Abstract
Diagnosing papillary thyroid carcinoma (PTC) remains challenging, particularly due to limitations in fine-needle aspiration biopsy (FNAB), which yields up to 10% nondiagnostic results. The objective of this study was to evaluate the diagnostic and prognostic potential of four candidate microRNAs (miR-21, miR-31, miR-187-3p, and miR-200a-5p) in PTC from multinodular goiter (MNG) and normal. Fresh tissue samples from PTC and MNG patients were analyzed using quantitative RT-PCR, followed by ROC analysis to assess diagnostic accuracy and correlation with clinical, histopathological, and hormonal parameters. Compared to normal tissue, miR-21 and miR-187-3p were significantly upregulated in PTC, while miR-31 and miR-200a-5p were downregulated. MNG samples showed similar but less pronounced trends. All four miRNAs differed significantly between PTC and MNG. ROC analysis revealed strong diagnostic performance, particularly for miR-187-3p (AUC = 0.937) and miR-21 (AUC = 0.914), with their combination achieving an AUC of 0.968. Expression levels correlated with age, tumor stage, surgical status, and thyroid hormones (TSH, ATG, TG), highlighting novel regulatory patterns. This miRNA panel offers promising diagnostic value and insight into PTC pathogenesis, suggesting potential for non-invasive diagnostics and targeted therapies.
Introduction
Cancer has gained global attention as the second leading cause of mortality in emerging countries. According to the International Agency for Research on Cancer (IARC) by 2030, cancer cases are projected to reach 21.7 million, with 13 million deaths. This growing burden is attributed to a combination of genetic, environmental and socioeconomic factors, amplified by globalization and lifestyle changes. 1 Thyroid cancer (TC), the predominant endocrine malignancy, exhibits a favorable prognosis when promptly diagnosed and treated. 2 Papillary, follicular, medullary, and anaplastic constitute the most prevalent histologic subtypes of thyroid cancer, accounting for approximately 80%, 10%, 5%, and less than 2% of cases, respectively. The incidence of thyroid cancer has been on the rise, with an estimated 53,990 new cases projected to be diagnosed in the United States in 2018, reflecting increasing rates spanning from 0.03% to 36%. 3 In Pakistan, thyroid cancer accounts for 2.1% of all malignancies, showing a higher prevalence among females with a female-to-male ratio of 2.6:1. 4 The rise in detected PTC cases is largely attributed to advances in detecting thyroid nodules particularly small asymptomatic ones, which require further evaluation. Fine-needle aspiration biopsy (FNAB), guided by ultrasound, is the global standard for PTC diagnosis, being low-risk, cost-effective, and highly accurate; however, up to 10% of FNAB results are non-diagnostic and provide limited information on tumor characteristics, such as angioinvasion. 5 Thus, identifying additional effective diagnostic biomarkers is essential to complement current thyroid cancer diagnostic tools. MicroRNAs (miRNAs) have recently emerged as promising biomarkers with significant diagnostic potential in this area. 6
MicroRNAs (miRNAs or miRs) are small endogenous molecules, approximately 18–22 nucleotides in length that regulate gene expression at the post-transcriptional level by complementary binding to target mRNAs. They modulate gene expression either through mRNA degradation or by inhibiting translation. 7 MicroRNAs (miRNAs) play critical roles in regulating cellular functions, including proliferation, development and apoptosis. Dysregulated miRNA expression is implicated in various cancers, including the development and progression of PTC. The clinicopathological features of PTC are associated with specific molecular alterations, such as changes in miRNA expression, prompting extensive research into miRNA profiles for their potential as therapeutic targets and prognostic biomarkers in PTC.8–10
The diagnostic application of miRNAs holds significant potential to improve the accuracy of PTC diagnosis, thereby reducing the need for invasive procedures in patient management. 11 For instance, microRNA-21 is among the earliest identified oncomirs shown to be upregulated across various malignancies, including breast cancer, PTC and gastric cancer.12,13 miR-31 exhibits dual roles in cancer, with reduced expression in ovarian, breast, prostate cancers, and PTC, correlating with poor prognosis and survival. In contrast, its overexpression in head, neck, esophageal, lung and colorectal cancers suggests oncogenic properties. This highlights the context-specific behavior of miR-31 in tumorigenesis.14–16 miR-187, located at 18q12.2, has garnered significant attention due to its variable expression across cancers. While it shows upregulated expression in PTC and FTC with correlations to somatic mutations, its role remains controversial in non-small cell lung and stomach cancers. Conversely, miR-187-3p exhibits downregulation in renal, colorectal, lung, and prostate cancers, indicating a tumor-suppressive function.17–19 The Hsa-miR-200 family is pivotal in cancer biology, with miR-200a suppressing renal carcinoma via SIRT1 inhibition and apoptosis induction. Elevated miR-200a-5p in PTC complicates its diagnostic value, while downregulation in nasopharyngeal cancer highlights its diverse roles. Monitoring miRNA expression, including miR-200a, offers potential in evaluating malignancy risks in multinodular goiter.20–23
This study explores the expression and role of key miRNAs (miR-21, miR-31, miR-187-3p, and miR-200a-5p) in papillary thyroid carcinoma (PTC) and multinodular goiter (MNG), addressing critical gaps in thyroid cancer research. It is the first study to systematically assess these miRNAs expression in relation to clinicopathological features and key hormonal markers (TSH, TG, and ATG), offering novel molecular insights into miRNA dysregulation in thyroid disorders. Although individual miRNAs have been studied for their diagnostic relevance, the combined expression profile of these miRNAs remains unexplored. This study for the first time presents a ROC-AUC analysis of these particular miRNAs and their collective diagnostic accuracy, highlighting the potential of a multi-miRNA panel as a superior biomarker for distinguishing thyroid malignancies.
Present study also aimed to evaluate the diagnostic potential and clinical relevance of four microRNAs (miR-21, miR-31, miR-187-3p, and miR-200a-5p) in differentiating papillary thyroid carcinoma (PTC) from multinodular goiter (MNG). Using quantitative real-time PCR (qRT-PCR), we analyzed miRNA expression in fresh thyroid tissue samples from Pakistani patients, and further explored associations with clinical and histopathological characteristics. These findings hold implications for regional and global thyroid cancer research particularly the most common histopathological subtype PTC, contributing to improved risk assessment, early diagnosis and personalized management.
Materials and methods
A retrospective case-control study was conducted based on fresh tissue samples obtained from pathology departments of tertiary care hospitals in Pakistan between 2019 and 2024. Samples were selected through purposive sampling, focusing on cases with complete clinical and histopathological information. The inclusion criteria were limited to patients diagnosed with papillary thyroid carcinoma (PTC) or multinodular goiter (MNG), confirmed through histopathology, and with available clinical and hormonal profiles. Patients with prior chemotherapy, radiotherapy, autoimmune thyroiditis, or insufficient tissue for RNA extraction were excluded.
Clinical and biochemical data (age, gender, tumor stage, and thyroid hormone levels) were collected using a structured data extraction form from hospital records and verified by two independent reviewers. Molecular data were generated using standardized qRT-PCR protocols following total RNA extraction from fresh tissue samples using TRIzol reagent. Fresh tissues were immediately preserved in Falcon tubes containing RNAlater and stored at −20 °C until processing. A total of 100 tissue specimens were analyzed, including 35 PTC tissues, 35 adjacent normal tissues, 15 MNG tissues and 15 adjacent normal tissues. The study design is described in Figure 1 (the flowchart of the study). In accordance with the recommendations of the Research Ethics Committee of PMAS Arid Agriculture University Rawalpindi, as well as from the relevant hospitals in Rawalpindi and Islamabad, Pakistan, informed written consent was obtained from all participants prior to their inclusion in the study. The research protocol was formally reviewed and approved under Ethics Approval Number Approval No. PMAS-AAUR/IEC/232. Furthermore, this study was conducted in full compliance with the ethical principles outlined in the Declaration of Helsinki (World Medical Association), ensuring adherence to international standards for research involving human subjects.
