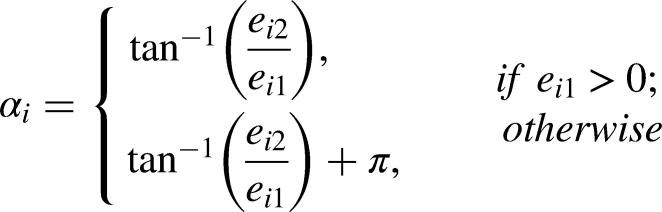Abstract
Background
Directional deep brain stimulation (DBS) provides more precise control of current spread than conventional ring-shaped electrodes. Whether this enhanced flexibility improves motor function is unclear.
Objective
Here we examine whether directional and circular stimulation differentially impact motor performance in patients with Parkinson's disease.
Methods
Motor behaviors were assessed in 31 patients who underwent unilateral subthalamic nucleus brain stimulation surgery (SUNDIAL, NCT03353688). Eight configurations, including 6 directional contacts and their corresponding rings), were evaluated during device activation. Objective measures of motor performance related to limb dexterity, gait, and overall mobility were evaluated in a double-blind fashion in the “off” medication state versus preoperative baseline, with stimulus amplitude at the center of the therapeutic window.
Results
Significant changes in performance were observed across each of five motor tasks between the best and worst directional contacts on a given DBS row (p < 0.001 each task). Certain stimulation directions led to functional declines versus baseline, whereas the best direction yields greater improvement than ring stimulation (p = 0.005, p = 0.001, p = 0.007, p < 0.001, respectively, across tasks). Directional DBS improves therapeutic window and side effect thresholds versus ring stimulation (0.40 ± 0.94 and 0.35 ± 0.51 mA, p < 0.001, respectively), but these variables correlated only modestly with motor performance at a given stimulation site.
Conclusions
Optimized directional subthalamic nucleus DBS yields better group-level motor performance than ring stimulation, in addition to its known advantages related to tolerability. Prospective studies should evaluate whether these improvements persist over longer time intervals.
Keywords
Introduction
Deep brain stimulation (DBS) is effective for motor symptoms of Parkinson's disease (PD) in patients who no longer respond optimally to pharmacologic therapies. 1 Major clinical goals after implant are to maximize motor improvements and avoid or reduce potential stimulation side effects. While conventional ring-shaped leads are beneficial at the group level, they generate a circular electrical field that can potentially interact with off-target structures. Recent advances in lead architecture provide more precise control over stimulation perpendicular from the lead, resulting in improvements in therapeutic window (the range of effective DBS amplitudes at a given stimulation site) and side effect thresholds.2–4 This increased flexibility might also improve motor performance, however novel lead designs increase the dimensionality and complexity of device programming. Therefore, better understanding of how directional leads impact objective motor performance is of significant clinical importance.
Motor improvements from DBS can vary widely from minimal5–7 to clinically significant. 8 This response variability arises from various factors including differences in neuroanatomy, stimulation site, electrode architecture, disease phenotype, assessment methods, and other factors.9,10 In a prior study, we used double-blind, randomized assessments and found reliable changes in walking speed within participants in response to changes unilateral subthalamic nucleus (STN) stimulation. Effect sizes on gait versus preoperative baseline were large within individuals and ranged from clinically meaningful improvement to worsening. 8 However, we did not investigate how motor performance related to stimulation direction, nor did we examine functional domains beyond walking speed. Addressing knowledge gaps on the effects of directional stimulation on motor performance could provide insights into how new DBS lead architectures might enhance patient-specific outcomes and overall efficacy.
A prior clinical study in ten participants with bilateral STN leads, showed that directional DBS improves Movement Disorders Society – Unified Parkinson Disease Rating Scale (MDS-UPDRS) items related to contralateral hand rotation to a greater extent than rigidity or repetitive finger tapping. 4 Other groups show minimal differences in clinical improvement with directional versus ring stimulation with optimal lead placement. 3 Despite its greater complexity, directional DBS is already adopted widely. 11 In one clinic over the course of 2.5 years approximately 2/3 of patients who were implanted received directional stimulation rather than conventional ring stimulation. 12 Understanding the full range of motor performance that arises from directional stimulation would have implications for both clinical programming and device design and parameterization. If directional DBS demonstrates superior group-level motor performance than ring stimulation, this would provide a more compelling rationale for expanding directional lead architectures and for utilizing directionality from the outset, rather than merely as a ‘backup’ strategy.
Despite its immense overall value, individual UPDRS motor items are coarse ordinal scores that are subjectively assigned by raters and sometimes only modestly correlate with quality of life.13,14 Capturing changes in limb rigidity and bradykinesia can be difficult, especially when measured repeatedly in a single session. Similarly, the items related to gait, mobility, and balance can be insensitive to important changes in function, and addressing declines in these domains is a major unmet need for therapy in patients with advanced motor symptoms from PD.15,16 To address questions, we systematically examined motor performance during initial DBS activation with a multidimensional battery of objective, validated motor tasks. We tested two interrelated hypotheses: (1) directional DBS yields significant within- and across-participant changes in motor performance, (2) optimized directional stimulation can yield greater motor improvements than conventional ring stimulation.
Methods
Enrollment and DBS surgery
We obtained prior Institutional Review Board approval and written informed consent from all participants and studied 31 PD patients at a preoperative screening visit and initial device activation after surgery as part of the SUNDIAL study (NCT03353688, IDE# G170063, Supplemental Table 1). Patients were approached for participation after our multidisciplinary committee recommended unilateral STN DBS for routine care. We and others have shown that unilateral STN DBS (followed by staged contralateral surgery when and if it is needed) improves multiple aspects of PD-related motor disability and quality of life for years following surgery.1,17 Inclusion required ≥30% improvement in the MDS-UPDRS part III on dopaminergic medications versus the practically defined “off” state (≥12 h off medications), Hoehn and Yahr score >1, and Dementia Rating Scale-2 score ≥130. Exclusion criteria included disease duration of <4 years, and no history of stroke or other significant neurological conditions. Full enrollment criteria are available at clinical.trials.gov. After screening, participants underwent unilateral DBS surgery to treat the most affected side of the body, implanting a Boston Scientific CartesiaTM ‘1-3-3-1’ lead. Targeting during surgery was informed by multi-pass microelectrode recordings, macrostimulation, intraoperative O-arm 2 imaging, and trial macrostimulation with the newly implanted DBS lead.
Motor battery
Motor performance was assessed in a biomechanics laboratory at baseline with a battery of objective, validated tasks, including elements from the NIH Toolkit. 18 Assessments were performed in the practically defined “off” state at a screening baseline visit and approximately 1 month after surgery on the day of initial device activation (monopolar review). 19 Various domains of function were evaluated, including upper and lower extremity dexterity, gait, balance, and overall mobility. The 9-hole pegboard test measures upper limb dexterity, incorporating repetitive, combined usage of fine motor object manipulation coupled with wrist, elbow, and shoulder movements. This is a timed task where individuals grasp pegs from a cup one at a time with the limb contralateral to the intended hemisphere for DBS, insert the pegs into the 9-hole pegboard, and then extract and place each peg back into the cup. We also created and validated an analogous timed task to assess lower limb dexterity. 20 Seated participants use the contralateral lower limb to touch the numbers 1 through 9 placed in a 3 × 3 grid on a force plate embedded in the floor. They begin the task with their foot on an external target just posterior to the grid and sequentially tap each number in an ‘S-shaped’ pattern on the grid, returning to the target between numbers. To assess gait, participants walked over a 5.5 m gait mat (Zeno, Protokinetics, Havertown, PA) with 2 m before and after the mat, allowing participants to reach and maintain comfortable walking speed. To assess overall mobility, we used the Timed Up and Go test. Participants stand from a seated position in a chair, walk 3 m, turn around, and return to a seated position, while avoiding using their hands to stand up or sit down. To assess static balance, individuals stood unshod on a force plate (AMTI, Watertown MA) with minimal voluntary movement while fixating visually on an ‘X’ on the wall. The center of pressure of the individual is quantified as an area measure in mm2. The presentation of tasks was randomized to avoid an ordering effect.
