Abstract
BACKGROUND:
There is an urgent need for early detection of lung cancer. Screening with low-dose computed tomography (LDCT) is now implemented in the US. Supplementary use of a lung cancer biomarker with high specificity is desirable.
OBJECTIVE:
To assess the diagnostic properties of a biomarker panel consisting of cytokeratin 19 fragment (CYFRA 21-1), carcinoembryonic antigen (CEA) and cancer antigen 125 (CA125).
METHODS:
A cohort of 250 high-risk patients was investigated on suspicion of lung cancer. Ahead of diagnostic work-up, blood samples taken. Cross-validated prediction models were computed to assess lung cancer detection properties.
RESULTS:
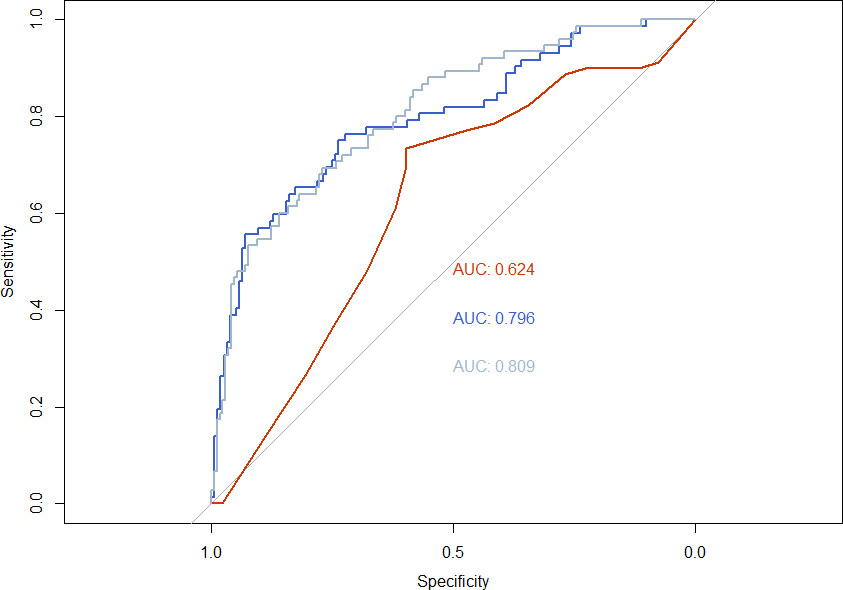
In total 32% (79/250) of patients were diagnosed with lung cancer. Area under the curve (AUC) for the three biomarkers was of 0.795, with sensitivity/specificity of 57%/93% and negative predictive value of 83%. When combining the biomarkers with US screening criteria, the AUC was 0.809, while applying only US screening criteria on the cohort, yielded an AUC of 0.62. The ability of the biomarkers to detect stage I-II lung cancer was substantially lower; AUC 0.54.
CONCLUSIONS:
In a high-risk cohort, the detection properties of the three biomarkers were acceptable compared to current LDCT screening criteria. However, the ability to detect early stage lung cancer was low.
Keywords
Introduction
Lung cancer remains the leading cause of cancer-related death worldwide [1]. The global incidence of lung cancer continues to increase due to aging, world population growth and smoking [2]. Lung cancer stage at diagnosis has a high impact on prognosis [3] and significant efforts to increase the detection of early disease and reduce mortality have been undertaken [4].
Following the results of the US National Lung Screening Trial (NLST) [5], the United States Preventive Services Task Force (USPSTF) has recommended annual low-dose computed tomography (LDCT) for people at high risk of lung cancer [6]. In 2021 the USPSTF changed the screening criteria and now recommends annual LDCT in adults aged 50 to 80 years who have a 20 pack-year smoking history and currently smoke or have quit smoking within the past 15 years [7]. European lung cancer screening trials have also been conducted [8, 9, 10, 11], and several European countries are currently in the process of implementing LDCT lung cancer screening [12].