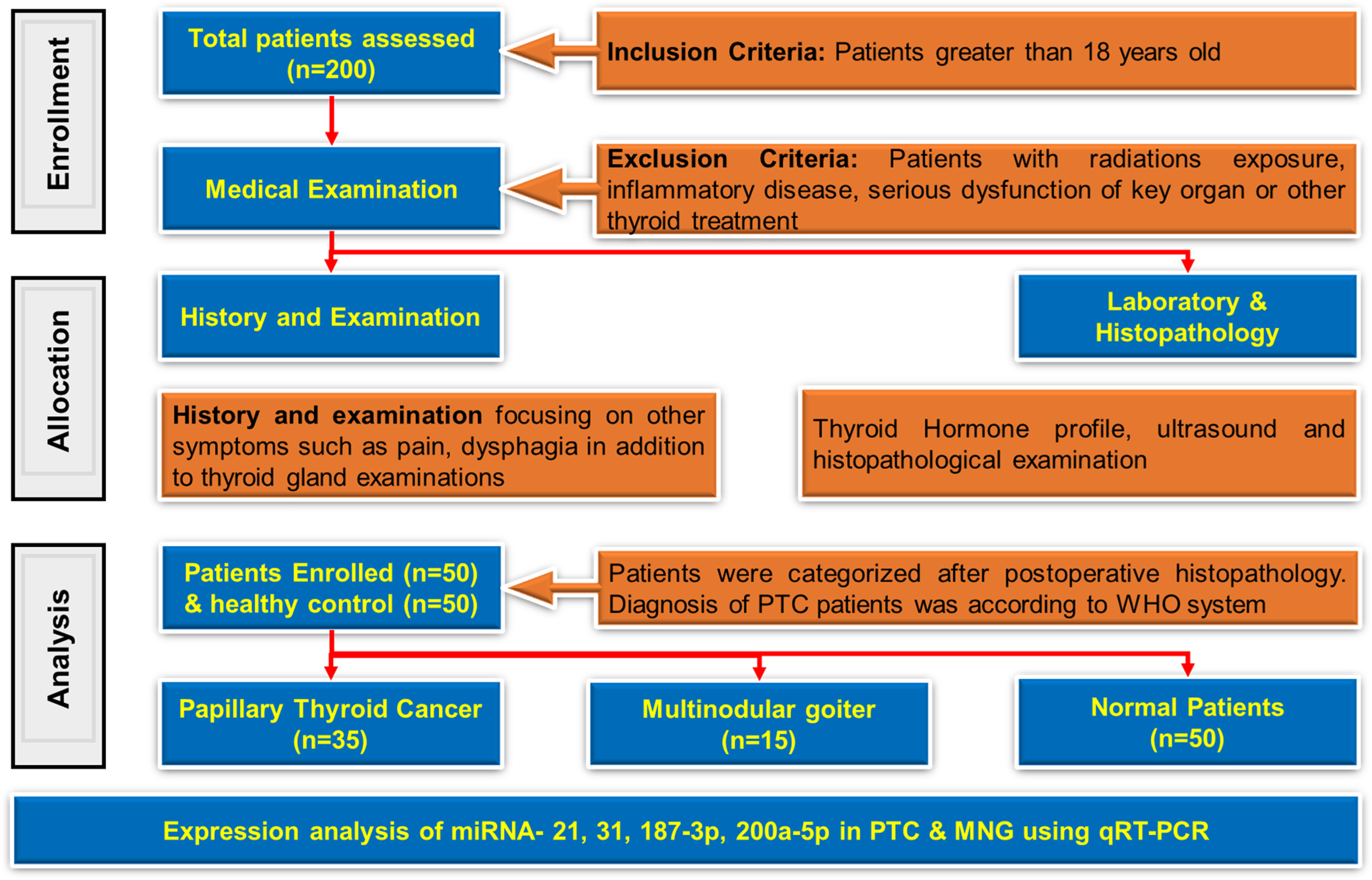
Flow diagram of the present study.
Hematoxylin and eosin staining (H&E) analysis
Four-micrometer tissue sections from PTC sample was prepared, stained with hematoxylin and eosin (H&E), and examined under Olympus CX23 microscope, with images captured via built-in software. The major axis diameter of thyroid follicles was quantified using ImageJ software and pixel measurements were calibrated to millimeters with a 100 mm scale bar.
RNA extraction, quantification & cDNA synthesis
Total RNA was extracted from tissue samples using the TRIzol reagent (Invitrogen, USA). RNA concentration was measured using an RNA nanodrop spectrophotometer (Bio spec-nano Spectrophotometer for life science, SHIMADZU BIOTECH). Complementary DNA (cDNA) was synthesized from the extracted RNA utilizing random hexamer primers, following the manufacturer's protocol provided with the cDNA synthesis kit
Quantitative real-time polymerase chain reaction (qRT-PCR) for the selected miRNA
The miRNAs selected for expression analysis included miR-21, miR-31, miR-187-3p, and miR-200a-5p, with GAPDH serving as the housekeeping gene. The PCR protocol involved an initial denaturation at 95 °C for 10 min, followed by 40 cycles of 95 °C for 15 s, 60 °C for 60 s, and a final step at 95 °C for 15 s. 24
Statistical analysis
The fold change method was employed to evaluate the expression profiles of microRNAs in diseased versus healthy tissue samples. Statistical comparisons between disease and normal samples were conducted using a t-test, with a significance threshold set at p < 0.05. Correlation analysis was applied to examine the association between microRNA expression levels and patient hormonal levels. Diagnostic potential of each microRNA was assessed through Receiver Operating Characteristic (ROC) curves, calculating the Area Under the Curve (AUC). Data analysis was performed using Origin 8 and SPSS 16 software.
Results
Demographic and clinical data
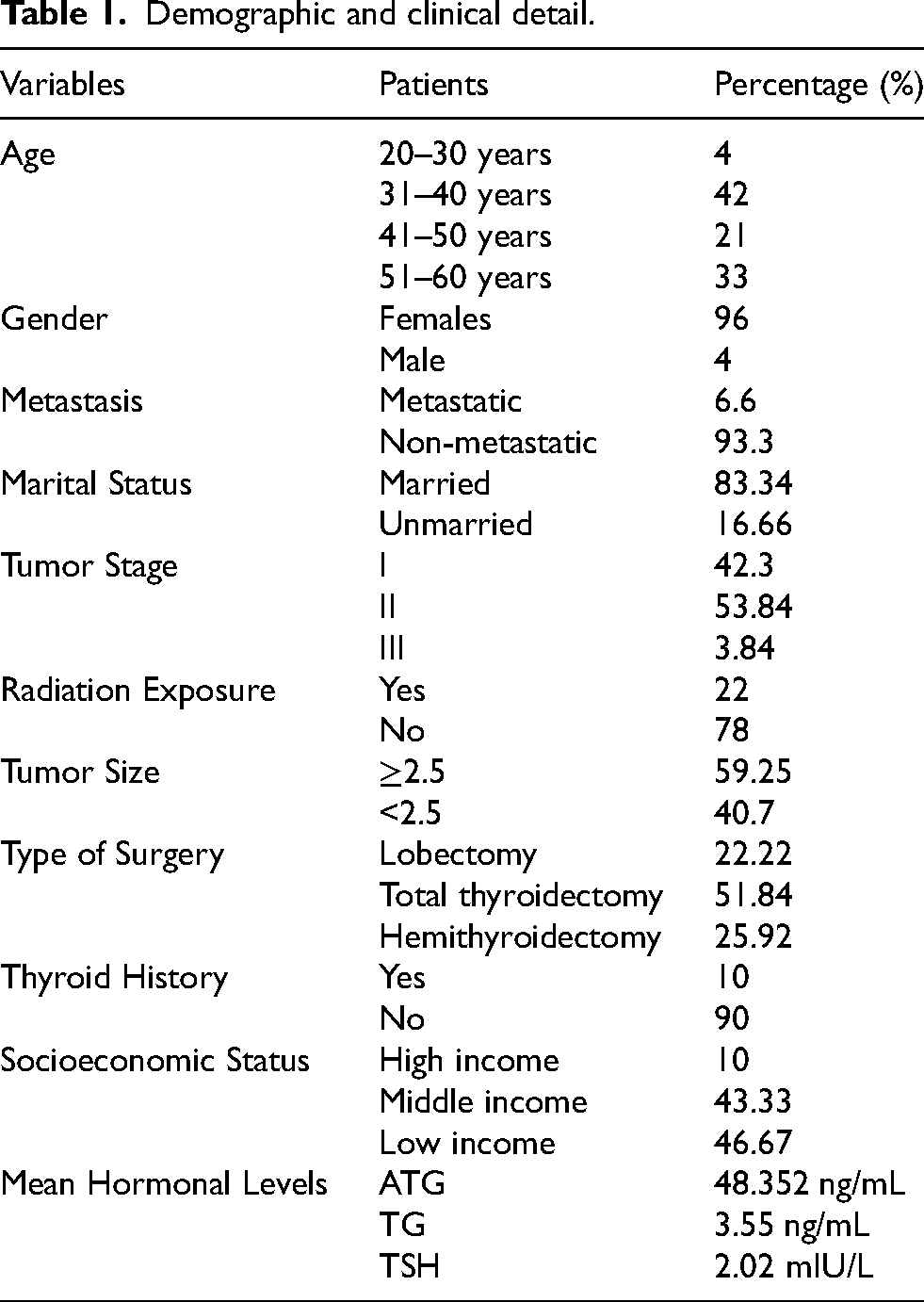
Table 1 presents the demographic and clinical characteristics of the study population, including age distribution, gender, metastasis status, marital status, tumor stage, tumor size, type of surgery, thyroid history, and socioeconomic status. The majority of patients are between 31–40 years old (42%) and predominantly female (96%). Most cases are non-metastatic (93.3%) and married (83.34%). Tumor staging shows that 53.84% are in stage II, while 59.25% have tumors ≥2.5 cm in size. Surgical interventions include lobectomy (22.22%), total thyroidectomy (51.84%), hemithyroidectomy (25.92%). Only 10% of patients have a prior thyroid history and only 22% (among confirmed PTC cases) had some radiation exposure before inclusion into the study. Socioeconomic distribution indicates that 46.67% belong to the low-income group. Mean hormonal levels for ATG, TG, and TSH are 48.352 ng/mL, 3.55 ng/mL, and 2.02 mIU/L, respectively.