Monopolar review
Our primary contrasts between directional and ring stimulation were tested during initial device activation approximately 1 month after device implant as contrasts in motor function versus pre-operative baseline. Throughout these sessions, both participant and rater were blinded to all stimulation parameters. Typical activation sessions for the directional lead in this research protocol were longer than routine care, typically lasting 3 to 4 h for a single lead. An experienced DBS clinician programmed in monopolar cathodal configuration with pulse width 60 µs and frequency 130 Hz. The directional lead in this study consists of 8 contacts arranged in a 1-3-3-1 pattern, with directional contact segments in the middle two rows. We contrasted behavioral responses to DBS from the 6 directional contacts versus their corresponding virtual ring configuration. Virtual ring stimulation is defined as simultaneous activation of the 3 directional contacts on a given row with each at 33% current fractionation each. Contacts were tested in a pre-specified, random order to mitigate potential order effects.
To define therapeutic window at any DBS contact, we first identify the floor of the therapeutic window. This is the minimum stimulus amplitude that provides significant improvement in contralateral motor symptoms, such as relief of rigidity and/or rest tremor. DBS amplitude is then gradually increased until a side effect emerged, such as contralateral muscle contraction, dysarthria, persistent or uncomfortable paresthesia, or worsening of parkinsonism. The therapeutic window ceiling is defined as 0.1 mA less than the amplitude threshold for this side effect. Therapeutic window size is then calculated as ceiling minus floor in mA. For motor performance testing at each stimulation site, DBS amplitude was set to the midpoint of the therapeutic window, or (ceiling + floor) / 2, for each contact. In instances where there was no symptomatic benefit at any stimulus amplitude, DBS amplitude was set to 0.2 mA beneath the threshold for side effects or 4 mA (whichever larger). We allowed a few minutes for acclimation to DBS at each site prior to administering the motor performance battery. Between settings, we deactivated DBS and did not proceed with subsequent testing until visible return of motor symptoms, which typically occurred within 5 min.
Here we tested each DBS configuration at the midpoint of its therapeutic window, building on previously described methods. 21 The reasons for examining motor performance at the therapeutic window midpoint are that (1) it is reproducible, (2) it is tailored within each DBS contact/configuration, which facilitates comparisons within and across participants, and (3) it avoids inherent limitations of other methods. One alternative approach is to use a uniform stimulus amplitude for all contacts. This neglects the anatomic and functional heterogeneity of the STN region, where a single DBS amplitude might be poorly tolerated at some sites or insufficient to provide benefit at others. Another method is to manually define an ‘optimal’ stimulation amplitude at each stimulation site by measuring iterative changes contralateral UPDRS sub-score across a range of amplitudes. While this has advantages, it requires repeated measures of one or more elements of the contralateral UPDRS sub-score, which can become subjective and less reliable with repeated testing. Moreover, this method commonly returns a range of effective amplitudes that yield similar subjective improvements. Therefore, we view the therapeutic window midpoint as a reasonable compromise between these approaches to facilitate reproducibility and behavioral comparisons with directional versus virtual ring stimulation.
Data analyses
Outcome variables of interest fall into two broad categories: performance on motor tasks and thresholds for stimulation effects (i.e., therapeutic window). Our primary hypothesis examines whether directional stimulation yields different motor performance versus conventional ring stimulation. We assessed differences between directional and ring stimulation within a given DBS row (i.e., dorsal and ventral) with the dependent variable expressed as a change versus preoperative baseline. To quantify the variance in motor performance within each row we rank-ordered outcomes from best to worst to assess the range in motor outcomes for each row. We used a 2 × 2 repeated measures ANOVA assessing row (i.e., dorsal versus ventral) by stimulation (i.e., directional versus ring) effects. We also noted how frequently changes in motor performance between comparisons met or exceeded the clinically important differences for each task, where known. For instance, previous clinical studies have defined clinically important differences for comfortable walk speed (0.06–0.22 m/s for a small to large effect).
22
timed up and go (3.5 s).
23
and 9-hole pegboard (2.6 s)
24
tasks. If a significant difference was found with the omnibus test, we assessed pairwise effects with the Bonferroni method. Secondary analyses also evaluated change in motor performance versus baseline on the day of device activation, anticipating some component of microlesion.
25
We used identical statistical methods to evaluate how directional stimulation changes therapeutic window on a given DBS row. Additionally, we examined the extent to which motor improvement is predicted by other motor tasks and stimulation thresholds. We analyzed these relationships with the R package ‘rmcorr’ which measures the strength and significance of correlations among variables with repeated measures. Elements in the correlation matrix visualization from ‘corrplot’ are ordered mathematically in an unsupervised manner based upon the correlation structure of their angular eigenvectors with
Results
Baseline motor performance “off” dopaminergic medications >12 h at the pre-operative assessment and the time of device activation showed microlesion effect on 4 of 5 motor tasks (Table 1). 25 Unilateral STN stimulation during device activation significantly improved objective motor performance versus “off” medications on all motor tasks versus both pre-operative baseline and baseline at the time of device activation. Based upon the hypothesized orientation of the directional contacts, directional and ring stimulation did not differ from one another in any systematic way. However, when we assessed the range in motor performance outcomes, we observed statistically significant improvements with directional stimulation versus virtual ring stimulation on each of our 5 motor tasks. Changes in motor performance were large in magnitude, particularly between the best and worst directional contact within an individual, and in most cases well within ranges that are considered clinically relevant. Notably, specific stimulation directions either did not change or even worsened upper limb dexterity and balance relative to the new baseline at the time of device activation. Finally, the magnitude of improvement with optimized directional stimulation exceeded that with virtual ring stimulation on each of the 5 motor tasks. Summary data for these analyses are provided in Table 2.
Baseline motor performance prior to surgery and at device activation.
