Abstract
Background
We previously utilized pretreatment tumor markers Carcinoembryonic Antigen (CEA) and Cancer Antigen 125 (CA125) for predicting lymph node metastasis (LNM) in endometrioid endometrial cancer (EC).
Objective
The aim of this study was to externally validate a nomogram developed in our previous single-center retrospective study.
Methods
A multi-center validation study was conducted to recruit endometrioid EC patients from four branches of Chang Gung Memorial Hospital between 2009 and 2021, with patients participating in the original research being excluded. The previously established nomogram was applied with optimal cut-off values for CEA 1.4 ng/ml and CA125 40 U/mL identified through receiver operating characteristic (ROC) curves. The concordance index (C-index) was calculated to assess discrimination, and comparative negative predictive value (NPV) and negative likelihood ratio (NLR) were determined. Decision curve analysis (DCA) was plotted to evaluate our predictive model's clinical utility and net benefit.
Results
Overall, 1271 patients were included in this external validation study. The results demonstrated a C-index of 0.727, indicating moderate discrimination ability of the nomogram in predicting LNM in this independent cohort. Comparative analysis of NPV 97.2% and NLR 0.36 revealed performance metrics consistent with the original study, reinforcing the nomogram's potential clinical utility in ruling out the possibility of LNM if both pretreatment CEA and CA125 were less than 1.4 ng/ml and 40 U/mL, respectively. The DCA indicated that the nomogram provided clinical utility.
Conclusion
The reproducible performance metrics in the independent large sample cohort underscore the robustness and generalizability of utilizing CEA and CA125 as predictors of LNM in endometrioid EC, suggesting its potential as a simple tool for clinicians in preoperative decision-making regarding lymphadenectomy.
Keywords
Introduction
As the most prevalent gynecologic malignancy globally, 1 the incidence of endometrial cancer (EC) continues to rise but varies according to factors such as age, obesity, hormonal factors, and geographic location. In developed countries with a higher life expectancy and a higher prevalence of obesity, the incidence tends to be higher. 2 While the majority of cases are diagnosed at an early stage, a subset of patients presents with lymph node metastasis (LNM), signifying an advanced disease state associated with a poorer prognosis. Accurately identifying LNM is pivotal in tailoring treatment strategies and improving overall outcomes. Moreover, identifying patients at negligible risk for LNM allows for the avoidance of pelvic lymphadenectomy, potentially averting post-operative complications such as lymphocele formation or lymphedema.
Efforts to refine risk stratification and optimize surgical decision-making have led to the exploration of various predictive models.3–7 In this context, tumor markers such as Carcinoembryonic Antigen (CEA) and Cancer Antigen 125 (CA125) have emerged as potential tools for identifying patients at a significant risk of LNM. These markers, easily measured in routine clinical practice, may offer valuable insights into disease aggressiveness and guide preoperative planning. Our previous single-center study addressed this need by developing a nomogram incorporating CEA and CA125 to predict LNM, specifically in endometrioid EC, the most common histological subtype. 8 We found that the risk of LNM for those with both markers below cut-off (CEA < 1.4 ng/ml, CA125 < 40 U/mL) was low, with a negative predictive value (NPV) of 96.6%. The nomogram constructed from these findings exhibited good discrimination and reliable calibration (concordance index 0.779) through our internal validation. This encouraging finding prompted the need for further external independent validation studies. By extending our investigation to diverse patient populations and healthcare settings, this study aimed to confirm the reliability of the nomogram and ensure its applicability beyond the initial study cohort.
