Abstract
Genome-wide association studies have identified SV2C as a Parkinson's disease (PD) risk locus, with a common missense variant p.Asp543Asn in the synaptic vesicle glycoprotein 2C (SV2C) protein significantly associated with PD. We examined if other rare SV2C variants also influence PD risk. We analyzed sequencing data of 9810 East Asian individuals comprising 4298 patients and 5512 controls and identified 55 rare nonsynonymous variants. There was no significant association of rare nonsynonymous or loss-of-function variants with PD. Our findings show that besides p.Asp543Asn, other rare coding variants in SV2C do not play a major role in PD susceptibility in East Asia.
Plain language summary
Plain language summary: In our previous study, we found a genetic change p.Asp543Asn in the synaptic vesicle glycoprotein 2C (SV2C) protein to be associated with higher Parkinson's disease (PD) risk. Here, we aim to determine if there are other genetic changes, especially those that alter the SV2C protein, that also influence PD risk. We analyzed sequencing data of 9810 East Asian individuals comprising 4298 patients and 5512 controls and identified 55 other genetic changes that were not associated with PD risk. Our findings show that within the SV2C gene, only p.Asp543Asn is associated with PD risk in East Asia.
Introduction
Parkinson's disease (PD) is a common, age-related neurodegenerative disease influenced by multiple rare and common genetic factors. A genome-wide association study in East Asian populations identified SV2C as a novel PD-associated locus. 1 This association was replicated in European-ancestry populations.1–3 The lead SNP within the locus rs246814 is in near complete linkage disequilibrium (r2 = 1) with a missense variant p.Asp543Asn (rs31244) that occurs within an extracellular/luminal domain of the encoded synaptic vesicle glycoprotein 2C (SV2C) protein. SV2C was previously investigated for a possible role in PD because of its restricted expression in PD-relevant brain regions, whereby knockout mice showed a significant reduction in striatal dopamine release 4 and impaired dopamine storage. 5 The introduction of a new asparagine residue by this amino acid substitution may affect N-linked glycosylation of this domain via the creation of a new glycosylation site (Asn543-X-Thr545). 6 This variant is common, present at allele frequency ∼10% in East Asians. Here, we aim to investigate whether rare loss-of-function variants in SV2C that are not tagged or represented on genome-wide SNP arrays are also associated with PD. We also evaluated whether any other rare missense variants may introduce or abolish other glycosylation sites in the SV2C extracellular/luminal domain.
Methods
We sequenced all coding exons of SV2C in 9810 East Asian individuals comprising 4298 PD patients (age at onset [mean ± standard deviation] 61.1 ± 10.9 years; age at sample collection 66.6 ± 9.8 years; 53.44% males) and 5512 healthy controls (65.2 ± 9.4 years; 46.88% males) from Singapore, Malaysia, Hong Kong, Taiwan and South Korea. 7 Samples were 151 bp paired end sequenced on Illumina sequencers (HiSeq 4000 and NovaSeq 6000). Patients were diagnosed with PD using the UK Brain Bank Criteria. Cases and controls were confirmed to be genetically well-matched in a principal component analysis. 7 All subjects gave informed consent, and the study received approval from the institutional ethics committee (SingHealth Centralized Institutional Review Board CIRB 2002/008/A and 2019/2334 and Nanyang Technological University Institutional Review Board IRB-2016-08-011). All 12 exons of SV2C were sequenced to a mean coverage of 67X with 97.9% of target bases covered by 10 or more reads. Variant calling was conducted using the Genome Analysis Tool Kit (GATK v3.7) on Hg19/GRCh37 8 following the Best Practices Guidelines. 