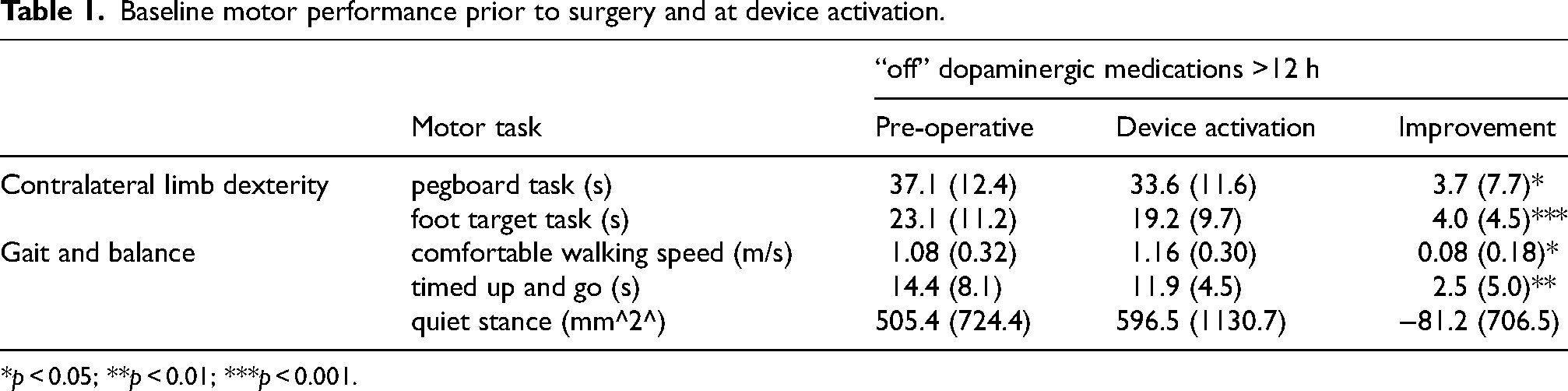
*p < 0.05; **p < 0.01; ***p < 0.001.
Change in motor performance by directional stimulation (n = 31 participants) a .
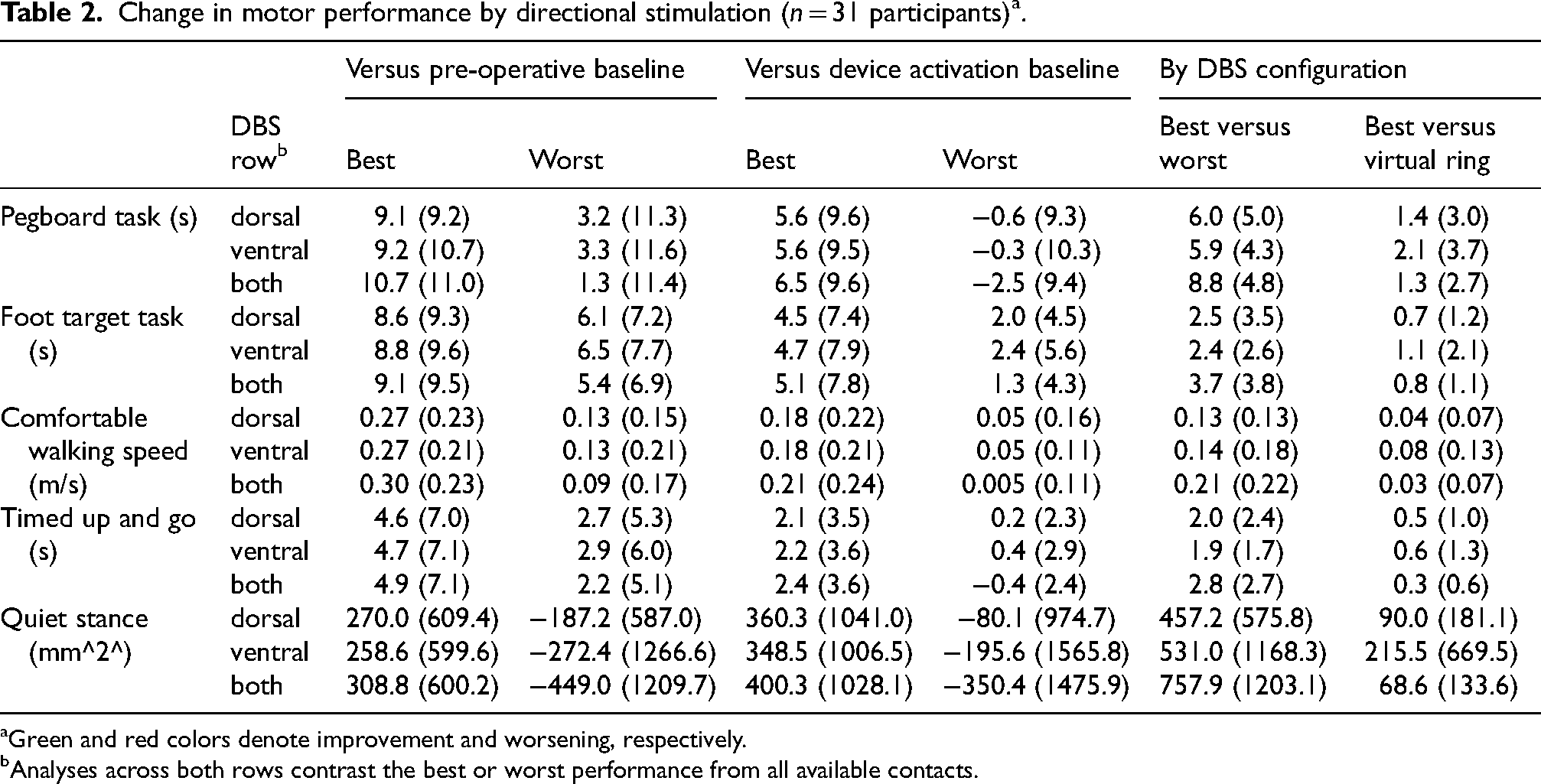
Green and red colors denote improvement and worsening, respectively.
Analyses across both rows contrast the best or worst performance from all available contacts.
Limb dexterity with directional versus ring stimulation
Based on assumed anatomical location, directional contacts improve upper limb dexterity similar to ring stimulation. The row by contact interaction (p = 0.34), main effect of row (p = 0.99) nor the main effect of contact (p = 0.41) were not significant. However, optimized directional contacts significantly modify contralateral upper limb in the 9-hole pegboard task (Figures 1 and 2). When comparing best versus worst directional contact on a given DBS row for each participant, we observed a clinically significant improvement in performance in 25/31 (80%) of dorsal contacts (6.0 ± 4.3 s) and 24/31 (77%) of ventral contacts (5.8 ± 4.3 s) (Figure 2). Contrasting directional versus ring contacts, the row by contact (ring versus directional) interaction and main effects for DBS row were non-significant (p = 0.63 and p = 0.37). Notably, 9-hole pegboard performance was faster on the best directional contact versus its corresponding virtual ring on both the dorsal and ventral rows (p < 0.01, respectively).