Demographic and clinical detail.
Histopathological characterization of papillary thyroid carcinoma using H&E staining
Hematoxylin and Eosin (H&E) staining was performed to microscopically assess the morphology of cancerous cells in PTC tissues as shown in Figure 1Sa (supplemental material). Hematoxylin highlighted the round to oval nuclei in dark blue or purple, revealing characteristic “Orphan Annie eye” nuclei distinctive of PTC as shown in Figure 1Sb (supplemental material), due to intranuclear inclusions and grooves. Eosin counterstained the cytoplasm and extracellular matrix in pink, with cells organized into papillary structures formed by malignant follicular cells surrounding fibrovascular cores.
Comparative analysis of miRNA biomarkers in papillary thyroid carcinoma and multinodular goiter
Expression profile and fold change study of miRNAs in papillary thyroid cancer
Figure 2 presents the expression levels and fold-change analysis of four key miRNAs (miR-21, miR-31, miR-187-3p, and miR-200a-5p) in PTC and normal tissues, as determined by qRT-PCR. All four miRNAs exhibited significant dysregulation in PTC. The mean expression levels of miRNA-21, miRNA-31, miRNA-187-3p, and miRNA-200a-5p were 19.927, 0.1384, 20.167, and 0.2068 respectively, in PTC tissues, compared to 1.867, 2.461, 2.204 and 2.136 in normal samples, as depicted in Figure 2(a). The corresponding average fold change for miRNA-21, miRNA-31, miRNA-187-3p, and miRNA-200a-5p were 32.714, 0.0831, 18.071, and 0.3238 respectively, as shown in Figure 2(b).
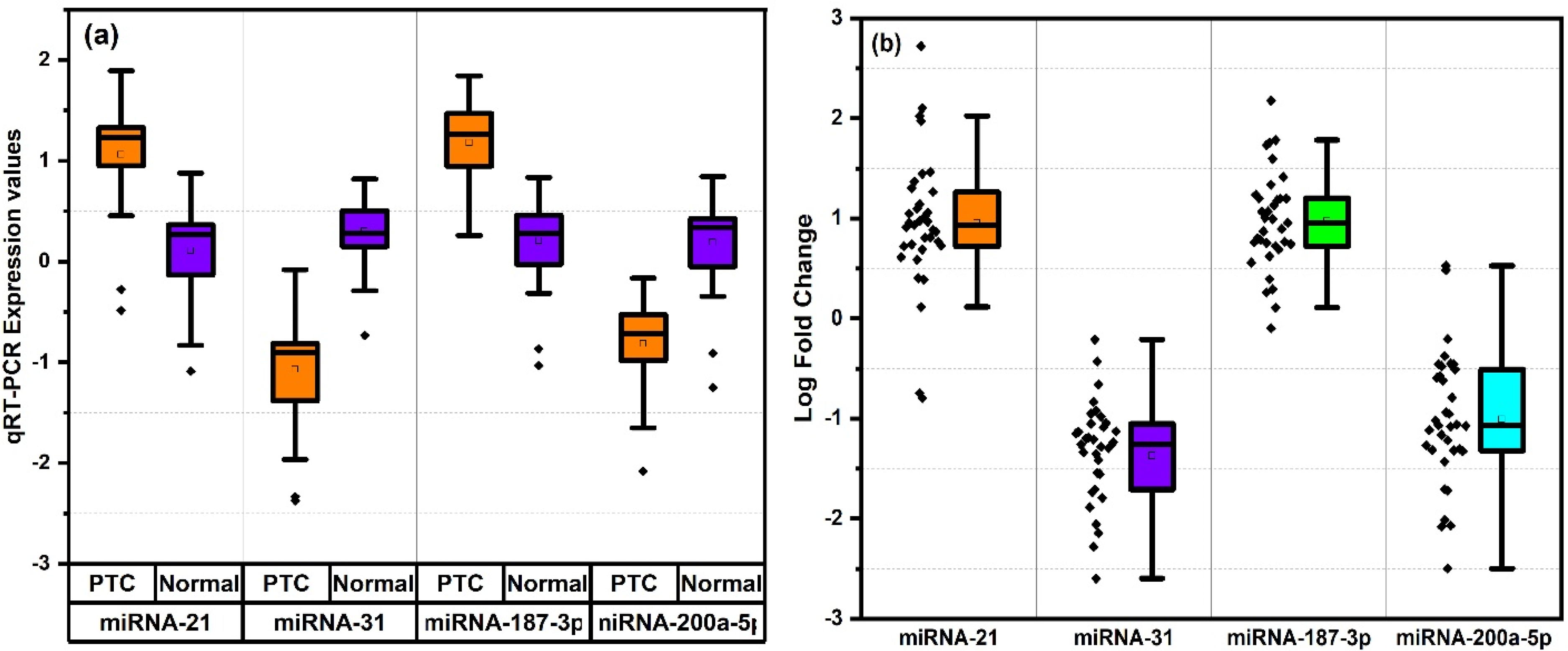
(a) Expression analysis and (b) fold change rise in PTC for miRNA (−21, −31, −187-3p, −200a-5p) with values plotted on y-axis as log scale.
Expression profile and fold change study of miRNAs in multi nodular goiter
Figure 3 illustrates the expression levels and fold-change analysis of miR-21, miR-31, miR-187-3p, and miR-200a-5p in MNG and normal tissues, highlighting significant dysregulation in MNG. The mean expression levels of miR-21, miR-31, miR-187-3p, and miR-200a-5p in MNG tissues were 6.968, 0.203, 5.273, and 3.781, respectively, compared to 1.672, 0.2355, 2.793, and 0.2145 in normal samples (Figure 3(a)). The corresponding average fold changes for miR-21, miR-31, miR-187-3p, and miR-200a-5p were 5.814, 2.731, 2.419, and 4.189, respectively (Figure 3(b)).