Materials and methods
Patient selection and data collection
The development cohort consisted of 405 patients who underwent pelvic lymphadenectomy with or without para-aortic lymphadenectomy for endometrioid EC at Kaohsiung Chang Gung Memorial Hospital (CGMH) between 2015 and 2022. 8 Within this cohort, 11.9% of patients exhibited LNM. Notably, among the five clinical variables assessed, only pretreatment levels of CEA and CA125 emerged as significant predictors of LNM. Tumor grade was not included as a clinical variable because our database relies on hysterectomy specimens. Curettage specimens, which may show discrepancies with post-surgical findings, were not considered reliable for use as a preoperative predictor. 9
For the validation cohort, data were collected from the Chang Gung Research Database (CGRD) spanning from January 2009 to December 2021 for analyses. The CGRD combines original medical records from all four prominent medical institutes across Taiwan: Keelung CGMH, Linkou CGMH, Chiayi CGMH, and Kaohsiung CGMH. Notably, these four institutions, as per the National Cancer Registry, collectively manage approximately 20% of EC cases in Taiwan. The CGRD provides comprehensive patient information, encompassing cancer registry data such as the date of cancer diagnosis, laboratory results, disease stage, pathological findings, treatment modalities, dates of recurrence, and mortality events. Extensive details about the database are available in a separate study. 10 To ensure independence between the development and validation cohorts, patients from Kaohsiung CGMH between 2015 and 2021 were excluded, preventing any overlap of study cases that could bias validation. Consequently, our validation cohort exclusively comprised endometrioid EC patients with pretreatment CEA and CA125 data from the four CGMH institutes, establishing a robust and distinct dataset for external validation. We defined LNM as having at least one positive lymph node, whether pelvic or para-aortic. Differences in lymphadenectomy or sampling between centers were difficult to assess due to the lack of specific codes for “sampling” in our raw data. This study was approved by the Institutional Review Board of CGMH (IRB No.202400193B0) in February 2024. Informed consent was waived due to the retrospective nature of this study.
Statistical analysis
The identical methodology and models were directly applied to the external independent cohort without undergoing retraining. A rigorous assessment of comparability between the two sets was conducted. Frequency distributions among categorical variables were analyzed using the chi-squared test, while comparisons of median/mean values and ROC curves were carried out using the two-sample t-test and DeLong test, respectively. Multiple logistic regression analysis was used to determine independent variables predicting LNM. It is crucial to note that the construction of the nomogram relied on data from the training set, and the validation set's data were subsequently utilized for the nomogram's validation. The nomogram's predictive accuracy was assessed using the Area Under the Curve (AUC) of the Receiver Operating Characteristic (ROC) curve and the Concordance Index (C-index) in both the training and validation sets. The alignment between actual outcomes and predicted probabilities was evaluated through calibration curves and the sensitivity of the model was determined using the Hosmer–Lemeshow goodness-of-fit test. The clinical usefulness of the nomogram was assessed using the decision curve analysis (DCA). The sensitivity, specificity, positive predictive value (PPV), positive likelihood ratio (PLR), NPV, and negative likelihood ratio (NLR) to estimate LNM were calculated. Data management and analysis were performed using SPSS (software version 22, IBM, Armonk, NY, USA) and SAS (software version 9.4, Cary, NC, USA). A p-value less than 0.05 was considered to be statistically significant.
Results
Demographic characteristics
Figure 1 shows the enrollment flowchart, detailing the number of patients at each institute. Initially, 2974 patients newly diagnosed with EC during the study period were included. Subsequently, patients treated at Kaohsiung CGMH between 2015 and 2021 were excluded. Further refinement involved the exclusion of non-endometrioid histology and those without lymphadenectomy, followed by the exclusion of patients lacking pretreatment CEA and CA125 evaluations. Ultimately, a qualified cohort of 1271 patients was retained for comprehensive inclusion and analysis. The comparison of baseline characteristics between the training and external validation cohorts of the studied patients is listed in Table 1. Of the entire cohort, the distribution of age, BMI (Body Mass Index), FIGO (International Federation of Gynecology and Obstetrics) stage, and CA125 level were similar between the two cohorts, while the percentage of the subjects with LNM and CEA < 1.4 ng/ml rates were significantly lower in the external validation cohort (11.9% vs. 7.4% and 65% vs. 57%, respectively).