9 Variant quality control was conducted by removal of variants that (i) did not pass Variant Quality Score Recalibration (VQSR), (ii) had low quality genotype calls (DP < 8, GQ < 20), (iii) had call rates < 95% in cases, controls and all samples, (iv) failed Hardy-Weinberg equilibrium test (p < 1 × 10−6), and (v) had significant differential missingness between cases and controls (p < 1 × 10−4). Sample quality control was conducted by removal of samples with (i) gender discordance between patient records and sex genetically inferred from whole-exome sequenced data, (ii) poor genotyping concordance (< 95%) between whole-exome sequenced data and previously conducted genome-wide association arrays, 1 (iii) high heterozygosity (>3.5 standard deviations from the mean), high exome-wide singleton counts, and low genotyping call rates (< 95%), (iv) with the lower call rate in related sample pairs when identity-by-descent analysis (IBD > 0.125), and (v) not of East Asian genetic ancestry from ancestry principal component analysis (PCA) against 2141 samples from the 1000 genomes (1KGP) reference panel. 10 Variants were also annotated based on canonical transcript with Ensembl Variant Effect Predictor (VEP, version 104). 11
We then identified SV2C variants (GRCh37 genomic region: chr5:75,378,997-75,649,764) which were rare in East Asians (minor allele frequency (MAF) ≤ 1% in Genome Aggregation Database (gnomAD) EAS) and were predicted to be pathogenic as defined by probably damaging/possibly damaging classification of SNPs by PolyPhen-2 12 and high-confidence classification of indels and splice variants by LOFTEE. 13 These variants were further assessed for pathogenicity in ClinVar 14 and Varsome, 15 with pathogenic variants defined by Varsome classification of “PP3: Pathogenic Very Strong” and “BP4: Supporting”. Visualization of variants across SV2C protein domains was conducted with Protter (https://wlab.ethz.ch/protter/start/).
Gene burden testing of SV2C rare pathogenic variants carrier count between PD cases and controls was conducted using stratified Cochran-Mantel Haenszel test (four strata: Singapore/Malaysia, Hong Kong, Taiwan and South Korea; Supplemental Figure 1) 7 without continuity correction. A similar test was done on allele counts for single variant association testing of common variants. Further gene-based tests were also conducted using sequence kernel association tests (SKAT), SKAT-O (optimal) and Burden tests 16 with adjustment for PCs 1-3.
Results
A total of 4298 PD cases and 5512 matched controls were collected from cohort studies in Singapore, Malaysia, South Korea, Hong Kong, and Taiwan. PCA analysis indicated that these 9810 samples were of East Asian descent (Supplemental Figure 1A). All samples from Singapore, Malaysia, Hong Kong and Taiwan that were retained in our analysis are of Han Chinese Ancestry, with Singapore and Malaysia sharing similar genetic background (Supplemental Figure 1B). 7
We identified a total of 92 variants in the coding exons of SV2C out of which 51 were missense variants, 2 were stop-gain variants, 3 were frameshift variants and 1 was a splice site variant. 89 of these variants were rare (MAF ≤ 1%) and 3 variants were common (Figure 1, Supplemental Tables 1 and 2). Of the missense variants, 28 were predicted to be pathogenic by PolyPhen-2 (Table 1). Another 4 variants were predicted to cause loss-of-function at high confidence by LOFTEE (Table 1). Of the 4 variants occurring within the extracellular/luminal domain of SV2C (Figure 1), none occured at the known glycosylation sites within that domain. Similarly, none of the variants introduced new asparagine residues within the extracellular/luminal domain.