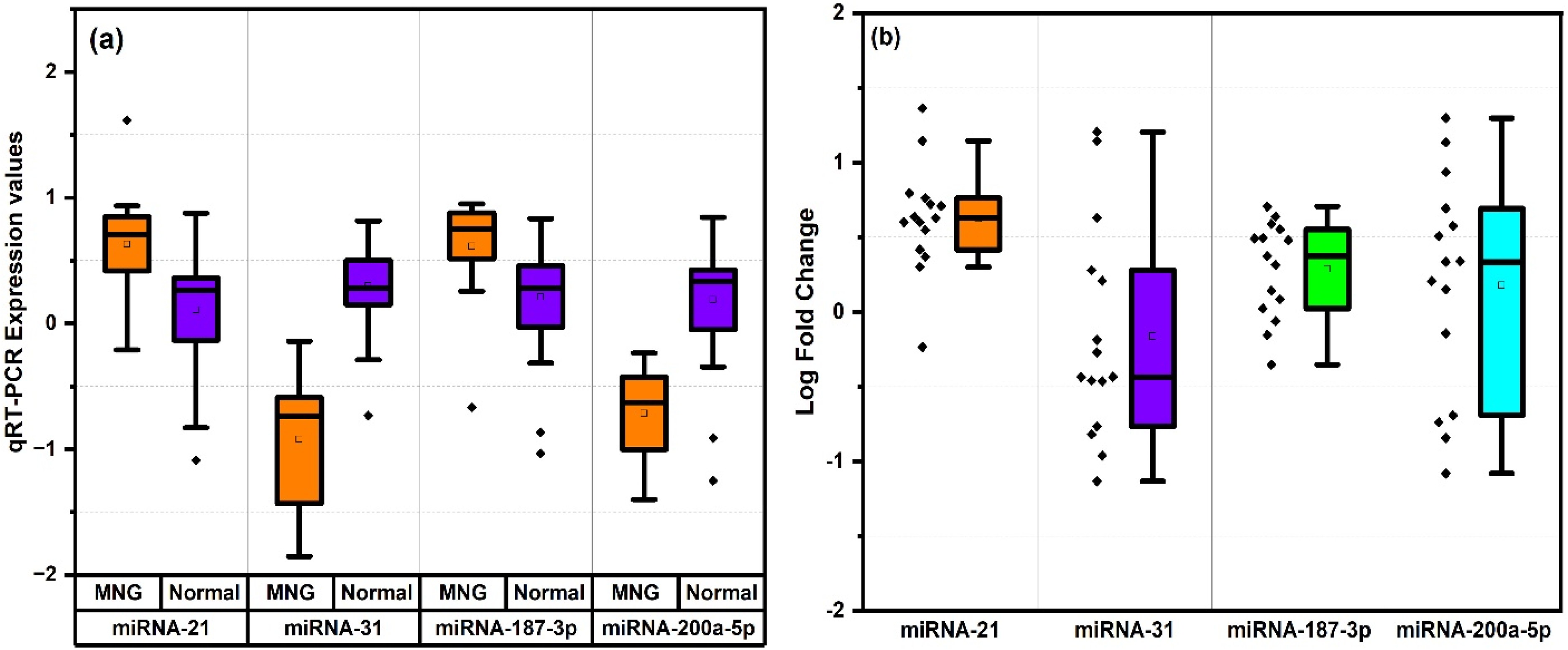
Expression analysis (a) and (b) fold change rise in MNG for miRNA (−21, 31, 187-3p, 200a-5p) with values plotted on y-axis as log scale.
Relative fold change study between PTC and MNG
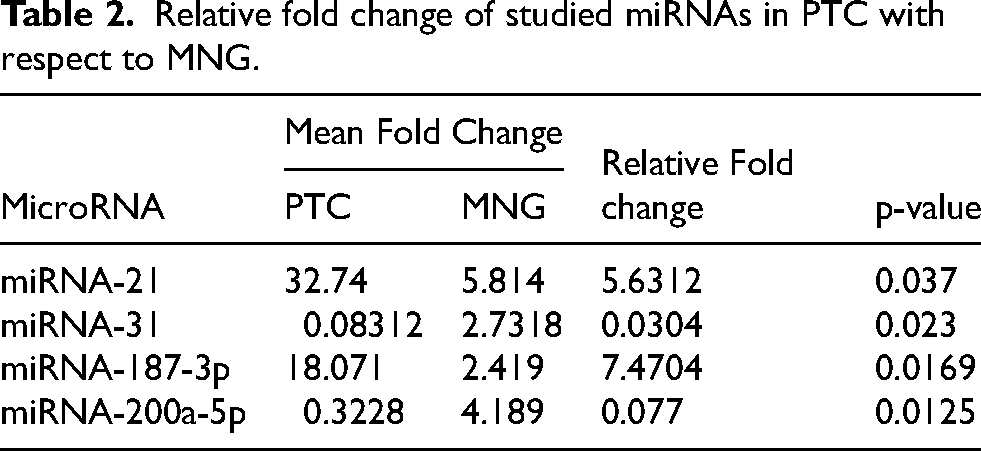
The Table 2 presents the relative fold change of the studied microRNAs in PTC and MNG. The results show that miRNA-21 and miRNA-187-3p are significantly upregulated in PTC, with relative fold changes of 5.63 and 7.47, respectively, and p-values of 0.037 and 0.0169. In contrast, miRNA-31 and miRNA-200a-5p are downregulated in PTC, with relative fold changes of 0.03 and 0.077 and p-values of 0.023 and 0.0125 respectively.
Relative fold change of studied miRNAs in PTC with respect to MNG.
Hormonal profiling in PTC and MNG and its correlation with miRNA biomarkers
The boxplot in Figure 4(a) illustrates the distribution of Anti-Thyroglobulin Antibody (ATG), Thyroglobulin (TG), and Thyroid-Stimulating Hormone (TSH) levels in PTC and MNG patients. ATG levels appear comparable between the two groups (mean ATG 48.532 ng/mL in PTC vs 48.125 ng/mL in MNG). In contrast, TG levels are elevated in PTC (mean, 3.63 ng/mL) compared to MNG (mean, 3.48), with a narrower interquartile range (IQR). TSH levels exhibit a slightly higher mean (2.12 ng/mL) in PTC than MNG (1.928 ng/mL).
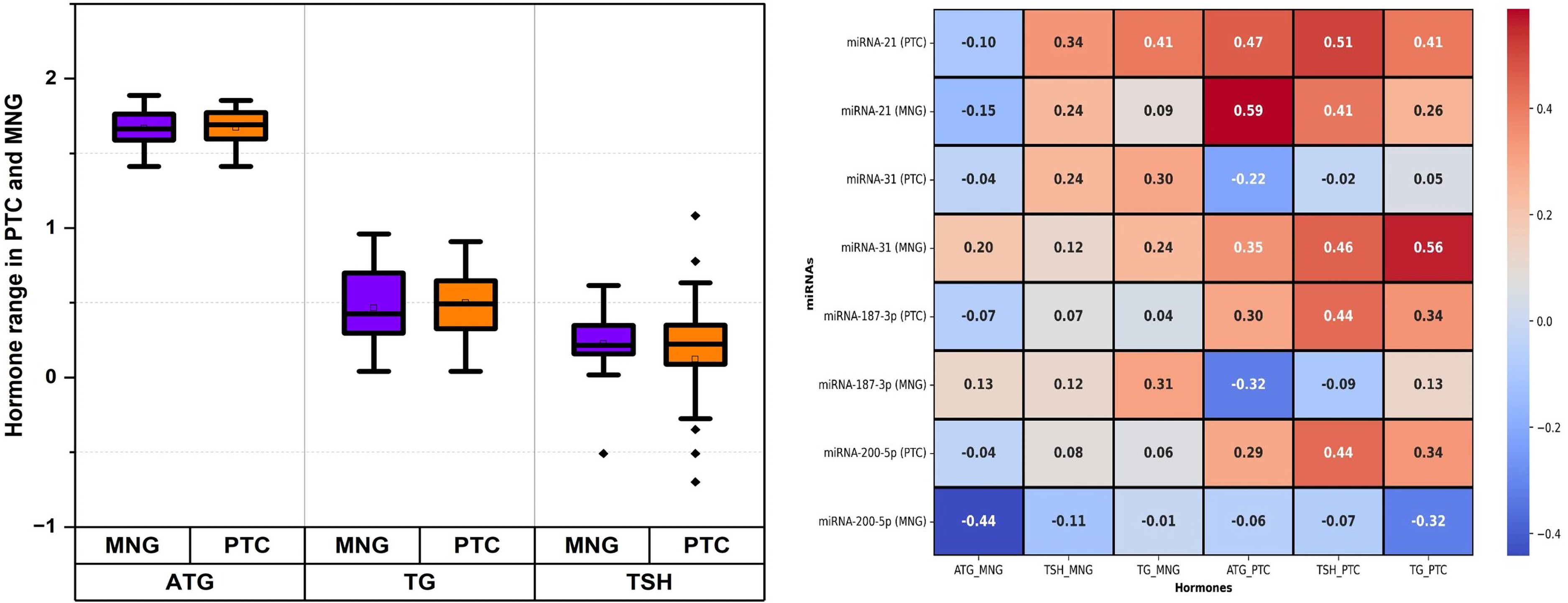
(a) Hormone levels in patients of PTC & MNG with values plotted on y-axis as log scale and (b) correlation of hormones with miRNA-21, −31, −187-3p and −200a-5p in PTC and MNG.