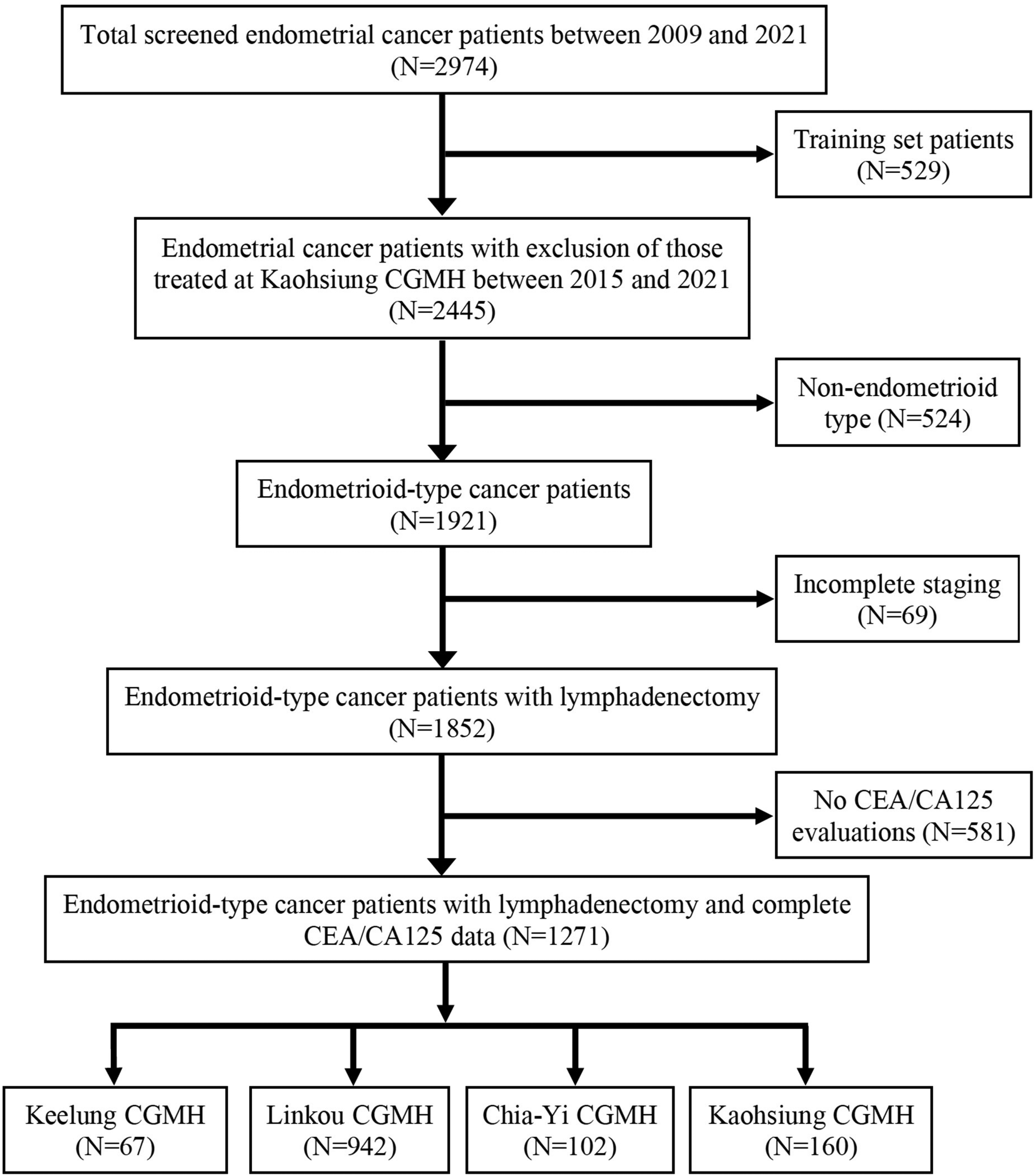
Consort flow diagram.
