D, typically occur in the later stages of the disease (i.e. 10-12 years from diagnosis), 16 it may be that the time assessed in the current study was not sufficient to assess the long-term trajectory of cognitive and affective function change in PD. Furthermore, complete data were only available on a relatively small subset of the total cohort (i.e. 113 out of 422 individuals initially enrolled), which may have further impacted our ability to detect predictors of longitudinal change within the domains of interest.
Within the linear regression model, cognitive ability and affective dysfunction at baseline were, unsurprisingly, significant predictors of cluster membership, since performance on these same measures at 5-year follow-up provided the basis for the fuzzy C-means clustering of the PD patients. That is, it is not surprising that PwP with more pronounced impairment on these measures at time of diagnosis would continue to show impairment on these measures at 5-year follow-up (and, hence, be members of the second, more impaired cluster). In line with this, Santos-García et al. have previously shown that cognitive ability at time of PD diagnosis can predict more severe longitudinal non-motor declines using aggregated scores of cognitive, affective and autonomic functions. 65 More recently, Pourzinal and colleagues (2024) used the Montreal Cognitive Assessment as the outcome measure in latent class mixed models and identified four cognitive subtypes in two large longitudinal cohorts (PPMI and ICICLE-PD). Notably, baseline performance on the Judgment of Line Orientation and category fluency tasks were predictors of steady cognitive decline in the PPMI and ICICLE-PD cohorts, respectively. 66 Similarly, affective dysfunction in early PD has been shown to predict severe affective impairments over disease duration, including PD-associated depression67,68 and overall quality of life. 69
In addition, however, both reduced CSF levels of amyloid-beta and increased CSF levels of phosphorylated-tau181 significantly predicted membership in the cluster with more pronounced cognitive/affective dysfunction (i.e. cluster 2) at 5-year follow-up. Importantly, both of these are well-documented biomarkers of both Alzheimer’s Disease (AD) and PD. AD has been associated with an overaccumulation of both amyloid-beta and phosphorylated-tau, 70 and similar pathology can be seen in PD, with increased CSF concentration of amyloid-beta aggregates and phosphorylated-tau consistently reported in de novo PD.71,72 Previously, in a population-based, prospective study of 210 participants with new-onset idiopathic parkinsonism, Bäckstörm et al found reduced CSF levels of Aβ42 to be a significant predictor of PD-D development within 10 years. 33 Similarly, Siderowf et al. showed that low CSF levels of Aβ42 predict more rapid cognitive decline at 2-year follow-up in a prospective cohort study of 45 individuals with well-established PD (mean years since onset was 11). 73 Within the PPMI cohort, Andersson et al. (2021) used a multivariate latent class linear mixed model to explore longitudinal cognitive performance data in 349 de novo PwP, and showed that CSF amyloid-beta42 levels were higher in a small subgroup (10%) of individuals who showed rapid decline in all cognitive performance measures over a 5-year period. 74
In contrast to the current work, however, in Siderowf et al.'s study, CSF levels of total tau and p-tau181 were included in their models, but failed to predict cognitive trajectory. 73 Aggregations of phosphorylated-tau increase in response to the C-terminal synaptic pathology that occurs throughout PD progression, 75 and have been demonstrated to significantly predict altered visuospatial and verbal learning functioning in PwP. 76 To date, however, the link between p-tau and affective dysfunction in PD remains largely unexplored. Interestingly, p-tau181 concentrations in the CSF have been shown to not significantly differ between PD and age-matched controls 3.5 years after diagnosis, 77 with Baek et al. reporting that p-tau181 concentrations steadily increased at a rate of 0.06/pg/mL/year in early PD and elevate exponentially 15 years following diagnosis. 78 Thus, it may be that levels of phosphorylated-tau were not yet significantly elevated in the prior study by Siderowf and colleagues (2010), which investigated a relatively narrow window post-diagnosis. In line with this, in the current work, phosphorylated-tau181 levels were similar in the two groups, not only at baseline but even at 5-year follow-up. Additionally, when only the baseline biomarkers were included in the regression model (Supplemental Table 4), CSF levels of p-tau181 were no longer a significant predictor of cluster membership, indicating that further work is needed, particularly at longer follow-up timepoints, to fully elucidate how p-tau181 levels may be related to progression of cognitive and affective impairment in PD. Nevertheless, the results of the current work highlight the potential prognostic utility of both biomarkers for improving prediction of cognitive and affective function trajectory in PD, currently a major area of concern for PwP. 79