The relationship between miRNA expression levels and TSH, ATG, and TG levels in PTC and MNG patients were also analyzed as depicted in Figure 4(b). For miRNA-21, the correlation coefficients between TSH, TG and ATG were 0.51, 0.41 and 0.47 correspondingly in PTC patients, while the coefficients were 0.24, 0.09 and −0.15 in MNG patients. For miRNA-31, the correlation coefficients between TSH, TG and ATG, were −0.02, 0.05 and −0.22 respectively in PTC patients, while the coefficients were 0.12, 0.24 and 0.2 in MNG patients. In the case of miRNA-187-3p, the correlation coefficients between TSH, TG and ATG were 0.44, 0.34 and 0.3 respectively in PTC patients, while the coefficients were 0.12, 0.31 and 0.13 in MNG patients. For miRNA-200a-5p, the correlation coefficients between TSH, TG and ATG were 0.44, 0.34 and 0.29 respectively in PTC, while the coefficients were −0.11, −0.01 and −0.44 in MNG patients.
Combined panel of miRNA as a diagnostic biomarker of PTC
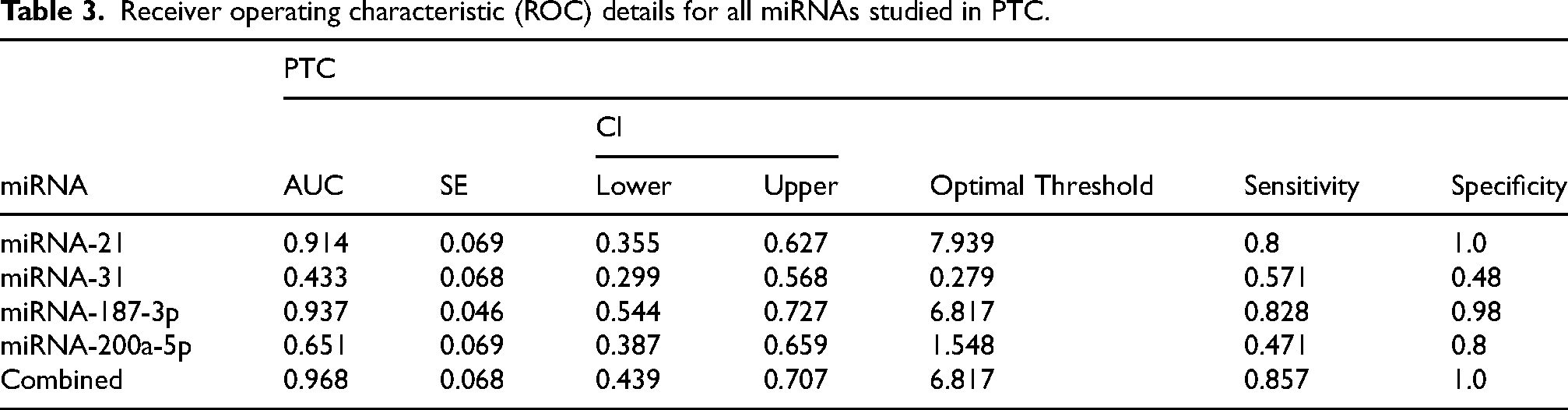
Table 3 and Figure 5 presents the Receiver Operating Characteristic (ROC) analysis for the studied miRNAs in PTC, evaluating their diagnostic performance based on Area Under the Curve (AUC), sensitivity, specificity, and optimal threshold values. Among the individual miRNAs, miRNA-187-3p exhibited the highest diagnostic accuracy with an AUC of 0.937, a sensitivity of 0.828, and a specificity of 0.98. miRNA-21 demonstrated a high AUC of 0.914, with an optimal threshold of 7.939, achieving a sensitivity of 0.8 and perfect specificity (1.0). miRNA-31 showed limited diagnostic utility with an AUC of 0.433. miRNA-200a-5p exhibited moderate diagnostic potential with an AUC of 0.651, a specificity of 0.8 and sensitivity (0.471). While the combined analysis of all miRNAs significantly enhanced the diagnostic performance, achieving the highest AUC of 0.968, sensitivity of 0.857, and specificity of 1.0.
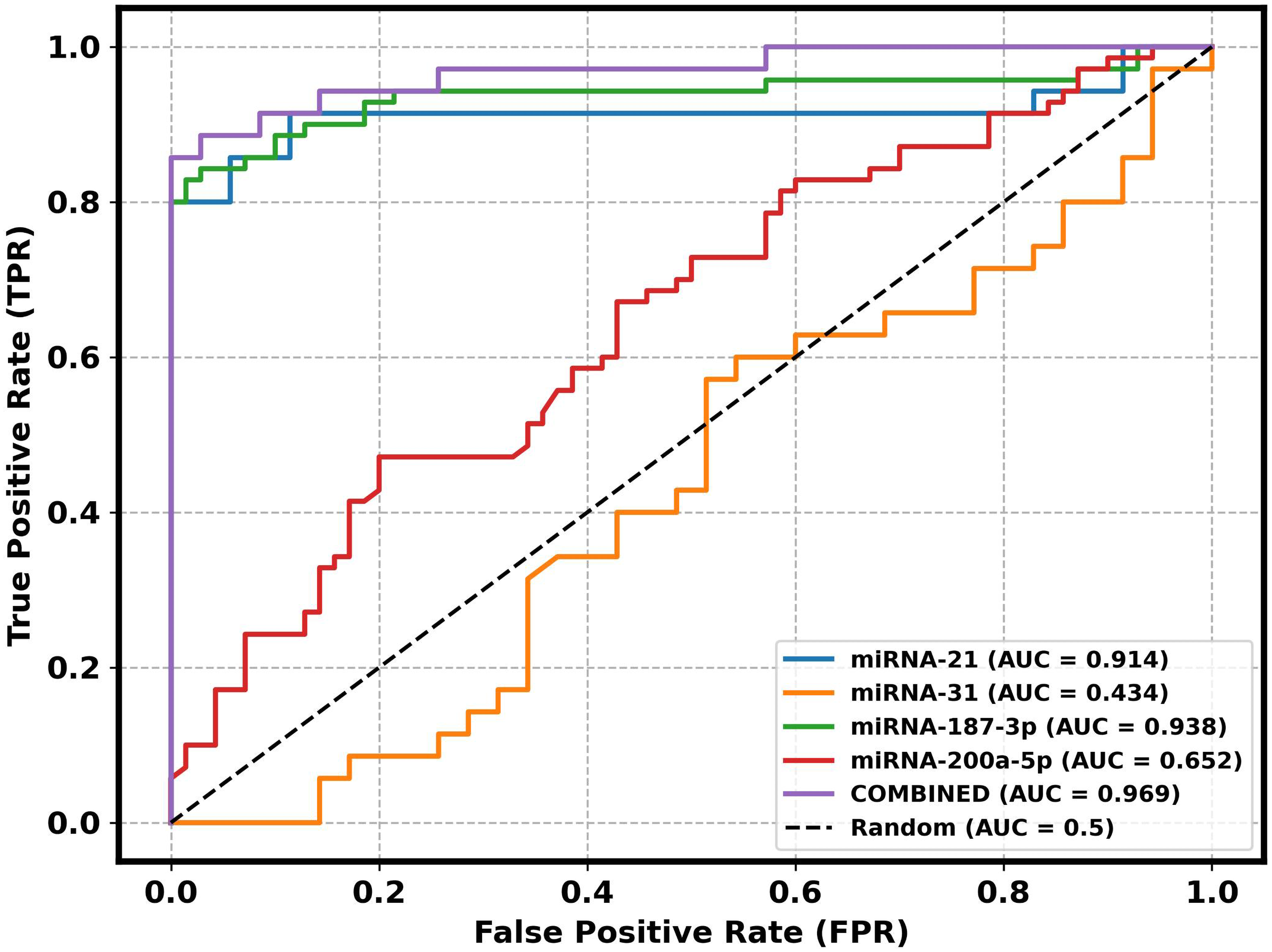
Receiver operating characteristic (ROC) curve for PTC patients of miRNA-21, miRNA-31, miRNA-187-3p, miRNA-200a-5p and their combination.