Comparison of clinicopathological characteristics between training and external validation cohort.
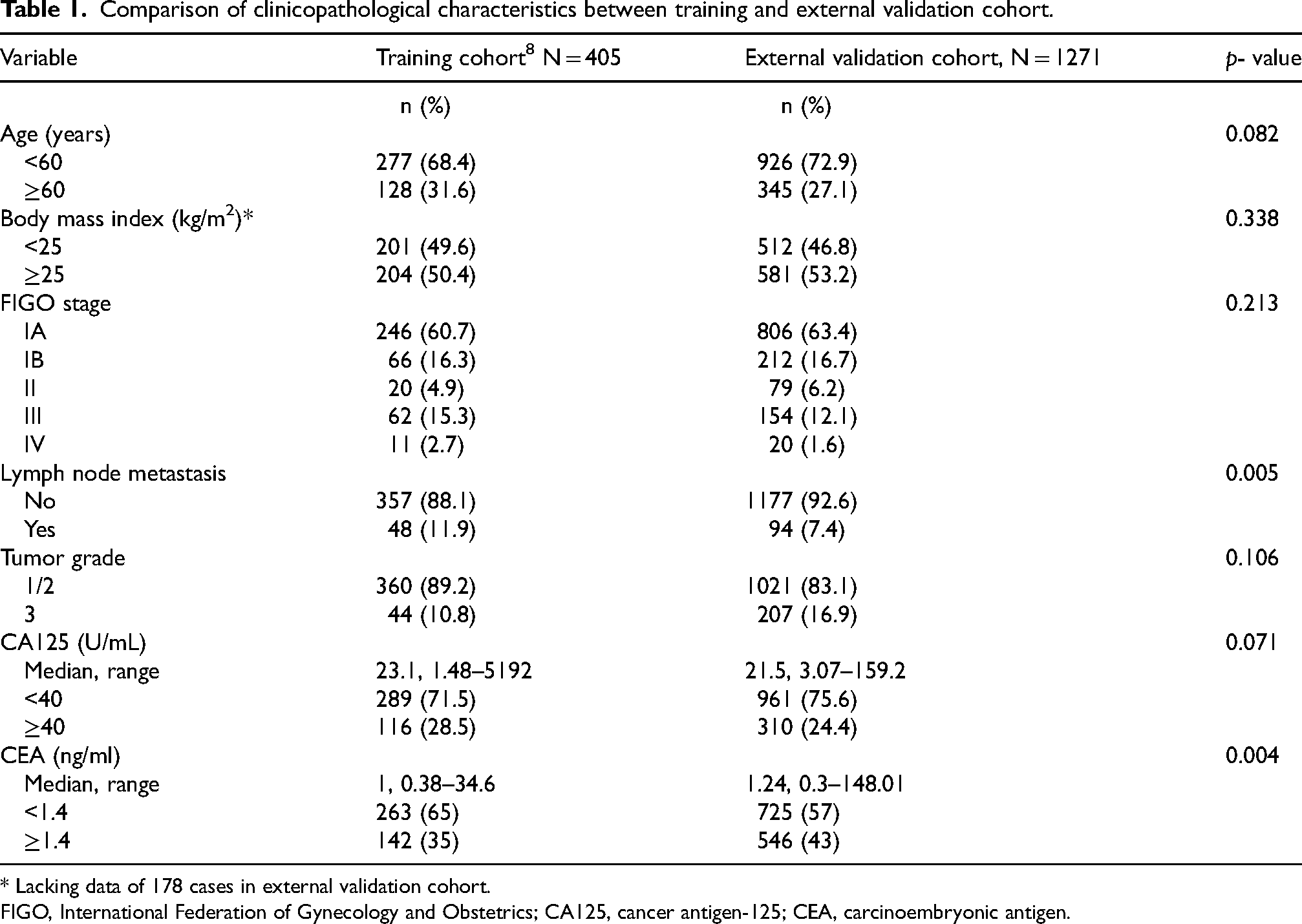
* Lacking data of 178 cases in external validation cohort.