Several Variables Previously Established as Predictors of Cognitive/Affective Function Trajectory in PD Were Not Significant Predictors in Our Models
Interestingly, several other biomarkers previously linked with cognitive decline in PD were not significant predictors of cluster membership at 5-year follow-up in this study. This included CSF levels of alpha-synuclein and serum concentrations of IGF1, which both have previously been shown to be associated with cognitive decline in PD across multiple previous studies80-82 (although their link with progression of affective symptoms in PD is less clear). However, this previous evidence for their association with cognitive decline in PD used general screening tools to assess cognition, including the Mini-Mental State Examination (MMSE) 81 or only tracked domain-specific cognitive functioning for up to two years beyond the CSF sampling. 82 In contrast, we analysed scores on a battery of cognitive tests during a five-year follow-up period, which may explain the discrepancy in results observed.
Similarly, we were surprised that neither our baseline proxy measure of SN hypointensity nor baseline striatal binding, as measured by DaTScan, were significant predictors of cluster membership, given the critical role of DA in the presentation of cognitive and affective impairments in PD. 83 This was particularly noteworthy given that members of cluster 2 did have significantly lower levels of striatal DaT binding at baseline (although data for Year 5 follow-up were not available for this measure). A previous study utilising the PPMI cohort showed associations between striatal DaT binding and the development of cognitive impairment at two-year follow-up, although this was localised to the caudate. 84 In contrast, global measures of striatal DaT, as were used in this current work, may mask subtle regional differences. Similarly, previous work using the same proxy marker for SN hypointensity that was used in the current work has shown it to be significantly positively correlated with cognitive impairments in two independent PD cohorts with mean disease duration of 5.15 years (r = 0.23) 30 ; however, segmenting in that study was done using automated thresholding techniques, rather than manual masking, which may account for the differences seen in the current work. Further, that study did not explore the relationship of this proxy SN hypointensity marker to affective dysfunction, making it impossible to comment on its utility for predicting disease trajectory within this domain. It is also worth considering whether other subcortical nuclei that are DA-rich, such as the ventral tegmental area (VTA) of the mesocortical/mesolimbic pathways, may be more relevant areas to assess when investigating cognitive and affective function trajectories in PD. In line with this, a recent study analysing the PPMI dataset found that the right VTA, as assessed on structural MRI scans, was a significant predictor of PD-MCI at Year 5 follow-up, 85 which is also consistent with findings reported by Aarsland et al. on the prognostic role of the VTA in both PD-MCI and PD-associated depression. 86
Of note, prodromal PD symptom presentation also did not predict cluster 2 membership at 5-year follow-up, although the clusters significantly differed in all three prodromal variables (i.e. REM behaviour disorder; olfactory dysfunction and autonomic function) at baseline. This was surprising, as these prodromal symptoms, particularly olfactory dysfunction, have previously been reported to be significant predictors of cognitive impairment in the early stages of PD.84,87 Anang et al further showed that REM behaviour disorder is an independent predictor of PD-D development, 88 with this finding widely supported in the literature by multiple other studies.88-90 Additionally, prodromal PD markers, particularly sleep disturbances, have also been correlated with depression presentation in PD, with analysis of the PPMI dataset showing that RBDSQ scores significantly correlated with depression scores as measured by the GDS. 91 These discrepancies may be due to the fact that other variables in the model had much greater relative importance for predicting cluster membership. In support of this, in a post-hoc linear regression that included the three prodromal variables as the sole predictors in the model, all three were significant in predicting cluster 2 membership (Supplemental Table 4), suggesting that they may have at least some prognostic utility in predicting cognitive and affective symptom trajectory in PD, although perhaps not to the same extent as the other predictors explored in the current work (i.e., baseline cognition and affective function; CSF levels of Aβ42 and p-tau181).