Receiver operating characteristic (ROC) details for all miRNAs studied in PTC.
Clinical and demographic association of miRNAs dysregulation in papillary thyroid carcinoma
Table 4 presents the mean expression levels of miRNA-21, miRNA-31, miRNA-187-3p, and miRNA-200a-5p in PTC patients compared to normal samples, stratified by demographic and clinical characteristics. Overall, miRNA-21, miRNA-31, miRNA-187-3p, and miRNA-200a-5p were significantly dysregulated in PTC across most subgroups, with a few non-significant differences (n.s.). Age-wise, patients aged 51–60 years exhibited the highest miRNA-21 expression, while miRNA-187-3p was most elevated in the 31–40 age group. Gender analysis showed significantly higher miRNA expression in females than males, except for miRNA-31 and miRNA-200a-5p, which exhibited non-significant differences in males. Metastatic cases had higher miRNA-31 and miRNA-187-3p expression than non-metastatic cases, although the differences in miRNA-31 and miRNA-200a-5p were non-significant. Unmarried patients displayed notably higher miRNA-21 levels than married individuals. Tumor stage III patients exhibited the most pronounced increase in miRNA-21 and miRNA-187-3p expression, though miRNA-31 and miRNA-200a-5p differences were non-significant. Patients with no thyroid history showed generally higher miRNA levels than those with a history. Tumors <2.5 cm showed increased miRNA-21 expression, while larger tumors had higher miRNA-187-3p levels. Among surgical types, total thyroidectomy cases exhibited the highest miRNA-21 and miRNA-187-3p expression.
Mean expression status of studied miRNAs in demographic and clinical characteristics of PTC patients.
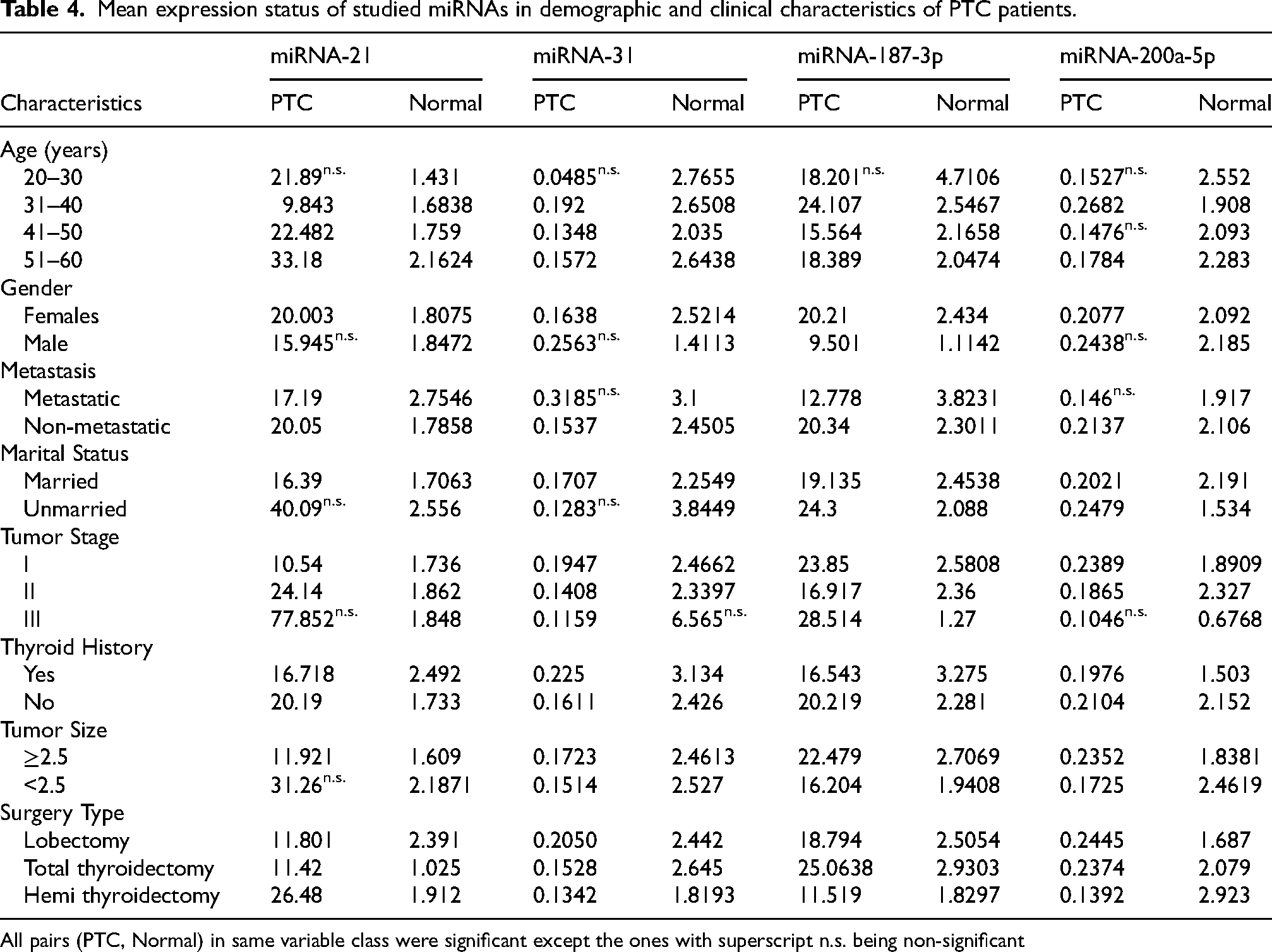
All pairs (PTC, Normal) in same variable class were significant except the ones with superscript n.s. being non-significant
Clinical and demographic association of miRNAs dysregulation in multinodular goiter
miRNA expression levels in MNG were generally higher than in normal samples, though many comparisons were non-significant (n.s.) (Table 5). Age-wise, miRNA-21 and miRNA-31 showed moderate upregulation in the 31–40 age group, while miRNA-187-3p expression peaked in the 41–50 age group. Gender-based analysis was limited, as male-specific data were unavailable. In non-metastatic cases, all four miRNAs exhibited increased expression, though miRNA-21 and miRNA-187-3p differences were non-significant. Marital status influenced miRNA expression, with unmarried patients showing a notable increase in miRNA-21 levels. Tumor stage I and II cases demonstrated mild upregulation of miRNA-31 and miRNA-200a-5p, but these differences were not statistically significant. Patients without thyroid history had slightly higher miRNA expression than those with a history, although these differences were mostly non-significant. Tumor size <2.5 cm was associated with higher miRNA-21 expression, whereas miRNA-187-3p was more elevated in tumors ≥2.5 cm. Among surgical categories, total thyroidectomy cases exhibited the highest miRNA-21 expression, whereas miRNA-31 and miRNA-200a-5p levels varied inconsistently.
Mean expression status of studied miRNAs in demographic and clinical characteristics of MNG patients.