FIGO, International Federation of Gynecology and Obstetrics; CA125, cancer antigen-125; CEA, carcinoembryonic antigen.
Logistic regression analysis
Results of univariate and multivariate logistic regression analyses in both the training and external validation cohorts are shown in Table 2. Both CEA and CA125 remained significant independent variables for predicting LNM, with odds ratio and 95% confidence interval of 2.02 (1.27–3.21) and 5.48 (3.44–8.73), respectively.
Univariate and multivariate logistic regression analyses of clinical risk factors in endometrioid-type endometrial cancer for predicting lymph node metastasis.

BMI, body mass index; CA125, cancer antigen-125; CEA, carcinoembryonic antigen; CI, confidence interval; OR, odds ratio.
Nomogram's predictive accuracy
The initial C-index for the nomogram developed in our previous study after internal validation was 0.779 (95% CI: 0.709–0.848) (Supplementary Figure 1). Upon external validation with present data, the C-index was 0.727 (0.674–0.781) [Figure 2A]. DeLong's test showed no significant difference between the two ROC curves (p = 0.251). The calibration plots and results from the Hosmer-Lemeshow goodness of fit tests are shown in Figure 2 B showing that the data fit the model reasonably well in both cohorts (p = 0.915).
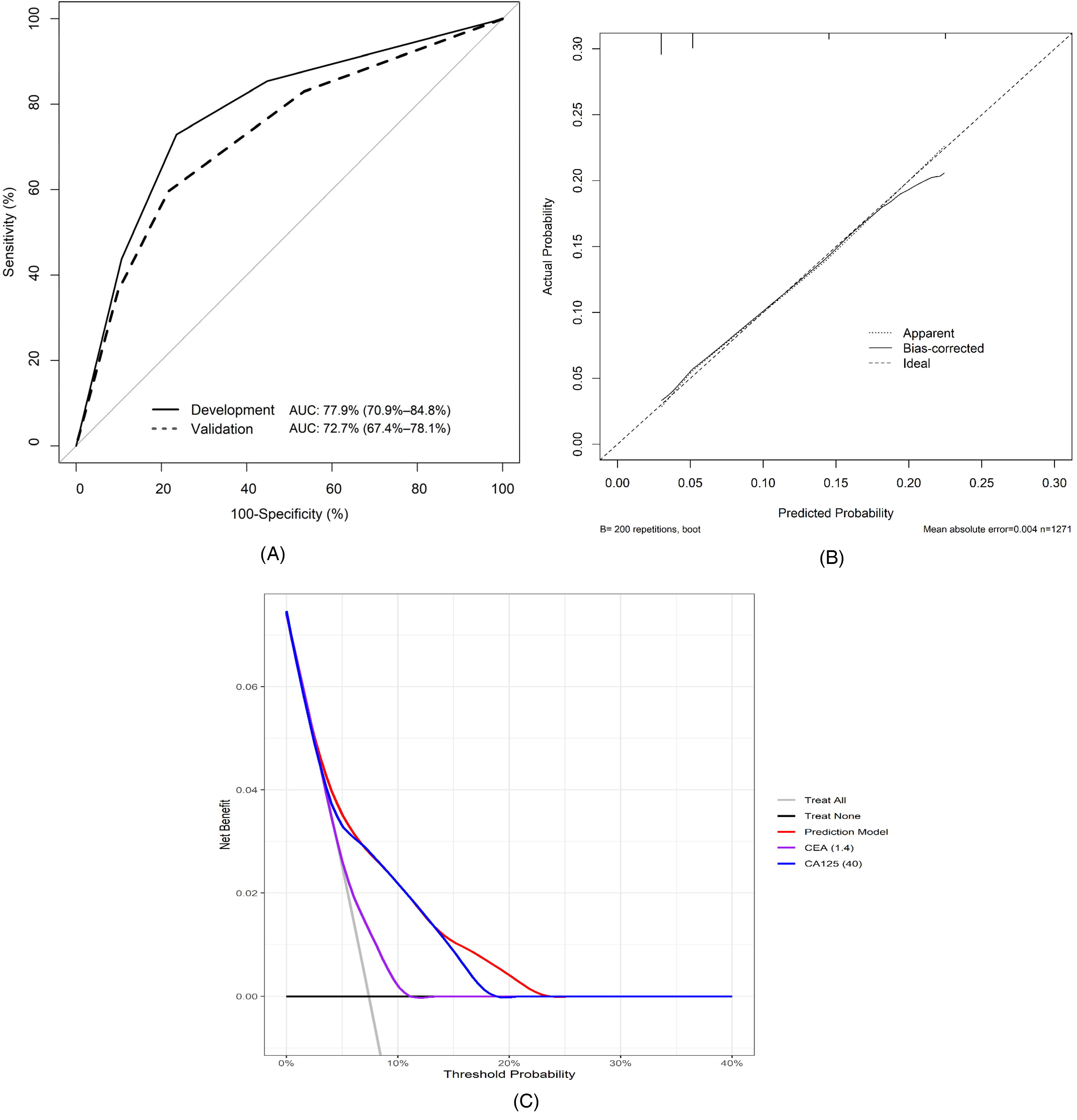