Although LDCT screening proved beneficial in the NLST with reduced overall mortality, the largest European screening trial (NELSON) [10] has only found reduced lung cancer-specific mortality. Furthermore, issues such as overdiagnosis, false positives, nodule management and complications cannot be ignored [13]. Therefore, it has been suggested to add a blood biomarker panel to either screening criteria or as part of the screening result to improve discrimination between malignant or benign indeterminate nodules [14].
Currently, several types of cancer biomarkers are being investigated [15]. The Scottish ECLS trial [16] used a positive autoantibody as part of the inclusion criteria of a two-year biannually LDCT screening trial program. The intervention resulted in a significant diagnostic stage shift. However, the control group received usual clinical care, and thus, the incremental contribution of the autoantibody test to LDCT screening cannot be assessed. Other potential lung cancer biomarkers that are yet to be tested in conjunction with LDCT screening include several types of protein panels and circulating tumor DNA (ctDNA) [15].
In this study, the potential utilization of a 3-biomarker panel consisting of cytokeratin 19 fragment (CYFRA 21-1), carcinoembryonic antigen (CEA) and cancer antigen 125 (CA125) was tested in a high-risk cohort. These three biomarkers have been tested previously in conjunction with surfactant protein B [19], showing promises results regarding optimization of inclusion criteria in LDCT. However, surfactant protein B is not commercially available and, hence, the study aimed to assess the diagnostic properties without surfactant protein B. The primary aim of the study was to assess the ability of the biomarker panel to detect lung cancer at any stage. The secondary aims included assessing the detection of stage I-II lung cancer using the biomarker panel, evaluating the performance of the US LDCT screening criteria if applied on the cohort and the performance of a combination of the biomarker panel with current US LDCT screening criteria or tobacco pack years.
Materials and methods
Inclusion and exclusion
At the Department of Internal Medicine, Lillebaelt Hospital Vejle, Denmark, a total of 250 patients were consecutively included in the study. They were all referred from their family doctor on suspicion for lung cancer, aged 18 years or above and able to participate in diagnostic procedures. Patients with a previous cancer diagnosis (except non-melanoma skin cancer) were excluded from the study. Lung cancer diagnosis and staging were obtained using the IASLC 8
Biomarker analysis
Prior to diagnostic work-up, blood samples were taken in BD Vacutainer
Statistics
Normally distributed values are presented as means and standard deviation, while non-normally distributed values are described as median and IQR. Categorical values are presented as frequencies and percentages. Cross-validated logistic regression prediction models were computed using K-fold cross-validation technique (5-fold) in R, Caret package [18]. Results are presented as receiver operating characteristics (ROC) curves with area under the curve (AUC) and confidence intervals (CI).
Ethical considerations
The study was observational and diagnostic work-up or treatment of a potential lung cancer were not influenced by participation in the study. The study was approved by the Danish Data Protection Agency (18/33058) and the Regional Committee on Health Research Ethics in Southern Denmark (S-20180052). All participants gave informed consent to participate. The study conforms with The Code of Ethics of the World Medical Association (Declaration of Helsinki), printed in the British Medical Journal (18 July 1964).
Results
Lung cancer diagnosis
Participants’ characteristics
Participants’ characteristics
Presented as frequencies (%), mean (standard deviation) or median [IQR].
Of the 250 included patients, 79 (32%) were diagnosed with lung cancer. Background information on patients is given in Table 1. Age and sex distribution did not differ substantially between groups while lung cancer patients more often were smokers compared to controls (
Median concentrations of the markers in lung cancer patients vs. controls were: CA125: 10 kU/L vs. 6 kU/L; CEA 1837:
The lung cancer detection abilities of the three potential biomarkers individually and a cross-validated biomarker model containing all three markers are presented as ROC curves in Fig. 1. The AUC values of the three biomarkers are presented in Table 2.