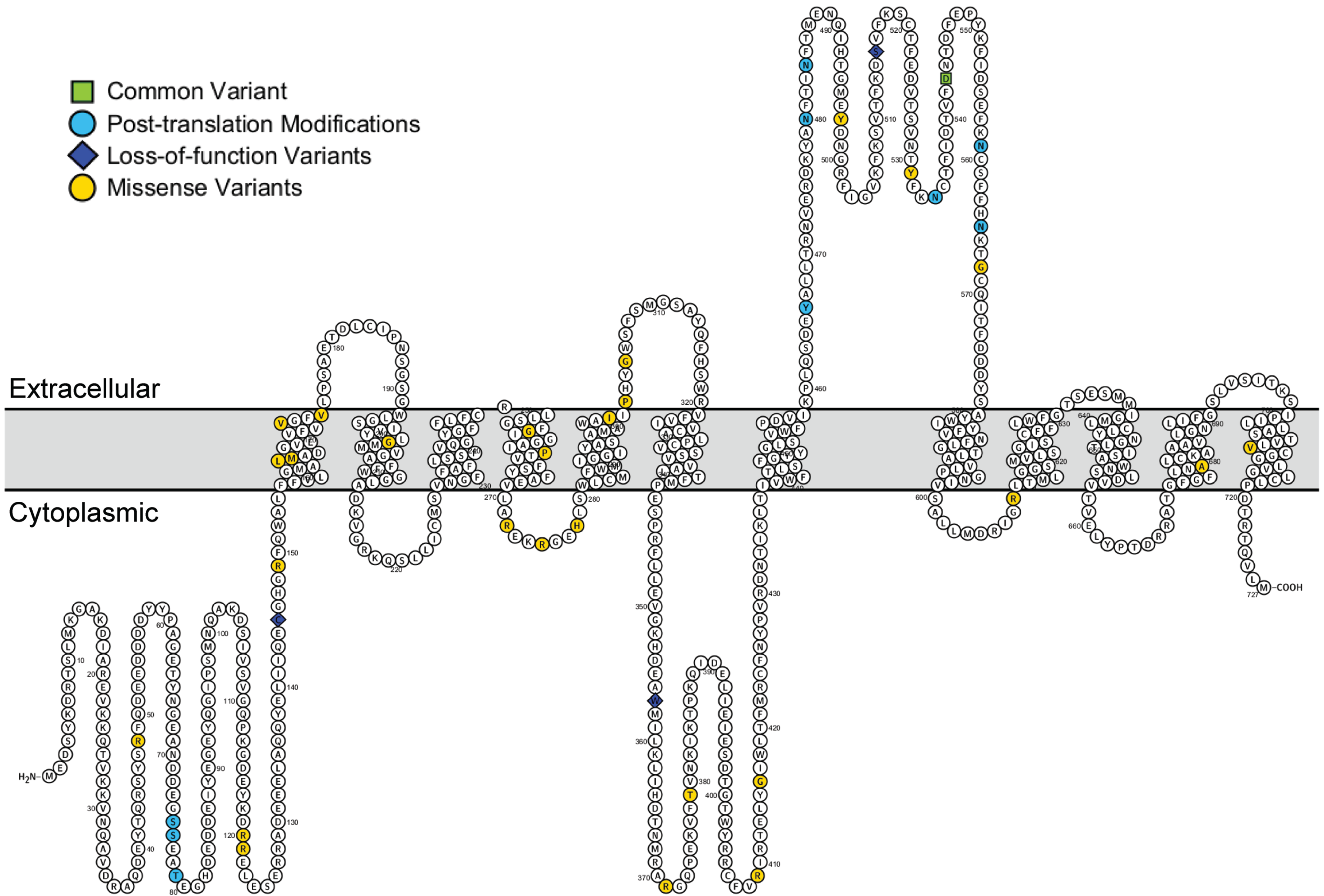
Annotation of rare predicted pathogenic variants and common missense variant (p.Asp543Asn) along the SV2C protein.
Rare variants in the coding exons of SV2C that were predicted to be pathogenic by PolyPhen-2/LOFTEE.