(A) Comparison of area under ROC curves between developmental and external validation cohorts. (B) The calibration plot demonstrates satisfactory concordance with reality, with a mean absolute error of 0.004 and a concordance index of 0.727. (C) Decision curve analysis of the prediction model using the external validation cohort indicates a net benefit when the threshold probability ranges from 3% to 23%.
Nomogram performance characteristics for training and external validation data are shown in Table 3. The DCA of the external validation group was presented in Figure 2C, which showed net benefit when the threshold probability was between 3% and 23%. In this external validation cohort, both the CEA and CA125 below cut-off levels represented about 44% of our study cases. The sensitivity and specificity were 83.0% and 46.6%, respectively. The NPV was 97.2% and NLR was 0.36 (95% CI, 0.23–0.57), with moderate evidence to rule out the possibility of LNM. Comparative analysis revealed performance metrics consistent with those in the original study, reinforcing the nomogram's potential clinical utility in ruling out the possibility of LNM.
Comparison of predictive performance between training and external validation cohort.
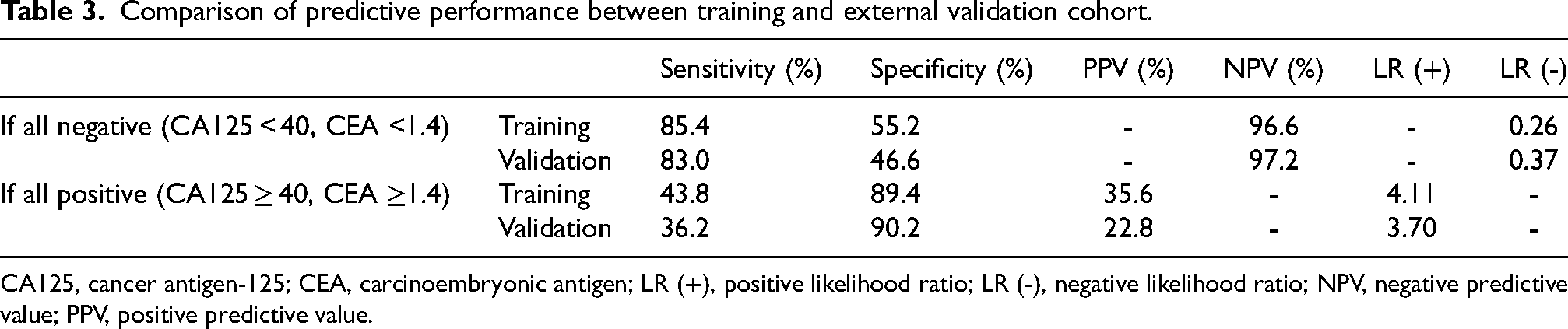
CA125, cancer antigen-125; CEA, carcinoembryonic antigen; LR (+), positive likelihood ratio; LR (-), negative likelihood ratio; NPV, negative predictive value; PPV, positive predictive value.