Biomarker detection abilities
Biomarker detection abilities
AUC: Area under the curve; 95% CI: 95% confidence interval; CA125: Cancer antigen 125; CYFRA 21-1: Cytokeratin 19 fragment; CEA: carcinoembryonic antigen.
AUC of ROC curves: CA125: 0.685, CI 0.609–0.761 (red); CYFRA 21-1: 0.686, CI 0.616–0.756 (green); CEA: 0.752, CI 0.680–0.824 (gray); biomarker panel: 0.795, CI 0.729–0.861 (blue).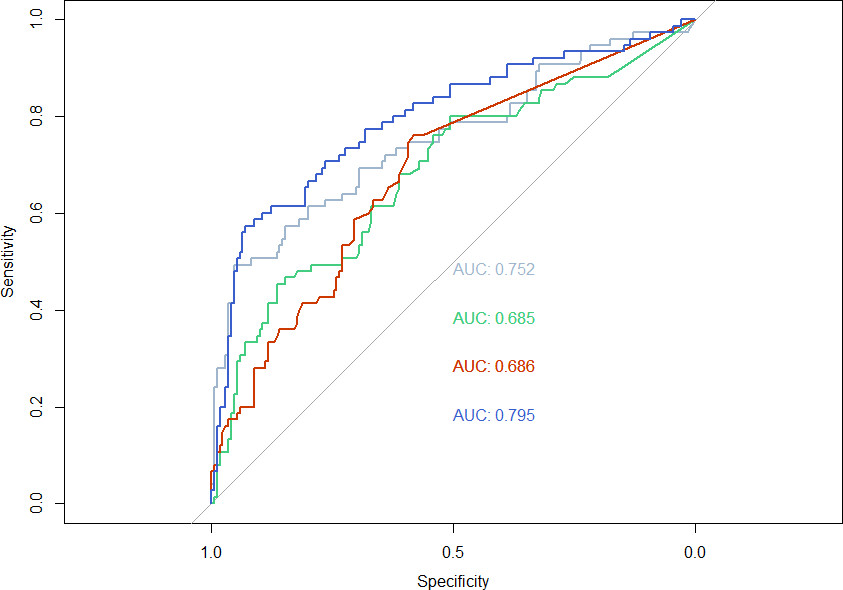
The cross-validated biomarker prediction model yielded a sensitivity and specificity of 57% and 93%, respectively. The negative predictive value was 83%.
Prediction models detecting stage I-II or III-IV lung cancer
Prediction models detecting stage I-II or III-IV lung cancer
Footnotes
Acknowledgments
The authors would like to thank Kristina Kock Hansen for identifying potential participants and administrating the database, Anne-Mette Gintberg, Marianne Mikkelsen, Rikke Maria Iversen and Marianne Kammer for enrolling the participants. Karin Larsen for administrating funding and ethics board approval, Nilosa Ushanthan, Sara Egsgaard and Camilla Davidsen for performing the laboratory analyses and Sandra Esperanza Rubio-Rask for entering data into the database. This work was supported by grants from Region of Southern Denmark [grant number: J.nr. 20/14276, Efond 481]; Gangstedfonden [grant number A35818], and Lillebaelt Hospital Research Foundation. The sponsors had no role in study design, in the collection, analysis and interpretation of data, in the writing of the report, and in the decision to submit the article for publication.
Author contributions
Conception: Morten Borg, Torben Frøstrup Hansen, Sara W.C. Wen, Anders Jakobsen, Rikke Fredslund Andersen, Line Nederby.
Interpretation or analysis of data: Morten Borg, Ole Hilberg, Ulla Møller Weinreich.
Preparation of the manuscript: Morten Borg.
Revision for important intellectual content: Morten Borg, Torben Frøstrup Hansen, Sara W.C. Wen, Anders Jakobsen, Rikke Fredslund Andersen, Ole Hilberg, Ulla Møller Weinreich, Line Nederby.
Supervision: Ole Hilberg, Ulla Møller Weinreich, Line Nederby.