Another unexpected finding of the current work was that neither baseline muscle rigidity nor tremor served as a significant predictor of cluster membership at Year 5 follow-up, even when considered in isolation as the sole predictors in a linear regression model (Supplemental Table 4). This is in contrast with previous literature, which supports that the severity of motor symptoms in PwP are a risk factor for both PD-D 92 and depression up to 10 years following diagnosis. 93 Further, the risk of these impairments has been shown to differ based on motor subtype, with PwP with a more rigid-dominant presentation being more susceptible to both cognitive impairment94,95 and depression 96 compared to those with a more tremor-dominant presentation. The discrepancies observed in the current work may be due, at least in part, to the fact that there were no baseline differences in these motor measures between clusters, which may reflect the homogenous nature of the PPMI cohort as a whole. Further, motor presentation in PwP is not fixed, but rather can fluctuate with disease progression and with medication treatment. 97 This has previously been shown to be a factor in the PPMI cohort, with Luo et al. showing that, of the 162 PwP analysed over a 5-year period in the PPMI cohort, 40 participants shifted from a tremor-dominant presentation to a PIGD-dominant presentation, and vice versa. 98 Thus, this instability, at least in the first few years following diagnosis, may limit the prognostic utility of these variables for predicting cognitive and affective dysfunction trajectory.
Finally, in our analyses, we also did not see the expected relationships with several demographic variables that have been well-established for their association as predictors of cognitive and affective symptom progression in PD, including age, sex and education. Age has frequently been reported to be an independent predictor of both PD-MCI and PD-D. 99 In a recent meta-analysis of 42 PD studies, increased age consistently significantly predicted the development of cognitive impairment, with subgroup analyses showing its significant role in predicting both PD-MCI and PD-D as independent outcomes. 100 A similar relationship has been noted between the presence of affective dysfunction and age, with findings from a study of 127 PwP showing significantly greater prevalence of anxiety disorders in older PwP. 101 Notably, age did significantly differ between our clusters at baseline (and consequently at Year 5 follow-up) and was a significant predictor when demographic variables were considered in isolation (Supplemental Table 4), suggesting that further exploration of the link between age and prediction of cognitive/affective trajectory in PD is warranted. Interestingly, similar to our findings, a prior meta-analysis highlighted that sex and education showed no significant utility in the prediction of cognitive impairment in PwP, 100 although these findings contrast with those reported elsewhere in the literature. For example, males with PD have been shown to be more susceptible to more severe cognitive declines and increased anxiety-like symptoms, as demonstrated in a study of 232 (149M/83F) de novo PwP. 102 Similarly, education has been shown to be an independent predictor of cognitive performance in PwP, with baseline years of education demonstrated to predict baseline MoCA performance. 87 Thus, further work is needed to resolve these discrepancies in the literature.
Potential Limitations of the Current Findings
In interpreting these results, it is important to consider whether our findings could have been impacted by attrition due to loss to follow-up. Five-year follow-up data was only available for 309 of the 422 individuals originally enrolled in PPMI (representing a dropout of 113 individuals or ∼27%). To understand the potential effect of this on our cluster analysis, we carried out a baseline comparison of those who continued to follow-up vs those who were lost to Year 5 follow-up. The only significant difference noted between the two groups was age, with the median age of those lost to follow-up being significantly higher than those who remained. As increased age negatively correlates with the capability of PwP to complete activities of daily living and engage with the community, 103 it may be that those who did not return for follow-up were affected by age-related challenges that impacted their ability to complete the study requirements. This may have impacted our ability to explore the prognostic role of age for predicting cognitive and affective function trajectory in PD. Importantly, the comparisons of baseline cognitive and affective function between those included for analysis and those lost to follow-up were not significantly different, suggesting that this did not systematically bias our assessment of the presentation and progression of these symptoms.