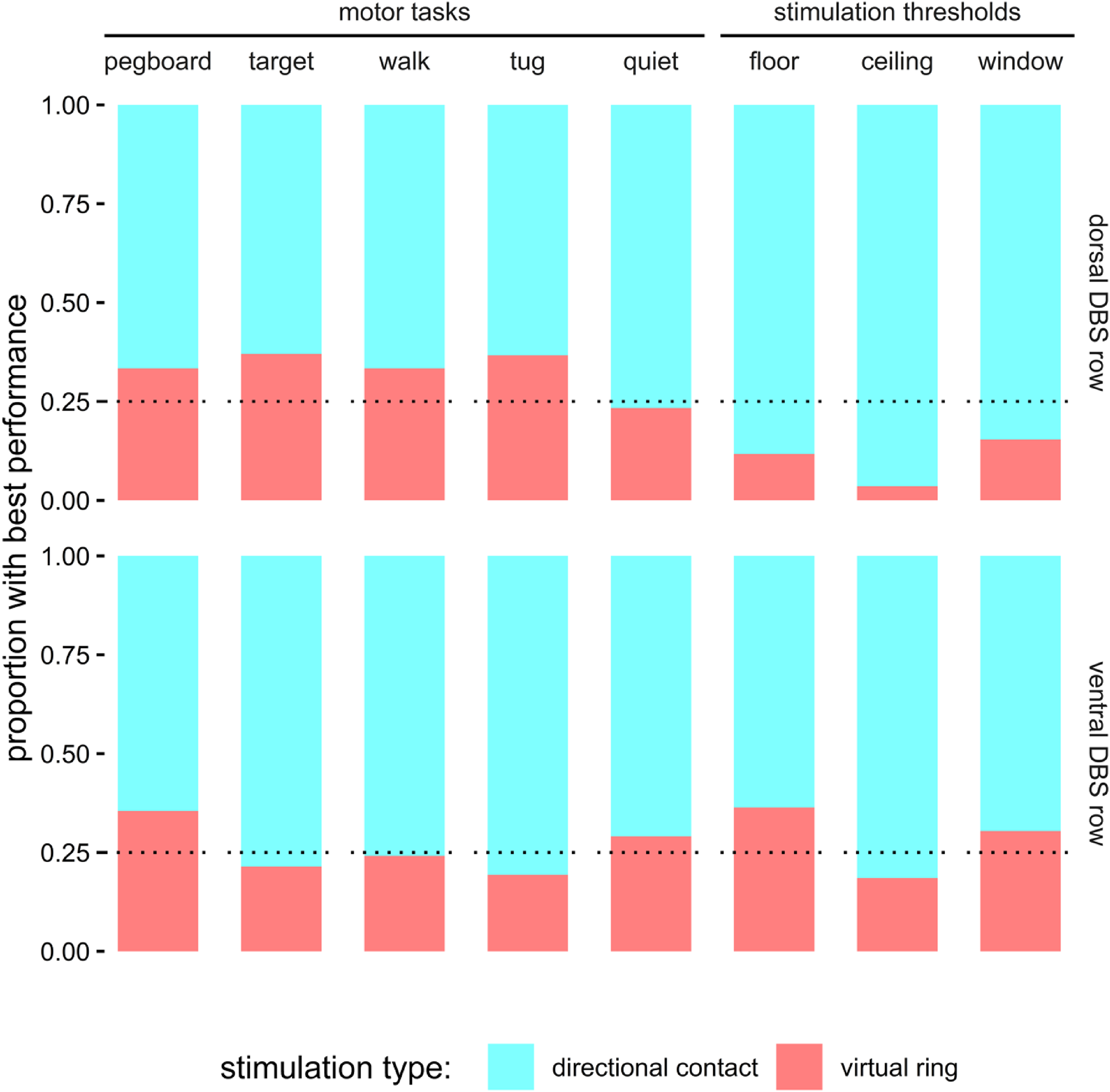
Optimized directional stimulation (lighter color) yielded the best behavioral outcomes more frequently than ring stimulation (darker color) across multiple task demands. Dotted line represents chance within each directional DBS row (i.e., 1 in 4, where one configuration is selected among the three directional contacts plus their virtual ring).
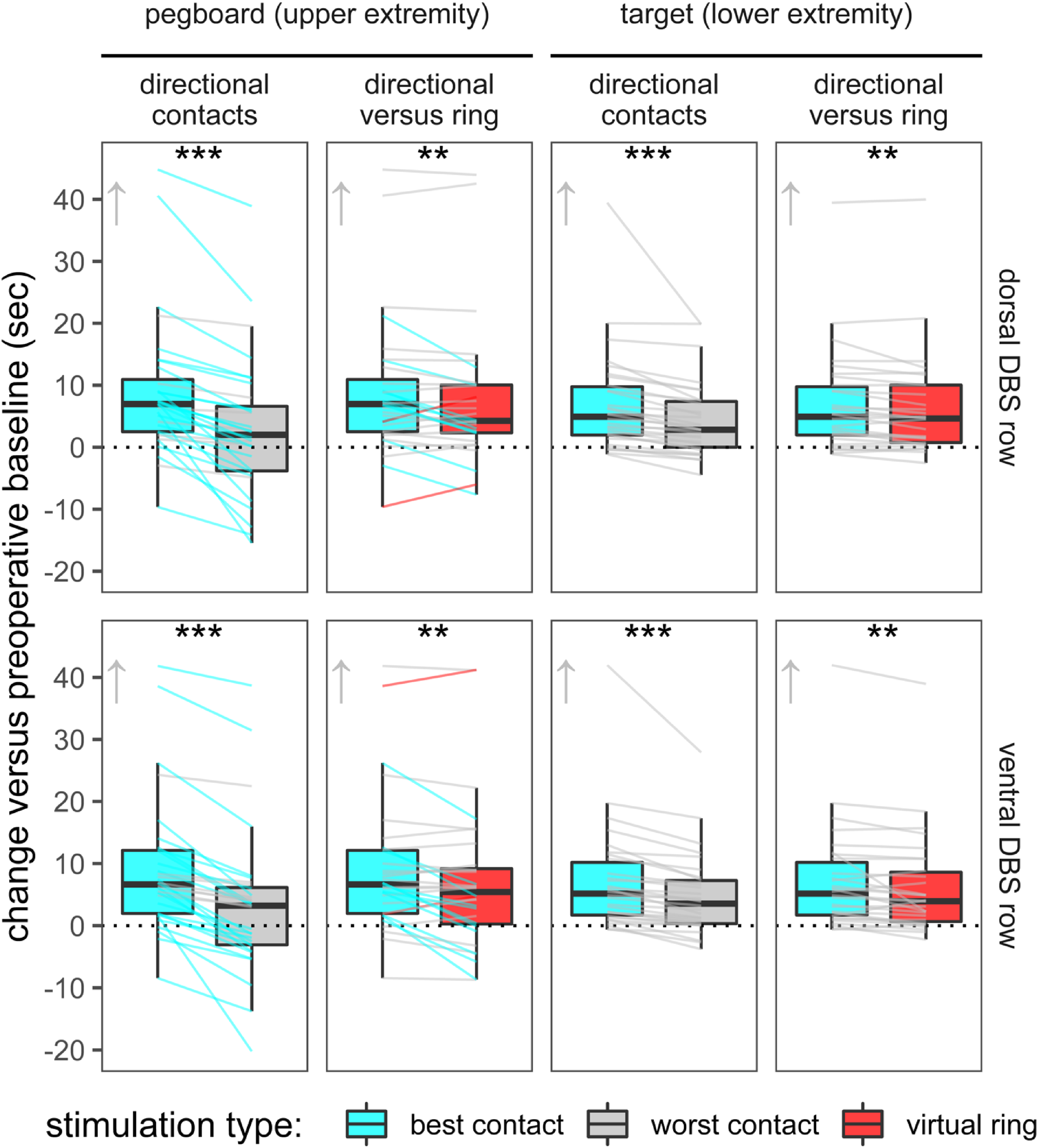
Dexterity tasks measure bradykinesia and coordination in the contralateral limbs. Lines and box plots show within-participant and group changes, respectively. Optimized directional DBS yields greater group-level improvement than ring stimulation on both tasks (arrows indicate improvement). Individual changes that exceed the minimal clinically significant change with directional or ring stimulation are colored cyan and red, respectively. In the second column, 18 instances favor directional stimulation whereas 4 favor ring stimulation, based on this behavioral threshold. Notably, some configurations worsen performance, and optimized directional DBS often mitigates this worsening. Minimal significant change has not been normed for the lower extremity task. *p < 0.05, **p < 0.01, and ***p < 0.001 for group-level contrasts.