Discussion
Our study aimed to validate the utility of a nomogram incorporating pretreatment CEA and CA125 levels in predicting LNM among endometrioid EC patients. Our primary findings indicate promising outcomes for the external validation of the nomogram across a multi-center cohort. In our external validation, a total of 1271 patients were recruited from four branches of CGMH and a moderate but consistent C-index of 0.727 was obtained. This indicates the nomogram's ability to discriminate between patients with or without LNM, affirming its reliability across different patient populations and healthcare settings. Additionally, the results of the comparative analysis of NPV and NLR further confirm the nomogram's potential as a valuable tool for ruling out the possibility of LNM in endometrioid EC patients. By leveraging data from multiple institutions, the applicability of the predictive model was enhanced and potential biases associated with single-center studies were mitigated.
Several studies have explored the utility of pre-treatment CA125 levels for predicting LNM in EC. Our team first showed that CA125 levels above 40 U/mL correlate with retroperitoneal LNM, although having a sensitivity and specificity of 77.8% and 81.0%, respectively, its NPV was only 94.4%. 11 By enhancing its predictive accuracy, Kang et al. combined CA125 with specific MRI criteria to identify EC patients at low LNM risk, achieving a NPV close to 99%. 12 However, MRI's cost limits its use in lower-resource settings. Other researchers have paired CA125 with immunohistochemical markers like hormone receptors to predict LNM, yielding good results.13,14 Yet, the lack of standardized methods for interpreting these markers leads to variability in results, making them less practical for routine clinical use. In contrast, the measurement of serum tumor markers like CEA and CA125 is more standardized, making them accessible worldwide.
The inclusion of pretreatment CEA and CA125 levels as predictive factors in the nomogram yields significant clinical implications. These tumor markers, readily accessible in routine practice, offer valuable insights into the likelihood of LNM to facilitate informed decision-making regarding surgical management and adjuvant therapy. Caution is warranted as multiple immunoassay methods are available for measuring serum levels of CEA and CA125. Although there are international standardizations for tumor marker measurement to ensure accuracy, precision, comparability, and clinical utility,15,16 the absence of strict assay methodologies for tumor marker measurement contributes to variability in assay results across different laboratories. While enzyme-linked immunosorbent assay (ELISA) is widely utilized, radioimmunoassay (RIA) and chemiluminescent immunoassay (CLIA) are also commonly accepted in clinical and research settings. Subtle differences in accuracy may exist between ELISA, RIA, and CLIA methods. Previous studies demonstrated that CLIA exhibited superior precision and accuracy compared to RIA and ELISA.17,18 Consequently, the optimal cut-off values of CEA and CA125 for estimating LNM should be determined individually for each laboratory to ensure reliable and consistent results.