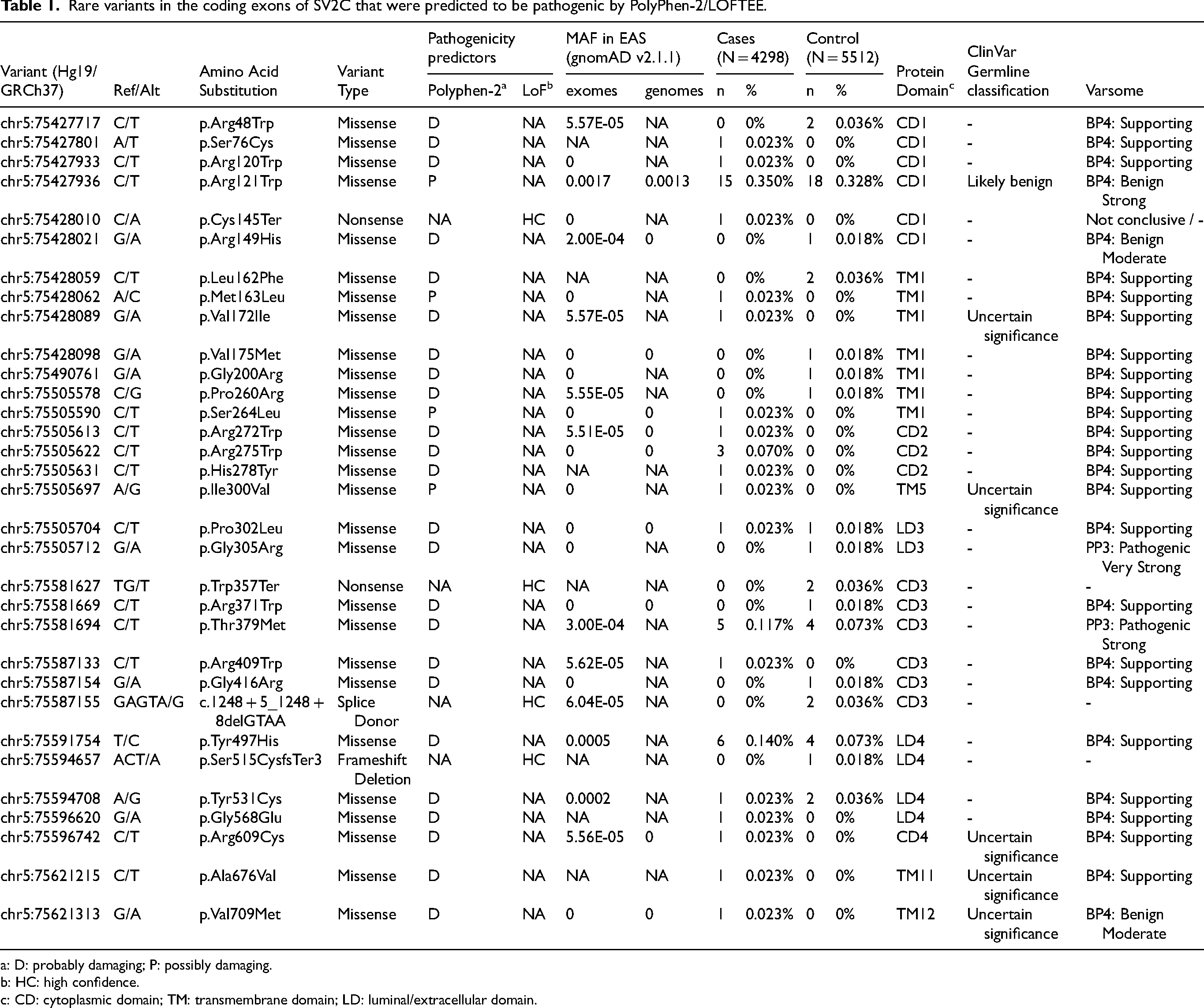
: D: probably damaging; P: possibly damaging.
: HC: high confidence.
: CD: cytoplasmic domain; TM: transmembrane domain; LD: luminal/extracellular domain.