Based on assumed anatomical location, directional contacts improve lower limb dexterity similar to ring stimulation. The row by contact interaction (p = 0.34), main effect of row (p = 0.99) nor the main effect of contact (p = 0.41) were not significant. However, optimized directional contacts significantly modify contralateral lower limb dexterity in the lower extremity target task (Figures 1 and 2). When comparing directional contacts on a given DBS row for each participant, the best versus worst directional contact yielded a difference of 2.5 ± 3.5 s (33% improvement) for the dorsal row and 2.6 ± 2.6 s (22% improvement) for the ventral row. Again, the row by contact interaction and main effects of DBS row were non-significant (p = 0.36 and p = 0.87 respectively). Directional contacts versus virtual rings also led to better lower extremity dexterity at the group level on both the dorsal and ventral DBS rows (p < 0.01, respectively).
Gait, overall mobility, and balance with directional versus ring stimulation
Based on assumed anatomical location, directional contacts improve gait similar to ring stimulation. For gait, the row by contact interaction (p = 0.27), main effect of row (p = 0.37) nor the main effect of contact (p = 0.68) were not significant. However, optimized directional contacts significantly impacted gait. Walk speed improved with the best versus worst directional contact on the dorsal and ventral DBS row in 19/31 (61%) and 24/31 (77%) of cases, respectively (Figures 1 and 3). Walk speed differed by 0.13 ± 0.14 and 0.11 ± 0.11 m/s on the best versus worst dorsal and ventral directional contacts, respectively. The row by contact interaction and main effects of DBS were non-significant (p = 0.11 and p = 0.16). However, we detected a significant main effect of contact type (directional versus virtual ring) with faster walk speeds during directional stimulation on both the dorsal and ventral rows (p < 0.01 each, Figure 3).
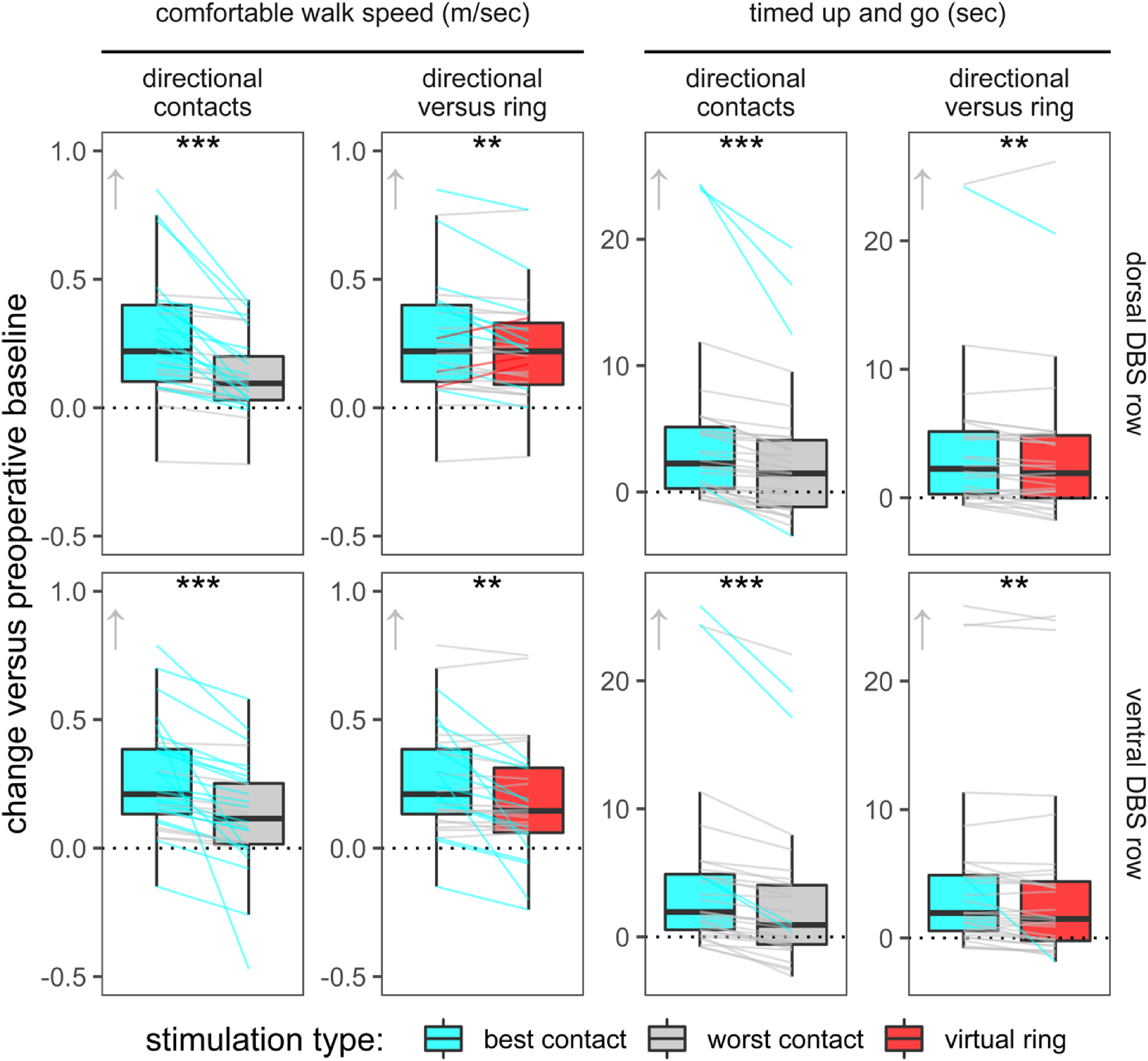
Comfortable walk speed and timed up and go measure gait and overall mobility. Optimized directional stimulation yields more group-level motor improvement than ring stimulation on both tasks (arrows indicate direction for improvement). Individual changes that meet or exceed the minimal clinically significant difference with directional or virtual ring stimulation are colored cyan and red, respectively. In the second column, 21 instances favor directional stimulation whereas 3 favor virtual ring stimulation, based upon this behavioral threshold. Specific directional and ring stimulation configurations sometimes worsen gait and mobility, and optimized directional DBS often mitigates this worsening. *p < 0.05, **p < 0.01, and ***p < 0.001 for group-level contrasts.
Based on assumed anatomical location, directional contacts improve mobility similar to ring stimulation. Regarding the timed up and go task, the row by contact interaction (p = 0.15), main effect of row (p = 0.49) nor the main effect of contact (p = 0.24) were not significant. However, optimized directional contacts significantly impacted mobility. The best directional contact exceeded the threshold for minimal clinically significant improvement (≥3.5 s change) versus the worst contact in 4/31 of contact (13%) on both the dorsal and ventral DBS rows (Figures 1 and 3). Mean differences between best and worst contacts were 2.0 ± 2.4 and 1.9 ± 1.7 s on the dorsal and ventral rows (Figure 3). There was a non-significant row by contact interaction and main effect of DBS row (p = 0.60 and p = 0.98, respectively), but group level performance was faster for the best directional contact versus virtual ring on each DBS row (p < 0.01 each).