For a considerable duration, there has been a lack of consensus regarding the necessity of tumor marker evaluation in patients with EC. 19 While some clinicians advocate for the routine measurement of tumor markers such as CEA and CA125 as part of the diagnostic workup and disease monitoring, others question their clinical utility due to the limited sensitivity and specificity of these markers, as well as the absence of standardized cut-off values and international guidelines for their interpretation. This is evidenced by the lack of substantial CEA and CA125 evaluations within our study cohort. Additionally, the heterogeneity of EC subtypes and individual patient characteristics further complicates the utility of tumor markers in clinical practice. As a result, the decision to evaluate tumor markers in EC patients remains a subject of ongoing debate, highlighting the need for further research and consensus-building on this issue. In our previous study, mesenchymal tumors were excluded since both CEA and CA125 are considered epithelial markers and are primarily expressed in various adenocarcinomas. Patients with type II epithelial carcinoma were also excluded due to their higher risk of LNM, as prediction models for avoiding lymphadenectomy have not been deemed feasible for this group of patients in several prior studies.20–23 Through these exclusions, we successfully determined optimal cut-off values for predicting LNM in unique endometrioid-type EC patients. In this validation cohort, these novel cut-off values for CEA and CA125 were further validated. These values differ from their conventional reference values, specifically for preoperative prediction of LNM in endometrioid EC patients. Our findings provide compelling evidence supporting the inclusion of CEA and CA125 assessment in the initial diagnostic workup following EC diagnosis, offering valuable insights for clinical decision-making.
We believe that our prediction model could provide a clear enhancement and transformative application in clinical practice in several ways. First, improved decision-making: By confirming the accuracy of the CEA and CA125 thresholds (1.4 ng/ml and 40 U/mL, respectively) for predicting LNM, our study provides clinicians with a simple and effective tool for preoperative decision-making. This can help in identifying patients who are at low risk for LNM, allowing for more personalized treatment plans. Second, reduction of unnecessary procedures: The study highlights the model's ability to rule out the possibility of LNM with high NPV, reducing the need for unnecessary lymphadenectomy in patients who are unlikely to benefit from it. This can minimize surgical risks and postoperative complications, improving patient outcomes. Third, cost-effectiveness: By potentially reducing the number of unnecessary surgeries, the predictive model can contribute to cost savings in healthcare, making it a cost-effective approach for managing EC. Fourth, integration into clinical practices: The performance metrics of the nomogram support its integration into clinical protocols for EC management. This can lead to standardized practices across institutions, improving the quality of care.
Despite the strengths of our study, including large sample size and multi-center validation, several limitations should be acknowledged. Firstly, the retrospective study nature introduces inherent biases and limitations in data collection, potentially affecting the accuracy and completeness of clinical information. Additionally, the reliance on retrospective data led to the fact that CEA and CA125 values of a substantial proportion of patients were unavailable, which could introduce selection bias to affect the generalizability of our findings. Furthermore, the use of different commercialized CLIA systems across decades may have introduced variability in the measurement of CEA and CA125 levels, potentially influencing the accuracy of our predictive model. Lastly, the lack of external validation beyond the CGMH system limits the transferability of our findings to broader patient populations.
Conclusion
Our results emphasize the value of incorporating pretreatment CEA and CA125 levels into a nomogram to rule out the possibility of LNM in endometrioid EC. The nomogram's performance in external validation reaffirms its potential as a clinically valuable tool for risk assessment and treatment decision-making in this patient population. Further prospective validation with the inclusion of data from different geographic regions and healthcare systems and integration into clinical practice is warranted to fully ensure the benefits of this predictive model.
Footnotes
Acknowledgments
All authors extend their gratitude to the Biostatistics Center of Kaohsiung Chang Gung Memorial Hospital for their invaluable assistance with the statistical analysis.
Ethics approval and consent to participants
This study was conducted under the Declaration of Helsinki and approved by the Institutional Review Board of Chang Gung Memorial Hospital (approval number: 202400193B0). Patient consent was waived due to the retrospective nature of the study.
Author contributions
Conception: Y.C.O. and H.L.
Interpretation or analysis of data: S.Y.H, S.C.W. and H.C.F.
Preparation of the manuscript: H.L.and C.H.W.
Revision for important intellectual content: All authors reviewed the manuscript for important intellectual content and provided final approval of the manuscript.
Supervision: H.C.F., C.H.W., and Y.C.O.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Chang Gung Memorial Hospital Research Project Grant (CORPG8M0391, 0401, 0411, 0421, CORPG8N0381).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