Among the common variants, only p.Asp543Asn (rs31244) showed significant association with PD (Supplemental Table 2) (allele frequencies: 10.72% cases, 9.14% controls, OR = 1.156, 95% CI = 1.049–1.273, p = 0.003). The other two common variants (Supplemental Table 2) showed no significant association or nominal association with PD (p.Arg120Arg [rs10070440]: allele frequencies 33.79% cases, 32.27% controls, OR = 1.028, 95% CI = 0.967–1.094, p = 0.372; p.Thr482Ser [rs2270927]: allele frequencies 19.69% cases, 17.63% controls, OR = 1.079, 95% CI = 1.002–1.163, p = 0.045) and were not in high linkage disequilibrium with the lead GWAS SNP rs246814 (r2 < 0.5) in East Asians. We did not observe significant gene burden of rare pathogenic SV2C variants (Table 1) in PD cases (0.512% carriers) compared to controls (0.408% carriers) using stratified CMH (OR = 1.213, 95% CI = 0.799–1.844, p = 0.356; Supplemental Table 3). Testing with sequence kernel association test also showed a lack of association between SV2C rare pathogenic variants gene burden with PD (SKAT-O p = 0.483; SKAT p = 0.458; Burden p = 0.319; Table 1). Similar results were obtained when Varsome was used to classify rare pathogenic variants (CMH: OR = 1.451, 95% CI = 0.869–2.423, p = 0.145; SKAT-O p = 0.271; SKAT p = 0.204, Burden p = 0.174). The 4 variants predicted to cause loss-of-function at high confidence were found in 1 PD case and 5 controls (CMH: OR = 0.287, 95% CI = 0.033–2.454, p = 0.224).
Discussion
Overall, we observed no evidence for association of rare missense variants in SV2C with PD in a sequencing study of 9810 East Asian samples. SV2C knockout mice were shown to exhibit mild motor deficits and impaired dopamine release. 4 However, we identified only 1 patient with a high confidence heterozygous loss-of-function variant in SV2C, suggesting that loss-of-function mutations in SV2C do not play a major role in PD risk in the East Asian population. Consistent with our observations, rare pathogenic (MAF < 0.1%, LoF High Confidence + CADD > 20) SV2C variants were not enriched (p > 0.05) in PD cases (0.226%) compared to controls (0.567%) in European-ancestry populations (UK Biobank).17,18
The common missense SNP p.Asp543Asn does not appear to cause loss-of-function of the SV2C protein. The mechanisms through which it contributes to PD susceptibility remain to be investigated, although it is predicted to introduce a new glycosylation site in the extracellular/luminal domain of SV2C. None of the other rare missense variants we identified in SV2C are likely to influence glycosylation of this domain. Overall, our results show that rare variants in SV2C do not play an important role in PD susceptibility in the East Asian population. Further work is needed to understand the effects of the GWAS-associated SNP on SV2C gene function and PD susceptibility.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X241300298 - Supplemental material for Rare SV2C coding variants in Parkinson's disease risk
Supplemental material, sj-docx-1-pkn-10.1177_1877718X241300298 for Rare SV2C coding variants in Parkinson's disease risk by Chu Hua Chang, Elaine Guo Yan Chew, Michelle Mulan Lian, Moses Tandiono, Zheng Li, Sun Ju Chung, Louis CS Tan, Wing-Lok Au, Kumar M Prakash, Azlina Ahmad-Annuar, Ai Huey Tan, Vincent Mok, Anne YY Chan, Juei-Jueng Lin, Beom S Jeon, Chiea Chuen Khor, Shen-Yang Lim, Eng-King Tan and Jia Nee Foo in Journal of Parkinson's Disease
Footnotes
Acknowledgments
We thank Shin Hui Ng (National Neuroscience Institute), Wei Ling Beh, Winnie Tay, Ken Wong and Andrew Ang (Nanyang Technological University Singapore) for administrative support.
ORCID iDs
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the National Medical Research Council, Singapore under the Open Fund Large Collaborative Grant (MOH-000207; to E.-K.T) and the Open Fund Individual Research Grant (MOH-000559; to J.N.F.), Open Fund Young Individual Research Grant (MOH-001329 to E.GY.C) as well as the Singapore Ministry of Education Academic Research Fund Tier 2 (MOE-T2EP30220-0005; to J.N.F.) and Tier 3 (MOE-MOET32020-0004; to J.N.F.). C.C.K is supported by the Singapore National Research Foundation Investigatorship (NRF-NRFI2018-01). S.-Y.L. and A.H.T. are supported by the University of Malaya Parkinson's Disease and Movement Disorders Research Program (PV035-2017).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Eng-King Tan is an Editorial Board member of this journal but was not involved in the peer-review process of this article nor had access to any information regarding its peer review.
The remaining authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data supporting the findings of this study are available within the article and/or its supplemental material.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