Based on assumed anatomical location, directional contacts improve balance similar to ring stimulation. Regarding the quiet stance task, the row by contact interaction (p = 0.19), main effect of row (p = 0.40) nor the main effect of contact (p = 0.09) were not significant. However, optimized directional contacts significantly impacted balance. For the quiet stance task, we omitted observations from one participant from the group-level analyses as they were each >5.5 standard deviations from the mean. Sway area differed by 403.7 ± 562.4 mm2 for best versus worst dorsal contacts (40% improvement) and 345.6 ± 439.0 mm2 for best versus worse ventral contacts (41% improvement) (Figures 1 and 4). Row by contact interaction and main effect of DBS row were non-significant (p = 0.47 and p = 0.73 respectively) but best directional contact again outperformed virtual ring on each DBS row (p < 0.01 each).
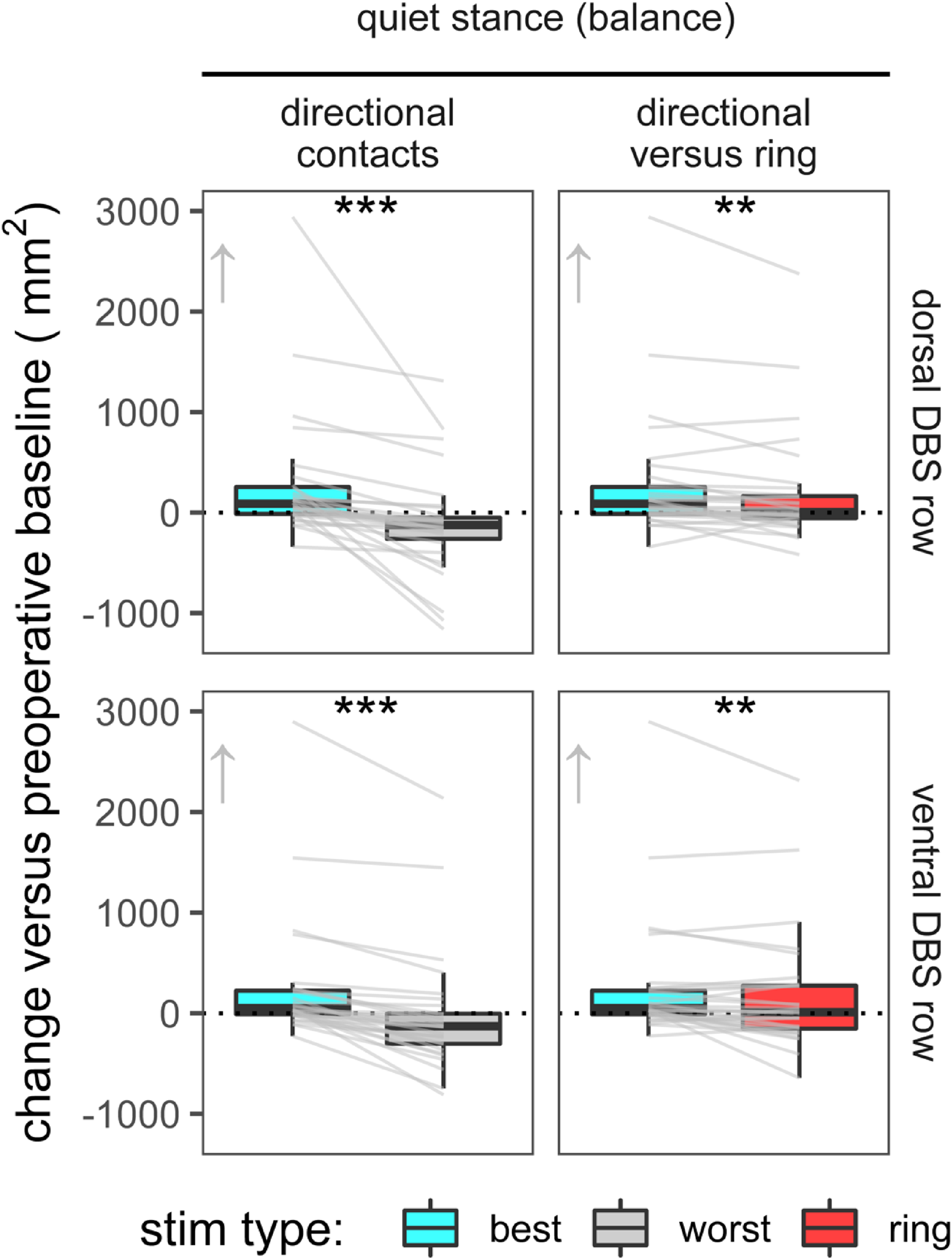
Static balance measured by an embedded force plate in the floor. Optimized directional stimulation yields greater improvement than ring stimulation in static balance at the group level (boxplots). Despite overall improvements versus baseline, directional and ring stimulation can both worsen static balance, and optimized directional DBS often mitigates this worsening (line segments). Minimal clinically significant change is not established for this task. *p < 0.05, **p < 0.01, and ***p < 0.001 for group-level contrasts.
Stimulation thresholds with directional versus ring stimulation
Stimulation thresholds include therapeutic window, floor, and ceiling. These measures differed among directional contacts and when comparing the best directional contact to its corresponding ring (Figure 5). Significant main effects were found for side effect threshold on both DBS rows, regardless of whether comparing among directional contacts or between the best directional contact and its virtual ring (p < 0.01 and p = 0.07, respectively). However, we observed non-significant effects of row and row by contact interaction for both side effect threshold and therapeutic window (p = 0.59, p = 0.28, p = 0.81, p = 0.74, respectively).