Nevertheless, we acknowledge that there are limitations in this study, arising, in part, from the imaging modalities available in the PPMI database. Within the PPMI database, structural T2-weighted MRI scan were the best option available for delineating the SN. While T2-weighted MRI scans provide higher image contrast compared to other MRI modalities when viewing deep subcortical structures, 104 several drawbacks must also be acknowledged, including that T2-weighted MRI is highly susceptible to image quality interference due to physiological noise (e.g. circulation and respiration). 105 Other imaging modalities have been optimised for the viewing of the SN, with SN segmentation utilising T1-Neuromelanin MRI scans shown to be a useful marker for dopaminergic neurons in the SN. 106 Future work using these more sensitive measures may allow for the true predictive utility of this measure to be more comprehensively explored.
The use of multiple linear regression may also represent a methodological limitation in our work. Logistic regression may have been more appropriate, given the nature of our data-driven clustering, which grouped the PwP into two clusters and created a categorical outcome. However, as our clusters were not well differentiated, we deemed the continuous probability of each PwP belonging to cluster 2 (the more impaired cluster) more important to explore. Additionally, we aimed to maximise the predictability by including as many predictor variables as deemed necessary in our multimodal panel, resulting in an increased risk of attrition, as only participants with full data could be included in the final analyses. From the initial sample of 309 PwP, our models used only a sample of 113 due to missing data, with biomarkers, specifically within the CSF, being the largest contributors to this attrition and reduction of statistical power.
Conclusions
In this study, we highlighted that, in addition to baseline cognitive and affective function, reduced amyloid-beta and elevated phosphorylated-tau181 in the CSF at baseline significantly predicted membership in the more impaired cluster at five-year follow-up. Interestingly, DA markers, including our proxy marker of SN hypointensity and striatal DaT binding, did not play a significant role in this prediction, despite the known modulation of cognitive and affective functioning by DA. Similarly, other markers, such as prodromal symptom presentation, motor subtype and key demographic variables, were also not significant predictors of cluster membership. Nevertheless, our models accounted for a large amount (45%) of variance seen in the model.
Taken together, this study highlights the utility of moving beyond clinical symptom presentation alone and instead incorporating assessment of a multimodal panel of biomarkers in the clinical management of PD. This might allow for improved prediction of risk of development and progression of both cognitive and affective impairment in PD, currently an unmet need in PD, where disease presentation can differ significantly between individuals. Such enhanced understanding of disease trajectory has the potential to significantly impact clinical management of PD, leading to enhanced monitoring, such as earlier referral for neuropsychological assessment, and more personalised management strategies.
Supplemental Material
Supplemental Material - Predicting Cognition and Affective Changes in Newly Diagnosed Parkinson’s Disease Through Longitudinal Data-Driven Clustering
Supplemental Material for Predicting Cognition and Affective Changes in Newly Diagnosed Parkinson’s Disease Through Longitudinal Data-Driven Clustering by Benjamin Ellul, Angus McNamara, Stephan Laurenz, Irina Baetu, Mark Jenkinson, Lyndsey E. Collins-Praino in Journal of Geriatric Psychiatry and Neurology
Footnotes
Acknowledgements
Data used in the preparation of this article were obtained on May 2021 from the Parkinson’s Progression Markers Initiative (PPMI) database (https://www.ppmi-info.org/access-dataspecimens/download-data), RRID:SCR 006431. For up-to-date information on the study, visit ![]() .
.
Author Contribution
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Full financial disclosures for the previous 12 months: BE and AM are supported by an Australian government research training programme scholarship. MJ receives royalties from Oxford University Innovations for licensing of the FSL software for commercial, non-academic use.
Funding
This work was funded by grants awarded to LCP from the James and Diana Ramsay Foundation and the NeuroSurgical Research Foundation. The authors declare that there are no conflicts of interest relevant to this work. PPMI – a public-private partnership – is funded by The Michael J. Fox Foundation for Parkinson’s Research and funding partners, including funding partners found at: ![]() . BE and AM are supported by an Australian Government Research Training Programme Stipend (RTP).
. BE and AM are supported by an Australian Government Research Training Programme Stipend (RTP).
Data Availability Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