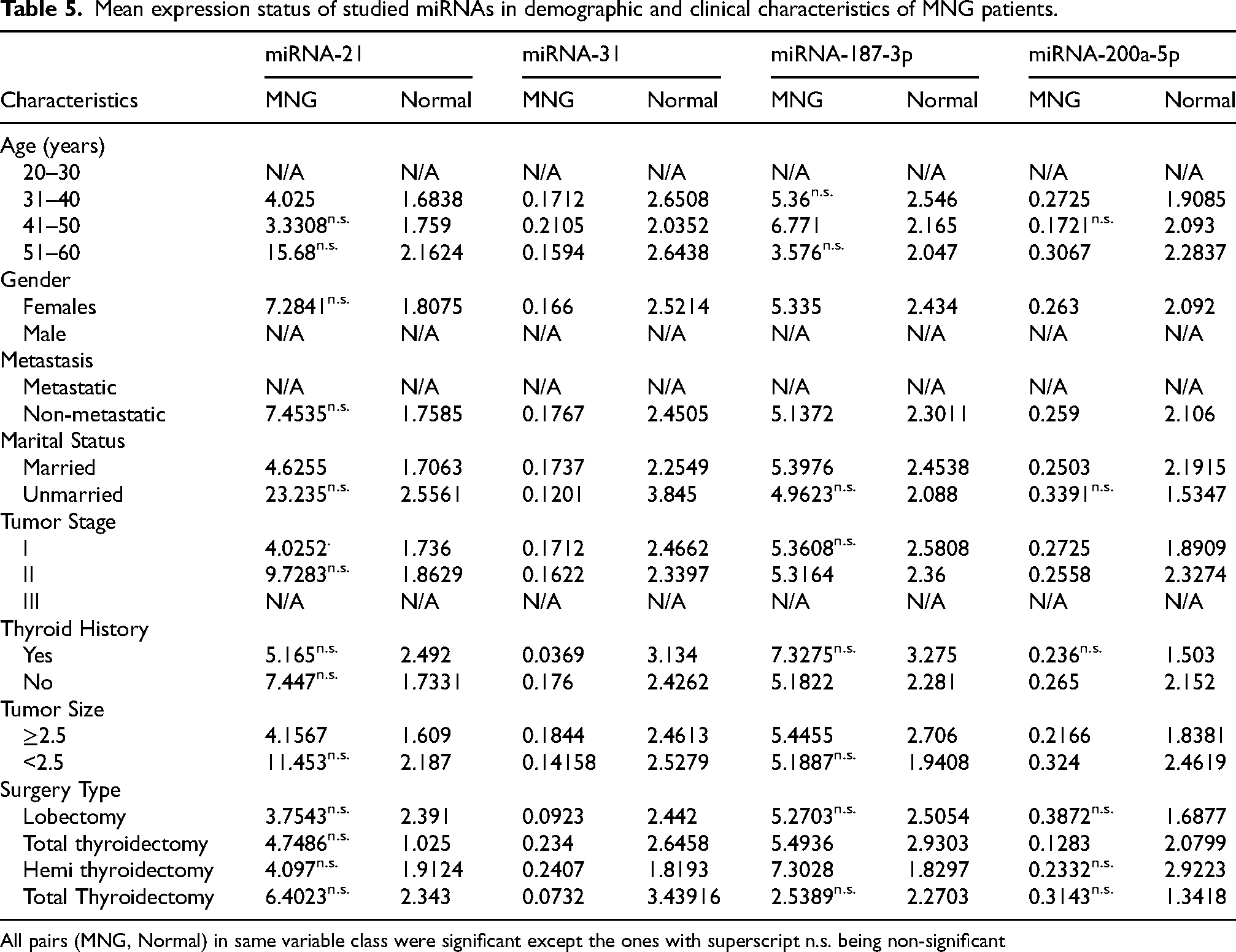
All pairs (MNG, Normal) in same variable class were significant except the ones with superscript n.s. being non-significant
Discussion
Papillary thyroid carcinoma (PTC) generally has a favorable prognosis; however, aggressive variants are associated with poor outcomes. The advancement of PTC therapies is hindered by inadequate diagnostic biomarkers and therapeutic strategies. Fine-needle aspiration biopsy (FNAB), despite its high accuracy and cost-effectiveness, presents several complications including hematoma, pain, ecchymosis, and inadvertent punctures to vital structures. Moreover, approximately 10% of FNAB results are nondiagnostic, highlighting the urgent need for novel screening and diagnostic markers. 25
Blood testing of thyroid hormones, including TSH (Thyroid-Stimulating Hormone), ATG (Anti-Thyroglobulin Antibodies), and TG (Thyroglobulin), serves as an initial indicator of thyroid pathology. Recent studies have associated TSH values greater than 0.9 IU/mL with higher risk of PTC malignancy, 26 while serum TG has been utilized as a monitoring strategy for low-risk PTC patients. 27 ATG levels are valuable for monitoring differentiated thyroid cancer patients, particularly those with undetectable or rising TG levels despite treatment. Present research work has also considered these hormones for relevancy with PTC.
In present study, mean hormonal values between PTC and MNG patients showed minimal differences: TSH (PTC: 2.12 mIU/L, MNG: 1.92 mIU/L), ATG (PTC: 48.53 mIU/L, MNG: 48.12 mIU/L), and TG (PTC: 3.63 mIU/L, MNG: 3.48 mIU/L). These values differ from those reported by kaleem Khan 28 in northern Pakistan, likely due to population diversity and methodological variations. Despite the association of TSH with increased PTC risk, its specificity is limited due to fluctuations in benign thyroid conditions. Similarly, ATG's diagnostic accuracy is hindered by its presence in autoimmune thyroid diseases like Hashimoto's thyroiditis. TG, while crucial for detecting PTC recurrence and metastasis post-thyroidectomy, has compromised reliability when ATG antibodies interfere with its measurement. 29 The overlapping hormonal profiles between malignant and benign cases in our study underscore the limitations of using hormones alone as definitive biomarkers for PTC.
Growing evidence has demonstrated the pivotal role of miRNAs in thyroid cancer pathogenesis. Current study investigated the expression patterns of four previously reported miRNAs (miR-21, miR-31, miR-187-3p, and miR-200a-5p) in Pakistani patients with PTC and MNG. We observed distinct miRNA expression profiles in these conditions compared to normal tissues, highlighting their potential diagnostic utility. In PTC tissues, miR-21 and miR-187-3p were significantly upregulated, while miR-31 and miR-200a-5p were markedly downregulated. MNG tissues also exhibited dysregulation of these miRNAs, albeit with less pronounced differences compared to PTC.
miR-21, commonly overexpressed in human malignancies, influences PI3K/Akt and MAPK/ERK pathways, inhibiting apoptosis while promoting cell migration and proliferation in thyroid cancer cells. The findings of present work corroborate previous studies by Kondrotienė et al. 10 and Pamedytyte et al. 30 who reported elevated miR-21 expression in PTC compared to normal tissues. In our cohort, miR-21 expression was substantially higher in PTC (19.927) compared to normal tissues (1.867) and MNG (5.631-fold difference). Enhanced miR-21 expression was particularly evident in patients aged 51-60 years (33.18), unmarried individuals (40.09), those with stage III tumors (77.85), and patients who underwent total thyroidectomy (11.42). ROC analysis yielded an AUC of 0.914, indicating excellent diagnostic potential for distinguishing PTC from normal tissues, surpassing the AUC values of 0.83 and 0.774 reported by Park et al. 31 and Kondrotienė et al. 32 respectively.
miR-187-3p has emerged as a cancer-related miRNA with variable expression across tumor types. 17 High levels of miR-187-3p in tumors were correlated with poor prognosis and higher levels of peripheral inflammation-related blood markers. 33 Ng et al. 33 associated overexpression of miR-187-3p in colorectal cancer (CRC) cell lines with impaired colony formation, cell migration and invasion, and induced chemosensitivity. Our study revealed elevated miR-187-3p levels in PTC tissues (20.167) compared to normal tissues (2.204), particularly in patients aged 31-40 years (24.10), females (20.21), metastatic cases (12.77), stage III tumors (28.51), tumors >2.5 cm (22.47), and patients who underwent total thyroidectomy (25.06). These findings align with Celakovsky et al. 34 who reported miR-187 upregulation in PTC compared to adjacent normal thyroid tissue. The diagnostic utility of miR-187-3p in our study (AUC = 0.937) surpassed its performance in hepatocellular carcinoma (AUC = 0.84) as reported by previous researchers, 35 underscoring its potential as a robust biomarker for PTC.