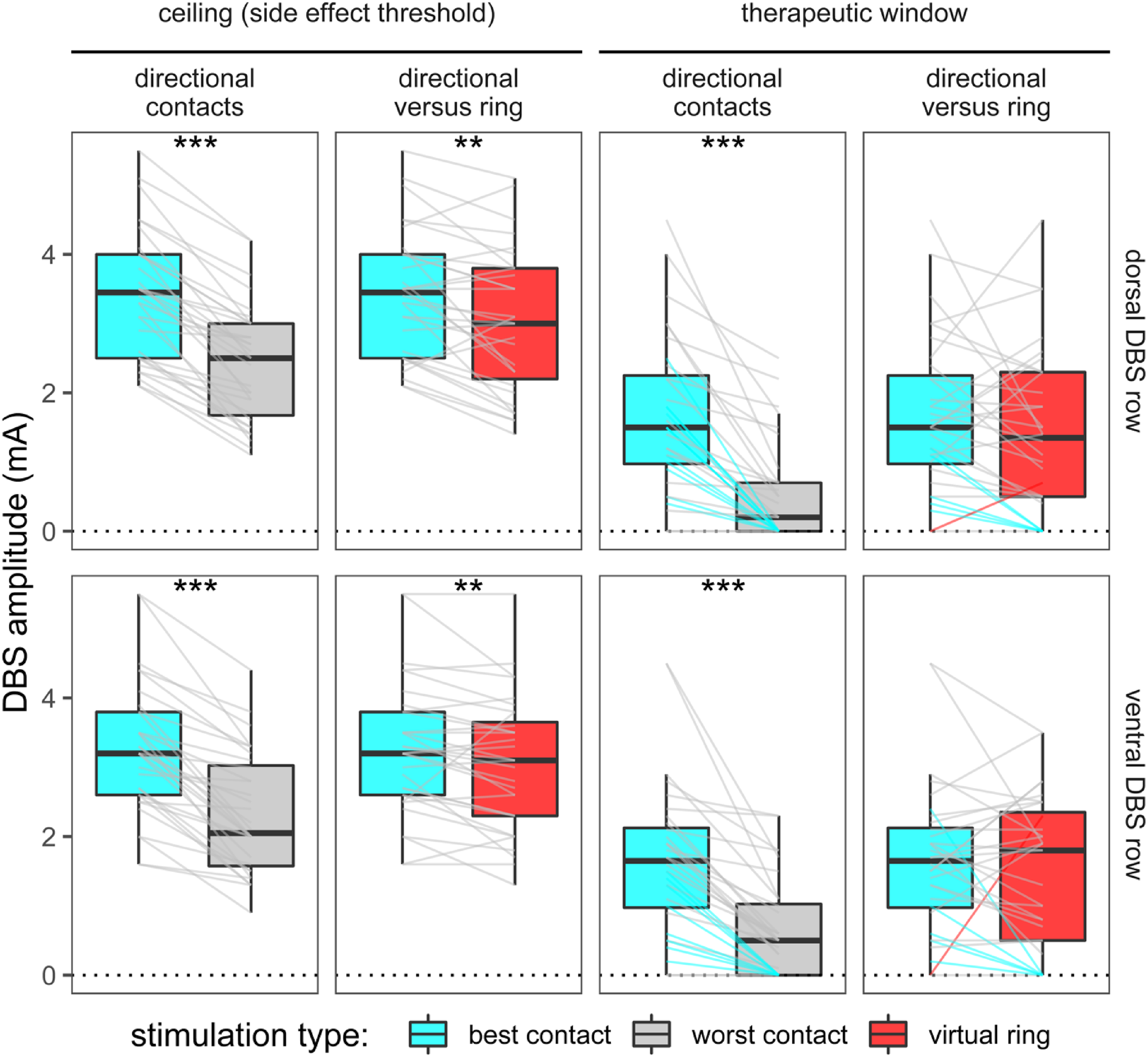
Directional stimulation improves amplitude thresholds for dose-limiting side effects and therapeutic window. Optimized directional DBS often rescues both virtual ring and directional configurations that display no therapeutic effects on the same DBS row (cyan line segments). *p < 0.05, **p < 0.01, and ***p < 0.001 for group-level contrasts.
Predictors for motor improvement by directional stimulation
Given the complex and time-intensive nature of directional DBS programming, we sought to identify predictors for improvements in motor performance at the group level (Figure 6). For this, we used R statistical packages ‘rmcorr’ to calculate repeated measures correlations across all directional contacts and tasks and ‘corrplot’ to generate matrix visualizations of these relationships. Notably, motor performance on other tasks emerged as the most robust predictor for motor function at a given directional contact. In contrast, stimulation threshold such as ceiling and therapeutic window exhibited strong internal correlations but predicted motor performance only moderately.
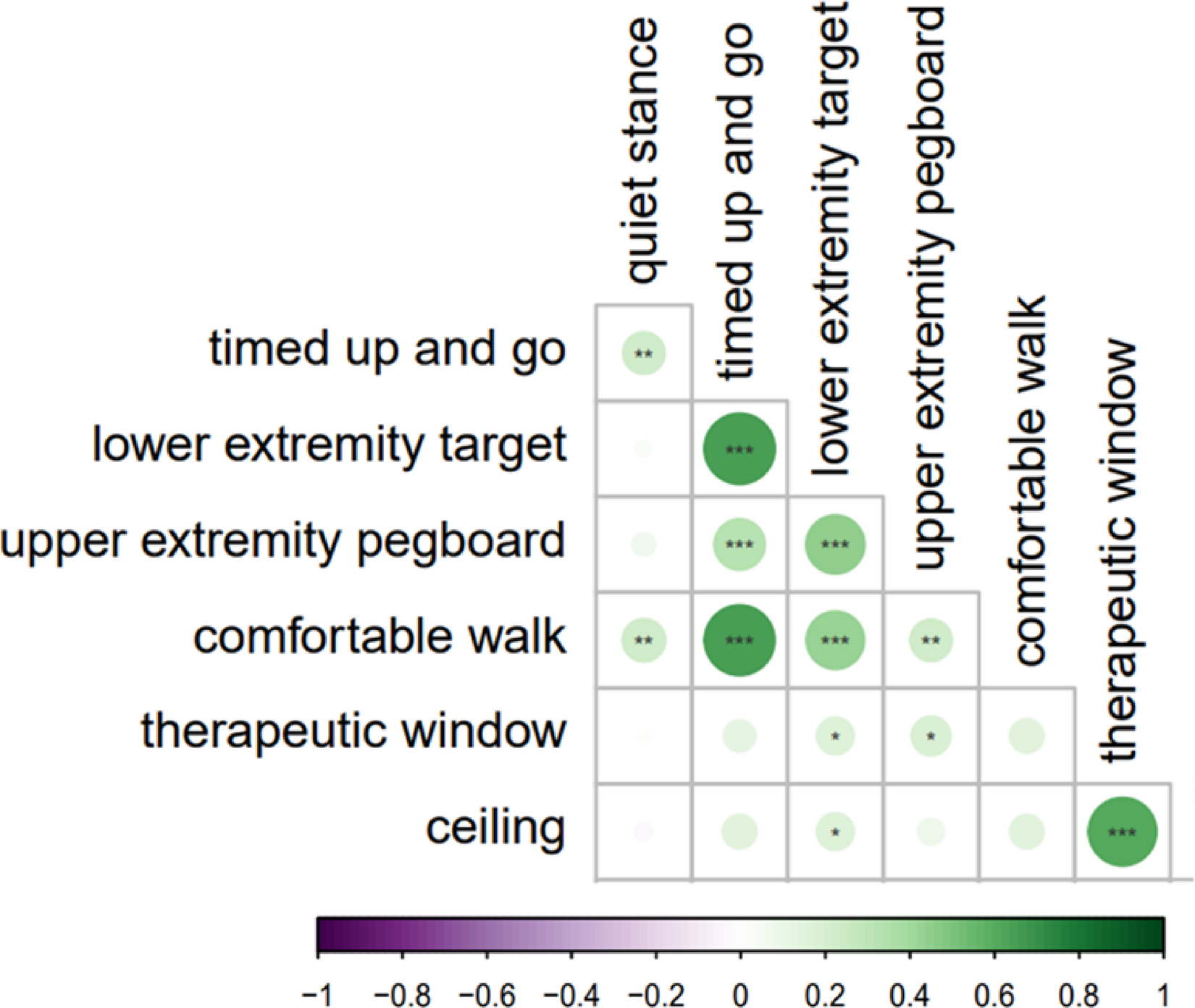
Matrix of repeated measures correlations across all dependent variables, organized in an unsupervised manner by ‘corrplot’ based upon their covariance structure. The best predictor for motor improvement from directional DBS is performance on the other motor tasks. Stimulation thresholds (floor, ceiling, and therapeutic window) correlate strongly internally but predict motor performance only modestly. Color and dot size represent the correlation coefficients, and asterisks correspond to p-values (*p < 0.05, **p < 0.01, and ***p < 0.001).
Discussion
Altering stimulation direction in the STN region elicits meaningful changes in objective, quantitative measures of motor performance. Our double-blind, randomized behavioral assessments during device activation showed these changes both at the group level and within individuals. Although the largest changes related to stimulation direction occurred with the best versus worst directional contacts within individual, optimized directional DBS yielded statistically significant improvements compared to virtual ring stimulation, as well. Furthermore, directional DBS often mitigated or rescued suboptimal motor improvements from ring stimulation within individuals, across multiple domains of motor function (Figures 1–4). Collectively, our findings suggest that the spatial flexibility of directional leads not only enhance tolerability but, in some instances, can also enhance motor performance.
Optimized directional stimulation improved motor performance compared to virtual ring stimulation across each of five objective motor tasks at the group level. These tasks various functional domains including contralateral upper and lower limb dexterity, gait, mobility, and balance. And while, on average, the improvement in performance between optimized directional and virtual ring were statistically significant, in some instances they are modest at the group level. However, DBS programming is a personalized and tailored therapy for an individual, and our data showed large clinically meaningful improvements, and this can greatly impact quality of life. These assessments were with a first-generation segmented lead, a crucial step towards understanding the full range of clinical outcomes that might arise from new lead architectures. Previous research on directionality mainly assessed motor efficacy with a limited set of UPDRS items, focusing predominantly on rater assessment of contralateral arm rigidity.3,26,27 Earlier studies assessing ring-shaped contacts using similar methods found effect sizes ranging from small or equivocal.28–31 to large and clinically significant.8,22 Importantly, this response variability might in part relate to the coarser stimulation fields of earlier generation leads, among additional variables such as disease phenotype, lead location, stimulation parameters, and others. One strength of this study is that our results rest largely on within-participant contrasts, which minimize or eliminate many of these potential confounding factors.
Prior research indicates that directional stimulation improves amplitude thresholds for DBS side effects. Our study confirms and expands on this finding, suggesting similar findings related to motor efficacy. As with side effect thresholds, motor efficacy among the 3 contact segments in a DBS row typically ranges from improvement to no significant change, to worsening versus virtual ring stimulation, as shown in Table 2. Notably, different DBS configurations, whether directional or ring, sometimes lack a therapeutic window, and specific stimulation directions can improve motor performance in these instances (Figures 2–4). Together, these findings raise questions about the rationale for ring stimulation at the STN target, particularly when at least one segment in the virtual ring contributes minimally, if at all, to therapy in isolation (Table 2). Moreover, our finding that specific stimulation directions can worsen motor performance was somewhat unexpected, especially given testing occurred within the therapeutic window and/or beneath the side effect threshold. Thus, while reduction in contralateral rigidity and/or rest tremor define therapeutic window, this might not always translate into optimal motor performance, especially for complex or repetitive tasks. Hence, novel algorithm-, neuroimaging-, or biomarker-based methods to guide directional DBS programming are needed. Our findings emphasize that rigorous and systematic motor testing should be conducted prior to widespread clinical adoption of such methods, as specific stimulation directions can yield measurable declines in motor function.
While DBS generally improves UPDRS motor items and more integrated functional tasks.5,7,8,32 outcomes can vary.8,28 One contributor to this variance might be the lack of explicit optimization of stimulation parameters based on motor performance. Directional stimulation opens new avenues for more detailed measurement of performance changes across more diverse functional domains. Of note, directional DBS generally improves limb dexterity, gait, and overall mobility across participants, but upper extremity dexterity and balance (quiet stance) tended to worsen more readily compared to the other tasks. Previous authors have proposed distinct functional sub-regions in the subthalamic region.33,34 and noted that STN stimulation does not improve balance as consistently as gait or overall mobility, whether measured in laboratory or clinical settings.30,35 Additionally, the STN is near fibers of passage that can worsen function such as the corticospinal and corticobulbar tracts. Given these considerations, one future approach to directional programming might be to identify contact segments that worsen or do not change function and exclude them from the stimulation field. Collectively, our findings suggest that directional stimulation provides opportunities to exploit the anatomy of the local brain circuit to improve efficacy or prevent subtle, unintended functional declines.
How then do we harness potential advantages of directional stimulation? One challenge is that directional leads greatly expand therapeutic options within a given patient, which in turn increases the time requirements for device programming. This underscores the need for robust predictive biomarkers to guide DBS therapy. In our study, the best predictor for motor efficacy at a given stimulation site was performance on the other motor tasks, whereas elements of the therapeutic window predicted efficacy more modestly (Figure 6). While sensible and internally consistent, these results do not address the unmet need for dimension reduction in clinical DBS programming. Our approach differs from previous studies in that we defined efficacy based on functional performance on validated, quantitative tasks rather than more subjective elements from the UPDRS. 36 We opted for these measures in lieu of 10 serial repetitions of a small number of the UPDRS part 3 items related to contralateral upper extremity function (predominantly rigidity), anticipating that this would provide more diverse task demands, greater objectivity, and better sensitivity to change.
Our study has potential limitations, some of which open new avenues for inquiry. Repeated motor assessments might introduce lingering behavioral responses across conditions; however we randomized the order of contact evaluation, and our prior work demonstrates large and consistent changes in comfortable walk speed across multiple task repetitions. 8 In any case, serial assessments of motor function are important to the larger clinical context, as they remain the gold standard for decision-making related to DBS programming. Another consideration is that our studies at the time of device activation are relatively acute, so future work should examine whether motor improvements are sustained over longer time intervals. Third, we only studied directionality with single contact segments. Finer gradations of current fractionation across multiple contact segments should be evaluated, as well, especially given that at least one of the three contact segments on a given DBS row tended to worsen motor performance (Table 2). Fourth, two of our tasks (foot target task and quiet stance) were impacted significantly by stimulation direction but do not have established norms for clinically meaningful change. Both however are validated, and the quiet stance task is a component of the NIH motor toolkit. 18 Future studies should define the clinical thresholds for these tasks, especially given the need for more therapy options for gait, balance, and mobility in these patients. Finally, we only investigated PD patients who received DBS in the STN, which typically has less favorable side effect thresholds than other stereotactic targets for movement disorders. 37 Directionality may offer different advantages or disadvantages for other brain targets or diseases, therefore further research in more diverse patient populations is warranted.
Conclusions
Optimized directional STN DBS can yield better motor performance than conventional ring stimulation, in addition to known advantages related to tolerability. Robust clinical programming algorithms and predictive biomarkers are needed to efficiently identify optimized parameters for directional stimulation.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X241301071 - Supplemental material for Pointing in the right direction: Greater motor improvements with directional versus circular subthalamic nucleus deep brain stimulation for Parkinson's disease
Supplemental material, sj-docx-1-pkn-10.1177_1877718X241301071 for Pointing in the right direction: Greater motor improvements with directional versus circular subthalamic nucleus deep brain stimulation for Parkinson's disease by Christopher P Hurt, Daniel J Kuhman, Alyson Moll, Barton L Guthrie and Joseph W Olson, Arie Nakhmani, Melissa Wade, Sarah A Brinkerhoff, Marshall T Holland, Harrison C Walker in Journal of Parkinson's Disease
Footnotes
Acknowledgements
We acknowledge the patients who donated considerable time and effort to this work.
ORCID iDs
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We are grateful for research funding from the National Institutes of Health (UH3NS100553, UG3NS130202, and R01NS119520).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Walker serves as a scientific consultant for Varian. The remaining authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.