Our study demonstrated significant downregulation of miR-31 in PTC tissues (0.138) compared to normal tissues (2.461), consistent with findings of Wang et al. 16 and its tumor suppressor role in bladder cancer (BCa) was also linked to BCa progression and a poor prognosis. 36 In studied cohort, this downregulation was most pronounced in patients aged 41-50 years (0.134), those with stage III tumors (0.115), no thyroid history (0.161), tumor size <2.5 cm (0.151), and patients who underwent hemithyroidectomy (0.134). The fold change of 0.030 relative to MNG further highlights its potential as a discriminatory marker. The AUC value for miR-31 in our study (0.43) was lower than previously reported values by Rogucki et al. 37 (0.58) and Lu et al. 38 (0.661), suggesting that its diagnostic utility may be context-dependent and warrants further investigation in larger cohorts.
miR-200a-5p regulates epithelial-to-mesenchymal transition (EMT) and functions as both a tumor suppressor and pro-metastatic factor in various epithelial cancers. 39 Contrary to report of Wang et al. 20 of miR-200a-5p upregulation in PTC, current study observed decreased expression (0.206) compared to normal tissues (0.323). This downregulation was particularly evident in patients aged 51-60 years (0.178), married individuals (2.2), those with thyroid history (0.197), tumor size <2.5 cm (0.172), and patients who underwent hemithyroidectomy (0.139). The diagnostic performance of miR-200a-5p (AUC = 0.651) was moderate. Although downregulation of miR-200a/b was suggested as candidate biomarker of central pelvic recurrence by Nilsen et al., 40 more comprehensive studies are needed to elucidate its specific role in PTC pathogenesis across different populations.
Using a panel of miRNAs improves diagnostic accuracy by integrating multiple biomarkers, reducing the influence of variability in any single miRNA. Panels also provide greater robustness and reproducibility, making them more reliable for clinical application than individual miRNAs. 37 Multiple miRNA panels have been proposed as diagnostic biomarkers for papillary thyroid carcinoma (PTC), with varying degrees of diagnostic performance depending on the selected miRNAs, sample type, and study population. For instance, a four-miRNA panel comprising miR-152-3p, miR-221-3p, miR-551b-3p, and miR-7-5p demonstrated a combined AUC of 0.841 for distinguishing PTC from benign thyroid lesions in a previous study. 37 Similarly, a plasma-based three-miRNA panel including miR-346, miR-10a-5p, and miR-34a-5p achieved an AUC of 0.877 when differentiating PTC from nodular goiter. 41 Notably, a serum-derived panel of miR-25-3p, miR-296-5p, and miR-92a-3p reached an AUC of 0.969, indicating very high discriminatory power. 42 In another study, a three panel (hsa-miR-301a-3p, hsa-miR-195-5p in plasm) was used to evaluate the diagnostic potential for discriminating PTC from healthy patients, obtaining an AUC of 0.978. 43 In comparison as mentioned in Table S1, our tissue-based four-miRNA panel (miR-21, miR-31, miR-187-3p, and miR-200a-5p) achieved a combined AUC of 0.968, which is comparable to the performance of several previously reported panels. The strong diagnostic capability of our panel may be attributed to the selection of miRNAs representing distinct molecular pathways implicated in PTC tumorigenesis, including epithelial-mesenchymal transition, inflammatory signaling, and thyroid hormone regulation. Furthermore, the high AUC observed in our cohort underscores the potential of this panel to serve as a reliable adjunct diagnostic tool, particularly in regions where access to commercial molecular assays is limited. These findings align with and, in some cases, surpass the diagnostic performance of previously published miRNA panels, reinforcing the potential utility of multi-marker strategies over single-marker approaches in improving diagnostic accuracy for PTC.
Our study pioneers the investigation of correlations between miRNA expression and thyroid hormone levels in PTC and MNG. The differential correlation patterns observed between these conditions provide novel insights into the regulatory mechanisms underlying thyroid pathology. In MNG, miR-21 showed a strong positive correlation with TSH (r = 0.59), while this correlation was moderate in PTC (r = 0.47), suggesting a more pronounced role of TSH in benign conditions. Similarly, miR-31 exhibited a stronger positive correlation with TG in MNG (r = 0.56) compared to PTC (r = 0.30), implying that TG may more substantially impact miR-31 regulation in non-malignant conditions. miR-187-3p displayed a moderate positive correlation with TG in MNG (r = 0.30) but a negative correlation in PTC (r = −0.32), suggesting a potential shift in TG-miRNA interactions during malignant transformation. Notably, miR-200a-5p demonstrated a significant negative correlation with ATG in MNG (r = −0.44), a trend absents in PTC, highlighting potential immune-related differences between benign and malignant thyroid conditions. Although no direct correlation between these specific miRNAs and TSH, TG, or ATG has been reported in the literature, dysregulated miRNA expression is known to play a crucial role in hormone synthesis and regulation.44,45
The translational potential of these miRNAs extends beyond diagnostics. miR-21's central role in PI3K/Akt signaling positions it as a therapeutic target, with antisense oligonucleotides (ASOs) already in trials for other cancers. Similarly, miR-187-3p inhibitors could sensitize advanced PTC to radioiodine therapy, while miR-31 mimics might restore chemosensitivity in metastatic disease. However, challenges remain, including delivery efficiency and off-target effects. Furthermore, population-specific differences, such as the higher baseline TSH levels in Pakistani cohorts compared to other studies, emphasize the need for regionally tailored reference ranges. The discordance between this study's miR-200a-5p findings (downregulated) and Wang et al.'s report of upregulation may reflect ethnic variability in miRNA expression or differences in tumor microenvironment composition, necessitating multiethnic validation. Although present study suggests probable role of the miRNA considered in this work, one limitation of the present study is the lack of sub-classification of PTC variants due to incomplete histopathological annotation of fresh tissue samples. Future studies should aim to incorporate variant-specific analyses to refine miRNA biomarker associations. Furthermore, correlation analysis between miRNA expression and more specific histopathological features such as capsular invasion and lymphovascular invasion might also be considered.
Conclusions
This study provides novel insights into the molecular landscape of papillary thyroid carcinoma (PTC) and multinodular goiter (MNG) by quantifying dysregulated miRNA expression in the Pakistani population. The integration of miRNA profiling into thyroid nodule evaluation demonstrated a diagnostic accuracy of AUC = 0.968 for combined panel of miR-21, miR-31, miR-187-3p and miR-200a-5p
Future perspectives and recommendations
This study validates a four-miRNA panel with high diagnostic accuracy for PTC, offering greater accuracy than single-miRNA approaches, particularly for indeterminate thyroid nodules. Multi-center studies with larger, diverse cohorts are needed for external validation, along with long term follow up and longitudinal monitoring to assess treatment response and recurrence. The distinct, context-dependent miRNA–hormone interactions observed in PTC and MNG warrant further mechanistic investigation. Advancing these findings toward clinical application could enable personalized, molecularly guided strategies that improve diagnostic precision and patient outcomes in thyroid oncology.
Supplemental Material
sj-docx-1-cbm-10.1177_18758592251392827 - Supplemental material for Characterization of a novel four-miRNA signature in papillary thyroid carcinoma: Integrating molecular profiling, hormonal regulation, and diagnostic implications in populations with rising PTC incidence
Supplemental material, sj-docx-1-cbm-10.1177_18758592251392827 for Characterization of a novel four-miRNA signature in papillary thyroid carcinoma: Integrating molecular profiling, hormonal regulation, and diagnostic implications in populations with rising PTC incidence by Afshan Afzal, Nafeesa Kainat, Aayesha Riaz, Afia Malik, Azhar Ejaz, Yasmeen Sher, Zaineb Tariq, Kainat Abbasi, Rashida Khan and Ruqia Mehmood Baig in Cancer Biomarkers
Footnotes
Ethical approval statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Institutional Review Board of PMAS-Arid Agriculture University, Rawalpindi (Approval No. PMAS-AAUR/IEC/232).
Informed consent statement
This study does not involve any identifiable patient data; therefore, informed consent was not required.
Author contribution statement
Conceptualization, AA and RMB; Formal analysis, NK, SAA and RMB; Methodology, AA and NK; Resources, RK and YS; Software, YS; Supervision, RMB; Validation, RK and SAA; Visualization, YS; Writing – original draft, AA; Writing – review & editing, AR, AE, and RMB.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All the data is available within the manuscript. No additional file was prepared.
Supplemental material
Supplemental material for this article is available online.
